Abstract
This was a study of the microscopic, ultrastructural, immunohistochemical, and enzyme cytochemical features of giant eosinophilic granules encountered in pancreatic acinar cells of alloxan-induced diabetic rats. Seven male F344 rats with diabetes induced by a single i.v. dose of alloxan were sacrificed after twenty-five weeks of treatment. Histologically, the pancreatic acini were diffusely atrophied, and the islets showed marked atrophy or had disappeared, and giant eosinophilic granules and small vacuoles were observed in almost all acinar cells. The eosinophilic granules showed negative reactions for periodic acid-Schiff (PAS) and acid phosphatase, as well as fat stains such as Nile blue, Oil red O, and Sudan III. Ultrastructurally, the giant eosinophilic granules were huge structures surrounded by a double membrane containing many irregular cristae. A large amount of small lipid droplets was also apparent in the basal area of the acinar cells. Immunohistochemical analysis of prohibitin, a kind of protein located in the mitochondrial inner membrane, was partially positive in the marginal area of some giant eosinophilic granules, but negative for the central area. The enzyme activity for succinic dehydrogenase (SDH), one of the mitochondrial enzymes, showed a localizing pattern similar to that of prohibitin. These findings confirmed that the giant eosinophilic granules in the exocrine pancreas of alloxan-induced diabetic rats were giant mitochondria.
Introduction
Giant cytoplasmic eosinophilic granules have often been observed in various cells under some pathological conditions and age-related changes. They are formed when cytoplasmic organelles such as endoplasmic reticulum, mitochondria, lysosome, secretory granules, and cytoskeleton are altered or increased (Herdson et al. 1964; Horinouchi et al. 1997; Ito et al. 1998; Kambara et al. 2009; Ozaki et al. 1994; Remmer and Merker 1963). Eosinophilic inclusions or granules have also been detected as viral inclusion bodies and nuclear invagination (Kim et al. 2002; Weller and Stoddard 1952).
Pancreatic acinar cells have rarely detected giant cytoplasmic eosinophilic granules derived from enlarged mitochondria under a light microscope (Darle, Ekholm, and Edlund 1970; Kugino and Kishino 1989; Tasso, Clop, and Sarles 1971; Watari and Baba 1968). Mitochondria are ubiquitous cytoplasmic organelles that effectively generate large amounts of energy. They are morphological indicators of the state of a cell’s activity and health, and they occasionally increase in size in response to chemical or metabolic injury (Darle, Ekholm, and Edlund 1970; Coleman et al. 1995; Hexeberg et al. 1995; Lorente et al. 2002; Ludatscher and Silbermann 1976; Rohr and Riede 1973; Tandler and Hoppel 1986; Tasso, Clop, and Sarles 1971).
Alloxan, a cyclic urea analog, is selectively toxic for B-cells in pancreatic islets and induces insulin-dependent diabetes (Malaisse et al. 1982; Scarpelli 1989; Watanabe et al. 2004). It is well known that the endocrine pancreas is remarkably damaged by alloxan treatment (Scarpelli 1989). In addition, some researchers have reported the disturbed function of the exocrine pancreas, particularly pancreatic juice secretion, in alloxan-induced diabetic dogs and rabbits (Alvarez and Lopez 1989; Tiscornia, Janowitz, and Dreiling 1968). Recently, we unexpectedly found giant eosinophilic granules in the cytoplasm of pancreatic acinar cells in rats with alloxan-induced diabetes. In this study, we investigated the morphological features of the exocrine pancreas and focused on these giant eosinophilic granules.
Materials and Methods
Animals and Diets
Five-week-old male F344 rats obtained from Japan SLC, Inc. (Shizuoka, Japan) were reared in a barrier-sustained animal room maintained at a temperature of 24°C ± 2°C and a relative humidity of 60% ± 20%, with 12-hour light/dark cycles and ventilated at least 12 times/hour by sterilized fresh air. All rats were housed three per aluminum mesh cage. To protect against infection, the cages were changed at least once each week. Rats were given a powdered standard diet (CRF-1; Oriental Yeast, Tokyo, Japan) and chlorinated water ad libitum.
All procedures for animal handling and experimental treatments were in accordance with the Guidelines for the Care and Use of Laboratory Animals of the Committee for Animal Experiments of Setsunan University and the Japanese Association for Laboratory Animal Science. The level of care provided to the animals met the basic requirements outlined in the National Institutes of Health guidelines (NIH; 1996).
Glucosuria and Glycemia Monitoring
Urinary glucose levels were measured semiquantitatively using urine test paper (Wako Pure Chemical Industries, Osaka, Japan) every day from day 1 to day 3 after dosing, once every week for one month after the first week, and once every month thereafter from fresh urine obtained from the alloxan-treated rats. Blood glucose levels were measured semiquantitatively by the glucose oxidase method (Glutest E; Sanwa Kagaku, Aichi, Japan) once every month from the fourth week after dosing and using blood samples from the tail vein. Samples of blood from the tail vein and fresh urine were collected from 1:00 to 4:00 PM. We decided that the definitions of hyperglycemia and glucosuria are more than 300 mg/dL and 500 mg/dL, respectively.
Experimental Design
A total of seven 6-week-old rats were given a single dose of alloxan (Sigma-Aldrich Japan, Tokyo, Japan) through the tail vein at a dosage level of 35 mg/kg body weight. Alloxan-treated rats and three non-treated rats were sacrificed at 31 weeks of age. The dose of alloxan was determined as the given dose at which a rat survives for a long period after diabetes symptoms and induces glucosuria continuously.
The diabetic rats and nontreated rats were sacrificed by exsanguination from the abdominal aorta under deep anesthesia at the end of each scheduled period. The organs were fixed by immersion in 10% phosphate-buffered neutral formalin.
Light Microscopy, Histochemistry, and Electron Microscopy
Two diabetic rats died during the examination period, and their organs were unavailable for histopathological examination owing to cannibalism or autolysis. The cause of death was attributed to urinary tract infection. The remaining five diabetic rats were examined by light microscopy. In addition, three of them were examined by electron microscopy.
Fixed organs were trimmed, dehydrated by automated processor, and embedded in paraffin. Sections (4-µm thick) of tissue specimens were stained with hematoxylin-eosin for histopathological examination. Frozen sections (7 µm) were cut in a CM3000 cryostat (Leica Instruments GmbH, Germany) at a cabinet temperature of −23°C. The sections were stained with Nile blue, Oil Red O, and Sudan III. Enzyme histochemical staining was performed for succinic dehydrogenase (SDH) according to Nachlas's method (Nachlas et al. 1957) and acid phosphatase (ACP) according to Gomori’s method (Gomori 1952).
For electron microscopy, small fragments of pancreatic tissue were fixed with 2.5% glutaraldehyde in 0.1 M phosphate buffer, pH 7.4, post-fixed in 1% osmium tetroxide solution (pH 7.4) for 2 hours, and processed into resin. Semithin sections were cut and stained with toluidine blue. Ultrathin sections were cut and stained with uranyl acetate and lead citrate, and then examined under an electron microscope (JEM1200EX, JEOL, Tokyo, Japan).
Immunohistochemistry
The expression of prohibitin, a kind of proteins located in the mitochondrial inner membrane (Ikonen et al. 1995; McClung et al. 1995), amylase and insulin were examined immunohistochemically in the pancreas of all rats. Routinely processed formalin-fixed, paraffin-embedded tissue specimens were cut into 4 µm–thick sections. The sections were deparaffinized in xylene and rehydrated through graded ethanol at room temperature. Antigen retrieval was performed in a pressure cooker for fifteen minutes in citrate-buffered solution (pH 6.0) for prohibitin, by a microwave for ten minutes in citratebuffered solution (pH 6.0) for amylase. Antigen retrieval was not performed for insulin immunohistochemistry.
All slides were rinsed with 0.05 M Tris–buffered saline (TBS, pH 7.6) plus 0.01% Tween 20, treated with 1% hydrogen peroxide in methanol, and again rinsed with TBS plus Tween 20. Slides were incubated in bovine serum albumin for 5 minutes and with anti-prohibitin rabbit polyclonal antibodies (diluted 1:50, Fitzgerald Industries International, Inc., Acton, MA, USA), anti-amylase goat polyclonal antibodies (diluted 1:100, Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) and anti-insulin guinea pig polyclonal antibody (diluted 1:200, Dako, Carpinteria, CA, USA) overnight at 4°C. The slides were subsequently rinsed with TBS plus Tween 20, treated for thirty minutes at room temperature with N-Histofine MAX PO (R) and (G) (Nichirei, Tokyo, Japan), rinsed with TBS plus Tween 20, incubated in diaminobenzidine solution containing 0.01% hydrogen peroxide for the peroxidase coloring reaction, and counterstained with Mayer’s hematoxylin.
Results
General Conditions and Monitoring of Glucosuria and Glycemia
For all alloxan-treated F344 rats, severe hyperglycemia (>300 mg/dL) and glucosuria (>500 mg/dL) continued for approximately 25 weeks from the day after alloxan injection to the time of scheduled necropsy or when a rat was found moribund. The average body weight 25 weeks after alloxan treatment was 155.7 ± 6.9 g.
Light Microscopy and Histochemistry
Pancreatic tissues were diffusely atrophied and characterized by reduced size and decreased amounts of zymogen granules in each cell along with severe atrophy or loss of islets as a direct effect of alloxan treatment. The atrophy of pancreatic acini seemed less severe around the residual islets (Figure 1a). The most characteristic changes in the exocrine pancreatic tissue were the appearance of giant eosinophilic homogeneous granules and small vacuoles in the basal area of acinar cells in all alloxan-induced diabetic rats (Figure 1b). Eosinophilic granules were detected as single or multiple discrete cytoplasmic inclusions in the acinar cells and were mostly round in shape. The size of these granules varied and was often larger than the nucleus of the host acinar cell. The numbers of eosinophilic granules and small vacuoles were also much smaller around the residual islets where the atrophic change of the acinar cells was less severe. The eosinophilic granules remained unstained for lipotropic dye, such as Oil Red O, Sudan III, and Nile blue (Figures 2a–2c), whereas small vacuoles were positive for Oil Red O, Sudan III, and Nile (Figures 2a–2c). Eosinophilic granules were negative for PAS reaction and ACP by enzyme histochemistry (Figures 2d and 2e).
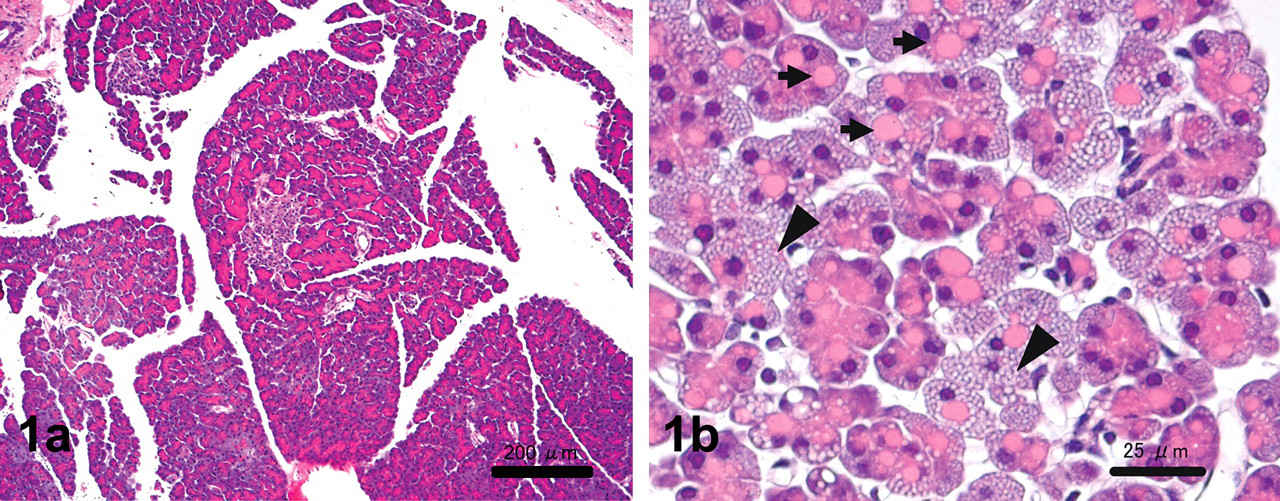
(a) Pancreatic acini are atrophied except around the residual islets. Hematoxylin and eosin stain. Scale bars, 200 µm. (b) Pancreatic acinar cells have eosinophilic homogeneous granules (arrow) and small vacuoles (arrowheads) in the basal area. Hematoxylin and eosin stain. Scale bars, 25 µm.
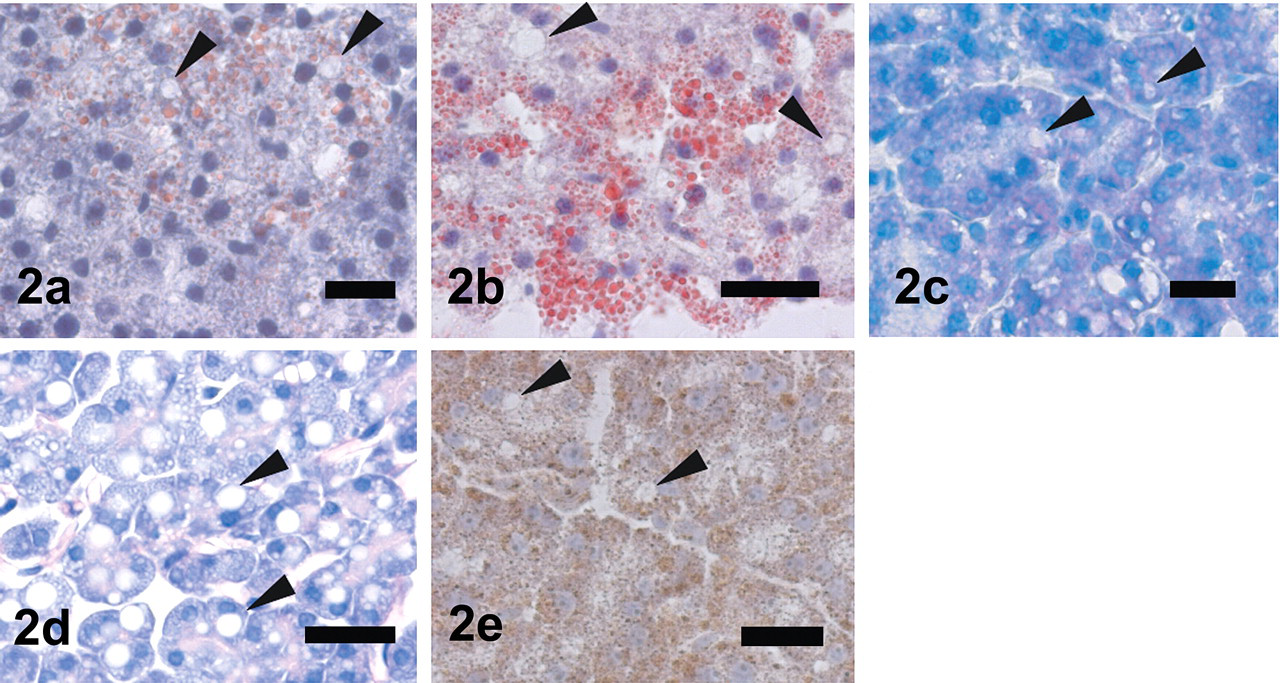
Representative staining of giant eosinophilic granule and small vacuoles in pancreatic acinar cells. Scale bars, 25 µm. Eosinophilic granules are negative for Sudan III (a), Oil Red O (b), Nile blue (c), PAS (d), and ACP (e). Small vacuoles are positive for Sudan III (a), Oil Red O (b), and Nile blue (c).
Electron Microscopy
Ultrastructurally, huge, round globules corresponding to the microscopical eosinophilic granules in the pancreatic acinar cells were surrounded by a double membrane and consisted of fragmentations of cristae into tubular structures (Figures 3a and 3b). The inner limiting membrane showed infolding to form a small number of cristae, suggestive of mitochondrial origin in the periphery (Figures 3a and 3b). The matrix density was similar to normal-sized mitochondria, up to 5 μm (much larger than the nucleus), and sometimes contained extremely long crista and several electron-dense granules (Figure 3a). Most of the globules were located throughout the whole cytoplasm, but they were most frequent between the nucleus and apical side where many electron-dense zymogen granules occupied in the normal acinar cells. Mitochondria with normal size and normal ultrastructure were also observed, however, the number of total mitochondria was not increased even if included in the huge globules. Corresponding to a large amount of small vacuoles in acinar cells at light microscopy, small lipid droplets of low density frequently contained myelin-like figures (Figure 3c). Smaller-sized zymogen granules with medium density accumulated near the lumen (Figure 3d).
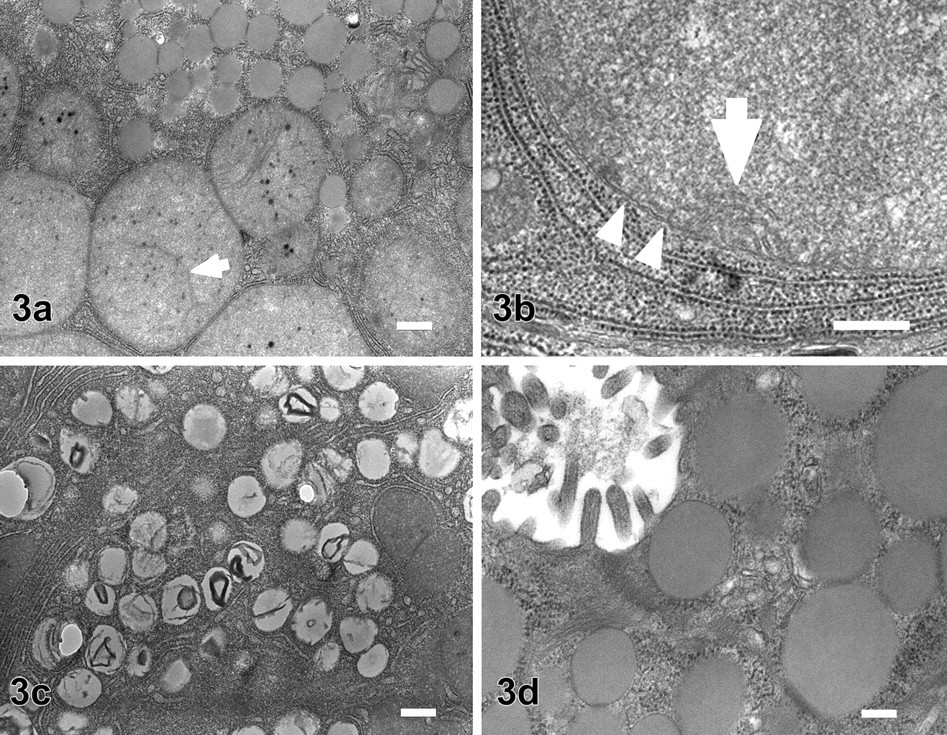
Electron micrograph of pancreatic acinar cells. (a) Huge, round globules corresponding to microscopical eosinophilic granules (lower part of figure) and smaller-sized zymogen granules with low density (upper part of figure). Scale bars, 500 nm. (b) Higher magnification of huge, round granule. Granules are surrounded by double membrane (arrowheads) and consist of fragmentations of cristae into tubular structures. The inner limiting membrane showed infolding to form a small number of cristae (arrow). Scale bars, 500 nm. (c) Lipid droplets corresponding to the large number of small vacuoles. Lipid droplets have low-density materials with myelin-like figures. Scale bars, 1 μm. (d) Smaller-sized zymogen granules with medium density accumulate near the lumen. Scale bars, 200 nm.
Immunohistochemistry and Enzyme Histochemistry
Immunohistochemical analysis of prohibitin, a 30-kDa protein located in the inner membrane of mitochondria, was partially positive in the marginal area of some eosinophilic granules, but negative for the central area (Figure 4a). The enzyme activity for SDH, one of the mitochondrial enzymes, showed a localizing pattern similar to the immunohistochemical prohibitin–positive area (Figure 4b).
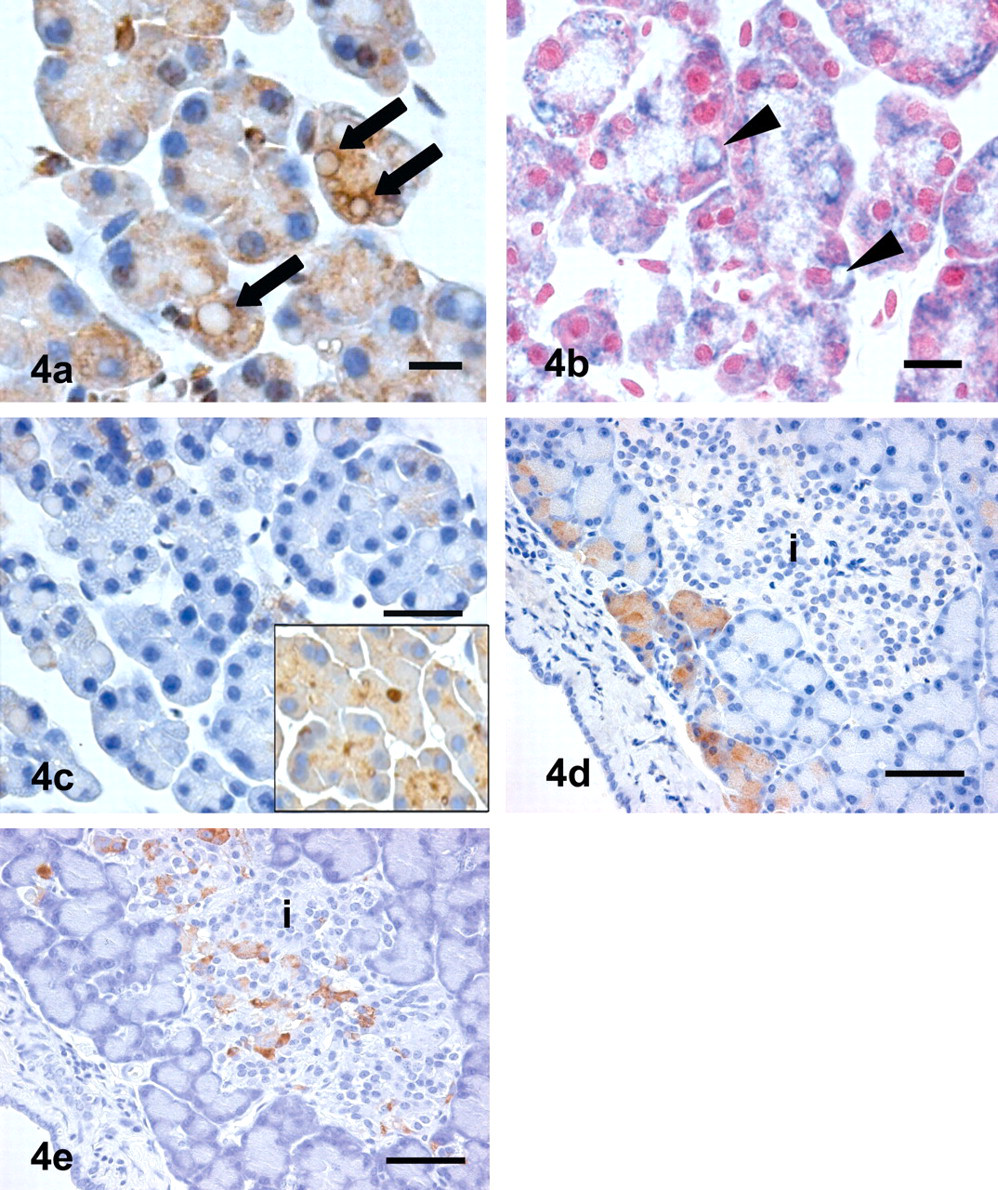
(a) Prohibitin is partially positive in the marginal area of some giant granules (arrow) in acinar cells, but negative in the central area of these granules. Scale bars, 10 µm. (b) Enzyme activity for succinic dehydrogenase of giant granules shows a localizing pattern similar to that of prohibitin (arrowheads) (nuclear red). Scale bars, 25 µm. (c) Almost all acinar cells of alloxan-induced diabetic rats are negative for amylase. All acinar cells of control rats show immunopositivity for amylase (insert). Scale bars, 50 µm. (d) Limited number of pancreatic acini are positive for amylase around the residual pancreatic islets (i). Giant eosinophilic granules and small vacuoles in acinar cells were not observed in this area. Scale bars, 50 µm. (e) Center of atrophied pancreatic islet shows immunopositivity for insulin. Scale bars, 50 µm.
Immunohistochemically, acinar cells were negative amylase except for a few cells, compared to the constant positive reaction on the apical side of the cytoplasm in a normal pancreas (Figure 4c). A few pancreatic acini were positive for amylase around the residual islets (Figure 4d). The center area of the residual islets was positive for insulin (Figure 4e).
Discussion
Electron microscopic, immunohistochemical, and enzyme histochemical findings in this study strongly suggest that the eosinophilic granules in the pancreatic acinar cells of alloxan-induced diabetic rats were giant mitochondria. To the best of our knowledge, there are no reports on giant mitochondria in any kind of cells of alloxan-induced diabetic dogs and rats, even after a long-term diabetic condition (Satoh 1966; Watanabe et al. 2004).
It is well known that the administration of some chemicals and a variety of nutritional manipulations induce the appearance of giant mitochondria in experimental animals (Ludatscher and Silbermann 1976; Rohr and Riede 1973; Satake et al. 2008; Tandler and Hoppel 1986; Suzuki 1969; Wakabayashi, Asano, and Kawamoto 1979). In humans, giant mitochondria have been reported in a variety of diseases such as steatohepatitis and mitochondrial neurogastrointestinal encephalopathy (Caldwell et al. 1999; Perez-Atayde et al. 1998). Also, giant mitochondria have been observed in normal cells such as secretory and ciliated cells of the gerbil trachea and the bronchioles and serous acinar cells in the submandibular gland of the fruit bat, and the retinal cone inner segments of the tree shrew in a physiological state (Knabe and Kuhn, 1996; Spicer et al. 1990; Tandler, Nagato, and Phillips 1997). In pancreatic acinar cells, lysine has the direct effect of depolarization of the mitochondrial membranes, resulting in mitochondrial swelling (Kugino and Kishino 1989). After prolonged exposure to ethanol in drinking water, rats have some altered pancreatic mitochondria that are somewhat enlarged (Darle, Ekholm, and Edlund 1970; Tasso, Clop, and Sarles 1971). The occurrence of huge, degenerated mitochondria was also reported in acinar cells six hours after alloxan treatment in a dog (Watari and Baba 1968). Our immunohistochemical and enzyme cytochemical observation of prohibitin and SDH revealed the decreased function of giant mitochondria because many were negative or only partially positive in localized parts for both proteins. In addition, electron microscopic examination of giant mitochondria detected an abnormal cristae structure, suggesting decreased function of mitochondria. The giant mitochondria formations in our study may indicate reduced function of mitochondria, but the mechanism and pathogenesis to form such giant mitochondria remain unclear.
Alloxan is a chemical that induces loss of insulin-producing islet β-cells and causes a hypoinsulinemic condition and resultant diabetes mellitus in animals. This effect is thought to be mediated by a sequence of redox reactions involving the production of superoxide anion radicals in or near the β-cells (Heikkila and Cabbat 1978; Jorns et al. 1999). The specific toxicity of alloxan to the β-cell accounted for the rapid and extensive cellular uptake through glucose transport owing to the structural similarity between alloxan and D-glucose (Malaisse et al. 1982). In addition, the activity of glutathion peroxidase was much less in islets than in other tissues (Malaisse et al. 1982).
In in vitro studies, giant mitochondria have been induced by various chemicals generating free radicals in hepatocyte-derived cell lines and kidney cell lines (Karbowski et al. 1997; Karbowski et al. 1999; Wakabayashi and Karbowski 2001). Thus, alloxan-producing superoxide anion radicals might be involved in induction of giant mitochondria. However, to our knowledge, the direct toxicity of alloxan in relation to pancreatic acinar cells is unknown. Starvation has induced giant mitochondria in hepatocytes (Rohr and Riede 1973) and gastric parietal cells (Ogawa et al. 2003), and the formation of giant mitochondria resulted from fusion of several mitochondria. In the present study, the number of giant eosinophilic granules was much smaller around the residual islets that secreted insulin. Thus, insulin may be involved in the formation of giant mitochondria.
The increase in fat droplets and an increase both in the number and size of mitochondria may suggest impaired energy metabolism. Although the number of mitochondria did not increase in the present rats, the enlargement of mitochondria and accumulation of cytoplasmic lipid droplets were similar to those in juvenile visceral steatosis mice with congenital carnitine deficiency (Kaido et al. 1997; Narama et al. 1997). In the present study, rats were kept in a severe, prolonged diabetic condition with hypoinsulinemia. Unsuccessful transport of blood sugar into the pancreatic acinar cells despite hyperglycemia is a possible cause of impaired energy metabolism and the formation of giant mitochondria similar to nutritional deficiency.
It has been reported that damage to B cells by alloxan profoundly decreases the synthesis and release of amylase from the exocrine pancreas of the rat owing to the close relationship of the vascular network between endocrine and exocrine cells (Henderson, Daniel, and Fraser, 1981). These changes can be restored to a normal level by insulin treatment (Henderson, Daniel, and Fraser 1981). The significant depression of amylase expression and atrophy of acinar cells in our diabetic rats reflected the loss of amylase-positive zymogen granules. The decreased electron density and size of zymogen granules observed by electron microscopy also suggested the impairment of zymogen maturation in pancreatic exocrine cells. In our study, the atrophic change of acinar cells was less severe around the residual islets, suggesting near-normal insulin secretion. These findings imply that the insulin secretion level is closely related to exocrine pancreatic function.
Further study is needed to clarify the true mechanism of formation of giant mitochondria in pancreatic acinar cells on a molecular level.
