Abstract
The present study investigated the hepatoprotective role of selenium during alloxan-induced diabetes in rats. Male Wistar rats were divided into four groups, namely, normal control, selenium treated, diabetic, and selenium-treated diabetic. Diabetes was induced in the animals by injecting alloxan intraperitoneally at a dose rate of 150 mg/kg body weight. Selenium in the form of sodium selenite was supplemented to rats at a dose level of 1 ppm in drinking water, ad libitum for two time durations of 2 and 4 weeks. The effects of different treatments were studied on various parameters in rat liver, which included serum glucose levels, serum insulin levels, alkaline phosphatase (ALP), aspartate aminotransferase (AST), alanine aminotransferase (ALT), lipid peroxidation (LPO), glutathione reduced (GSH), oxidized glutathione (GSSG), total glutathione (TG), superoxide dismutase (SOD), catalase (CAT), glutathione reductase, glutathione peroxidase, metallothionein (MT), and histoarchitecture. A significant increase in the serum glucose levels, LPO levels, and in enzyme activities of ALP, ALT, and AST was observed in diabetic rats which, however, got decreased significantly upon supplementation with selenium. On the contrary, decreased enzyme activities of GSSG, SOD, and CAT and depressed levels of GSH as well as serum insulin levels were observed in diabetic rats which got improved following selenium supplementation. Interestingly, MT levels were increased both in diabetic and selenium-treated diabetic rats. Further, marked alterations in histoarchitecture were seen in diabetic rats with the prominent features being congestion in sinusoids, lipid accumulation, and centrilobular hepatocyte degeneration. However, selenium treatment to diabetic rats showed overall improvement in the hepatic histoarchitecture.
Introduction
Diabetes is associated with altered metabolic processes and enzymatic defenses that adversely affect functioning of the various organs. Diabetes is the interplay of many factors that lead to multiple complications including oxidative stress. Increase in oxidative stress has been directly linked with the pathogenesis of diabetes. 1 Oxidation of lipoproteins, alterations in polyol pathways, and glucose autoxidation lead to oxidative stress. 2,3 Diabetic patients have been reported to show significant decrease in antioxidant enzyme concentrations when compared with healthy individuals. 4 Apart from regulation of blood glucose levels in diabetes, alleviation of free radical insult in the diabetic tissues is also a matter of concern. An imbalance in the levels of micronutrients (trace elements) leads to diabetic conditions and vice versa is also true. 5
In the present study, selenium is the trace element of interest. Selenium has been reported to prevent varied pathological states, including carcinogenesis both in humans and experimental animal models. 6,7 A recent study showed protective effects of selenium on antioxidant enzymes and histological changes during colon carcinogenesis. 8 Selenium also plays important role in preserving membrane integrity and stimulating apoptosis. 9 –11 Moreover, there are studies that evidenced the role of selenium as an effective protector against DNA damage and inflammation. 12,13 The present study was conducted to investigate the possible curative action of selenium on the antioxidant defense system, liver functions, and altered histoarchitecture during alloxan-induced diabetic rats.
Methods
Chemicals and equipment
All the chemicals used in the present study were of analytical grades. Sodium selenite was purchased from E.Merck (Germany). Nicotinamide adenine dinucleotide phosphate (NADPH), reduced glutathione (GSH), nitro blue tetrazolium (NBT), and 5,5′-dithiobis (2-nitrobenzoic acid) (DTNB) were purchased from Sigma Chemicals Co. (St Louis, Missouri, USA).
Experimental design
Grouping of animals
Male Wistar rats weighing 150–200 g were housed in polypropylene cages under hygienic conditions strictly in accordance with the guidelines as outlined by the institutional animal ethics committee. After acclimatizing for at least 1 week in the departmental animal house, the animals were randomly and equally divided into four groups containing eight animals each. Animals in control group were given normal diet. In diabetic group, animals were administered a single intraperitoneal injection of alloxan at a dose level of 150 mg/kg body weight. 14 In selenium group, animals were given selenium in the form of sodium selenite in drinking water ad libitum at a dose level of 1 ppm drinking water 15 for two time durations of 2 and 4 weeks. In combined group, animals were given a single intraperitoneal injection of alloxan at a dose of 150 mg/kg body weight and selenium in the form of sodium selenite in drinking water ad libitum at a dose level of 1 ppm drinking water. Animals in all groups were fed standard laboratory feed and water ad libitum for two time durations of 2 and 4 weeks. Diabetic status of rats was analyzed by urine reagent strips (Tech Diagnostic, Las Vegas, USA). Animals with urine glucose higher than 500 mg/dl at 3rd day of injection were considered diabetic.
Glucose and insulin levels in serum
Glucose levels were measured in serum using Abcam glucose assay kit (Cambridge, UK). Insulin levels were measured using Abcam enzyme-linked immunosorbent insulin assay kit.
Antioxidant defense status
Lipid peroxidation
Lipid peroxide formation was assayed by the method followed by Wills.
16
Since malondialdehyde (MDA) is a degradation product of peroxidized lipids, a thiobarbituric acid (TBA)-MDA chromophore was taken as an index of lipid peroxidation (LPO), which produced pink color having absorption maximum at 532 nm. The amount of MDA formed was calculated on the basis of molar extinction coefficient of MDA-TBA chromophore, and the results were expressed as
Reduced glutathione
Glutathione content was estimated according to the method of Ellman.
17
GSH is a nonprotein sulfydryl compound. DTNB is a disulfide compound, which is readily reduced by sulfydryl compounds (SH) to form 1 mole of 2-nitro-5-mercaptobenzoic acid per mole of SH. The nitromercaptobenzoic acid anion released has an intense yellow color that was used to measure –SH groups at 412 nm, and the results were expressed as
Glutathione reductase
The assay of glutathione reductase (GR) activity was performed according to the method by Carlberg and Mannervik. 18 GR is a flavoprotein that catalyzes the NADPH-dependent reduction of glutathione disulfide (oxidized glutathione; GSSG) to GSH. The oxidation of NADPH was followed spectrophotometrically at 340 nm.
TG content
The total glutathione (TG) content was measured by the method of Zahler and Cleland. 19 The method is based on the reduction with dithierythritol and determination of the resulting monothiols with DTNB. The absorbance resulting from the monothiols was determined by extrapolating the linear plot of the curve at the time of addition of DTNB and subtracting the blank value from a sample containing no disulfide.
Oxidized glutathione
GSSG was estimated by subtracting the value of GSH from TG levels.
Glutathione peroxidase
Glutathione peroxidase (GPx) was assayed by the method of Flohe and Gunzler. 20 This enzyme catalyzes the formation of GSSG from GSH and hydrogen peroxide (H2O2). GSSG was reduced by excess of GR and simultaneous oxidation of NADPH was measured at 340 nm. The activity of enzyme was expressed as micromoles of NADPH oxidized per minute per milligram protein.
Catalase
Catalase (CAT) was estimated spectrophotometrically by following the method described by Luck. 21 The optical absorption of H2O2 solution was measured at 240 nm on decomposition of H2O2 with CAT. The amount of H2O2 decomposed was calculated on the basis of molar extinction coefficient of H2O2, and the results were expressed as micromoles of H2O2 decomposed per minute per milligram protein.
Superoxide dismutase
Superoxide dismutase (SOD) assay was performed according to the method of Kono. 22 The reduction of NBT to blue formazon mediated by hydroxylamine hydrochloride and was measured under aerobic conditions. The addition of SOD inhibited the reduction of NBT by superoxide anions, which were generated by photooxidation of hydroxylamine hydrochloride. The extent of inhibition was taken as measure of enzyme activity.
MT estimation
Liver metallothionein (MT) was estimated by the biochemical method of Viarengo et al. 23 MT (20 cysteine groups) is a protein sulfydryl (–SH) compound and DTNB is a disulfide compound, which is readily reduced by SH to form 1 mole of 2-nitro-5-mercaptobenzoic acid per mole of SH. The nitromercaptobenzoic acid anion released has an intense yellow color and was used to measure –SH groups at 412 nm.
Liver marker enzymes
Aspartate aminotransferase
Hepatic aspartate aminotransferase (AST) estimation was performed by the method of Rietman and Frenkel. 24 AST-catalyzed reaction produced oxaloacetate, which was coupled with 2,4 dinitrophenyl hydrazine (DNPH) to produce corresponding hydrazone that formed color in alkaline medium and was measured at 510 nm.
Alanine aminotransferase
Hepatic alanine aminotransferase (ALT) was done by the method of Reitman and Frankel. 24 ALT catalyzes the formation of pyruvate which was coupled with DNPH to produce corresponding hydrazone that formed color in alkaline medium and was measured at 510 nm.
Alkaline phosphatase
The alkaline phosphatase (ALP) enzyme activity was estimated by following the method of Kind and King.
25
The hydrolysis of
Histopathlogical studies
For various histological studies, small sections of liver from the normal control and treated animals were taken, washed with ice-cold 0.9% sodium chloride and were fixed in the 10% formal saline for about 24 h. After fixation, the tissues were embedded in wax according to the standard technique. 26 Paraffin sections with 5–7 µm thick were cut and then stained with hemotoxylin and eosin and finally were observed under the light microscope at 200× as well as 400×.
Statistical analyses
The statistical significance of the data was determined using one-way analysis of variance, followed by a multiple post hoc test least significant difference with 5% considered significant. The results are represented as mean ± SD.
Results
All the results of various treatment groups have been compared with the normal controls and additionally the results from selenium-treated diabetic group have been compared with the results of diabetic group.
Serum glucose and insulin levels
A statically significant increase was observed in the glucose levels in serum (Table 1) of diabetic rats, which, however, got significantly decreased upon selenium supplementation. Also there was significant decrease in the serum insulin levels (Table 2) in diabetic rats which got elevated after selenium supplementation.
Effect of selenium supplementation on serum glucose levels and insulin levels in normal and diabetic rats.a
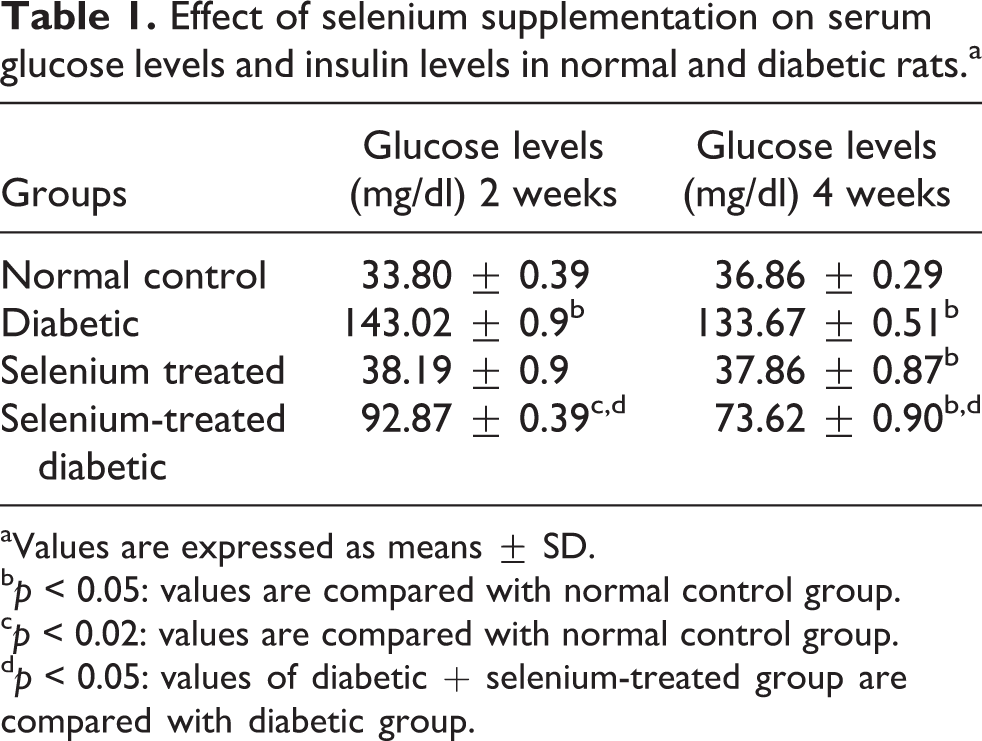
aValues are expressed as means ± SD.
b
c
d
Effect of selenium supplementation on serum insulin levels in normal and diabetic rats.a
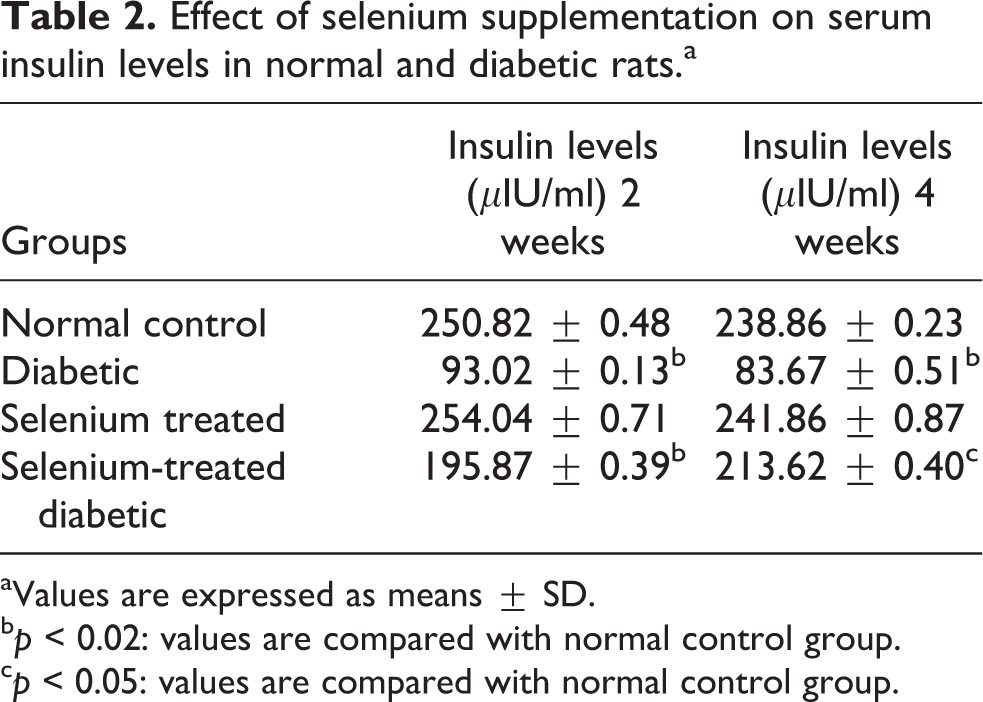
aValues are expressed as means ± SD.
b
c
Antioxidant status in liver
A statistically significant increase was observed in the formation of MDA products at the intervals of 2 and 4 weeks (
Effect of selenium on hepatic LPO levels and SOD and CAT activities of normal and diabetic rats.a

LPO: lipid peroxidation; SOD: superoxide dismutase; CAT: catalase; MDA: malondialdehyde; H2O2: hydrogen peroxide.
aValues are expressed as means ± SD.
b
c
d
e
f
g
A significant decrease in SOD activity (
The CAT activity was significantly decreased in diabetic rats (
Glutathione system
GSH contents in various treatment groups are shown in Table 4. Diabetic rats showed significant decrease (
Effect of selenium on hepatic GSH, TG, and GSSG levels of normal and diabetic rats.a

GSH: reduced glutathione; TG: total glutathione; DTNB: 5,5′-dithiobis (2-nitrobenzoic acid); GSSG: oxidized glutathione.
aValues are expressed as means ± SD.
b
c
d
e
TG content was significantly increased in diabetic rats as compared to normal rats as shown in Table 4. When selenium was supplemented to diabetic rats, the glutathione content decreased significantly (
GSSG levels (Table 4) were found to be increased significantly in diabetic rats at both the intervals (
GR activity was decreased significantly (
Effect of selenium supplementation on hepatic GR and GPx activities of normal and diabetic rats.a

GR: glutathione reductase; GPx: glutathione peroxidase; NADPH: nicotinamide adenine dinucleotide phosphate.
aValues are expressed as means ± SD.
b
c
Metallothionein
MT content in diabetic rats was found to be significantly increased at 4 weeks (
Effect of selenium on hepatic MT levels of normal and diabetic rats (mg/g).a

MT: metallothionein.
aValues are expressed as means ± SD.
b
c
d
Liver marker enzymes
Hepatic ALP activity in diabetic rats was significantly (
Effect of selenium supplementation on hepatic ALP, ALT, and AST activities in normal diabetic rats.a

ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase.
aValues are expressed in means ± SD.
b
c
d
e
Hepatic histoarchitecture
Histological investigations of liver as observed by light microscopy are shown in various photomicrographs (Figures 1 to 4). In the normal control animals, clear-cut lobules separated by interlobular septa, traversed by portal vein were observed. Plates of hepatic cells were seen radiating from the central canal and hepatic sinusoids were disposed irregularly. There was no portal tract and nuclei were round and their sizes were more or less the same except in binucleated cells (Figures 1 and 2). As evident from the light micrograph (Figures 3 and 4) of liver sections of diabetic animals, there was congestion in the sinusoids with signs of mild centrilobular hepatocyte degeneration. Lymphocytoplasmocytic infiltration in the portal tracts and lipid accumulation were also seen in the liver of diabetic rats. Although polyhedral shape of the cells was intact, binucleated cells were increased in number in the diabetic liver. Liver sections from the selenium-treated animals (Figures 5 and 6) showed more or less the normal structure. Selenium treatment to diabetic rats resulted in overall improvement in histoarchitecture of the liver, as there was less lipid deposition (Figures 7 and 8). Lymphocytic infiltration was still visible but much reduced than the diabetic rats.

Liver section from a normal control rat. H&E staining (×200). H&E: hematoxylin and eosin.

Liver section from a normal control rat. H&E staining (×400). H&E: hematoxylin and eosin.

Liver section from diabetic rat. H&E staining (×200). H&E: hematoxylin and eosin.

Liver section from diabetic rat. H&E staining (×400). H&E: hematoxylin and eosin.

Liver section from selenium-treated normal rat. H&E staining (×200). H&E: hematoxylin and eosin.

Liver section from selenium-treated normal rat. H&E staining (×400). H&E: hematoxylin and eosin.

Liver section from selenium-treated diabetic rat. H&E staining (×200). H&E: hematoxylin and eosin.

Liver section from selenium-treated diabetic rat. H&E staining (×400). H&E: hematoxylin and eosin.
Discussion
Hyperglycemia is the characteristic feature of diabetes that enhances nonenzymatic binding of glucose to proteins, which, in turn, causes functional changes in the antioxidant enzymes. In the present study, significant increase in the levels of LPO levels as well as glucose levels in serum indicated the coexistence of the oxidative stress in diabetic rats. The observation is in sync with the recent study by Mallick et al. that reported clear association between lipid peroxides and glucose concentrations in diabetes mellitus. 27 The observed decrease in the SOD activity in the present study might be due to the direct damaging effect of free radicals on the enzyme. 28 –30 Further, the decrease in the activities of CAT could be owed to the increased stress conditions and loss of micronutrients. 31 The observed decrease in the GSH content and GR activity is the result of defective functioning of γ-glutamyl cysteine synthase due to its excessive glycation and defective in vivo transportation. 32 Also the observed elevated levels of glucose in serum could have contributed toward accelerated glycation of GR that markedly affected its enzyme activity. As reported by Robert Blakytny and John Harding, 33 suppression of enzyme activity of GR further caused decrease in GSH contents, thus providing one possible explanation for the early loss of GSH in diabetic rats.
MT is a transition metal-binding protein and is found in all eukaryotes and in some prokaryotes. 34,35 An increase in MT in livers of diabetic rats was noticed in the present study, which is in corroboration with a previous study. 36 The elevation of MT during diabetes is understandable through its antioxidant action. 37 The rise in MT levels could also be the result of the natural protective response of the liver tissue so as to scavenge free radicals. 38,39 Selenium supplementation to diabetic animals significantly modulated the levels of GSH, thus proved its protective efficacy in restoring GSH levels. 40 The vulnerability of diabetes to oxidative stress is due to the lower antioxidant micronutrients. 41 However, supplementation with selenium restored various anomalies by preventing oxidation of proteins due to stabilization of SH groups in glutathione system.
Accumulation of fat and hyperlipidemia has been reported during diabetes by Ohno et al. 42 ALT and AST activities are used as the indicators of hepatocyte damage. 43 In our study, diabetic rats showed increased activities of these enzymes, which established hepatic damage and deficiency of insulin. Moreover, the activity of ALP was increased in diabetic rats as compared to normal control rats, which could be due to altered liver functions during diabetes. 44 It might be a consequence of narrowing of biliary channels due to fatty infiltration and congestion of sinusoids. 45,46 These changes in liver marker enzymes are substantiated by our observations in histopathology of the liver. The increased protein catabolism accompanying gluconeogenesis and urea formation could have contributed toward the elevation of these transaminases. 47 Selenium supplementation to the diabetic animals revealed a significant decrease in the elevated activities of these enzymes that could be due to the ability of selenium to regulate membrane integrity and antioxidant defense enzymes.
Histoarchitecture of diabetic liver showed plasmolytic infiltration causing distortion of usual concentric arrangements of hepatocytes and congestion of sinusoids. There was occasional inflammation due to the infiltration of lymphocytes and monocytes in the liver of the diabetic animals as reported in previous studies. 48,49 The observed accumulation of lipid droplets is due to increased influx of fatty acids into the liver as a consequence of hypoinsulinemia as well as low capacity of excretion of lipoprotein secretion because of deficiency of apolipoprotein B synthesis. 42,50 Administration of selenium to diabetic rats resulted in appreciable moderation of the histoarchitectural changes with regard to integrity of the cells as a whole as well as the cell organelles. The observed modulatory effects of selenium can well be attributed to its capacity in maintaining the membrane integrity either by some direct mechanism or by indirectly scavenging the free radicals responsible for increased LPO. In other words, the improvement in histoarchitecture in diabetic rats by selenium is due to its capacity to manage the membrane dynamics of cell. The present study, therefore, concludes that selenium helps in maintaining the integrity of the hepatic structure by containing the damage due to free radicals onslaught and also restores the altered antioxidative defense system in diabetic rats.
Footnotes
Authors’ Note
The authors CZ and HC contributed equally to this work.
Conflict of interest
The authors declared no conflicts of interest.
Funding
The research work by JL was sponsored by Jiangsu Provincial Bureau of Health Foundation (H201356) and International Exchange Program, The 4th “333” Scientific Research Project of Jiangsu Province (BRA2014058), and Jiangsu Six Talent Peaks Program (2013-WSN-013). It was also supported by the Xuzhou Outstanding Medical Academic Leader project and Xuzhou Science and Technology Grant (XM13B066, KC14SX013).
