Abstract
One of the major hazards arising from recycling sites is the generation of leachate containing mixed metal. This study evaluated the toxic effects of leachate obtained from Elewi Odo municipal auto-battery recycling site (EOMABRSL) on male liver functions using hepatic indices and biomarker of cellular adenosine triphosphate (ATP) in rat via the oral route. Concentrations of heavy metals analysis showed that lead, cadmium, nickel, chromium, manganese, and iron were 1.5-, 2-, 2.5-, 1.36-, 19.61-, and 8.89-folds, respectively, higher than acceptable limits set by regulatory authority World Health Organization. Copper, zinc, and cobalt were 5.9-, 300-, and 1.02-folds, respectively, lower than permissible limits. The EOMABRSL was administered at 20, 40, 60, 80, and 100% concentrations to adult male rats for 60 days. Following exposure, plasma and livers were collected for several biochemistry assays. Exposure of animals to EOMABRSL resulted in 27.51, 28.14, 63.93, 28.42, and 40.16% increase in aspartate aminotransferase activity, whereas it elevated alanine aminotransferase activity by 5.35, 22.33, 88.68, 183.02, and 193.08%, respectively, when compared with the control. Similarly, γ-glutamyl transferase activity increased by 111.22, 114.19, 122.96, 573.14, and 437.02%, respectively, when compared with the control. EOMABRSL administration significantly decreased catalase activity and reduced glutathione level and superoxide dismutase with concomitant increase in malondialdehyde and hydrogen peroxide levels. Also, significant (p < 0.05) decrease in lactate dehydrogenase (LDH) activity (marker of cellular ATP) was observed. Taken together, the hepatotoxicity of EOMABRSL could be due to the depletion of LDH and induction of oxidative damage, which may suggest possible health hazards in subjects with occupational or environmental exposure.
Introduction
One of the major hazards arising from recycling sites is the generation of leachate containing mixed metal. The production of leachate is caused principally by precipitation percolating through waste deposited in a site. The precipitation may result from rain, melted snow, or the waste itself. Once in contact with decomposing solid waste, the percolating water becomes contaminated. 1 Small quantities of leachate had been implicated in polluting large volume of ground and surface waters, making them useless for domestic exploitation. 2 It is because of the increased use of automobile cars that led to the multiplicity of auto car batteries, which consequently result into the high levels of solid waste generation. Meanwhile, the facilities and modern scientific methods required for proper recycling of auto-batteries are not bio-tolerable. In some areas where recycling sites are designated for the salvaging and reprocess of the materials are open lands. The supposed leachate resulting from the decomposition of the biodegradable portion of the batteries finds its way into the ground/surface water. 3 This is because there are no better ways to prevent and/or collect the leachate produced. Hence, mixed metal contamination of the water resources may result.
Leachate containing mixed metals are assimilated by aquatic species and pass through the food chain and bioaccumulate upon long-term exposure. 4 Much of the damage produced by toxic metals stems from the generation and proliferation of oxidative free radicals. 5 Li et al. 6 reported the oxidative damage induced in hearts, kidneys, and spleens of mice by landfill leachate. Similarly, from our previous reports, exposure to leachate from municipal auto-battery recycling site inhibited steroidogenic enzymes and induced prostate cancer in rats. 7 Also, leachate prepared from municipal sludge dose dependently inhibited the mitotic index and increased the frequency of chromosome aberrations in the bone marrow of mice. 8 In addition, increased frequencies of micronucleated polychromatic erythrocytes and DNA damage were shown in mouse bone marrow cells. 8,9 Genotoxicity of leachate from the sediment of river Matanza-Riachuelo, one of the most polluted rivers of Latin America, has been reported. 10 Lastly, recent study showed that leachate activated key enzymes linked to non-insulin-dependent diabetes and hypertension as well as increased malondialdehyde (MDA) level in rat pancreas and testes. 11,12
More so, exposure to leachate has been shown to increase lipid peroxidation and activities of antioxidant enzymes such as superoxide dismutase (SOD), glutathione (GSH) peroxidase, and catalase (CAT) in heart, kidney, and spleens in mice. 6,13,14 Tire leachates have been shown to have profound effects both on cytochrome P450 A1 content and on ethoxyresorufin-O-deethylase activity as well as upregulation of antioxidant defenses in mice. 15 The possible deleterious effects of these heavy metals among occupationally exposed painters in Nigeria have also been documented. 16 –19 Widespread contamination of the environment with lead (Pb) is consistent with the results of epidemiological studies that have found elevated blood Pb levels in a large proportion of Nigerian children. 16,18
In Nigeria, the recycling means of rechargeable batteries containing Co, Ni, Cr, and Cd does not conform to the standard regulatory authorities as the approach to material recovery currently taking place is labor intensive and inimical to both ecosystem and environment. Recent study on the waste portable rechargeable batteries used in mobile phones revealed that the Co and Ni concentrations of the batteries are so high and exceeded the toxicity threshold limit concentrations by about 40-folds the limit concentrations of each metal. 20 Some inorganic substances have no known function in human biochemistry or physiology and do not occur naturally in living organisms. Hence, intake of metal-contaminated food and water or occupational exposure even at very low concentrations can be very harmful because of its bioaccumulation. 7,12
Most recycling sites receive a mixture of municipal, commercial, and mixed industrial batteries. Several xenobiotics enter the body through the gastrointestinal tract and after absorption are transported by the hepatic portal vein to the liver; thus, the liver is the first organ perfused by chemicals that are absorbed in the gut. 21 To date, research has largely concentrated on hepatic cells, since the liver plays a major role in the metabolism of xenobiotics and is consequently the primary target of most toxic responses. This study investigated the subchronic effects of leachate (mixed metal), obtained from municipal battery recycling site in Oyo State of Nigeria, on hepatic functions, cellular adenosine triphosphate (ATP), and some biomarkers of oxidative stress in male rats.
Materials and methods
Sampling site and leachate preparation
Leachate was obtained from the Elewi Odo municipal auto-battery recycling site (EOMABRL), located at Ibadan North Local Government Area of Oyo State, Nigeria (latitude 7°25.08′N and 7°25.11′N; longitude 3°56.45′E and 3°56.42′E). It is nearby a stream and located in residential estate. It covers about 1080 m2 and 200 × 120 ft of land. A randomized sampling technique 22,23 was used to collect the first horizon solid soils (0–15 cm deep) from five different points in the municipal auto-battery recycling site. Five randomly collected samples from each point were pooled to make a single representative sample.
Leachate (100%) was prepared from homogenous mixture according to a standard procedure 24,25 by adding 100 g of sample to 100 ml of distilled water (w/v) and shaken for 48 h at 32°C. Thereafter, the sample was left to sediment for 30 min, the supernatant was filtered with a 2.5-μm filter paper, and the filtrate was stored at 4°C until use. Water samples were collected from well nearby the site. They were designated as WELL-A and WELL-B. The water from the stream was regarded as STREAM and control drinking water was designated as POW.
Heavy metal analysis
The heavy metal contents of EOMABRSL were determined in accordance with the standard methods. 26 –28 Exclusion of interferences from extraneous sources of heavy metals was put in place to avoid heavy metal contamination during the analysis. Some metals, namely, copper (Cu), Pb, cadmium (Cd), cobalt (Co), chromium (Cr), zinc (Zn), iron (Fe), nickel (Ni), and manganese (Mn) were determined in the sample. Briefly, 100 ml of each leachate was digested by heating with concentrated nitric acid (HNO3) and the volume was reduced to 2–3 ml. This volume was made up to 10 ml with 0.1 N HNO3, and the concentrations of the metals were estimated using an atomic absorption spectrophotometer. 29
Chemicals and reagents
Epinephrine, GSH, 5,5-dithio-bis-2-nitrobenzoic acid, hydrogen peroxide (H2O2), and thiobarbituric acid were purchased from Sigma Chemical Co. (St Louis, Missouri, USA). All other reagents were of analytical grade and were obtained from the British Drug Houses (Poole, Dorset, UK). Kits for plasma aminotransferase activities and lactate dehydrogenase (LDH) were purchased from Random Laboratory Limited (UK).
Experimental protocol
Subacute exposure
Healthy adult male Wistar rats weighing approximately 175–220 g obtained from the Department of Biochemistry, Federal University of Technology, Akure, Nigeria, were randomly assigned to four groups. They were housed in plastic suspended cages placed in a well-ventilated rat house, provided with rat pellets and water ad libitum, and subjected to natural photoperiod of 12-h light:12-h dark cycle. All the animals received humane care according to the criteria outlined in the Guide for the Care and Use of Laboratory Animals prepared by the National Academy of Science and published by the National Institute of Health. Ethic regulations have been followed in accordance with National and institutional guidelines for the protection of animal welfare during experiments. 30
Three different concentrations (20, 40, and 80%) of EOMABRSL were prepared according to the groups, and the rats in each group were administered 1 ml of EOMABRSL via oral route for seven consecutive days. 31 Corresponding group of animals were administered with the same volume of distilled water via the same route and served as control. Rats were killed by cervical dislocation 24 h after the final treatment. The livers were quickly removed and weighed.
Subchronic exposure
A total of 30 healthy adult male Wistar rats weighing approximately 175–220 g were randomly assigned to five groups of five animals per group. Five different concentrations (20, 40, 60, 80, and 100%) of EOMABRSL were prepared according to the groups, and the rats in each group were administered 1 ml of EOMABRSL via oral route for 60 consecutive days. 31 Control group were administered with the same volume of distilled water via the same route. Rats were killed by cervical dislocation 24 h after the final treatment and blood was collected by retina–ocular technique. The livers were quickly removed, weighed and were placed on ice for biochemical analysis. Some fragments were prepared for routine histology, while other pieces were placed on ice for biochemical analysis. The blood was put into sample bottles containing ethylenediaminetetraacetic acid and centrifuged at low speed (3000g) at room temperature for 15 min.
Biochemical assay
Liver samples were homogenized in 50 mM Tris–hydrochloric acid buffer (pH 7.4) containing 1.15% potassium chloride, and the homogenate was centrifuged at 10,000 g for 15 min at 4°C. The supernatant was collected for the estimation of CAT activity using H2O2 as substrate according to the method of Clairborne 32 and H2O2 was also determined. Briefly, 50 μl of the test sample was added to a reacting mixture containing 500 μl of 59 mM H2O2and 950 μl of 50 mM phosphate buffer (pH 7.0). The reaction was carried out at 25°C and the decrease in absorbance at 570 nm was monitored for 3 min at 150 s interval. A unit of the enzyme activity is defined as the amount of enzyme catalyzing the decomposition of 1 μmol of H2O2 per minute at 25°C and pH 7.0 under specified condition. SOD was assayed by the method described by Misra and Fridovich. 33 Briefly, 0.1 ml of the liver sample was diluted in 0.9 ml of distilled water to make 1 in 10 dilutions. An aliquot of 0.2 ml of the diluted homogenate was added to 2.5 ml of 0.05 M carbonate buffer pH 10.2 to equilibrate in a cuvette and the reaction started by the addition 0.3 ml of 0.3 M of adrenaline. The reference cuvette contained 2.5 ml of carbonate buffer, 0.3 ml of substrate (adrenaline), and 0.2 ml of distilled water. The increase in absorbance at 480 nm was monitored every 30 s for 150 s. Protein concentration was determined by the method of Lowry et al. 34 Reduced GSH was determined at 412 nm using the method described by Jollow et al. 35 Briefly, 1 ml of supernatant was treated with 500 μl of Ellman’s reagent (19.8 mg of 5,5′dithiobisnitrobenzoic acid in 100 ml of 0.1% sodium citrate) and 3.0 ml of 0.2 M phosphate buffer (pH 8.0). The absorbance was read at 412 nm in a spectrophotometer. Lipid peroxidation was quantified as MDA according to the method described by Farombi et al. 36 and expressed as micromoles of MDA per milligram of tissue. Activities of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) in plasma samples were determined by the method of Reitmann and Frankel. 37 Activity of γ-glutamyl transferase (GGT) in plasma samples was determined by the method of Szasz. 38
LDH assay
The liver homogenate was assayed for LDH activity using commercially available kit (Randox Laboratories, UK). Assay was carried out according to the manufacturer’s instructions. 39
Histopathological examination
The livers were fixed in 10% formalin. The sections were mounted on albumenized glass slides and stained with eosin and hematoxylin. Morphological study of liver was done with the help of ocular micrometer scale under a light microscope.
Statistical analysis
The results of the replicates were pooled and expressed as mean ± SD. A one-way analysis of variance was used to analyze the results and Duncan multiple test was used for the post hoc. 40 Statistical package for Social Science Version 17.0 for windows was used for the analysis and the least significance difference was accepted at p < 0.05.
Results
Heavy metal concentration in EOMABRSL, WELLs, STREAM, and POW
The results of some heavy metal contents (Cu, Zn, Pb, Cd, Mn, Co, Cr, Fe, and Ni), obtained from Elewi Odo municipal auto-battery recycling site leachate (EOMABRSL), water samples (STREAM and WELL) around the site, and portable water sample (control) are presented in Table 1. The concentrations of heavy metals in EOMABRSL, WELL, and STREAM were significantly (p < 0.05) higher than the POW and acceptable limits set by regulatory authority World Health Organization (WHO) (Table 1). Firstly, EOMABRSL, Cd (0.006 mg/l), Cr (0.068 mg/l), Fe (2.667 mg/l), Ni (0.05 mg/l), Pb (0.015 mg/l), and Mn (7.842 mg/l) exceeded WHO permissible limits by 2.00-, 1.36-, 8.89-, 2.50-, 1.50-, and 19.61-folds, respectively. Secondly, STREAM, Fe (1.076 mg/l), Ni (0.048 mg/l), and Pb (1.548 mg/l) were higher than WHO permissible limits by 3.59-, 2.40-, and 154.8-folds, respectively. Thirdly, WELL-A, Ni (0.044 mg/l), and Pb (0.068 mg/l) exceeded the WHO permitted limits in the drinking water by 2.20- and 6.80-folds, respectively. A similar trend was observed in WELL-B as Ni (0.049 mg/l) and Pb (0.306 mg/l) exceeded WHO permissible limits in drinking water by 2.45- and 30.6-folds, respectively. As observed in this study (Table 1), levels of Co, Cu, and Zn were lower in all the samples than the permissible level set by WHO.
Characterization of heavy metals in EOMABRSL, water samples (STREAM and WELLs) around the site, and POW as control.a

EOMABRSL: Elewi Odo municipal auto-battery recycling site leachate; POW: drinking water sample used as control; LOEC: least observable effective concentration; CDW: drinking water sample; BDL: below detection level; WHO: World Health Organization.
Hepatic biochemical indices
Plasma activities of AST, ALT, and GGT were significantly (p < 0.05) increased following subchronic exposure of rats to EOMABRSL (Figures 1 to 3). Exposure of animals to 20, 40, 60, 80, and 100% EOMABRSL resulted in significant (p < 0.05) increase (Figure 1) in AST activity by 27.51, 28.14, 63.93, 28.42, and 40.16%, respectively, when compared with the control. ALT activity (Figure 2) significantly (p < 0.05) increased by 5.35, 22.33, 88.68, 183.02, and 193.08%, respectively, when compared with the control. Similarly, GGT activity (Figure 3) significantly (p < 0.05) increased by 111.22, 114.19, 122.96, 573.14, and 437.02%, respectively, when compared with the control.
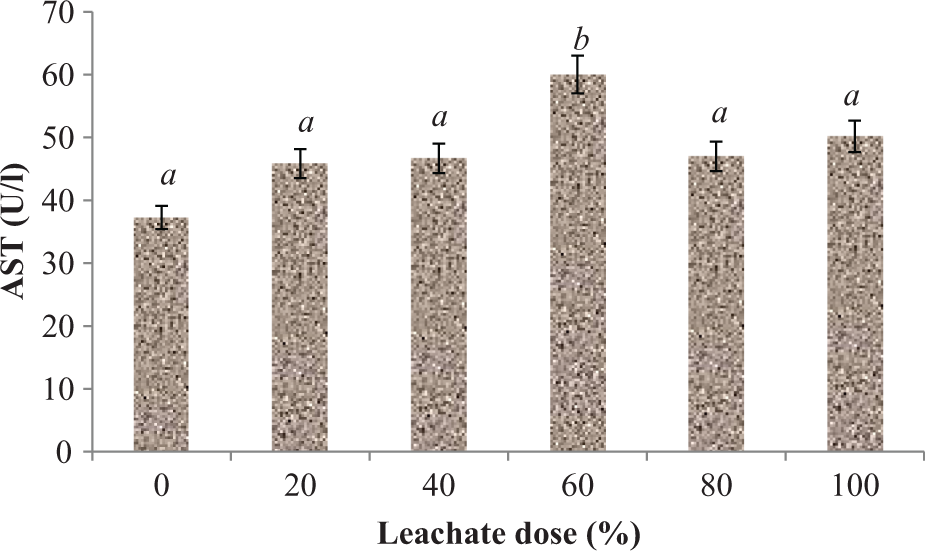
Effect of EOMABRSL on the activity of aspartate aminotransferase in a 60-day (long-term exposure) experiment. Group 1 received 0%, group 2 received 20%, group 3 received 40%, group 4 received 60%, group 5 received 80%, and group 6 received 100%. Values represent mean ± SD, n = 5. Values with different letters are significantly (p < 0.05) different. EOMABRSL: Elewi Odo municipal auto-battery recycling site leachate.
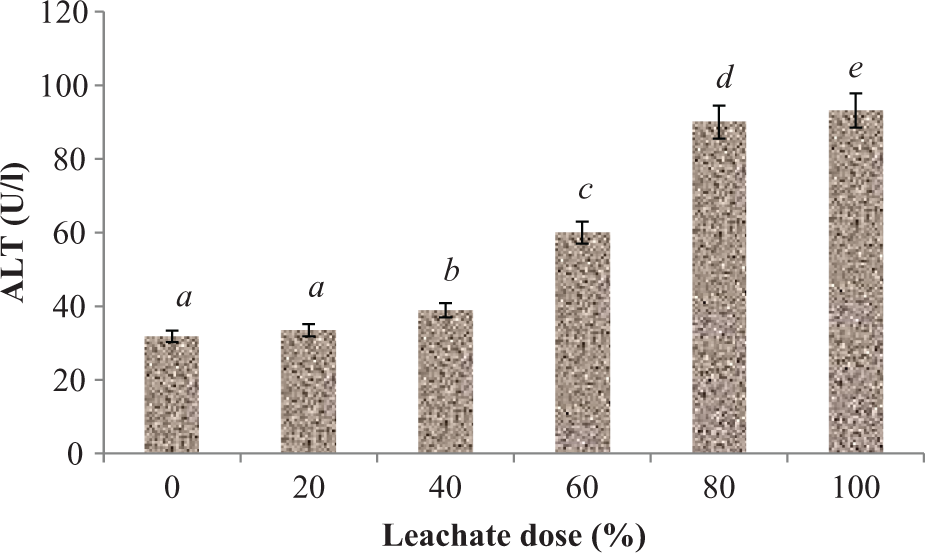
Effect of EOMABRSL on the activity of alanine aminotransferase in a 60-day (long-term exposure) experiment. Group 1 received 0%, group 2 received 20%, group 3 received 40%, group 4 received 60%, group 5 received 80%, and group 6 received 100%. Values represent mean ± SD, n = 5. Values with different letters are significantly (p < 0.05) different. EOMABRSL: Elewi Odo municipal auto-battery recycling site leachate.
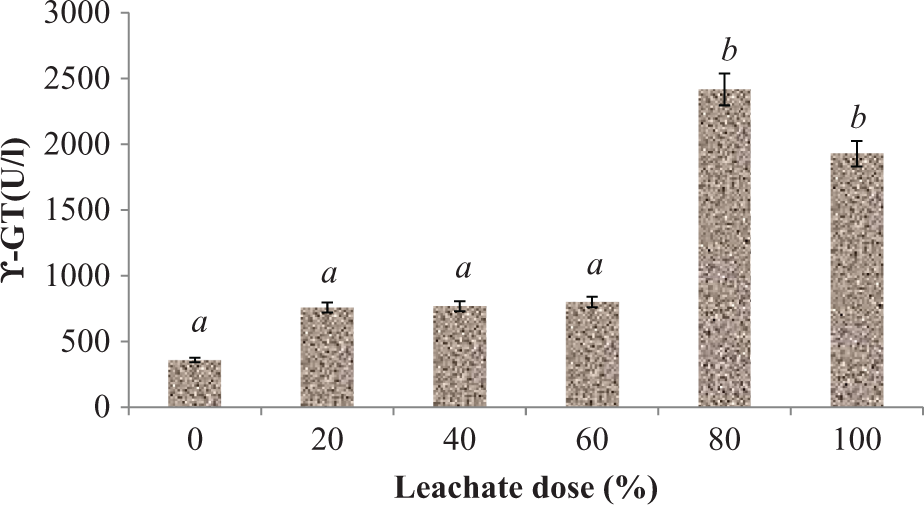
Effect of EOMABRSL on the activity of GGT in a 60-day (long-term exposure) experiment. Group 1 received 0%, group 2 received 20%, group 3 received 40%, group 4 received 60%, group 5 received 80%, and group 6 received 100%. Values represent mean ± SD, n = 5. Values with different letters are significantly (p < 0.05) different. EOMABRSL: Elewi Odo municipal auto-battery recycling site leachate; GGT: γ-glutamyl transferase.
Antioxidant status in the liver
The MDA content in liver homogenates of the rats treated with EOMABRSL was significantly (p < 0.05) elevated when compared with their corresponding control rats (Figure 4) during short-term exposure (7 days) by 89.58, 143.34, and 146.59%, respectively. As observed, there was no significant increase (p > 0.05) between 40% and 80% doses of the treated rats following subacute exposure. As shown in Figure 5, the rats exposed to EOMABRSL for 60 days (long-term exposure) had a significant (p < 0.05) increase in MDA content by 16.06, 14.79, 48.56, 100.28, and 59.94%, respectively, when compared with the control group. Effects of EOMABRSL on hepatic antioxidant status are shown in Figures 6 to 9. Following exposure to EOMABRSL, a significant (p < 0.05) decrease in hepatic GSH level and in activities of SOD and CAT were observed in all treated groups. While 20, 40, 60, 80, and 100% EOMABRSL treatment resulted in decreased GSH level by 38.65, 9.39, 45.07, 43.04, and 41.94%, SOD activity decreased by 3.16, 18.44, 59.15, 63.46, and 51.67%, respectively. H2O2 levels were markedly elevated in a non-dose-dependent manner following EOMABRSL administration. However, CAT activity was inhibited by 22.62, 22.32, 17.82, 19.70, and 21.09% after dosing the animal with 20, 40, 60, 80, and 100% EOMABRSL, respectively.

Effect of EOMABRSL on lipid peroxidation in a 7-day (short-term exposure) experiment. Group 1 received 0%, group 2 received 20%, group 3 received 40%, and group 4 received 80%. Values represent mean ± SD, n = 5. Values with different letters are significantly (p < 0.05) different. EOMABRSL: Elewi Odo municipal auto-battery recycling site leachate.

Effect of EOMABRSL on lipid peroxidation in a 60-day (long-term exposure) experiment. Group 1 received 0%, group 2 received 20%, group 3 received 40%, group 4 received 60%, group 5 received 80%, and group 6 received 100%. Values represent mean ± SD, n = 5. Values with different letters are significantly (p < 0.05) different. EOMABRSL: Elewi Odo municipal auto-battery recycling site leachate.
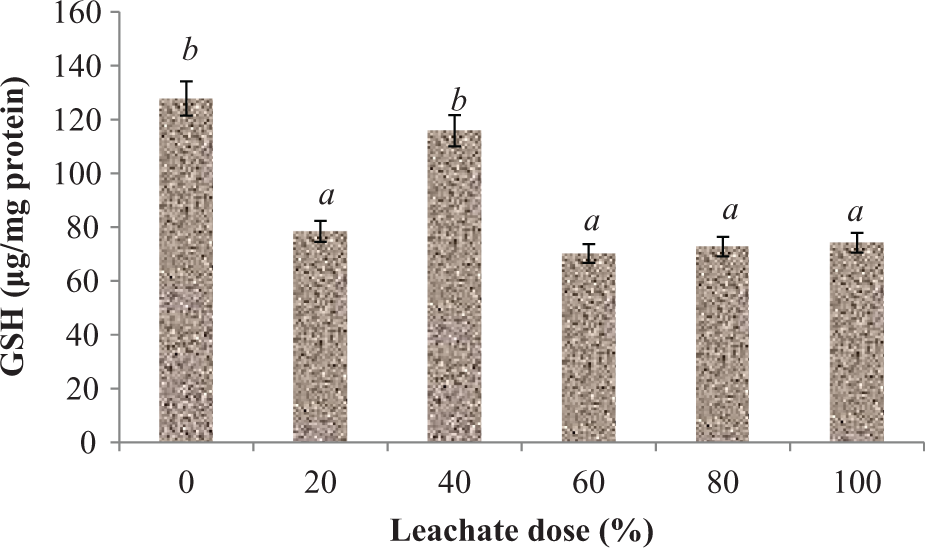
Effect of EOMABRSL on hepatic reduced GSH in a 60-day (long-term exposure) experiment. Group 1 received 0%, group 2 received 20%, group 3 received 40%, group 4 received 60%, group 5 received 80%, and group 6 received 100%. Values represent mean ± SD, n = 5. Values with different letters are significantly (p < 0.05) different. EOMABRSL: Elewi Odo municipal auto-battery recycling site leachate; GSH: glutathione.
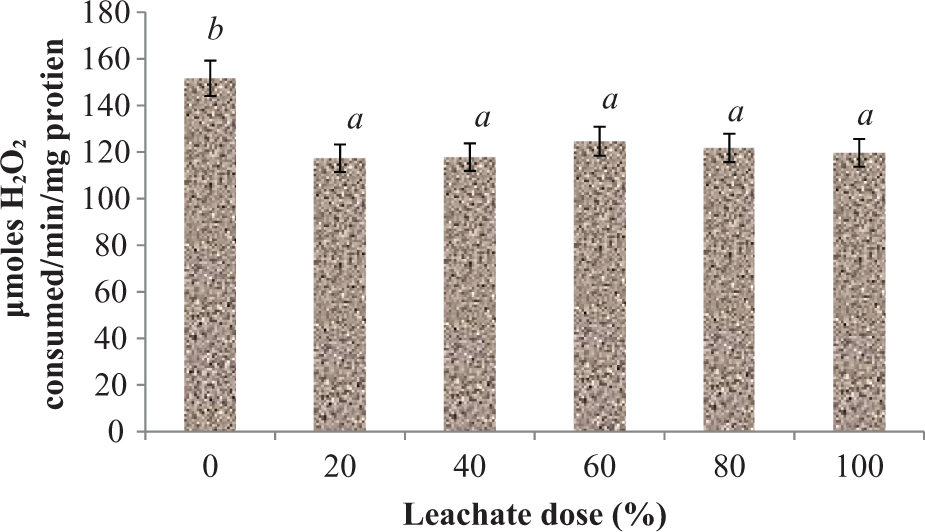
Effect of EOMABRSL on the activity of CAT in a 60-day (long-term exposure) experiment. Group 1 received 0%, group 2 received 20%, group 3 received 40%, group 4 received 60%, group 5 received 80%, and group 6 received 100%. Values represent mean ± SD, n = 5. Values with different letters are significantly (p < 0.05) different. EOMABRSL: Elewi Odo municipal auto-battery recycling site leachate; CAT: catalase.

Effect of EOMABRSL on the activity of SOD in a 60-day (long-term exposure) experiment. Group 1 received 0%, group 2 received 20%, group 3 received 40%, group 4 received 60%, group 5 received 80%, and group 6 received 100%. Values represent mean ± SD, n = 5. Values with different letters are significantly (p < 0.05) different. EOMABRSL: Elewi Odo municipal auto-battery recycling site leachate; SOD: superoxide dismutase.

Effect of EOMABRSL on H2O2 level in a 60-day (long-term exposure) experiment. Group 1 received 0%, group 2 received 20%, group 3 received 40%, group 4 received 60%, group 5 received 80%, and group 6 received 100%. Values represent mean ± SD, n = 5. Values with different letters are significantly (p < 0.05) different. H2O2: hydrogen peroxide; EOMABRSL: Elewi Odo municipal auto-battery recycling site leachate.
Cellular ATP level in the liver
Subchronic exposure to the rat of EOMABRSL significantly (p < 0.05) depleted the activity of LDH, functional marker linked to the production of energy (ATP) 42,43 by 54.61, 89.76, 76.25, 52.77, and 42.07%, respectively, when compared with the control group (Figure 10).
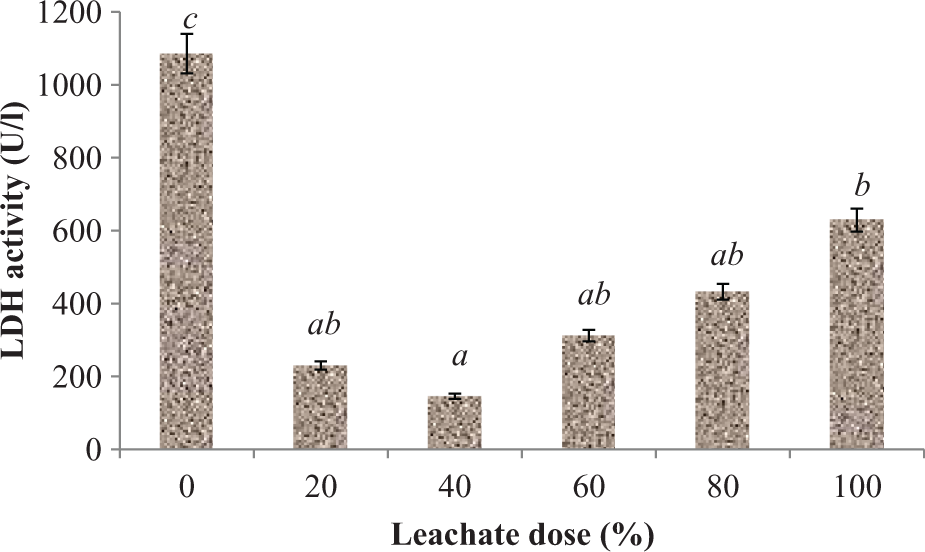
Effect of EOMABRSL on the activity of LDH in a 60-day (long-term exposure) experiment. Group 1 received 0%, group 2 received 20%, group 3 received 40%, group 4 received 60%, group 5 received 80%, and group 6 received 100%. Values represent mean ± SD, n = 5. Values with different letters are significantly (p < 0.05) different. EOMABRSL: Elewi Odo municipal auto-battery recycling site leachate; LDH: lactate dehydrogenase.
Hepatic cells damage
Figure 11 illustrates the different histopathological changes that were observed in the liver of animals that were given various doses in the study. The hepatic cells were arranged regularly and showed no visible lesions (Figure 10(a)). Administration of leachate at different doses as shown in Figure 10(b) to (f) caused severe hepatic lesions such as severe portal congestion and periportal fibroplasias, periportal cellular infiltration by mononuclear cells, and necrosis of hepatocytes.

(a–f) Changes of hepatic histopathology under subchronic exposure to leachate in rat (original magnification ×100): (a) 0% exposure showed no visible lesions to the hepatocytes or normal hepatocyte (NH); (b) 20% exposure of the leachate to the rat showed marked periportal cellular infiltration by mononuclear cells as indicated by arrows; (c) 40% exposure of the leachate to the rat exhibited severe portal congestion and periportal fibroplasias as indicated by arrows; (d) 60% exposure of the leachate to the rat showed periportal cellular infiltration by mononuclear cells as indicated by arrows; (e) 80% exposure of the leachate to the rat showed formation of periportal cellular infiltration by mononuclear cells as indicated by arrows; and (f) 100% exposure of the leachate to the rat showed severe formation of periportal cellular infiltration by mononuclear cells and hepatocyte necrosis as indicated by arrows.
Discussion
There is increasing concern in the society about the general health issues. Also, there is growing understanding of the role that recycling industries might be playing in the causation of certain health and related environmental problems. The use of land and water bodies to dispose hazardous or prescribed effluent is a significant contributing factor to the health risk faced by societies as a result of product recycling practices. Recycling sites have been reported to release large amounts of hazardous and deleterious chemicals to nearby groundwater via leachate. 1,2,44 Exposure to mixed toxicants may cause severe damage than single toxicant when exposed. Leachates consisting of mixture of many heavy metals may be a source of potential risk to human health. 45
An earlier report in our laboratory revealed that the heavy metal contents of EOMABRSL were significantly higher than acceptable limits set by the regulatory authority. 7,12,41 This indicates that the high concentration of heavy metals, namely, Cd, Cr, Cu, Pb, Co, Mn, Zn, Fe, and Ni in EOMABRSL is of great health risk. Heavy metals have been implicated in hepatocellular carcinoma. 46 Cd, Cu, Ni, and Fe produce free radicals and when present in an unbound form, they produce reactive oxygen species (ROS) that can cause DNA, protein, and lipid damage. 47 Co is an essential trace element present in vitamin B12. However, low intake of Co, Cu, and Zn has been linked with reproductive and developmental toxicity, neurotoxicity, carcinogenicity, and acute toxicity. 48,49 This study revealed non-dose-dependent increase in the activities of plasma ALT, AST, and GGT. These enzymes are localized in periportal hepatocytes, reflecting their role in oxidative phosphorylation and gluconeogenesis, and their plasma activities presumably increase as a result of cellular membrane damage and leakage. 50 This underlines their use as biochemical markers for early acute and subchronic hepatic damage. 51 Hence, the spontaneous elevated levels of AST, ALT, and GGT in the blood circulation were indicative of a hepatic injury after EOMABRSL exposure.
Evidence suggests that various enzymatic and non-enzymatic systems have been developed by the cell to cope with the ROS and other free radicals. However, when a condition of oxidative stress is established, the defense capacities against ROS become insufficient. 52 Administration of EOMABRSL during subacute and subchronic exposure resulted in increase in MDA level in treated animals. This result is consistent with previous study on oxidative damage induced in heart, kidney, and spleen of mice treated with landfill leachate. 6,12,14
The GSH plays a crucial role in protecting the cells from oxidative damage. 53 In our study, hepatic GSH content was considerably depleted following EOMABRSL administration. This decline in GSH content under the present experimental model suggests its overutilization to challenge the prevailing oxidative stress under the influence of ROS generated from EOMABRSL. Low levels of GSH were observed during increased oxidative stress. 54 Activities of SOD and CAT were markedly decreased by EOMABRSL treatment. Superoxide radicals by themselves or after their transformation to H2O2 caused oxidation of the cysteine in the enzyme and decreased the SOD activity. 54 The degradation of H2O2, a potent oxidant at high cellular concentration, is effected by CAT. The low activities of SOD and CAT could be linked to the excessive accumulation of H2O2 and O2− caused by EOMABRSL, resulting in oxidative stress. Our result is consistent with earlier report of Guangke et al. 13
LDH is an oxidoreductase enzyme that catalyzes the inter-conversion of pyruvate and lactate. Cells release LDH into the bloodstream after tissue damage. The level of cellular ATP during anaerobic condition had been widely assessed using LDH activity because it is a fairly stable enzyme. 42,43 As observed in the study, exposure to EOMABRSL significantly (p < 0.05) depleted LDH activity. This hypothesized that inhibition of LDH (a key enzyme of glycolytic pathway) caused by EOMABRSL in the absence of oxygen would slow down the metabolic pathway responsible for (ATP) energy production. This finding supports the previous discovery that patients with hepatic and renal problems exhibited low level of ATP. 55 In addition, under aerobic conditions, pyruvate is converted to acetyl coenzyme A catalyzed by pyruvate dehydrogenase, which may be oxidized in tricarboxylic acid cycle to yield ATP. It may also be used for the synthesis of fatty acids, cholesterol, and ketone bodies. As observed in the study, it is suggested that heavy metals in EOMABRSL would disrupt ATP production by inhibiting pyruvate dehydrogenase when competing with the phosphate group, thereby uncoupling oxidative phosphorylation. Also, energy-linked reduction of NAD+, mitochondrial respiration, and ATP synthesis of the liver are inhibited. This observation corroborates the work done by Klaassen et al. 56 that reported that arsenic and its compounds disrupt ATP production when exposed to animals via water drinking. This is also consistent with several studies that reported that cells exposed to arsenic showed a considerable depletion in ATP and glycogen levels in liver and other tissues such as heart and kidney. 57
From these findings, EOMABRSL induces hepatic oxidative damage and diminishes cellular ATP in rats. This may be due to the leachate or its metabolites. Humans are exposed to heavy metals by consuming contaminated food and water, which could result in various biochemical disorders. This could damage their structures and slow down their functions. 58 The concentration of Pb (0.015 mg/l) detected in the leachate of this investigation was far higher as compared to WHO permissible limits (0.01 mg/l). Previous studies showed that exposure to low inorganic Pb at 0.004 mg/l dose in blood impaired hepatocytes in male rats. 59 However, exposure to EOMABRSL (mixed metals) at the investigated doses exhibited profound depleted cellular ATP in a non-dose-dependent manner of the rat exposed. This effect may be linked to the elevated dose of the Pb that had implications in liver cirrhosis. 58,59 The toxic effect may also be linked to the individual, additive, synergistic, or antagonistic interactions of the metals with the hepatic biomolecules.
The main sources of Ni exposure to mammals had been traced to drinking water and contaminated foods. 60 As observed in the study, the level of Ni (0.05 mg/l) detected in EOMABRSL exceeded the least observable effective concentration when compared with WHO limits (0.02 mg/l). This suggests that Ni could pollute wells and stream water when situated close to battery recycling site. Similarly, Fe and Mn concentrations (2.667 and 7.842 mg/l) in the leachate exposed to the experimental rats were higher relative to WHO limits (0.3 and 0.4 mg/l), respectively. The rats exposed to Fe and Mn contained in EOMABRSL beyond WHO permissible limits showed reduced response of LDH activity in a dose-dependent manner. This finding is in agreement with the previous report, which stated that workers exposed to metals such as Fe and Mn had liver lesions and reduced response of LDH activity. 45 Mn and Fe in EOMABRSL may also be linked to alteration of antioxidant enzymes, 61 ROS production, and decreased GSH, which resulted into lipid peroxidation and oxidative damage of liver cells. 12,14
Reports had also shown that male rats that were intraperitoneally injected with Cr ranged between 1 mg/l and 2 mg/l showed severe portal congestion and periportal fibroplasias, periportal cellular infiltration by mononuclear cells, and necrosis of hepatocytes of treated rats. 62 The similar effect of the Cr in leachate-treated rats may be because of its large concentration (0.068 mg/l) relative to WHO permissible limits (0.05 mg/l).
Co is beneficial to humans because it is a part of vitamin B12, which is essential for human health and stimulates the production of red blood cells. However, large dose greater than 0.05 mg/l may damage liver cells. 63 Zn within the tolerable limits (3 mg/l) plays a critical role in male hepatic functions. 64 Copper occurs at the active site of SOD to exercise a redox function. 65 In the present findings, Co (0.049 mg/l), Zn (0.01 mg/l), and Cu (0.341 mg/l) detected in EOMABRSL were considerably lower than WHO exposure limits (0.05, 3, and 2 mg/l), respectively. The low level or the deficiencies of these metals (Co, Zn, and Cu) had been implicated in liver damage. This could cause sequential alteration of hepatic antioxidant enzymes, such as CAT and SOD. 63 –65 In addition, competitive interactions between Zn and other metallic ions (Cd, Pb, and Mn) might have occurred in the hepatic cells. This is because Zn has physicochemical properties similar to other ions and there is consumption of high doses of these other ions in EOMABRSL. Generally, the level of heavy metals in EOMABRSL was higher than STREAM, WELL-A, and WELL-B. Its high levels may be because soil can easily form ligands with metals or likely that it has high capacity to retain heavy metals than inorganic solvents. The considerably higher concentrations of Pb, 1.548, 0.068, and 0.306 mg/l in STREAM, WELL-A, and WELL-B, respectively, than the leachate (EOMABRSL), 0.015 mg/L of this study may be linked to the direct discharge of effluent from the factory into the stream. The previous study had implicated that when Pb passes through the soil, the complex ligand formation or adhesion capacity with soil and other materials may be weak. 1 In addition, the concentrations of Mn (7.842 mg/l), Fe (2.667 mg/l) in the leachate, and Pb (1.548 mg/l) in stream suggest that most of the waste batteries recycled at the site were made of electrolytes from Mn, Fe, and Pb sulfate. Collectively, the toxic effects of the whole leachate on rat hepatic biomarkers examined in this study are due to their individual, additive, competitive, synergistic, or antagonistic interference with the normal hepatic biomolecules.
Moreover, adverse histopathological changes were observed in the livers of treated animals, including severe portal congestion and periportal fibroplasias, periportal cellular infiltration by mononuclear cells, and necrosis of hepatocytes. However, the use of multi-stage treatment method of leachate has to be put into operation and enforced by Nigeria regulatory authorities to prevent large release of toxicants from battery recycling sites to the environment.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
