Abstract
In the present study, we have evaluated the chemopreventive effects of perillyl alcohol (POH) against diethylnitrosamine-initiated and 2-AAF (2-acetylaminofluorine)-promoted hepatocarcinogenesis in Wistar rats. Efficacy of POH against 2-AAF-induced hepatotoxicity was evaluated in terms of biochemical estimation of antioxidant enzyme activities, histopathological changes and expression levels of proliferative markers. 2-AAF is a potent hepatotoxicant and a hepatic carcinogen that induces its effect by causing oxidative stress. Pre-treatment of POH prevented oxidative stress and tumour incidences. POH suppressed 2-AAF-induced early tumour markers, namely ornithine decarboxylase activity, thymidine phosphorylase and proliferating cell nuclear antigen (PCNA) protein and also suppressed the expression of pro-apoptotic protein P53. Histopathological findings revealed that POH-pretreated groups showed marked recovery. From our results, it could be concluded that POH markedly protects against chemically induced liver cancer and acts possibly by virtue of its antioxidant and antiproliferative activities.
Introduction
Carcinogenesis is a multistep and complex process, which is a consequence of a single or combination of various chemical, physical, biological and genetic damage to cells.
1
Hepatocellular carcinoma (HCC) is the fourth important tumour world wide.
2,3
It is the fourth most form of tumour in the world.
3
To understand the mechanism of biotransformation of normal cell to malignant cell, chemical hepatocarcinogenesis is considered as a preferred model in rat. In experimental animals, different model systems have been used to develop cancer in liver.
4
Rat liver is the most widely studied model of carcinogenesis that has been described for the study of the development of cancer.
5
The prognosis of cancer patients is poor as recurrence is rapid. Therefore, there is a need to develop more efficient approach for the chemoprevention of HCC.
6
Diethylnitrosamine (DEN) is an environmental carcinogen and hepatotoxicant that belongs to
Materials and methods
Chemicals
Reduced glutathione (GSH), oxidized glutathione (GSSG), glutathione reductase (GR), bovine serum albumin (BSA), 1,2-dithio-bis-nitrobenzoic acid (DTNB), 1-chloro-2,4-dinitrobenzene (CDNB), reduced nicotinamide adenine dinucleotide phosphate (NADPH), flavine adenine dinucleotide (FAD), Tween-20, 2,6-dichlorophenolindo-phenol (DCPIP), DEN, 2-AAF) and POH were obtained from Sigma Chemical (St Louis, Missouri, USA). All other chemicals and reagents were of the highest purity and commercially available.
Animals
Male Wistar rats of 4–6 weeks old (130–150 g) were obtained from Central Animal House of Hamdard University, New Delhi, India. They were housed in a ventilated room at 25 ± 5°C under a 12-h light/dark cycle. Acclimatization was for 1 week before the experiments, and the rats were given free access to standard laboratory feed (Hindustan Lever Ltd, Bombay, India) and water ad libitum. The study was approved by the Committee for the purpose of Control and Supervision of Experimental Animals (CPCSEA). Registration number and date of registration is IAEC no. 173/CPCSEA and 2000, respectively. CPCSEA guidelines were followed for animal handling and treatment.
Experimental protocols
To study the effect of pretreatment with POH against 2-AAF-induced hepatic oxidative stress, 30 rats were randomly divided into five groups of six rats each (Table 1). Group I received only corn oil (2.5 ml kg−1 body weight (b.wt.)) from days 1 to 11. Group II served as toxicant group and was given 2-AAF (0.02%) in powdered diet from days 7 to 11. Groups III and IV were pretreated with POH dissolved in corn oil (2.5 ml kg−1 b.wt.) at a dose of 50 and 100 mg kg−1 b.wt., respectively, by gavage from days 1 to 6. Thereafter, from days 7 to 11, the animals were pretreated with POH and fed with 2-AAF (0.02%) in powdered diet. Group V received only POH (100 mg kg−1 b.wt.) by gavage once daily for 11 days. Group V was used only to ensure that the higher dose does not produce any kind of hepatotoxicity if given alone. All animals were killed by cervical dislocation on day 12 and liver samples were taken for various biochemical parameters and at the same time, blood was collected for the estimation of serum marker enzymes.
To study the effect of pretreatment of POH against 2-AAF-induced hepatic oxidative stress, 30 rats were randomly divided into five groups of six rats each.a

POH: perillyl alcohol; 2-AAF: 2-acetylaminofluorine; b.wt.: body weight; D1: 50 mg kg−1 b.wt.; D2: 100 mg kg−1 b.wt.
aGroup V was used only to ensure that the higher dose does not produce any kind of hepatotoxicity if given alone. All animals were killed by cervical dislocation on day 12 and liver samples were taken for various biochemical parameters processed and at the same time blood was collected for the estimation of serum marker enzymes.
To study the effect of pretreatment with POH on 2-AAF-mediated expressions of ODC and thymidine phosphorylase, the groupings of animals were as described above (Table 2). Group I received only corn oil (2.5 ml kg−1 b.wt.). Group II were administered 2-AAF (0.02%) in powdered diet for 14 days and subjected to partial hepatectomy (PH) on 7th day after 2-AAF diet initiation. Groups III and IV served as prevention groups and POH dissolved in corn oil (2.5 ml kg−1 b.wt.) at doses of 50 and 100 mg kg−1 b.wt., respectively, were administered daily subsequent to 2-AAF administration in diet and subjected to PH on day 7. All the animals were killed on day 14 and liver were excised from each group for immunohistochemical studies.
To study the effect of pretreatment with POH on 2-AAF-mediated expressions of ODC and thymidine phosphorylase, 24 rats were randomly divided into four groups of six rats each.a

POH: perillyl alcohol; 2-AAF: 2-acetylaminofluorine; ODC: ornithine decarboxylase; PH: partial hepatectomy; b.wt.: body weight; D1: 50 mg kg−1 b.wt.; D2: 100 mg kg−1 b.wt.
aAll the animals were killed on day 14 of 2-AAF administration in diet and liver were excised from each group for immunohistochemical studies.
For tumour inhibition studies, the experimental schedule of Solt and Farber was followed. Animals weighing 100–150 g were randomized into five different groups (
For tumour inhibition studies, the experimental schedule of Solt and Farber was followed.a

POH: perillyl alcohol; DEN: diethylnitrosamine; 2-AAF: 2-acetylaminofluorine; b.wt.: body weight; i.p.: intraperitoneal.
aAnimals weighing 100–150 g were randomized into five different groups (
Post-mitochondrial supernatant preparation
Post-mitochondrial supernatant (PMS) was prepared as described by Naghma Khan and Sultana. 24 Liver was removed and cleaned with ice-cold saline (0.85% sodium chloride). Liver tissues were homogenized in chilled phosphate buffer (0.1 M, pH 7.4) using a Potter Elvehjen homogenizer (Remi, Laboratory Instruments, Mumbai, India) and were centrifuged at 3000 r min−1 for 10 min at 4°C by Eltek Refrigerated Centrifuge (model RC 4100 D, Remi, Laboratory Instruments, Mumbai, India) to separate the nuclear debris. The aliquot so obtained was centrifuged at 12,000 r min−1 for 20 min at 4°C to obtain PMS that was used as a source of enzymes.
Measurement of liver toxicity markers serum AST and ALT
Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) activity were determined by the method of Reitman and Frankel.
25
Each substrate, 0.5 ml (2 mM α-ketoglutarate and either 200 mM
Assay for LDH activity
Lactate dehydrogenase (LDH) activity has been estimated in serum by the method of Kornberg. 26 The assay mixture consisted of 0.2 ml serum, 0.1 ml 0.02 M NADH, 0.1 ml 0.01 M sodium pyruvate, 1.1 ml 0.1 M phosphate buffer PH 7.4 and distilled water in a total volume of 3 ml. Enzyme activity was recorded at 340 nm and the activity was calculated as nanomoles of NADH oxidized per minute per milligram of protein.
Estimation of GSH
GSH was assessed by the method of Jollow et al. A 1.0 ml of 10% PMS mixed with 1.0 ml of 4% sulphosalicylic acid, then incubated at 4°C for a minimum time period of 1 h and then centrifuged at 4°C at 1200
Estimation of LPO
The assay of lipid peroxidation (LPO) was carried out according to the method of Wright et al.
28
The reaction mixture in a total volume of 1.0 ml contained 0.60 ml phosphate buffer (0.1 m, pH 7.4), 0.2 ml microsomes and 0.2 ml ascorbic acid (100 mm). The reaction mixture was incubated at 37°C in a shaking water bath for 1 h. The reaction was stopped by adding 1.0 ml of 10% trichloroacetic acid. Following the addition of 1.0 ml of 0.67% thiobarbituric acid, all tubes were placed in a boiling water bath for 20 min and then transferred to a crushed ice bath before centrifuging at 2500
Assay for GPx activity
The activity of glutathione peroxidase (GPx) was calculated by the method of Mohandas et al. 29 A total of 2 ml volume mixture consisted of 0.1 ml ethylenediaminetetraacetic acid (EDTA; 1 mM), 0.1 ml sodium azide (1 mM), 1.44 ml phosphate buffer (0.1 M, pH 7.4), 0.05 ml GR (1 IU ml−1), 0.05 ml GSH (1 mM), 0.1 ml NADPH (0.2 mM), 0.01 ml hydrogen peroxide (H2O2; 0.25 mM) and 0.1 ml 10% PMS. The depletion of NADPH at 340 nm was recorded at 25°C. Activity of the enzyme was calculated as nanomoles of NADPH oxidized per minute per milligram of protein with the molar extinction coefficient of 6.22 × 103 M−1 cm.−1
Assay for GR activity
The activity of GR was measured by the method of Carlberg and Mannervik. 30 The reaction mixture consisted of 1.65 ml phosphate buffer (0.1 M, pH 7.6), 0.1 ml NADPH (0.1 mM), 0.05 ml GSSG (1 mM), 0.1 ml EDTA (0.5 mM) and 0.1 ml 10% PMS in a total volume of 2 ml. Enzyme activity was assessed at 25°C by measuring the disappearance of NADPH at 340 nm and was calculated as nanomoles of NADPH oxidized per minute per milligram of protein using molar extinction coefficient of 6.22 ×103 M−1 cm−1 .
Assay for GST activity
The activity of glutathione-
Assay for CAT activity
Catalase (CAT) activity was carried out by the method of Claiborne. 32 In short, the reaction mixture comprised of 0.05 ml of PMS, 1.0 ml of H2O2 (0.019 M) and 1.95 ml of phosphate buffer (0.1 M, pH 7.4) in a total volume of 3 ml. Changes in the absorbance were recorded at 240 nm and the change in absorbance was calculated as nanomoles of H2O2 consumed per minute per milligram of protein.
Assay for QR activity
The activity of quinone reductase (QR) was measured by the method of Benson et al. 33 The 3-ml reaction mixture consisted of 2.13 ml Tris-hydrochloric acid buffer (25 mM, pH 7.4), 0.7 ml BSA, 0.1 ml FAD, 0.02 ml NADPH (0.1 mM) and 50 µl (10%) PMS. The reduction in DCPIP was recorded calorimetrically at 600 nm and the enzyme activity was calculated as nanomoles of DCPIP reduced per minute per milligram of protein using molar extinction coefficient of 2.1 × 104 M−1cm−1 .
Estimation of protein
The protein concentration in all the samples was determined by the method of Lowry et al. using BSA as standard. 34
Histopathological examination
After the rats were killed, and their livers were quickly removed and preserved in 10% neutral-buffered formalin for histopathological processing. Sections were stained with hematoxylin and eosin before being observed under an Olympus microscope (Tokyo, Japan) at 40× magnification.
Immunohistochemistry
Liver sections on polylysine-coated slides obtained were fixed in neutral-buffered formalin and embedded in paraffin. Following deparaffinization and rehydration, sections were irradiated in 0.1 mol l−1 sodium citrate buffer (pH 6.0) in a microwave oven (medium low temperature) for 20 min and exposed to 3% H2O2 for 10 min to bleach endogenous peroxidases, followed by rinsing three times in Tris buffer (pH 7.4) for 10 min and selectively incubated under humid conditions using an anti-ODC antibody (1:400; Thermo Fisher Scientific, USA), anti-thymidine antibody (1:200; Santacruz Biotechnology, Inc., USA), anti-P53 antibody (1:200; Santacruz Biotechnology, Inc., Dallas, Texas, USA) and anti-PCNA antibody (1:200; Thermo Fisher Scientific, Rockford, IL, USA) for overnight at 4°C. Next day, the slides were washed three times in Tris buffer for 10 min each. The specificity of the antibodies was tested by omission of the primary antibodies and a positive control of rat tonsil tissue. After washing in Tris buffer (pH 7.4), tissues were visualized with 3,3′-diaminobenzidine (DAB) and counterstained with hematoxylin, mounted with DPX and cover slipped. Positive and negative controls were conducted in parallel with ODC-, thymidine phosphorylase-, P53- and PCNA-stained sections. Staining of sections with commercially available antibodies served as the positive control. Negative controls included staining tissue sections with omission of the primary antibody.
Quantitative evaluation of ODC and thymidine phosphorylase
According to the diffuseness of the DAB staining, sections were graded as 0 (no staining), 1 (staining 25%), 2 (staining between 25% and 50%), 3 (staining between 50% and 75%) or 4 (staining >75%). According to the staining intensity, sections were graded as follows: 0 (no staining), 1 (weak but detectable staining), 2 (distinct staining) or 3 (intense staining). Immunohistochemical scoring values were obtained by adding the diffuseness and intensity scores.
Quantitative evaluation of PCNA and P53 protein
Percentages of PCNA-immunostained nuclei (PCNA labelling index, LI PCNA) and P53-immunostained nuclei (P53 labelling index, LI P53) were calculated in each selected section for control rats, toxicant-treated rats and rats given POH as a modulator, using the following formula: number of labelled nuclei × 100/total number (labelled + unlabelled) of nuclei. P53 and PCNA immunostained nuclei were considered positive regardless of staining intensity. All the slides were examined by two independent observers who were unaware of the experimental protocol. The slides with discrepant evaluations were re-evaluated, and a consensus was reached. Measurements were carried out using an Olympus BX51 microscope (Tokyo, Japan) using objectives with 40× magnifications.
Statistical analysis
Differences between the groups were analyzed using analysis of variance followed by Dunnet’s multiple comparisons test. All data points are presented as the treatment groups mean ± standard error.
Results
POH pretreatment modulates serum toxicity markers AST, ALT and LDH
Protective effect of POH on serum AST, ALT and LDH level was observed. Significant protection (
Results of pretreatment of POH on serum enzymes like AST, ALT and LDH.a

POH: perillyl alcohol; 2-AAF: 2-acetylaminofluorine; AST: aspartate aminotransferase; ALT: alanine aminotransferase; LDH: lactate dehydrogenase; b.wt.: body weight; SE: standard error; D1: 50 mg kg−1 b.wt.; D2: 100 mg kg−1 b.wt.
aResults represent mean ± SE of six animals per group.
b2-AAF treatment leads to significant elevation in the serum marker enzymes in group II when compared with group I (
cPretreatment of POH restored activity of these enzymes in group III is significant when compared with 2-AAF-treated II group (
dPretreatment of POH restored activity of these enzymes in group and IV is significant when compared with 2-AAF-treated II group (
POH pretreatment increased the hepatic antioxidant enzymes level
Tables 5 and 6 show the effect of pretreatment of rats with POH on 2-AAF-mediated hepatic glutathione content, its metabolizing enzymes and antioxidant enzymes. 2-AAF caused significant depletion in the GSH content (
Results of pretreatment of POH on antioxidant enzymes like reduced GSH, GST, GR and GPx.a

POH: perillyl alcohol; 2-AAF: 2-acetylaminofluorine; GSH: reduced glutathione; CDNB: 1-chloro-2,4-dinitrobenzene; NADPH: reduced nicotinamide adenine dinucleotide phosphate; GPx: glutathione peroxidise; GR: glutathione reductase; b.wt.: body weight; SE: standard error; GST: glutathione-
aResults represent mean ± SE of six animals per group. NS: compared with the corresponding value for treatment with 2-AAF.
bCompared to corresponding value for saline-treated control
cCompared to corresponding value for saline-treated control
dProphylactic treatment with POH at both doses significantly modulated the alterations induced by 2-AAF
eProphylactic treatment with POH at both doses significantly modulated the alterations induced by 2-AAF
fProphylactic treatment with POH at both doses significantly modulated the alterations induced by 2-AAF
Results of pretreatment of POH on quinone reductase, catalase and LPO.a

POH: perillyl alcohol; 2-AAF: 2-acetylaminofluorine; NADPH: reduced nicotinamide adenine dinucleotide phosphate; LPO: lipid peroxidation; MDA: malondialdehyde; b.wt.: body weight; SE: standard error; D1= 50 mg kg−1 b.wt. D2 = 100 mg kg−1 b.wt.
aResults represent mean ± SE of six animals per group.
b2-AAF treatment leads to significant depletion in the activities of antioxidant enzymes in group II when compared with group I (
c2-AAF treatment leads to significant depletion in the activities of antioxidant enzymes in group II when compared with group I (
dPretreatment of POH restored activity of these enzymes in groups III and IV is significant when compared with 2-AAF-treated group II (
ePretreatment of POH restored activity of these enzymes in groups III and IV is significant when compared with 2-AAF-treated group II (
fPretreatment of POH restored activity of these enzymes in groups III and IV is significant when compared with 2-AAF-treated group II (
POH pretreatment decreased MDA formation
MDA formation was measured to demonstrate the oxidative damage on LPO of 2-AAF-induced liver injury in rats. A significant (
Effect of POH on the expression of ODC and thymidine phosphorylase
Semi-quantitative evaluation of ODC and thymidine phosphorylase expressions is represented in Figures 1 and 2. Immunohistochemical evaluation showed significant (

(a) ODC protein expression in control group almost showing no staining, (b) ODC protein expression in 2-AAF-treated group showing intense and positive stained hepatocytes as shown by the arrows. (c) ODC immunostaining of liver treated with low dose of POH showing very weak and diffuse staining of hepatocytes as shown by arrows. (d) Almost negligible ODC expression at high dose of POH-treated rat hepatic tissue as shown by arrows. (e) Semi-quantitative analysis of ODC expression among four groups. Results represent mean ± SEM of six animals. Results obtained are significantly different from group I (***

(a) Thymidine phosphorylase protein expression in control group almost showing no staining, (b) thymidine protein expression in 2-AAF-treated group showing intense and positive stained hepatocytes as shown by arrows, (c) thymidine immunostaining of liver treated with low dose of POH showing very weak and diffuse staining of hepatocytes as shown by arrows, (d) almost negligible thymidine phosphorylase expression at high dose of POH-treated rat hepatic tissue as shown by arrows. (e) Semi-quantitative analysis of thymidine phosphorylase expression among four groups. Results obtained are significantly different from group I (***
Tumour incidences and histopathological observations in tumour study
Table 7 shows incidence of tumours in control and experimental animals. The data provide a summary of the percentage incidence of liver cell tumours in treatment groups. Group I did not show any incidence of liver tumours. However, treatment with 2-AAF of DEN-initiated animals enhanced the development of liver tumours in 78% of the animals studied. In comparison, tumour incidence in the group of animals co-treated with POH at a lower dose (50 mg kg−1 b.wt.) was 46%, whereas in the group receiving the higher dose of POH (100 mg kg−1 b.wt.), the tumour incidence was reduced to almost 29%.
Summary of tumour data on the effect of POH on DEN-initiated and 2-AAF-promoted liver tumours.a

POH: perillyl alcohol; DEN: diethylnitrosamine; 2-AAF: 2-acetylaminofluorine; PH: partial hepatectomy; b.wt.: body weight.
aGroup I: control group; group II: DEN + 2-AAF + PH group; group III: DEN + 2-AAF + PH + POH (50 mg kg−1 b.wt.); group IV: DEN + 2-AAF + PH + POH (100 mg kg−1 b.wt.).
POH was tested for its chemopreventive effect in the hepatocarcinogenesis assay induced by DEN as initiator and 2-AAF as a promoter. In the control group, a normal histological appearance was seen and the hepatic architecture was maintained. In group II, that is, DEN + 2-AAF + PH, development of tumour nodules formed of neoplastic hepatocytes was observed. The residual normal liver tissue was seen as a thin strip at the edge of the nodule and the edge of the tumour nodule showed infiltration by lymphocytes. The tumour cells have large hyperchromatic nuclei with prominent nucleoli. There was however a significant degree of lymphocytic infiltration around the portal triad area of the hepatic lobules. Oral administration of POH at doses 50 and 100 mg kg−1 b.wt. significantly caused regression of tumour formation in dose-dependent manner as evidenced by decreased nodule and carcinoma formation in Figure 3.

Histopathological examination of rat liver ×40. (a) Showing normal histology of untreated rat liver. (b) DEN + 2-AAF + PH group shows liver tumours with dark eosinophilic staining. The tumour cells have large hyperchromatic nuclei with prominent nucleoli as shown by arrows. (c) DEN+2-AAF + PH + POH (50 mg kg−1 b.wt.)-treated rat liver showing almost normal histology with mild hepatic congestion as shown by arrows. (d) DEN + 2-AAF + PH + POH (100 mg kg−1 b.wt.)-treated rat liver shows normal liver histology almost similar to control group as shown by arrows. POH: perillyl alcohol; DEN: diethylnitrosamine; 2-AAF: 2-acetylaminofluorine; PH: partial hepatectomy; b.wt.: body weight.
Effect of POH on the expression of PCNA and P53 proteins in tumour study
Semi-quantitative evaluation of PCNA is represented in Figure 4. The number of PCNA positive cells increased substantially (

(a) PCNA protein expression in control group. (b) PCNA protein expression in DEN + 2-AAF + PH-treated group showing intense and positive stained hepatocytes surrounding the central vein as shown by arrows. (c) PCNA immunostaining of liver treated with low dose of POH showing diffused and less intense staining of hepatocytes as shown by arrows. (d) PCNA expression at higher dose of POH-treated hepatic tissue showing very few immunopositive cells as shown by arrows. (e) Semi-quantitative analysis of PCNA expression among four groups. Results obtained are significantly different from group I (***
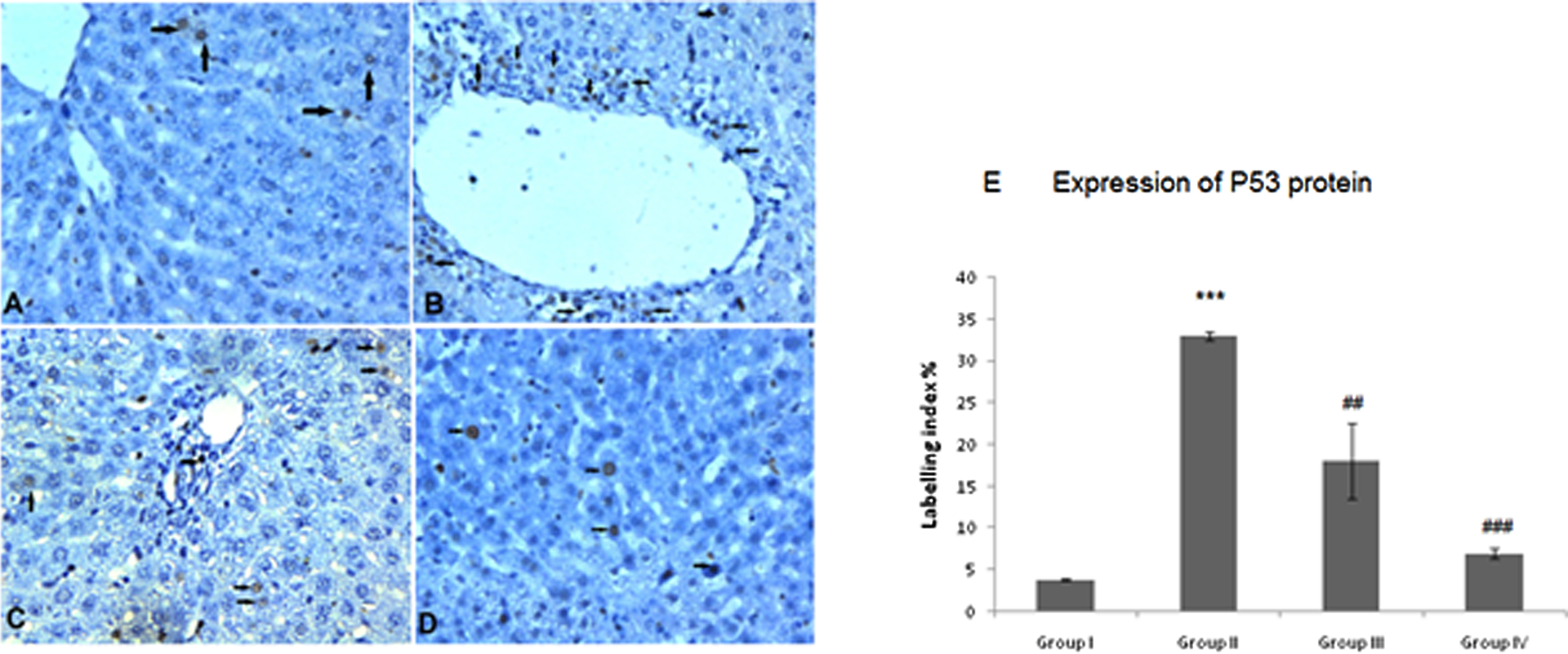
(a) P53 protein expression in control group, (b) P53 protein expression in DEN + 2-AAF + PH-treated group showing intense and positive stained hepatocytes near central vein as shown by arrows. (c) P53 immunostaining of liver treated with low dose of POH showing fewer staining around central vein as shown by arrows. (d) P53 expression in higher dose of POH-treated hepatic tissue showing very weak P53 staining as shown by arrows. (e) Semi-quantitative analysis of P53 expression among four groups. Results obtained are significantly different from group I (***
Discussion
In 2010, according to the data of American Cancer Society, 35 more than 24,000 new cases of liver cancer and intrahepatic bile duct cancer are reported and 19,000 deaths occurred in the United States. The present study was designed to investigate whether POH administration could inhibit the hepatic carcinogenesis in rats. POH is under phase I clinical trial testing of the cancer chemotherapeutic activity of POH, 36 which is in progress. Furthermore, since carcinogenesis is reversible until early promotion stage and entails a repetitive and prolonged exposure to promoter, its interruption at promotional stage is most appropriate and convenient. In the present study, we have demonstrated the role of POH to inhibit several events of 2-AAF-induced tumour induction and promotion response in rat liver. Chemopreventive activity of POH is related with a noticeable increase in tumour cell death by programmed cell death or apoptosis. 37 We have induced two-stage carcinogenesis by giving single i.p. administration of DEN and 2-AAF in diet and followed by PH in rats. PH acts as mitogenic stimuli. There was an induction of hyperplasia due to the enhancement of HCC development, which is caused by PH. 38 We observed a noticeable reversal of pathological expression, which gives sufficient evidence of modulating effect of POH on tumour development. We have observed tumours with neoplastic development in DEN- and 2-AAF-treated groups but with the pretreatment of POH to the rats, the number of tumours was less as well as the reversal of the pathological sequences with small foci of tumour nodule growth was also seen. The results we have obtained were further confirmed by the anti-tumour promotion activity of a POH against the toxicity of 2-AAF in rat liver by studying the oxidative stress and hyperproliferation markers. Oxidative stress occurs in all the stages of the progress of cancer with the genesis of other diseases. Antioxidant defence enzymes consists of GPx, GR and GSH, which protect the cellular macromolecules against oxidative damage by detoxifying carcinogens both by destroying their reactive centres or by conjugating with endogenous ligands facilitating their excretion. 39 The metabolism of 2-AAF occurs in rat liver, which results in the production of genotoxic metabolites and also initiates the formation of rat liver tumours. The microsomal generation of reactive oxygen by redox cycling of suitable 2-AAF metabolites is the possible mechanism responsible for tumour promotion by 2-AAF. 40 Hepatocarcinogenesis is promoted by the administration of 2-AAF in the diet by in vivo regulation of various proteins that are vital for a normal cell cycle progression. Due to the loss of cellular proliferation control, there is a loss of LPO in preneoplastic hepatocellular membranes. 41 POH ameliorated 2-AAF-induced inhibition of the activities of antioxidant enzymes, namely GPx, GR and phase-II metabolizing enzymes such as GST and QR. Our study shows induction of hepatic GST and QR activity by POH pretreatment. CAT, which is responsible for the detoxification of H2O2, has been found to be elevated in the POH-pretreated groups. Several chemopreventive compounds are well known to degrade electrophilic metabolites by the induction of QR and GST activities. One of the major enzymes of xenobiotics metabolism is QR that protects the cell against mutagenicity and carcinogenicity resulting from free radicals and toxic oxygen metabolites by carrying out two-electron reductions. It has been reported that there is a correlation between the induction of QR activity and prevention of cancer. 42 There was also a dose-dependent decrease in the 2-AAF-mediated susceptibility of hepatic microsomal membrane-induced LPO through decreased production of free radicals as revealed by ameliorated level of MDA. GSH is a first line of defence, neutralizes the hydroxyl radical and plays a key role against inflammatory responses and oxidative stress. 43 A significant restoration of glutathione and its dependent enzymes, such as GR and GPx, to normal levels in the dose-dependent manner in POH-pretreated groups was found. Due to the damage of the structural integrity of liver by 2-AAF, there is an increase in the level of serum transaminases in liver. 44 POH showed dose-dependent protection and maintained the structural integrity of hepatocytes. This was evident from the significant reduction in serum AST, ALT and LDH activities. It has been shown that 2-AAF induces ODC activity and rate of DNA synthesis by several folds in rat liver. 45 ODC is the rate-limiting enzyme in the biosynthesis of polyamines, which are essential for DNA synthesis and also for cell proliferation. There is usually an increase in ODC activity during tumourigenesis. Therefore, ODC induction is an important marker of tumour promotion and its inhibition is a promising tool for screening antitumourigenic agents. 46 In the present study, 2-AAF administration led to a marked increase in the ODC and thymidine phosphorylase expression in the liver, which was significantly and dose dependently ameliorated by pretreatment with POH, which suggested the antipromoting effect of POH. The results of histopathology were supported by the results of immunohistochemistry. Dysregulation of the cell proliferation is one of the essential features of carcinogenesis. PCNA is a highly conserved nuclear protein, which is considered to be a functional marker for the assessment of tumour cell proliferation and progression. 47 Gene alterations, which regulates cell cycle events, play a key role in the process of carcinogenesis. The expression of PCNA reaches its maximum during the synthesis phase (S phase), which depends on the cell cycle phase. 48 It has been reported that the PCNA was over expressed in various malignancies including liver cancers. 49 The number of PCNA positive cells increased substantially in DEN + 2-AAF + PH-treated group but POH suppressed the proliferation of liver cells at both the doses. Thus, we can propose that PCNA expression was downregulated by POH pretreatment, which inhibited the proliferation induced by 2-AAF administration. The P53 is a transcription factor, which helps in the suppression of tumour and regulation of cell cycle, DNA repair and apoptosis. 50 Our study revealed that DEN + 2-AAF + PH-treated group showed more P53 immunopositive staining when compared with control group, while pretreatment with POH significantly attenuated the P53 immunopositive staining. In the present study, hepatocytes of both control and POH-pretreated rats expressed low P53 protein level while DEN + 2-AAF + PH-treated rats experienced accumulation of P53 protein. Treatment of POH in rats with chronic exposure of DEN + 2-AAF + PH resulted in marked decline in protein expression of P53 and PCNA, induction of various antioxidant and phase II enzymes, scavenging reactive oxygen species, reduction in the levels of tumour promoter markers ODC and thymidine phosphorylase and decrease in the percentage incidence of tumours in two-stage hepatic carcinogenesis. Thus, our data suggest that POH is a potent chemopreventive agent and inhibits 2-AAF-induced hepatic carcinogenesis by delaying tumour formation and reducing the frequency of tumours and reducing the oxidative damage in Wistar rats.
Footnotes
Funding
This work was financially supported by the University Grants Commission (UGC), New Delhi, India.
Conflict of interest
The authors declared no conflicts of interest.
