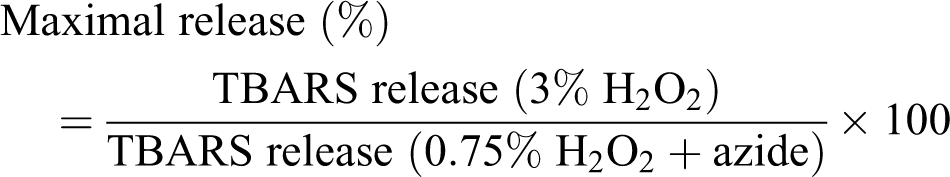Abstract
In the present study, we investigated the antioxidant effect of gallic acid (GA) on membrane lipid peroxidation and osmotic fragility in alloxan-induced diabetic Wistar rats. GA was administered orally at doses of 5, 10, and 20 mg/kg body weight for 45 days, after which liver and kidney tissues were analyzed for the degree of lipid peroxidation, reduced glutathione, and the activities of antioxidants such as catalase, superoxide dismutase, and glutathione peroxidase. Administration of GA to alloxan-induced diabetic rats reduced the blood glucose level with an increase in the level of insulin. Liver and kidney tissues from diabetic animals exhibited disturbances in antioxidant defense compared with normal rats. GA at a dose of 20 mg/kg b.w. showed a significant effect than that of the other doses. In addition, the results revealed that GA protected the integrity of erythrocyte membrane in diabetic rats as demonstrated by lower percentage of hemolysis and resistance to hydrogen peroxide-induced peroxidation. The anti-hyperglycemic activity of GA in alloxan-induced diabetic rats was also comparable with glibenclamide, a reference drug. These results suggest that GA could provide a beneficial effect on diabetes by decreasing oxidative stress-related diabetic complications.
Introduction
Diabetes mellitus, a metabolic disorder characterized by persistent hyperglycemia, is associated with defects in insulin secretion, insulin action, or both. Diabetes Atlas, International Diabetes Federation, reported that the number of people with diabetes worldwide is predicted to rise from 366 million in 2011 to 552 million by 2030. 1 In particular, the number of people with diabetes in India, which is currently around 42 million, is expected to rise to 80 million by 2030. Several pathogenetic processes associated with diabetes include hypoinsulinemia, dysregulated carbohydrate and lipid metabolism, formation of advanced glycation end products, 2 and oxidative stress. 3
Increasing evidences suggested that oxidative stress plays a major role in the pathogenesis of diabetes mellitus. 4 Persistent hyperglycemia in diabetes induces reactive oxygen species (ROS) production by glucose auto-oxidation, 5 activation of protein kinase C, 6 and increased flux through the hexosamine pathway. 7 This excess generation of free radicals damages the pancreatic β-cells through the induction of apoptosis. 8 Moreover, pancreatic β-cells are particularly vulnerable to the adverse effects of ROS due to its lower antioxidant capacity as compared to other tissues. 9 The impaired red blood cell (RBC) deformability due to oxidative stress plays a key role in the pathogenesis of chronic vascular complications of diabetes mellitus and progression of renal failure. 10 In diabetic condition, the antioxidant defense present in the system may not be adequate to neutralize the excess ROS produced. Hence, there is a need for an external source of antioxidants to neutralize the free radical load in the body. Antioxidants are considered as possible protective agents that reduce oxidative damage and retard the progress of many chronic diseases. 11 The traditional medicinal plants with various active principles and properties have been used to treat a variety of human diseases including diabetes, coronary heart disease, and cancer. 12 Wide array of plant-derived active principles has shown antidiabetic activity. 13,14 The main active constituents like alkaloids, glycosides, steroids, terpenoids, polyphenols, and inorganic ions have the following beneficial activities: manipulating the carbohydrate metabolism by multiple mechanisms, preventing and restoring integrity and functioning of β-cells, insulin release, improving glucose uptake and utilization, and antioxidant properties. 15,16 Gallic acid (GA; 3,4,5-trihydroxybenzoic acid), an endogenous plant phenol is present in a wide variety of plants such as tea, grapes, berries, fruits, and wine. It has been reported that among the numerous polyphenols tested, GA showed maximum absorption by the system. 17 It is a potent free radical scavenger and an inducer of differentiation and programmed cell death in a number of tumor cell lines 18,19 and exhibits various pharmacological actions such as anti-inflammatory, antimicrobial, and potent antioxidant effects. 20 –22 GA has been shown to protect the myocardium against isoproterenol-induced oxidative stress in rats, 23 It is recognized as safe by the Food and Drug Administration, and has been widely used as an antioxidant in food. 24 Toxicity studies in rodents showed that no observed adverse effect level was 120 mg/kg b.w., a dose equivalent to 2.9 g in a 70-kg man. 25 Our study hence aimed to evaluate the effect of GA on blood glucose, insulin, oxidative stress, enzymatic and nonenzymatic antioxidants, the erythrocytes osmotic fragility, and lipid peroxidation in alloxan-induced diabetic rats.
Materials and methods
Drugs and chemicals
All the biochemicals used in this experiment were obtained from Sigma Chemical Company (St Louis, Missouri, USA). All other chemicals were obtained from Hi Media (Mumbai, Maharashtra, India) and SD Fine Chem Limited (Mumbai, Maharashtra, India).
Animals and treatment
Male albino Wistar rats (170–200 g) were obtained from the Institutional Animal House, Bharathidasan University, Tiruchirappalli, Tamil Nadu, India, and were maintained under a constant 12-h light and dark cycle at 21–23°C. The animals were maintained in accordance with the guidelines of the National Institute of Nutrition, Indian Council of Medical Research, Hyderabad, Andhra Pradesh, India. The study was approved by the Institutional Animal Ethical Committee. Throughout the experimental period, the animals were fed with a balanced commercial pellet diet (Hindustan Lever Ltd, Mumbai, Maharashtra, India) with a composition of 5% fat, 21% protein, 55% nitrogen-free extract, and 4% fiber (wt/wt) with adequate mineral and vitamin contents. Diet and water were fed ad libitum.
Rats were injected intraperitoneally with freshly prepared alloxan monohydrate in normal saline at a dose of 150 mg/kg b.w.. 26 Because alloxan induces fatal hypoglycemia as a result of massive pancreatic insulin release, rats were treated with 20% glucose solution (5–10 mL) orally after 6 h. The rats were then kept for the next 24 h on 5% glucose solution bottles in their cages to prevent hypoglycemia. After 2 weeks, rats with moderate diabetes having glycosuria and hyperglycemia (i.e. with blood glucose levels of 200–300 mg/dL) 27 were chosen for the experiment.
Experimental design
In the present study, a total of 36 rats were used. The rats were divided into six groups with six rats in each group. Group 1: normal control rats treated with phosphate-buffered saline (PBS; vehicle control), Group 2: alloxan-induced diabetic rats, Groups 3–5: diabetic rats treated with graded concentrations of GA (5, 10, and 20 mg/kg b.w.), respectively, 28 and Group 6: positive control diabetic rats treated with glibenclamide (600 µg/kg b.w.). 29 GA and glibenclamide were given in aqueous solution daily using an intragastric tube for 45 days at a dosage level of 10 mL/kg b.w. Fasting blood glucose (FBG) was monitored every 7 days throughout the experiment. At the end of the experimental period, the animals were deprived of food overnight and then sacrificed by cervical decapitation. Blood samples were collected into a tube containing heparin for the estimation of blood glucose levels, and the plasma was separated for the estimation of insulin. Liver and kidney were dissected out, washed in ice-cold saline, patted dry, weighed, and subjected to biochemical estimation.
Preparation of hemolysate
Blood samples in ethylenediaminetetraacetic acid (EDTA) tubes were centrifuged at 3000 rpm for 5 min to remove the buffy coat. The packed cells obtained at the bottom were washed twice with PBS (0.9% NaCl in 0.01 M phosphate buffer, pH 7.4). A known amount of erythrocytes were lysed with hypotonic phosphate buffer. The hemolysate was obtained after removing the cell debris by centrifugation at 3000 rpm for 5 min and used for the determination of thiobarbituric acid reactive substances (TBARS), superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx) activities and reduced glutathione (GSH).
Analytical procedure
Determination of blood glucose, plasma insulin, and OGTT
Blood glucose was determined by glucose oxidase method. 30 Plasma insulin was assayed by enzyme-linked immunosorbent assay (ELISA) using the rat insulin ELISA kit (EMD Millipore Corporation, Billerica, Massachusetts, USA).
The oral glucose tolerance test (OGTT) was performed according to the method of Crowe et al. 31 After overnight fasting, “0”-min blood sample (0.2 mL) was taken from the normal and experimental rats by cyanacular puncture. Without delay, glucose solution (2 g/kg b.w.) was administered by intragastric gavage tube. Then, blood samples were taken at 30, 60, 90, and 120 min after glucose administration. The blood samples were collected with potassium oxalate and sodium fluoride, and their glucose content was estimated by glucose oxidase method as described previously.
Estimation of lipid peroxidation
Lipid peroxidation in tissues were estimated colorimetrically by TBARS, according to the methods of Nichans and Samuelson. 32 In brief, 0.1 mL of plasma and tissue homogenate (supernatant; Tris-hydrochloric acid (HCl) buffer, pH 7.5) was treated with 2 mL of (1:1:1 ratio) thiobarbituric acid (TBA)–trichloroacetic acid (TCA)–HCl reagent (0.37% TBA, 0.25 N HCl, and 15% TCA), placed in water bath for 15 min, cooled, and centrifuged at room temperature for 10 min. The absorbance of the clear supernatant was measured against reference blank at 535 nm and expressed in millimoles per 100 g of tissue.
Determination of CAT and SOD
CAT was assayed colorimetrically at 620 nm and expressed in moles of hydrogen peroxide (H2O2) consumed per minute per milligram of protein, as described by Sinha. 33 The reaction mixture (1.5 mL) contained 1.0 mL of 0.01 M (pH 7.0) phosphate buffer, 0.1 mL of tissue homogenate (supernatant), and 0.4 mL of 2 M H2O2. The reaction was stopped by the addition of 2.0 mL of dichromate acetic acid reagent (5% potassium dichromate and glacial acetic acid were mixed in a 1:3 ratio). SOD was assayed according to the technique described by Kakkar et al. 34 based on inhibition of the formation of nicotinamide adenine dinucleotide, phenazine methosulfate, and amino blue tetrazolium formazan. A single unit of enzyme was expressed as 50% inhibition of nitroblue tetrazolium reduction per minute per milligram of protein.
Determination of GPx and reduced GSH
GPx activity was measured by the method described by Rotruck et al. 35 Briefly, the reaction mixture contained 0.2 mL of 0.4 M phosphate buffer (pH 7.0), 0.1 mL of 10 mM sodium azide, 0.2 mL of plasma and tissue homogenate (supernatant; homogenized in 0.4 M phosphate buffer, pH 7.0), 0.2 mL of 10mM GSH, and 0.1 mL of 0.2 mM H2O2. The contents were incubated at 37°C for 10 min. The reaction was arrested by adding 0.4 mL of 10% TCA and centrifuged. The supernatant was assayed for GSH content by Ellman’s reagent (19.8 mg of 5,5-dithiobisnitro benzoic acid in 100 mL of 0.1% sodium nitrate). Reduced GSH was determined by the method followed by Ellman and Gan. 36 Briefly, 1.0 mL of supernatant was treated with 0.5 mL of Ellman’s reagent and 3.0 mL of phosphate buffer (0.2 M, pH 8.0). The absorbance was read at 412 nm. GPx activity was expressed in grams of GSH consumed per minute per milligram of protein and reduced GSH in milligrams per 100 g of tissue.
Determination of erythrocytes osmotic fragility and in vitro lipid peroxidation
The blood samples were freshly collected from each rat in heparinized tubes, centrifuged at 3000 rpm for 5 min, and the plasma was removed by aspiration. Erythrocytes were washed three times with equal volume of normal saline (0.9% NaCl); cells were collected and diluted with isotonic saline solution to get hematocrit of 10%. For each sample, 200 μL of erythrocytes suspension prepared previously was added to 2.8 mL of phosphate buffer solution in varying concentrations equivalent to NaCl solution (9–0 g/L). After homogenization, the tubes were incubated at ambient temperature for 15 min and then centrifuged at 3000 rpm for 5 min. The supernatant was decanted and the hemoglobin was measured spectrophotometrically at 540 nm. 37
Erythrocyte lipid peroxidation was determined according to the method described by Cynamon et al.
38
Malondialdehyde (MDA), a product of polyunsaturated fatty acid peroxidation, was measured as a TBA derivative in the supernatants after incubation of erythrocytes with two different concentrations of H2O2. When normal RBCs were exposed to peroxides in vitro, oxidation of antioxidants results in the release of TBARS that can be detected. Sodium azide, an inhibitor of CAT, prevents the destruction of hydrogen peroxide. The release of lipid peroxides without CAT inhibition can be considered as a reflection of erythrocyte membrane antioxidant protection and that with CAT inhibition as the maximum possible release. The percentage of maximal MDA release was calculated according to the following equation
Statistical analysis
All data were expressed as mean ± SD. The statistical significance was evaluated by one-way analysis of variance (ANOVA) using Statistical Package for the Social Sciences (SPSS) version 7.5 (SPSS, Cary, North Carolina, USA) and the Duncan’s multiple range test. A value of p < 0.05 was considered to indicate a significant difference between groups.
Results
Blood glucose and plasma insulin
Figure 1 shows the effect of GA on blood glucose level and plasma insulin of alloxan-induced diabetic rats. Diabetic control rats showed a significant elevation in FBG on successive days of the experiment as compared to their basal values, which was maintained over a period of 45 days, whereas the level of plasma insulin was significantly decreased. Daily oral treatment with GA at doses of 10 and 20 mg/kg b.w. daily showed a significant reduction in blood glucose with increase in insulin levels on successive days of the experiment as compared to their basal values, and the results were comparable with that of glibenclamide, a reference drug for diabetes. The administration of 20 mg/kg b.w. dose of GA elicited a more potent effect compared with the 5 and 10 mg/kg b.w. dose.

Effect of GA on the level of FBG and plasma insulin in normal and experimental animals. Data are mean ± SD values for six rats in each group. Values not sharing a common superscript letter differ significantly at p < 0.05 (DMRT).
Results of OGTT are presented in Figure 2. Although, in all treatment groups, 60 min after administration of glucose, the FBG levels reached a maximum and then started to decline, but in diabetic rats, it remained higher even after 2 h. However, in GA-treated rats, a dose-dependent effect on OGTT was observed and blood glucose level remained low at all sampling time points.

Effect of GA on OGTT in normal and experimental rats. Fasting blood glucose levels at 0, 30, 60, 90, 120 min for all the groups are given. Data are mean ± SD values for six rats in each group. Values not sharing a common superscript letter differ significantly at p < 0.05 (DMRT).
The preliminary dose-dependent studies showed pronounced result in 20 mg/kg b.w. when compared with low doses such as 5 and 10 mg/kg b.w. Hence, we restricted further studies with 20 mg/kg b.w.
Lipid peroxidation and antioxidant enzyme
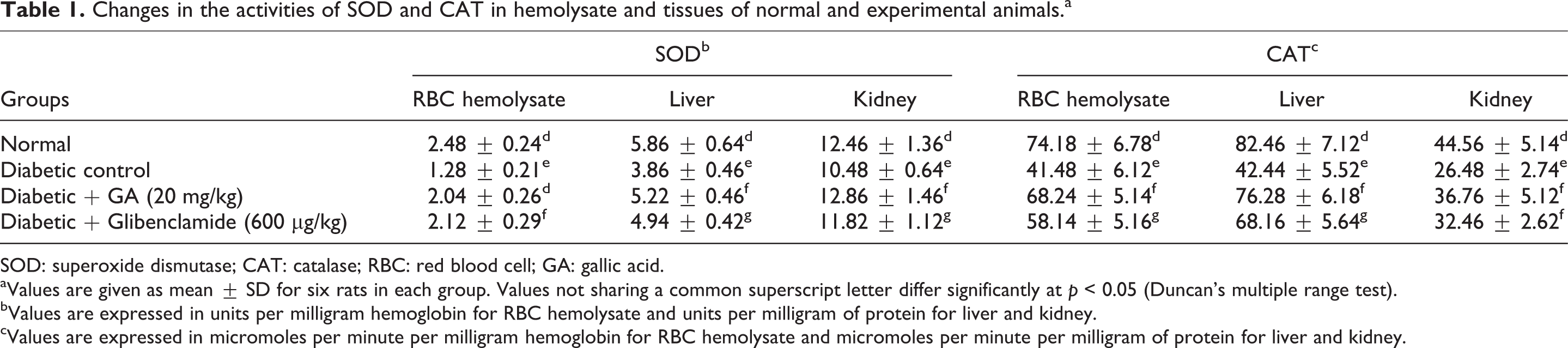
Lipid peroxidation was assayed by measuring the amount of TBARS formation. The increased concentration of TBARS was observed in the plasma, liver, and kidney of untreated diabetic rats (Figure 3). GA and glibenclamide treatment decreased the lipid peroxidation levels in alloxan-induced diabetic rats but remained higher than that of the nondiabetic control values. For studying the effect of GA on scavenging free radicals, the activities of SOD and CAT were measured in hemolysate and in liver and kidney of the control and experimental animals (Table 1). In diabetic rats, the levels of the enzymatic antioxidants were significantly (p < 0.05) decreased. Oral administration of GA significantly reversed these antioxidant levels in liver and kidney of diabetic rats. The modulatory effect of GA on antioxidant levels was observed to be even better than that of the reference drug, glibenclamide. Analysis of GPx and plasma GSH in hemolysate showed a significant reduction in diabetic control group (Table 2). Treatment with GA and glibenclamide tend to increase their levels to near normal values.

Effect of GA on the level of TBARS in plasma, liver and kidney of normal and experimental animals. Data are mean ± SD values for six rats in each group. Values not sharing a common superscript letter differ significantly at p < 0.05 (DMRT).
Changes in the activities of SOD and CAT in hemolysate and tissues of normal and experimental animals.a
SOD: superoxide dismutase; CAT: catalase; RBC: red blood cell; GA: gallic acid.
aValues are given as mean ± SD for six rats in each group. Values not sharing a common superscript letter differ significantly at p < 0.05 (Duncan’s multiple range test).
bValues are expressed in units per milligram hemoglobin for RBC hemolysate and units per milligram of protein for liver and kidney.
cValues are expressed in micromoles per minute per milligram hemoglobin for RBC hemolysate and micromoles per minute per milligram of protein for liver and kidney.
Changes in the activities of GPx and reduced GSH in circulation and tissues of normal and experimental animals.a

GPx: glutathione peroxidase; GSH: glutathione; GA: gallic acid; b.w.: body weight; RBC: red blood cell.
aValues are given as mean ± SD for six rats in each group. Values not sharing a common superscript letter differ significantly at p < 0.05 (Duncan’s modified range test).
bValues are expressed in units per milligram hemoglobin for RBC hemolysate and units per milligram of protein for liver and kidney.
cValues are expressed in milligram per deciliter for plasma and milligram per 100 mg tissue for liver and kidney.
Effect of GA on the osmotic fragility of erythrocytes in diabetic rats
The determination of osmotic fragility of the RBCs in a range of hypotonic concentrations of NaCl revealed that erythrocytes of diabetic control group were most resistant than that of GA-treated and normal groups (Figure 4). The concentration of NaCl inducing 50% of hemolysis in the control was found to be 4.3 g/L, whereas for alloxan-induced diabetic rats, it was 3 g/L. Different concentrations of GA such as 5, 10, and 20 mg/kg b.w. showed 50% hemolysis at 3.7, 4, and 4.2 g/L of NaCl, respectively.

Effect of GA on erythrocyte osmotic fragility of alloxan-induced diabetic rats. The degree of haemolysis of erythrocytes at different osmotic concentrations of sodium chloride in all groups are given. Data are mean ± SD values for six rats in each group. Values not sharing a common superscript letter differ significantly at p < 0.05 (DMRT).
Figure 5 shows hemolysis of erythrocytes in control and experimental groups. At 1 and 2 g/L of NaCl treatment, there was no significant difference observed in hemolysis of RBCs between treatment groups. The osmotic fragility of RBCs from rats treated with alloxan was significantly (p < 0.05) greater than that of diabetic rats treated with GA. The level of hemolysis was significantly improved in GA treatment compared to glibenclamide.
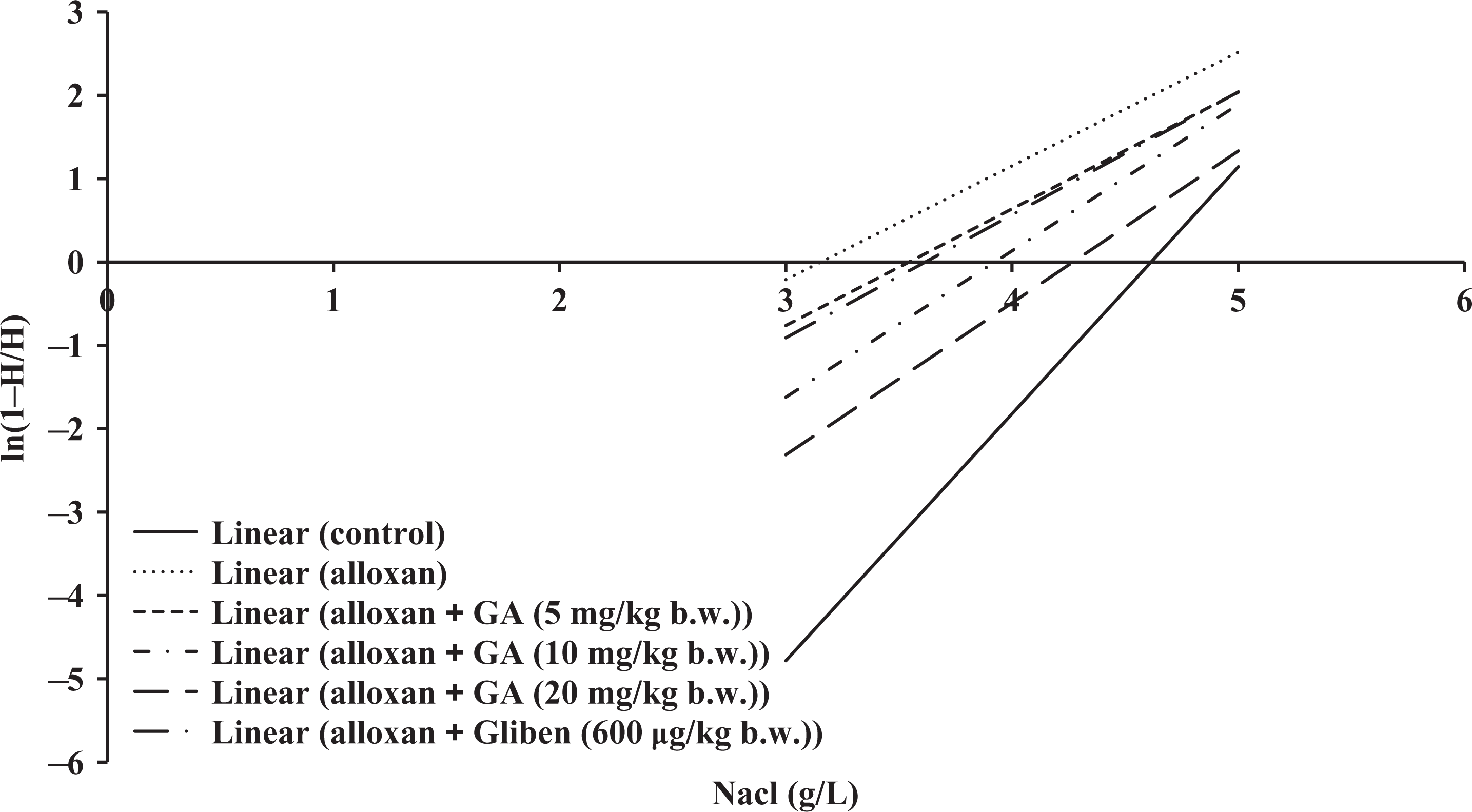
Linear representation of haemolysis curve for the effect of GA against alloxan-induced diabetes.
Effect of GA on in vitro RBC lipid peroxidation
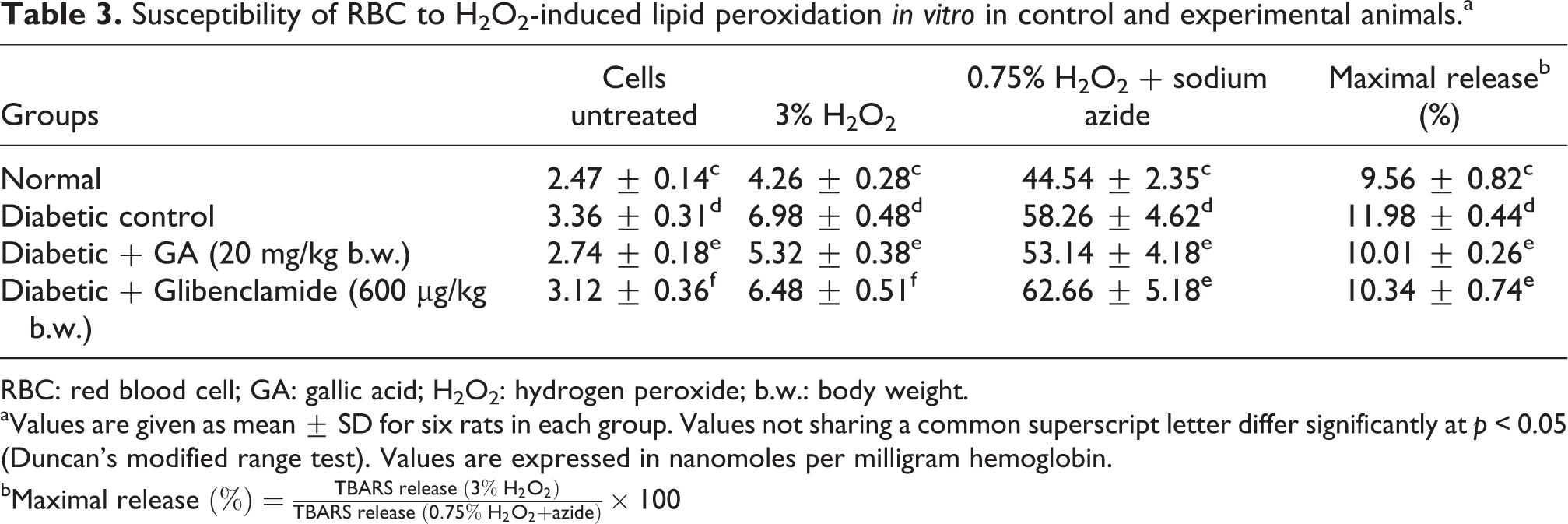
In vitro lipid peroxidation studies, in the presence of 3% H2O2, erythrocytes of the diabetic control, GA-treated animals, and glibenclamide-treated animals showed significantly greater release of lipid peroxides than the untreated nondiabetic control animals (Table 3). The level of lipid peroxidation was higher in the presence of sodium azide in the untreated diabetic rats. In GA-treated diabetic rats, lipid peroxidation in RBCs incubated with and without H2O2 was significantly lower compared with other groups. Erythrocytes of the diabetic rats showed significantly higher release of lipid peroxides than the other rats. The maximal release percentage of lipid peroxides was also significantly less in GA-treated rats than diabetic rats.
Susceptibility of RBC to H2O2-induced lipid peroxidation in vitro in control and experimental animals.a
RBC: red blood cell; GA: gallic acid; H2O2: hydrogen peroxide; b.w.: body weight.
aValues are given as mean ± SD for six rats in each group. Values not sharing a common superscript letter differ significantly at p < 0.05 (Duncan’s modified range test). Values are expressed in nanomoles per milligram hemoglobin.
b
Discussion
In the treatment of diabetes, many therapeutic strategies are directed for correcting both fasting and postprandial hyperglycemia and other metabolic parameters involved in the diabetes-associated complications. The antidiabetic drugs are targeted towards (i) stimulating pancreatic β-cell for releasing insulin, (ii) resisting activities of the hormones that involve in rising blood glucose levels, (iii) increasing the sensitivity of insulin receptors, (iv) decreasing the mobilization of glycogen and improving the utilization of glucose by the body, and (v) clearing free radicals and preventing lipid peroxidation. 16 Commercially available antidiabetic drugs have been reported to cause adverse effects including liver, kidney, and neurotoxicity. Hence, increasing research interest has been turned toward traditionally used natural medicine with limited or no side effects. 39
The natural products and compounds derived from natural products have a long history of clinical use as well as better patient tolerance and acceptance. There are several plant-derived compounds that have undergone development and have been marketed as drugs, including taxol from the bark of Pacific yew tree, Taxus brevifolia for lung, ovarian, and breast cancer; artemisinin to combat multidrug-resistant malaria; forskolin against allergies, respiratory problems, cardiovascular diseases, hypothyroidism, and weight loss; and silymarin and picroliv for liver diseases. 40 Over a 100 natural product derived compounds are currently undergoing clinical trials and at least 100 similar projects are in preclinical development. 41
GA, an endogenous plant phenol widely distributed in tea, berries, and fruits exhibited various pharmacological properties such as anti-inflammatory, anticancer, antimicrobial, and antioxidant effect. 42 Numerous evidences supported that GA supplements are safe to human use and has been used traditionally to treat a variety of ailments. 42,43 It has been documented that the bioavailability of GA was almost 40%. 24 Even though the effects of GA on serum glucose and insulin levels have been studied in experimental diabetes, 44 its effect on the integrity of erythrocyte membrane and oxidative stress has not been explored yet. Hence, our study focused on the modulatory effect of GA on the erythrocytes osmotic fragility and lipid peroxidation in association with diabetes along with the status of enzymatic and nonenzymatic antioxidants in diabetic rats.
Hyperglycemia-induced oxidative stress in diabetic conditions promotes alterations of the erythrocyte membrane function and other properties, which lead to adverse diabetic complications. 45 Several hematologic abnormalities have been defined in patients with diabetes such as increased erythrocyte aggregation, decreased deformability of erythrocytes, increased platelet aggregation, and endothelial damage and focal capillary occlusion. 46 Lipid peroxidation causes polymerization of membrane components and decreases cell deformability. 47 Products of lipid oxidation, such as oxidized unsaturated fatty acyl groups of phospholipids, reported to have an effect on membrane bilayer structure and function. Alterations in the phospholipid molecular species composition as a consequence of oxidant damage can be deleterious to the RBC membrane. 48 It has also been shown that peroxidation of erythrocyte membrane causes formation of high-molecular-mass protein aggregates within the membrane, which leads to membrane damage. 49
The increased production of free radicals and oxidative damage, the well-known causes of cellular dysfunction and apoptotic cell death were observed in tissue damages of alloxan-induced diabetic rats. 50,51 The increased levels of RBC lipid peroxidation observed in alloxan-induced diabetic rats may be associated with increased fragility of erythrocytes due to oxidative stress. 52 The effects of alloxan-induced diabetes have been demonstrated to be due to oxidative damage following the generation of superoxide radicals as a result of increased xanthine oxidase production, an enzyme involved in the generation of superoxide radicals. 27 Glucose auto-oxidation, protein glycation, and overproduction of superoxide radicals in mitochondria and via nicotinamide adenine dinucleotide phosphate oxidase have also been incriminated in the pathogenesis of oxidative damage in diabetes mellitus. 53 The occurrence of free radical-induced lipid peroxidation results in considerable changes in structural and functional organization of cell membrane and makes the membrane leaky. 49 The findings of this study demonstrated a significant decrease in erythrocyte lipid peroxidation in the diabetic rats supplemented with GA, which may be due to its free radical scavenging activity in the erythrocyte membranes.
Protection from oxidative stress is a potential form of treatment in regulating blood glucose levels and thus preventing diabetic complications. Hence, substances with antioxidant properties and free radical-scavenging abilities have been widely used for treating diabetes and its complications. 54 –56 Under hyperglycemic condition, excessive production of free radicals is associated with reduction in antioxidant enzymes and increase in lipid peroxidation. 57 –59 In our study, a significant improvement was found in the activities of SOD, CAT, and GPx in RBC of GA-treated alloxan-induced diabetic rats. Since the decreased activities of SOD and CAT during diabetes might be due to increased production of superoxide anion radical and H2O2, the present results suggest that GA may involve in free radical scavenging mechanisms thereby neutralizing excessive free radicals generated in diabetes.
SOD reacts with superoxide anion radical, which is the cellular oxidative metabolic product, and converts it to H2O2 and oxygen. This prevents the formation of reactive peroxynitrite, a ROS product of superoxide radical and nitric oxide in vivo reaction. 60 This H2O2 is neutralized by CAT defense system, 61 which protects the cells from ROS. GSH, an endogenous redox regulating free-radical scavenger, is a substrate for GPx, 62 which also detoxifies H2O2. In the present study, the supplementation of GA significantly increases the levels of antioxidants in the erythrocytes of diabetic rats thereby significantly reducing the levels of lipid peroxidation and oxidative agents.
The glucose-induced membrane lipid peroxidation and the increased osmotic fragility of RBC can change the properties of the RBC membrane. 63 Enhanced glycosylation by elevated glucose concentration may induce the formation of oxygen-derived free radicals through protein glycosylation, which releases early and late glycosylation end products, contributing to enhancement of oxidative stress seen in diabetes. 64 The plasma and RBC-MDA, cellular fragility (hemolysis), and lipoprotein metabolism were reported to be abnormal in type 1 diabetic patients. 65 Increased lipid peroxidation has been reported to cause an increase in osmotic fragility and a decrease in cell fluidity. 66 In the present study, while in alloxan-induced diabetic rats the erythrocyte membrane showed increased osmotic fragility, it was significantly reduced in GA-treated diabetic rats, which may also be due to the antioxidant effect of GA in the erythrocyte membranes.
In conclusion, our study demonstrated that GA exerts antioxidant and antidiabetic properties. The supplementation of GA reduced the levels of oxidative stress by inhibiting lipid peroxidation and enhancing the antioxidant defense system. The decrease in the level of hemolysis and osmotic fragility after GA treatment is hence an evidence for its activity in inhibiting diabetic complications. These findings of GA suggest its possible therapeutic application in the treatment of diabetes.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.