Abstract
Nilotinib is an oral potent tyrosine kinase inhibitor that has diverse biological activities. However, its effects on hypercholesterolemia and associated disorders have not been studied yet. The present study explored the effect of nilotinib on atherosclerosis progression, endothelial dysfunction, and hyperlipidemia-associated hepatic injury in high-cholesterol (HC)-fed rabbits. Rabbits were classified into four groups: control, nilotinib, HC, and HC + nilotinib groups. Rabbits were fed either a regular diet or an HC-enriched diet for 8 weeks. By the end of the eighth week, blood and tissue samples were obtained for biochemical, histological, immunohistochemical, and in vitro analyses. Results indicated that the HC diet induced a significant elevation in the serum lipid parameters (triglycerides, total cholesterol, low-density lipoprotein cholesterol, and high-density lipoprotein cholesterol), lactate dehydrogenase, and nitric oxide content. Endothelial dysfunction was evident through the impairment of acetylcholine-induced relaxation of isolated aortas and the histopathological lesions of the aortic specimen. Moreover, HC significantly increased serum malondialdehyde. Liver damage was clear through increase in serum transaminases and alkaline phosphatase, and it was further supported by histopathological examination. HC increased the expression of platelet-derived growth factor receptor (PDGFR)-B in both aorta and liver tissues. Interestingly, nilotinib administration retarded atherosclerosis progression and attenuated all of the aforementioned parameters. These data suggest that nilotinib may counteract atherosclerosis development, vascular dysfunction, and hepatic damage in HC-fed rabbits through interfering with PDGF-B.
Background
Hypercholesterolemia is one of the major risk factors for atherosclerosis and other cardiovascular diseases. Atherosclerosis is described by the deposition of the blood lipids in the subendothelial space of large blood vessels leading to the recruitment of circulating inflammatory cells, such as lymphocytes and monocytes and secretion of different inflammatory cytokines. 1 –3 The pathogenesis of atherosclerosis seems to be multifactorial. Lipoprotein oxidation, mainly low-density lipoprotein (LDL) oxidative modification with subsequent lipid-laden foam cells formation, plays the central role in atheromatous plaque formation in the intimal layer of the artery. 4,5 Endothelial dysfunction is the main feature in atherosclerosis. It results from decreased nitric oxide (NO) bioavailability either through decreasing NO synthesis and release from the endothelium or through NO reaction with superoxide radicals after its release from the endothelium. 6 Migration and proliferation of vascular smooth muscle cells (VSMCs) from the media to the subendothelial area contribute to intimal thickening during the arterial remodeling. Recent studies suggest that nuclear factor-κB induces the expression of platelet-derived growth factor (PDGF). PDGF is a well-known chemoattractant for VSMC, which activates neointima formation during arterial remodeling. PDGF and its receptor are both expressed by cells involved in the atherogenic process. PDGF-B promotes the uptake and degradation of modified LDL, and cholesterol accumulation by macrophages. Thus, it has been suggested that an overactivated PDGF system may be involved in atherosclerosis development. 7,8
Another important pathological state that is closely associated with hyperlipidemia is nonalcoholic fatty liver disease (NAFLD), which is a broad term used to describe a wide range of hepatic disorders. Simple steatosis is the earliest stage characterized by the deposition of triglyceride (TG) droplets in the hepatocyte cytoplasm and can develop into nonalcoholic steatohepatitis, with diffuse hepatic fatty infiltration and lobular inflammation as the main features. 2,9 –11 Prevention of hyperlipidemia may have a significant impact not only on atherosclerosis development and associated endothelial dysfunction but also on the NAFLD development.
Nilotinib, an oral highly potent tyrosine kinase inhibitor, is commonly used for BCR-ABL-positive chronic myelogenous leukemia (CML) treatment in both the frontline setting as well as for patients with resistance or intolerance to imatinib. 12 Besides the ability of nilotinib to suppress the tyrosine kinase activity of ABL/BCR–ABL, it also inhibits PDGF receptor (PDGFR), stem cell factor (KIT), colony stimulating factor-1 receptor, and discoidin domain receptor. 13,14 Recent studies have shown other beneficial pharmacological activities of nilotinib such as antifibrotic 15 and anti-inflammatory, 16,17 which were attributed to its ability to specifically inhibit PDGFR tyrosine kinase, transforming growth factor-beta1 (TGF-β1) and tumor necrosis factor-alpha (TNF-α). 15,17 Clinical studies have reported the abnormal expression of PDGF and its receptors in multiple disorders including fibroproliferative diseases, neoplasm, and atherosclerosis. 18 Furthermore, another tyrosine kinase inhibitor, imatinib, has showed efficacy in ameliorating diabetes-induced atherosclerosis. 7 Until now, not much has been discovered regarding its effects on atherosclerosis-associated vascular damage. Thus, this study attempted to explore nilotinib effects on vascular endothelial dysfunction as well as on liver damage in hypercholesterolemic rabbits and explore the possible underlying mechanisms.
Materials and methods
Chemicals
Nilotinib (Novartis Pharma AG, Basel, Switzerland) was suspended in 0.5% carboxymethyl cellulose solute. Cholesterol was purchased from Winlab, UK. Acetylcholine (ACh), phenylephrine (PE), and sodium nitroprusside (SNP) were purchased from Sigma-Aldrich Chemical Co. (St. Louis, Missouri, USA). All other chemicals and biochemicals used in this study are of high analytical grade.
Experimental animals
Twenty-four adult male New Zealand white rabbits (1.5–2.0 kg) were kept individually in standard stainless steel cages and acclimatized for 1 week before experiments. Rabbits were housed under standard conditions of temperature (25°C) and light (12-h light/12-h dark cycles). Rabbits received about a 100 g/kg/day normal rabbit chow diet, consisting of 40–50% carbohydrates, 15–25% fiber, 10% protein, and 2% vegetable fat or a high cholesterol (HC) diet prepared by incorporation of a normal chow diet with 0.5% cholesterol with free access to water. All experiment procedures described in this study follow the ethical principles and guidelines adopted by the “Research Ethics Committee, Faculty of Pharmacy, Mansoura University” which is in accordance with “Principles of Laboratory Animals Care, National Institutes of Health (NIH) publication no. 85–23, revised 1985.”
Experimental design
Rabbits were randomly assigned into four experimental groups (six rabbits each) and fed one of the following regimens: control group, a regular rabbit diet for 8 weeks; nilotinib group, rabbits were fed a regular rabbit diet for 8 weeks and administered nilotinib (7.5 mg/kg, orally) daily for only the last 4 weeks; HC group, where rabbits were fed a HC diet containing 0.5% cholesterol for 8 weeks; and HC + nilotinib groups, rabbits were fed a HC diet for 4 weeks and then started to be administered nilotinib (7.5 mg/kg, orally) along with the HC diet for another 4 weeks. The dose of nilotinib was selected based on the previous studies done on rats 17,19 and calculated using Paget and Barnes conversion table. At the end of the eighth week, rabbits were fasted for 12 h and then anaesthetized using IV ketamine (50 mg/kg) and xylazine (10 mg/kg). Animals were killed by carotid exsanguination after blood collection.
Biochemical analysis
Blood samples were collected prior to the treatment (baseline), after 4 weeks (w = 4), and at the end of the experiment (w = 8). Blood samples were obtained through marginal ear vein, and the collected sera were stored at −80°C until further analysis.
Estimation of serum lipid profile
Serum total cholesterol (TC) and TGs were determined enzymatically using commercial kits (Stanbio Laboratory, Boerne, Texas, USA, and Biocon Diagnostik, Germany) following the methods of Allain et al., 20 and Buccolo and David, 21 respectively. Serum high-density lipoprotein (HDL) was determined using HDL cholesterol (HDL-C) kit (Human, Germany) following the method of Izawa et al. 22 LDL concentration was calculated according to the Friedewald equation, LDL-C = TC − (HDL-C − TG/5). 23 Atherogenic index was computed using the following equation, Atherogenic index = (serum cholesterol level − serum HDL level)/serum HDL level. 24
Estimation of serum lactate dehydrogenase
Lactate dehydrogenase (LDH) was determined immediately using a commercial kit (Human) according to the manufacturer’s instructions.
Estimation of serum alanine aminotransferase, aspartate aminotransferase, and alkaline phosphatase
Aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP) were used as biochemical markers for hepatic damage. Serum activities of ALT, AST, and ALP were estimated using commercial kits (Human) following the manufacturer’s instructions.
Estimation of serum total nitrite/nitrate level
Briefly, equal volumes of samples and 0.5% zinc sulfate solution were mixed, incubated at 25°C for 5 min, and then centrifuged for 20 min. Nitrite/nitrate (NOx) level was estimated in the supernatant using NO assay kit (R&D Systems, Minneapolis, MN, USA), following the manufacturer protocol.
In vitro vascular reactivity experiments
Immediately after opening the chest, the thoracic aortas were carefully removed and cut into two portions. The first portion was used for in vitro study, while the other was used for histopathological and immunohistochemical (IHC) analyses.
The aortic rings (2–4 mm in length) were cut and placed immediately in a cold oxygenated physiological salt solution (PSS) of the following composition (mmol/L): sodium chloride 118, potassium chloride (KCl) 4.7, calcium chloride 2.5, monopotassium phosphate 1.2, magnesium sulfate·7H2O 1.2, sodium bicarbonate 25, and glucose 11.1, pH 7.4.
The aortic segments were mounted between two stainless steel hooks in an organ bath filled with PSS at 37°C and bubbled with a mixture of 95% O2 and 5% CO2. The segments were set under a passive tension of 2 g and equilibrated for 60 min with washing every 15 min. Using a force displacement transducer (Hugo Sachs Elektronik, Germany), the isometric tension generated by the aortic rings was measured and recorded with a PowerLab unit linked to a personal computer running Chart v4.2 software. After 60 min, the rings were exposed to KCl (80 mmol/L) for three times to produce the maximal contraction and then rings were washed until tension returned to the baseline.
Assessment of vascular reactivity of aortas
To measure vasocontraction, cumulative concentration–response curves (CRCs) to PE (10−9, 3 × 10−9, 10−8, 3 × 10−8, 10−7, 3 × 10−7, 10−6, 3 × 10−6, and 10−5 mol/L) were established. The contractile responses were calculated as percentage of KCl-induced contraction. Maximum effect (E max) and concentration inducing 50% of E max (EC50) were determined from CRC, and pD2 was calculated as –log (EC50). 25
To measure vasorelaxation, aortic rings precontracted with PE (10−6 mol/L) were exposed to either ACh (10−8, 3 × 10−8, 10−7, 3 × 10−7, 10−6, 3 × 10−6, and 10−5 mol/L) or SNP (NO releasing agent, 10−8, 3 × 10−8, 10−7, 3 × 10−7, 10−6, 3 × 10−6, and 10−5 mol/L) added to the bath cumulatively. Relaxant responses were described as percentage declines of the magnitude of the PE-induced contraction. E max and the inhibitory concentration 50% (IC50) were determined from CRC, and pD2 was calculated as −log (IC50). 25
Histopathological analysis
Aortic tissue histopathology
A part of aorta was fixed in neutral buffered formalin and stained with hematoxylin and eosin (H&E). Aortic specimens were examined, and the thickness of intimal and media (I/M) ratio was calculated. Another part of the aortic tissue was stained with Oil Red O for the quantification of lipid deposition in aortic tissues, and the positively stained area was expressed in square micrometers per section (10 μm; 100×). Specimens were examined using an image analyzer (Leica Microsystem Imaging solution Ltd., Cambridge, UK).
Liver histopathology
Livers were excised, washed with ice-cold, fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned (4 to 5 μm), and stained with H&E. The degree of steatosis (the percentage of liver cells containing fat) or necrosis (the percentage of the cross section containing necrotic foci) was scored as follows: 0 (0%), 1+ (<25%), 2+ (26–50%), 3+ (51–75%), and 4+ (>75%). 26
Measurement of oxidative stress
Serum malondialdehyde (MDA) content was estimated using a commercial kit (Biodiagnostic Company, Giza, Egypt). Briefly, MDA content was determined spectrophotometrically by measuring the level of thiobarbituric acid–reactive substances at 534 nm. MDA level was expressed as nanomoles per milliliter.
IHC estimation for PDGFR-β
Tissue miniarray (TmA) for aorta and liver blocks of all animals was constructed as previously described. 27 IHC staining was performed on 4-µm slices of paraffin-embedded TmA tissue sections using primary antibodies against PDGFR-β (Santa Cruz Biotechnology, Santa Cruz, California, USA) automatically in Ventana Bench Mark XT, Ventana Medical Systems, Inc., USA system. The avidin–biotin technique was done using diaminobenzidine for visualization, and sections were counterstained with hematoxylin. The slides were examined under a light microscope. IHC semiquantitative analysis was done using image analysis software (ImageJ, 1.46a; NIH, USA).
Statistical analysis
The data are displayed as the mean ± standard error of mean, where n is the number of rabbits. One-way analysis of variance (ANOVA) followed by Tukey–Kramer’s multiple comparisons post hoc test was used when appropriate to compare between groups. To estimate the general impacts of the HC diet on the measured indices over the whole period of the study (0, 4, and 8 weeks) among the different groups, ANOVA for repeated measurements followed by a Tukey–Kramer post hoc test was used. Nonparametric Mann–Whitney U-test (two-tailed) was used for the evaluation of histopathological scoring. For vascular reactivity experiments, E max was considered as the highest response obtained. Both E max and pD2 (= −log EC50) were estimated from CRCs using nonlinear regression analysis through Graphpad Prism software (Graphpad Software Inc., San Diego, California, USA). A p value of <0.05 was considered statistically significant.
Results
There was no significant difference between the nilotinib group and the normal control group in all the measured biochemical parameters.
Effects on serum lipid profile
As shown in Figure 1, the baseline levels of the measured serum lipid parameters were not significantly different among the different groups. At week 4, the HC diet induced significant elevations in serum TC, TGs, and LDL cholesterol (LDL-C), while HDL-C was significantly decreased compared to control or baseline levels. The HC group showed a significant increase in the atherogenic index compared to the control group. Interestingly, nilotinib succeeded to produce a significant reduction in serum TC, TGs, and LDL-C compared to the HC group at corresponding time and compared to 4 weeks levels leading to a significant decrease in atherogenic index. Furthermore, nilotinib increased the level of HDL-C to a value that was comparable to the control value.
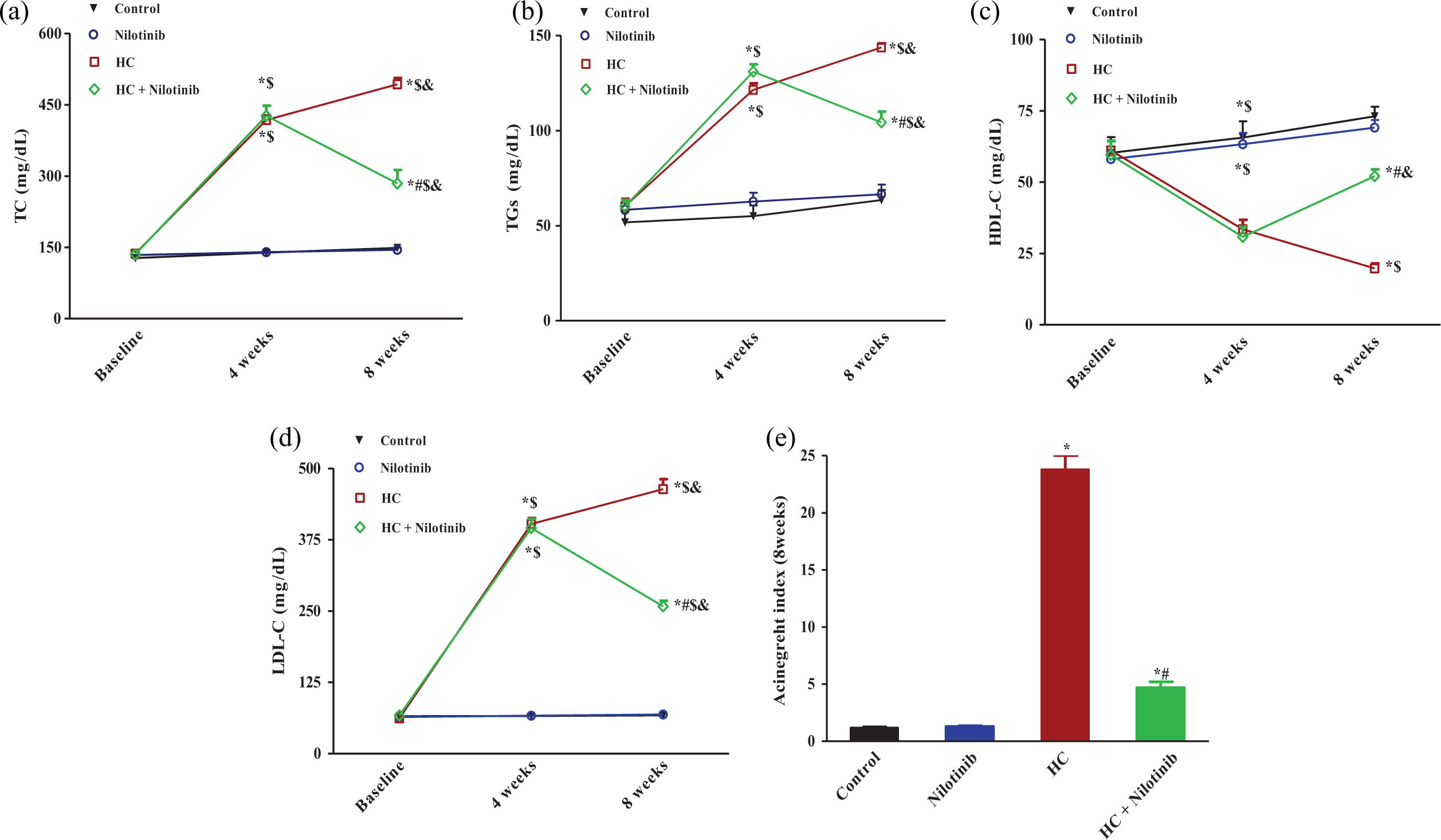
Effects of daily administration of nilotinib on serum lipid profile in HC-fed rabbits. Data are expressed as mean ± SEM (n = 6). *p < 0.05 as compared to the normal control group at experimental corresponding times. # p < 0.05 as compared to the HC group at experimental corresponding times. $ p < 0.05 as compared to serum baseline level within the same group. & p < 0.05 as compared to serum level at 4 weeks within the same group (repeated measurements for ANOVA followed by Tukey–Kramer’s multiple comparison test). HC: high cholesterol; SEM: standard error of mean; ANOVA: analysis of variance; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; TGs: triglycerides.
Effects on serum LDH, ALT, AST, and ALP levels
The HC diet induced a significant increase in LDH level compared to the control group at the corresponding times (Figure 2). Nilotinib treatment resulted in a significant attenuation of LDH level compared to the HC group at corresponding time and compared to 4 weeks level. Also, a significant increase in serum ALT, AST, and ALP was observed in the HC group compared to control or baseline levels (Figure 2). Daily administration of nilotinib caused a remarkable decrease in these elevated markers compared to the HC group at corresponding time and compared to 4 weeks level.

Effects of daily administration of nilotinib on serum LDH, AST, ALT, and ALP levels in HC-fed rabbits. Data are the mean ± SEM (n = 6). *p < 0.05 compared to the normal control group at experimental corresponding times. # p < 0.05 compared to the HC group at experimental corresponding times. $ p < 0.05 compared to serum baseline level within the same group. & p < 0.05 compared to serum level at 4 weeks within the same group (repeated measurements for ANOVA followed by Tukey–Kramer’s multiple comparison test). LDH: lactate dehydrogenase; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; HC: high cholesterol; SEM: standard error of mean; ANOVA: analysis of variance.
Effects on serum MDA and NOx levels
As illustrated in Figure 3, the HC diet induced a significant elevation in serum levels of MDA and NOx compared to normal control or baseline levels indicating the overproduction of reactive oxygen species (ROS) and NO. However, nilotinib treatment significantly ameliorated the levels of MDA and NOx compared to the HC group at corresponding time and compared to 4 weeks level.

Effects of daily administration of nilotinib on serum MDA and total NOx in HC-fed rabbits. Data are the mean ± SEM (n = 6). *p < 0.05 compared to the normal control group at experimental corresponding times. # p < 0.05 compared to the HC group at experimental corresponding times. $ p < 0.05 compared to serum baseline level within the same group. & p < 0.05 compared to serum level at 4 weeks within the same group (repeated measurements for ANOVA followed by Tukey–Kramer’s multiple comparison test). MDA: malondialdehyde; NOx: nitrite/nitrate; SEM: standard error of mean; HC: high cholesterol; ANOVA: analysis of variance.
Effect on aortic injury and atheroma development
Aortic specimen of the control group and nilotinib group showed normal histology. There were no signs of lesions or fat deposition. On the contrary, the HC group exhibited thickened intima, endothelial distortion, presence of foam cells in subendothelial layer (Figure 4(c)), and severe deposition of fats (Figure 4(h)). Furthermore, HC increased the mean atherosclerotic lesion area as well as I/M ration significantly compared to the control group. Nilotinib administration with HC reduced HC-induced lesions (Figure 4(d)) and the deposition of fats in aorta (Figure 4(i)), and there was a significant decrease in the mean atherosclerotic lesion area and in I/M ratio compared to the HC group.

Effects of daily administration of nilotinib on the development of aortic lesions and fatty streaks in HC-fed rabbits. The extent of aortic lesions and endothelial cell distortion stained with H&E ×200 (left panel) and the extent of fat deposition stained with Oil red ×100 (right panel). (a) and (f) Control group and (b) and (g) nilotinib group, showing normal histology with no signs of fat deposition. (c) and (h) HC group, presence of foam cells (yellow arrows) and fatty steaks (blue arrows). (d) and (i) HC + nilotinib group, showing improvement in atherosclerotic lesions and less fat deposition. (e) I/M thickness ratio in aorta. (j) Mean area of aortic atherosclerotic lesions induced by the high fat diet. Data are expressed as mean ± SEM (n = 6). *p < 0.05 as compared to the control. # p < 0.05 as compared to the HC group (one-way ANOVA followed by Tukey–Kramer’s multiple comparison test). HC: high cholesterol; H&E: hematoxylin and eosin; I/M: intimal and media; SEM: standard error of mean; ANOVA: analysis of variance.
Effects on vascular reactivity
As shown in Figure 5, PE (10−9–10−5 M)-induced contractions in isolated rabbit aorta were not altered among different groups. On the other hand, ACh (10−9–10−5 M)-induced relaxations were clearly impaired in the HC group indicating marked endothelial dysfunction. Both E max and pD2 were significantly decreased in the HC group compared to the control group. However, nilotinib treatment restored ACh-induced relaxation and increased E max and pD2 to a level nearby the normal value. The endothelium-independent relaxation induced by SNP (10−9–10−5 M) was not significantly changed among different treated groups (Figure 5(b)).
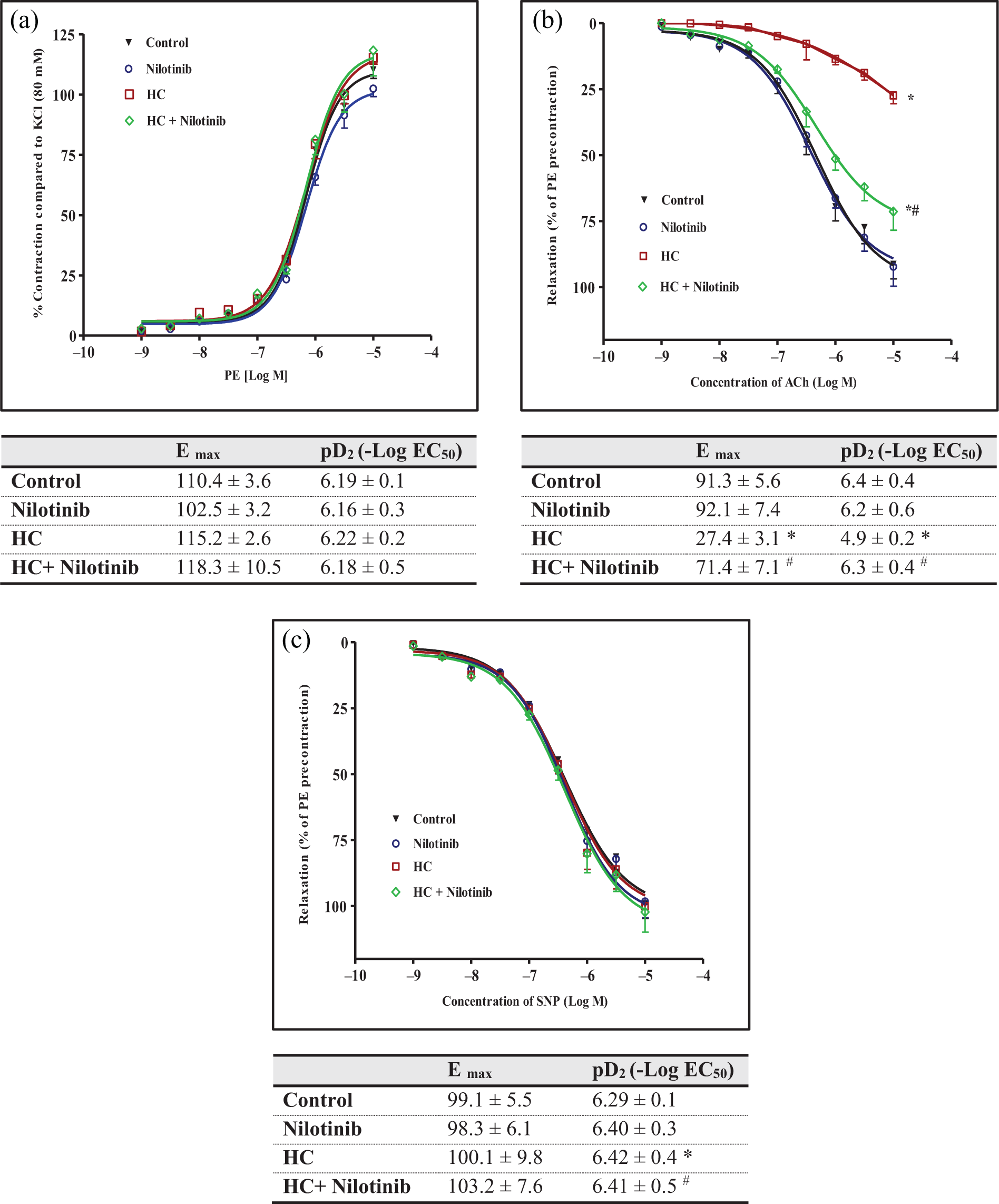
Effects of daily administration of nilotinib on HC-induced endothelial dysfunction. Data are the mean ± SEM (n = 6). Contraction to PE and pD2 values. Relaxation to ACh and pD2 values in PE (1 μM)-precontracted aortic rings of rabbits. Relaxation to SNP and pD2 values in PE (1 μM)-precontracted aortic rings of rabbits. *p < 0.05 as compared to the control. # p < 0.05 as compared to the HC group (ANOVA followed by Tukey–Kramer multiple comparison as a post hoc test). HC: high cholesterol; SEM: standard error of mean; PE: phenylephrine; ACh: acetylcholine; SNP: sodium nitroprusside; ANOVA: analysis of variance.
Effect on hepatic histology
As shown in Figure 6, both control and nilotinib groups exhibited hepatic lobules of normal structure and without fatty denaturalization. The HC group showed many lipid droplets vacuoles in hepatocytes which showed serious fatty denaturalization. There was a significant increase in the steatosis and necrosis scores compared to the control group. These pathological changes were remarkably attenuated in nilotinib + HC group.
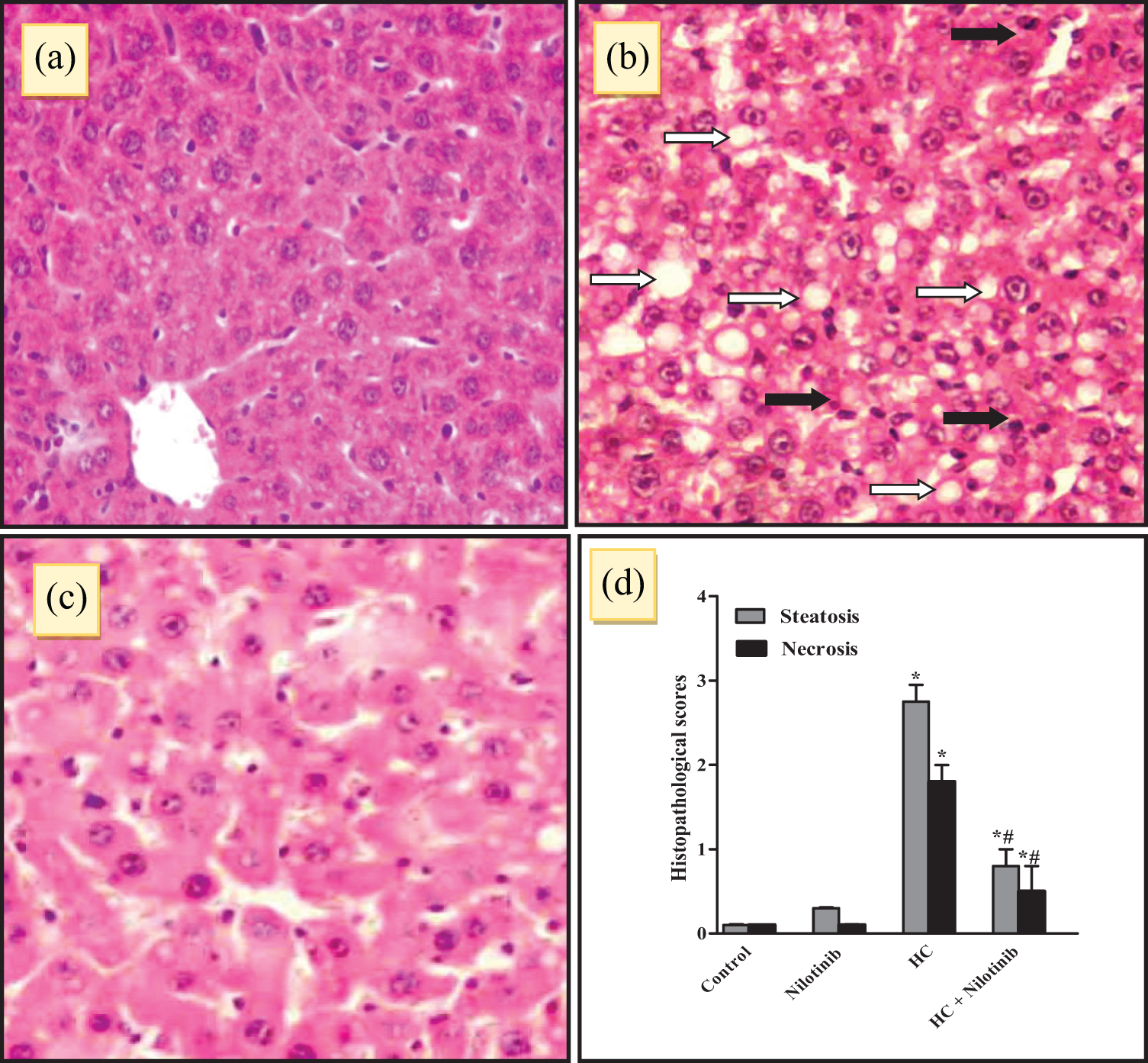
Effects of daily administration of nilotinib on HC-induced steatosis and necrosis of livers in rabbits. (a) Control group, show normal liver histology ×200. (b) HC group, marked micro and macrovesicular steatosis (white arrows) and minimal portal inflammation (black arrows) ×200. (c) HC + nilotinib group, significant attenuation of hepatic steatosis and inflammation ×200. (d) Steatosis and necrosis scores among different groups. Steatosis and necrosis were subjectively scored: 1+ (<25%), 2+ (26–50%), 3+ (51–75%), and 4+ (>75%). Data are the mean ± SEM (n = 6). *p < 0.05 as compared to the control. # p < 0.05 as compared to the HC group (nonparametric Mann–Whitney U-test, two-tailed). HC: high cholesterol; SEM: standard error of the mean.
Effect on IHC estimation for PDGFR-β
For aortic and hepatic tissues, IHC analysis revealed that PDGFR-β phosphorylation was significantly increased in the HC group compared to the control group. Daily administration of nilotinib significantly decreased the immunostaining compared to the HC group (Figure 7).

Effects of daily administration of nilotinib on IHC staining of liver and aorta in HC-fed rabbits. IHC staining of PDGFR-β in (a)–(d) liver and (e)–(h) aorta sections. (a) and (e) Control, (b) and (f) nilotinib, (c) and (g) HC, and (d) and (h) HC + nilotinib in HC-fed rabbits. Positive staining is shown in brown. Sections are counterstained with hematoxylin. Magnification ×200. (i) Semiquantitative analysis of PDGFR-β IHC staining results in liver and aortic tissues expressed as optical densities across 10 different fields for each TmA section. Data are the mean ± SEM (n = 6). *p < 0.05 as compared to the control. # p < 0.05 as compared to the HC group (ANOVA followed by Tukey–Kramer multiple comparison as a post hoc test). IHC: immunohistochemical; HC: high cholesterol; PDGFR-β: platelet-derived growth factor-β; OD: optical density; TmA: tissue miniarray; SEM: standard error of mean; ANOVA: analysis of variance.
Discussion
Hypercholesterolemia and atherosclerosis are known as the most risky inducers of ischemic cardiovascular disorders. This investigation has explored the potential beneficial role of nilotinib in attenuating atherosclerosis progression and its associated endothelial and hepatic damages.
In line with the previous investigations, results presented indicated that feeding a HC diet for 8 weeks caused hypercholesterolemia as indicated by the marked increase in serum levels of TC, TGs, LDL-C, and decrease in HDL-C level. 25,28 Additionally, HC induced a significant elevation of serum LDH level, which is a serum marker of cellular damage. As reported before, hypercholesterolemia normally progresses into atherosclerosis, which is mostly described by an aggregation of fatty streaks on the inner layer of arteries leading to endothelial dysfunction. 28 –30 This is in accordance with the presented histopathological results that showed distortion of the endothelial layer and heavily deposition of fats in the subendothelial layers in the HC group. Injured endothelial cells secrete adhesion molecules such as vascular cell adhesion molecule-1 and cell adhesion molecule-1 which recruit more macrophages and monocytes. These cells produce different pro-inflammatory mediators and induce migration and proliferation of smooth muscle cells from the media through the intimal endothelial layer. 31
As presented, the HC diet induced a significant increase in I/M ratio. Surprisingly, daily administration of nilotinib starting from week 4 till week 8 to rabbits fed HC diet significantly attenuated the elevated serum lipids and LDH with contaminant improvement in the aortic histopathological damages. This can lead us to suggest that nilotinib might antagonize the atherosclerosis development.
The endothelial dysfunction was further confirmed by the in vitro experiments. The isolated aortic segments from the HC group exhibited marked impairment of ACh-induced relaxation, which indicates an endothelial/NO dysfunction. On the contrary, endothelium-independent relaxations induced by the SNP were not significantly altered among different experimental groups. These results are in agreement with the previous studies. 25,32 Impaired endothelial function may be attributed to decrease NO bioavailability either due to decreased production from damaged endothelium or increased inactivation by ROS. 32 –34 However, daily administration of nilotinib resulted in restoration of the aortic vascular reactivity. Collectively, these results demonstrate that nilotinib may have the ability to counteract atherosclerosis progression and confer protection to the endothelial function.
Previous studies have demonstrated the important role of oxidative stress in atherosclerosis. 35,36 In subendothelial space of damaged artery, LDL is slowly oxidized through the action of resident vascular cells. This process plays a very important role in atherogenesis. Oxidized-low density lipoprotein (Ox-LDL) is taken up by macrophages that are accumulating in the endothelial wall as lipid-laden foam cells in the initial step of atherosclerotic fatty streak lesions. 1,25 In the present study, the oxidative stress marker, MDA, was significantly increased in the HC group. Previous studies have revealed that MDA level is increased in hypercholesterolemic state specifically with increased TGs and decreased HDL-C levels. This may be explained by overproduction of ROS in hypercholesterolemia. Nilotinib co-treatment counteracted the elevation of MDA indicating decrease in the oxidative burden. This characteristic effect of nilotinib has been reported before, and it may play an important role in the protective effect of nilotinib in the present context. 17,19 Additionally, the presented results showed a significant elevation in NOx level which is in accordance with the previous reports either in human or animals. 37,38 The increase in NOx level with ROS works together to enlarge the tissue damage and dysfunction. However, nilotinib treatment markedly attenuated the elevated NOx level which may contribute in part to the beneficial effect of nilotinib against HC-induced damages.
Many previous reports have focused on the close association between hypercholesterolemia and the development of hepatic steatosis/steatohepatitis. The presence of oxidizable fat within the liver has been reported to trigger lipid peroxidation. 9 As expected, 8 weeks of HC feeding to rabbits significantly increased liver transaminases and ALP levels indicating hepatocellular damages. Histopathological examination of liver specimen was in the same line of the biochemical results. It revealed marked steatosis, inflammation infiltrate, and steatohepatitis lesions. HC diet–induced hepatic injury and steatosis have been reported before in different animals. 39,40 Another important result of this investigation is the ability of nilotinib to significantly decrease the elevated transaminases and ALP levels as well as histopathological damage of liver indicating improvement of the HC-induced hepatic injury. Previously, nilotinib administration attenuated liver injury, fibrosis, and cirrhosis in other animal models. 19,41,42
PDGF is one of the main platelet-associated factors that plays a physiological role in the embryonic development of blood vessels, kidneys, and lungs. PDGF isoforms are expressed in multiple cells, including platelets, dysfunctional endothelium, and macrophage-derived foam cells. Overactivation of the PDGF system has a pivotal role in many inflammatory disorders such as different organ fibrosis and vascular wall proliferative diseases. During hypercholesterolemia, modified LDL uptake through a scavenger receptor–mediated pathway is a determinant step in atherosclerotic lesion development. Platelet proteins, including PDGF-BB, promote scavenger receptor activity in VSMCs and macrophages. 43 Furthermore, increased PDGFR-B signaling is involved in atherogenesis, hyperlipidemia, 44 –46 and in the steatohepatitis associated with a HC diet. 47 –49 Previous studies demonstrated that PDGF stimulates VSMC proliferation and migration leading to intimal thickening, which is a major pathological change in atherosclerotic lesions. 50 –54 Additionally, PDGF inhibitors can suppress the development of restenotic lesions in animal models. 55,56 Thus, it may be proposed that interfering PDGF pathway may interrupt atherosclerosis progression and hepatic injury during hyperlipidemia.
In accordance with the previous reports, results presented here have revealed the increased PDGF-B expression in aortic and hepatic tissues in HC-fed rabbits. On the other hand, daily administration of nilotinib decreased the expression of PDGFR-B. So, it is reasonable to presume that interfering with PDGF-B pathway is one of the underlying mechanisms of protective effects of nilotinib against HC-induced aortic and liver damage. The inhibitory effects of nilotinib on PDGF have been reported before in other model of experimental liver damage. 19,41 Previously, imatinib has been shown to have antiatherogenic effects in an experimental model of diabetes-associated atherosclerosis. 7 This effect was linked to imatinib ability to reduce PDGFR-B expression, which resulted in attenuation of prosclerotic and pro-inflammatory cytokine expression and prevention of cell proliferation in the plaque. 7,57 On the contrary, some previous studies have reported hypercholesterolemia in patients with CML receiving nilotinib but the mechanism is still unclear. 58 The discrepancy between experimental and clinical observations for nilotinib effects may be due to several factors. One of which is that clinical reports were performed on cancer patients who have different physiological and pathological conditions. However, this discrepancy warrants further investigations.
Collectively, the present study added new spectrum to nilotinib pharmacological activities. Nilotinib can attenuate the progression of atherosclerosis and reduce the endothelial dysfunction. In addition, it has hepatoprotective effects against HC-induced hepatic damage. The beneficial effects of nilotinib may be correlated with its ability to interfere with PDGF.
Footnotes
Acknowledgement
The author thanks the Deanship of Scientific Research, Taibah University, Al-Madinah Al-Munawwarah, Saudi Arabia, for providing assistance. The author acknowledges Dr Mohamed A Elkablawy, Department of Pathology, Faculty of Medicine, Taibah University, Al-Madinah Al-Munawwarah, Saudi Arabia, for performing the histopathological examination.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
