Abstract
Gastrointestinal risk factors after organ transplantation are prevalent, due to the chronic use of immunosuppressant. The immunosuppressive drugs such as tacrolimus/mycophenolate mofetil (TAC/MMF) association are the most commonly used therapy. TAC and MMF have been implicated in gastrotoxicity, but their direct effects, alone and combined, on intestinal cells are not completely elucidated. This study investigated the effect of TAC and MMF alone and combined on human colon carcinoma cells. Our results demonstrated that TAC and MMF individually inhibit clearly cells proliferation, enhanced free radicals, lipid peroxidation production, induced DNA lesions and reduced mitochondrial membrane potential. In this study, we also showed that the two molecules TAC and MMF combined at high concentrations amplified the cell damage. Furthermore, the TAC (5 µM) prevented cell death induced by MMF (half maximal inhibitory concentration (IC50)). Also, MMF (50 µM) induced cytoprotection in HCT116 cells against TAC (IC50) toxicity. Our findings provide additional evidence that oxidative damage is the major contribution of TAC and MMF combined toxicities. In fact, MMF and TAC exert a gastroprotective effect by modulating reactive oxygen species production. These data underscore the pleiotropic effect of TAC and MMF on HCT116 cells that play a preventive and critical role on intestinal function.
Introduction
Immunosuppressive treatment allowed organ transplantation to become a therapeutic use in transplant patients. To improve the effect of these immunosuppressive drugs and reduce their side effects, clinicians have used the combination of immunosuppressive drugs. Despite the different combinations used so far, the risk of rejection and side effects is a real problem. Tacrolimus (TAC) (Figure 1(a)) has become the most commonly used immunosuppressive macrolide, originating from

Chemical structures of TAC (a) and MMF (b). TAC: tacrolimus; MMF: mycophenolate mofetil.
All immunosuppressant medications are associated with toxicity, and much of the art and science of the clinical management of transplant recipients has been aimed to combine different immunosuppressant medications at a lower dose to improve the overall efficiency and safety profiles. 4 Mycophenolate mofetil (MMF) (Figure 1(b)) is an important and commonly used drug in the maintenance of immunosuppressive therapy for recipients of all types of organ transplants. In fact, the majority of patients are treated with the TAC in combination with MMF and this association is the protocol for maintaining the most commonly used immunosuppression. 5 –7 MMF is an antimetabolite drug, which blocks the de novo pathway of purine synthesis. Although it selectively inhibits B- and T-lymphocyte proliferation, 8 MMF has been proved to be capable of significantly reducing the side effects of TAC in the renal system. 8 The mechanism of action of MMF being blocked in de novo pathway 9,10 causes immunodeficiency without affecting other organs unlike the recycling channel. 11 Immunosuppressive drugs cause significant toxicities, such as, nephrotoxicity, neurotoxicity, cardiac toxicity, hematotoxicity, post-transplant diabetes, endothelial dysfunction, and gastrotoxicity. 12 –14 In the present work, we propose to study the effect of this association (TAC/MMF) in the intestinal cells. Although the intestinal epithelium is the first physiological barrier to these molecules, the effects of TAC on the gastrointestinal (GI) system are prevalent, so this treatment after transplantation causes frequent GI symptoms that are pathologic. 15 –19 Furthermore, MMF is associated with frequent GI complications. It has the potential to cause toxic injury throughout the GI tract. In fact, MMF appears to cause topical irritation and damage including esophageal and duodenal ulcers and reactive gastropathy. 20 Different studies have described histologic abnormalities, inflammatory bowel disease or Crohn’s-like changes as well as erosive or ulcerative enterocolitis in patients treated with MMF. 21 –26
To our knowledge, the effect of TAC/MMF association on the intestinal dysfunction is unknown. We hypothesized that MMF might protect against stress-induced cell death in response to TAC. Thus, the aim of this study is to investigate some of the intracellular signalling pathways that are affected by the combination of TAC/MMF on human carcinoma intestinal cells in vitro subjected to an oxidative stress, with a view to elucidate which signalling pathways may be up-regulated/down-regulated leading to detrimental effects on cell survival, free radicals, lipid peroxidation, DNA fragmentation and mitochondrial membrane potential (Δ
Materials and methods
Chemicals
TAC and MMF were purchased from Tocris Bioscience (France), dissolved in dimethyl sulphoxide (DMSO) and then stored at −20°C. MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium-bromide) was obtained from Sigma-Aldrich (St Louis, Missouri, USA). Roswell Park Memorial Institute 1640 cell culture medium (RPMI 1640), foetal calf serum,
Cell culture and treatment
The human colon cancer cell line HCT116 (wild type) was gifted by Professor Olivier Micheau (Faculty of Medicine and Pharmacy, University of Burgundy, Dijon). HCT116 cells were incubated in a humidified air/carbon dioxide (95:5) mixture at 37°C. Cells were cultured in RPMI 1640 medium supplemented with 10% foetal calf serum, 1%
Measurement of cell survival rate
Cytotoxicity of TAC and MMF was determined by using the colorimetric method described by Mosmann. 27 This method assesses the ability of viable cells to convert MTT into formazan by the mitochondrial enzyme succinate dehydrogenase. Exponentially growing HCT116 cells were seeded at a density of 2 × 104 cells per well in 96-well plates in 200 μL of fresh culture medium containing RPMI. Cells were treated with increasing concentrations of TAC (0–40 µM) or MMF (0–400 µM) and combined TAC/MMF for 24 h at 37°C. For this combination, we fixed two concentrations of TAC (5 and 25 µM) associated with different concentrations (25–400 µM) of MMF. Then the culture medium was replaced by 200 µL fresh medium containing 1 mg/mL MTT and the plates were incubated for 4 h at 37°C. The medium was then removed and replaced by 100 µL of 0.04 M hydrochloric acid/isopropanol for 1 h. The absorbance was determined using a spectrophotometer microplates reader (Stat Fax 3200, Awareness Technology, Palm City, Florida, USA) at a wavelength of 560 nm. Cell viability was expressed as the relative formazan formation in treated sample as compared to control cells. Half maximal inhibitory concentration (IC50) values were defined as the concentration inducing 50% loss of cell viability.
Measurement of ROS production
ROS are essential intermediates in oxidative metabolism. Nevertheless, when oxidative stress occurs, ROS are generated in excess and consequently may damage cells by oxidizing lipids and disrupting DNA and proteins. The intracellular amounts of ROS were measured by a fluorometric assay with DCFH-DA. This method was used extensively to monitor oxidation in biological systems as a well-established compound to detect and quantify intracellular production such as superoxide radical, hydroxyl radical and hydrogen peroxide (H2O2). 28 –30 The conversion of the nonfluorescent DCFH-DA to the highly fluorescent 2,7-dichlorofluorescein (DCF) product (max = 522 nm) happens in many steps. The fluorescent probe, after diffusing in the cell membrane, is hydrolysed by intracellular esterases to nonfluorescent dichlorofluorescin (DCF), which is trapped inside the cells, then oxidized to fluorescent DCF through the action of peroxides in the presence of ROS. 31 HCT116 cells were seeded on 24-well culture plates (Polylabo, France) at 105 cells/well for 24 h of incubation. The cells were incubated with 20 µM DCFH-DA for 30 min at 37°C, then without removing the DCF, cells were treated with TAC alone (5 and 25 µM) and MMF alone (50 and 330 µM). Each TAC concentration (5 and 25 µM) was associated with two concentrations of MMF (50 and 330 µM) for 24 h. H2O2 (75 µM) was used as a positive control. Intracellular production of ROS was measured after 24 h incubation at 37°C by fluorometric detection of DCF oxidation on a fluorometer (BioTek FL×800, Winooski, Vermont, USA) with an excitation wavelength of 485 nm and emission wavelength of 522 nm. The DCF fluorescence intensity is proportional to the amount of intracellular ROS. Results are expressed as the ratio of DCF-induced TAC and MMF alone and their association fluorescence/DCF-induced control fluorescence.
Lipid peroxidation
Lipid peroxidation was assayed by the measurement of malondialdehyde (MDA) according to the method of Ohkawa et al.
32
The cells were seeded in 6-well plates at 6 × 105 cells/well. After 24 h of incubation, they were exposed to different concentrations of TAC (5 and 25 µM), MMF (50 and 330 µM) individual and their combination. Indeed, each TAC concentration was associated with two concentrations of MMF for 24 h. Then, cells were incubated with H2O2 (75 µM) for 30 min. The cells were then washed with cold PBS, scraped and lysed by homogenization in ice-cold 1.15% potassium chloride. Samples containing 100 μl of cell lysates were combined with 0.2 mL of 8.1% sodium dodecyl sulphate, 1.5 mL of 20% acetic acid adjusted to pH 3.5 and 1.5 mL of 0.8% thiobarbituric acid. The mixture was brought to a final volume of 4 mL with distilled water and heated to 95°C for 120 min. After cooling to room temperature, 5 mL of mixture of
Comet assay
Comet assay, also known as single cell gel electrophoresis, is a visual and sensitive technique for measuring DNA breakage in individual mammalian cells. At 50% confluence, HCT116 cells were exposed to TAC (5 and 25 μM), MMF (50 and 330 µM) and the TAC/MMF association (5 and 25 µM) with two concentrations of MMF (50 and 330 µM) at 24 h. A positive control was treated with 75 μM H2O2. Negative control corresponds to cells treated by vehicle. Approximately 75 × 104 cells were mixed with 1% LMA in PBS and spread on a microscope slide previously covered with a 1% NMA in PBS layer. After agarose solidification, cells were treated with an alkaline lysis buffer (2.5 M sodium chloride, 0.1 M EDTA, 10 mM tris(hydroxymethyl)aminomethane (Tris), pH 10, 1% (v/v) Triton X-100 and 10% (v/v) DMSO) for 1 h at 4°C, then the DNA was allowed to unwind for 40 min in the electrophoresis buffer (0.3 M sodium hydroxide and 1 mM EDTA, pH > 13). The slides were then subjected to electrophoresis in the same buffer for 30 min at 25 V and 300 mA. Slides were then neutralized using a Tris buffer solution (0.4 M Tris, pH 7.5) for 15 min. After staining the slides with ethidium bromide (20 μg/mL), the comets were detected and scored using a fluorescence microscope Nikon Eclipse TE 300 fluorescence microscope (Nikon, Tokyo, Japan). The experiment was done in triplicate. The damage is represented by an increase of DNA fragments that have migrated out of the cell nucleus during electrophoresis and formed an image of a ‘comet’ tail. A total of 100 comets on each slide were visually scored according to the intensity of fluorescence in the tail and classified by one of five classes as described by Collins et al.
33
The total score was evaluated according to the following equation:
Determination of mitochondrial membrane potential
HCT116 cells were treated with TAC (5 and 25 µM), MMF (50 and 330 µM) individually and their combination for 24 h. Cells from different groups were incubated with Rh 123 at a final concentration of 5 µM for 30 min in the dark at 37°C, washed with PBS and then centrifuged at 500
Statistical analysis
Statistical analyses were performed using GraphPad prism Version 5. Data are represented as mean ± standard deviation (SD). The Mann–Whitney
Results
Determination of the effect of immunosuppressive drugs alone and combined on HCT116 viability
The results on cells’ viability assessed using the MTT assay after exposure of 24 h is reported in Figure 2. Results showed a concentration-dependent inhibition of cell viability by TAC and MMF alone at concentration ranging from 0 to 40 µM of TAC and 0 to 400 µM of MMF compared with untreated cells. The IC50 values of TAC and MMF are respectively 25 µM and 330 µM (Figure 2(a) and (b)). In addition, in this study, we have analysed the effects of the combination of two molecules at different concentrations. Figure 2(c) shows that the treatment of HCT116 cells with combined TAC at 5 µM and MMF (25–330 µM) was significantly associated by an increase in survival cells especially at MMF concentrations over 200 µM. The co-treatment of HCT116 cells with TAC (5 µM) and MMF (330 µM) ameliorates the survival cells with 23% compared with cells treated only with MMF. These results suggest that TAC at 5 µM is able to protect HCT116 cells from MMF cytotoxicity (Figure 2(b) and (c)). However, HCT116 cells were treated with TAC at 25 µM (IC50) and various concentrations of MMF (25–330 µM) for 24 h. Figure 2(c) clearly shows that MMF significantly restores the cell viability from 30% to 43% for 25 µM to 165 µM, respectively. We also observed that MMF concentrations ranging from 50 µM to 165 µM present the optimal preventive effect. The MMF over 200 µM presents a cytotoxic effect on the HCT116, amplified by the presence of TAC (25 µM) (Figure 2(d)). To investigate the combined effects we selected two concentrations of TAC (5 µM) and MMF (50 µM), which do not display any cytotoxic induction. Also, we chose two toxic concentrations of 25 and 330 µM for further experimentations in our study.

Concentrations response curves of TAC and MMF alone (a and b) and their association (c and d) induced HCT116 cell death after 24 h. Data are expressed as mean ± SD of independent experiments (
Oxidative stress status
ROS generation
To check the potential of TAC or MMF and their combination to generate oxidative stress, we measured the production of fluorescent DCF after 24 h. As shown in Figure 3(a) and (b), the highest relative intensity of fluorescence was observed in HCT116 cells treated separately with TAC at 25 µM and MMF at 330 µM. HCT116 cells co-stimulated with TAC at 5 µM and MMF at 330 µM showed a change in the redox status in response to this treatment. The TAC (5 µM) and MMF (330 µM) association significantly decrease ROS generation with about 1.4-fold (Figure 3(a)). Furthermore, the combination of TAC (5 µM) and MMF (50 µM) did not cause any change in ROS generation compared with control cells. In contrast, the TAC (25 µM) and MMF (50 µM) association decreased intracellular ROS with twofold as compared to cells treated with 25 µM of TAC. In this study, H2O2 has been used only as positive control. As shown in Figure 3(c), TAC at 5 µM and MMF at 50 µM were able to reduce ROS generation caused by H2O2.
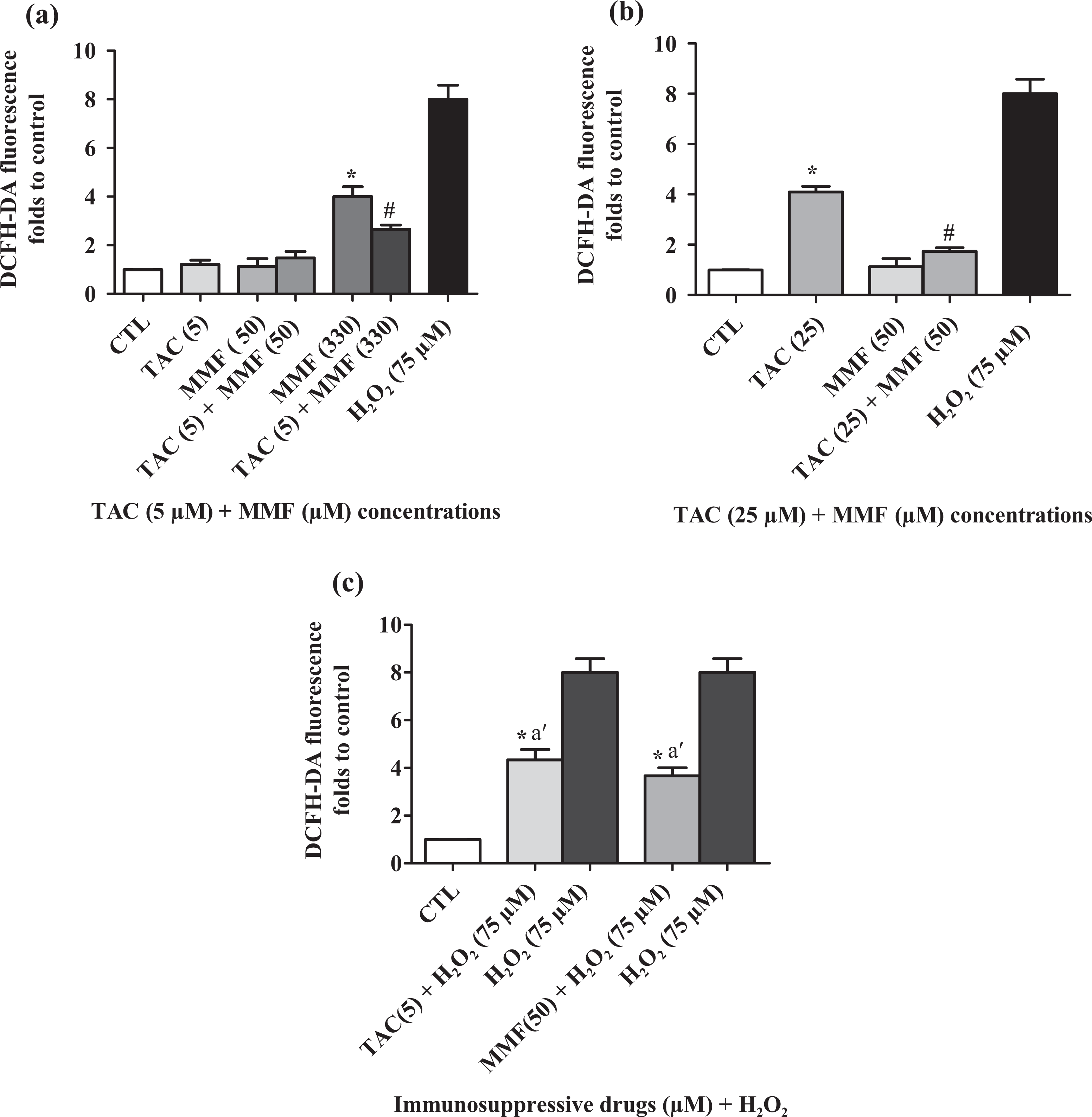
Modifications of the oxidative statute induced by TAC, MFF individually and their co-stimulation (a and b) after 24 h. Cells treated with H2O2 considered as a positive control. Cells treated with H2O2 + TAC or MMF (c). Data are expressed as mean ± SD of independent experiments (
Evaluation of lipid peroxidation
Results of lipid peroxidation after 24 h of incubation of HCT116 cells in the presence of TAC (5 and 25 µM) and MMF (50 and 330 µM), individually or combined, are illustrated in Figure 4(a) and (b). Indeed, TAC and MMF at low concentrations (5 and 50 µM) do not display any change of MDA formation compared with untreated control. When TAC was added at a fixed concentration of 5 µM in addition to MMF at 330 µM, MDA production decreased from 5.5- to 2.7-folds as compared to untreated cells. Similar results were found in the combined TAC at 25 µM and MMF at 50 µM from 4- to 1.8-folds (Figure 4(a) and (b)).

Induction of lipid peroxidation as measured by MDA level in HCT116 cells incubated for 24 h with TAC, MMF individually and their combination (a and b). Cells treated with H2O2 are considered as a positive control. Cells treated with H2O2 + TAC or MMF (c). Data are expressed as mean ± SD of independent experiments (
We noticed that HCT116 cells treated with the low concentrations of TAC or MMF (5 µM and 50 µM) incubated with H2O2 caused also a significant decrease in MDA level compared to H2O2 alone (Figure 4(c)). Our results confirm that TAC and MMF at low concentrations present a cytoprotective effect against MMF and TAC toxicity, respectively, at high concentrations.
Effect of TAC and MMF alone and their combinations on DNA damage
Analysis of the genotoxicity of TAC and MMF alone and associated in the HCT116 cells was performed by studying the level of DNA fragmentation using the comet assay. Cells exposed to different concentrations of drugs (TAC at 25 µM and MMF at 330 µM) exhibited significantly higher DNA fragmentation than those of control cells (Figure 5(a) and (b)). Our results indicated also that TAC and MMF alone and combined at low concentration at 5 and 50 µM, respectively, induced a similar rate of DNA fragmentation compared with the negative control. Our results also indicate that TAC at 5 µM significantly reduces DNA fragmentation caused by MMF at 330 µM and H2O2 at 75 µM with 35 and 36% (Figure 5(b) and (c)), respectively. However, MMF at 50 µM has a prevention percentage of 73 and 50%, which was sufficient to reduce the genotoxicity induced respectively by TAC at 25 µM and H2O2 at 75 µM in HCT116 cells (Figure 5(b) and (c)). It is possible that TAC and MMF free radical scavenging activity may have an important role to play in modulating ROS and DNA fragmentation, thereby acting as a cytoprotectant and genoprotectant.

Total DNA damage observed on HCT116 cells after treatment with TAC, MMF individually and in combination for 24 h of treatment. Cells treated with H2O2 are considered as a positive control. Cells treated with H2O2 + TAC or MMF (c). Data are expressed as mean ± SD of independent experiments (
Mitochondrial membrane potential
For further testing whether mitochondria function changed upon TAC and MMF individually and combined treatments, we explored changes of Δ

Loss of Δ
Discussion
In solid organ transplantation, a variety of immunosuppressive drugs are used to avoid rejection of graft. 6 TAC and MMF, two powerful immunosuppressive drugs, are our particular interests and their combination has drastically improved on the short-term graft survival rate of solid organ transplantation. 34 –36 Immunosuppressive molecules including the anti-calcineurin and antimetabolites are connected with significant toxicity especially gastrotoxicity. 10,20 Many studies have shown the effect of the combination of the two immunosuppressive molecules (TAC and MMF) on graft rejection, whilst the detailed mechanisms of this association are not yet clarified in the intestinal cells. In fact, previous studies indicated that oxidative stress is a cause of major pathogenic role in a number of GI tract diseases, in the pathogenesis of gastric damage, peptic ulcer diseases and likely gastric cancer. 37 –39
Consequently, the aim of this study was to investigate the molecular pathway of the toxicity of TAC and MMF alone and combined on human colon carcinoma HCT116 cells and the involvement of an eventual oxidative stress. Indeed, in a first set of experiment, we have assessed the effect of TAC and MMF individually and combined on the inhibition of cell proliferation using MTT assay.
For this purpose, we have demonstrated that the decrease of cell viability was induced in HCT116 exposed to TAC and MMF in a concentration-dependent manner with IC50 of 25 and 330 µM, respectively. As far as we have noticed, their combination, at high concentrations, decreases vigorously cell viability more than each molecule; this combination seems to be synergistic. Our findings are in accordance with previous studies showing that TAC and MMF have a cytotoxic effect in other cell lines. 40 –44 Our study showed that TAC at 5 µM and MMF at 50 µM were not toxic concentrations. These TAC concentrations corroborate approximately with those reported in various studies using various cell lines. 45 –49 Interestingly, TAC at low concentration (5 µM) presents a cytoprotective effect against MMF (at 330 µM) toxicity and inversely.
This observed cell death may be due to diverse cellular dysfunctions and different biochemical damages in response to TAC and MMF alone or combined. In this report, we have investigated the role of oxidative stress in the toxicity induced by combined immunosuppressive drugs in intestinal cells. ROS are mediators of intracellular signalling cascades and induce several changes at the cellular level and loss of cell function and lipid peroxidation. 50,51 MDA is generally considered to be an excellent index of lipid oxidation. 52 Our findings showed that both immunosuppressive drugs induced oxidative damage by enhancing ROS generation and increase of MDA level in HCT116 cells for TAC (25 µM) and MMF (330 µM), taken individually. These findings are in agreement with several studies showing that TAC induces lipid peroxidation in transplanted patients and MMF ameliorates the increase of MDA level in different animal models. 53 –57 Our results indicate that ROS production was probably the cause of the observed cell death induced by TAC and MMF at high concentration in HCT116 cells. However, TAC restores viability and presents an antioxidant effect at 5 µM when HCT116 cells were treated by MMF at high concentration (330 µM). The same results were observed with MMF at 50 µM when cells were treated with TAC at high concentration (25 µM). To confirm the involvement of stress oxidative in the observed cell death, we examined the protective effects of these two immunosuppressive drugs at low concentrations against H2O2 damage. Indeed, H2O2 was known to be an excellent inductor of ROS. 58 Hence, TAC (5 µM) exerts an important antioxidant effect by reducing ROS generation and MDA production induced by H2O2 at 75 µM. The same results were obtained with MMF at 50 µM. It is possible that TAC and MMF free radical scavenging activity have an important role to play in modulating ROS, thereby acting as a cytoprotectant. In light of these data, we can suggest that oxidative stress is a major contributor to TAC and MMF toxicity at high concentrations. Nevertheless, TAC and MMF play a dual role, they induced oxidative stress and antioxidant effect depending on the concentration. However, several studies indicate that TAC used at low concentration reduced MDA after injury/reperfusion and small intestinal ulceration in vivo. 59,60 Gabryel et al. 61 have demonstrated the neuroprotective effect of TAC in oxidative injury after hypoxic damage in the central nervous system, with an increase in GSH and a decrease of MDA levels. TAC has also a cardioprotective effect on ischaemia–reperfusion-induced myocardial damage which is shown by the reduction of superoxide radical formation. 62 However, cellular mechanisms of the cytoprotective effect of this compound are not fully understood. Herr et al. 63 have demonstrated the protective effect of TAC in human neuroblastoma cell line through reduction of ROS generation associated with reduction of ceramide release and activation of c-Jun. On the other side, MMF at 40 mg/kg/day reduces MDA formation in rat treated by cisplatin. 57 Also, our results are in accordance with the results of Krötz et al. 64 in which the antioxidant effect of MMF is shown by the inhibition of nicotinamide adenine dinucleotide phosphate activity and formation of superoxide radical in endothelial cells.
To explore the mechanism of TAC and MMF individually and combined-induced DNA damage, we further measured DNA migration performed by comet assay. Oxidative stress may cause DNA damage, repress the activity of cellular enzymes and induce cell death through the activation of kinases and caspase cascades. 65 The oxidative stress damage caused by TAC and MMF alone at high concentration might be one of the underling mechanisms for the two immunosuppressive molecules induced cell injury. Indeed, we demonstrated that an increase in DNA damage was vigorously induced in HCT116 cells exposed to the two immunosuppressive molecules, TAC (25 µM) and MMF (330 µM) concentration individually. This observed genotoxicity of TAC seems to be in agreement with previous reports showing that TAC is genotoxic compound in various human cell lines. 66,67 However, no study was available regarding the genotoxicity of MMF alone and its combination with TAC. The combined treatment of these immunosuppressive drugs remarkably reduced DNA fragmentation. Therefore, it can be considered that TAC and MMF at low concentrations play an important part in the protection against DNA damages in HCT116 cells exposed to MMF and TAC at high concentrations, respectively, which emphasize the involvement of oxidative stress as a mediator in the induction of DNA lesions.
Mitochondria are an important source of ROS within most mammalian cells. Excessive ROS production can lead to free radical attack of mitochondrial membrane depolarization and contributes to mitochondrial damage in a wide range of pathologies.
68,69
Our findings revealed that TAC and MMF at high concentration alone induced a loss of Δ
Conclusions
Our study is the first report linking the impact of immunosuppressive combination (TAC and MMF) in vitro. Present findings demonstrate that TAC and MMF probably play a dual role (oxidative stress and antioxidant) depending on their combination. These data contribute to better understanding of the genotoxic effects of TAC and MMF alone and associated at high concentration, which strongly implicate oxidative stress in HCT116 cell death.
Footnotes
Conflict of interest
The authors declared no conflict of interest.
Funding
This research was supported by the ‘Ministère Tunisien de l’enseignement Supérieur, de la Recherche Scientifique et des Technologies de l’Information et de la Communication’ through the ‘Laboratoire de Recherche sur les Substances Biologiquement Compatibles: LRSBC’.
