Abstract
In this study, possible thyrotoxicosis-related histological changes in testicular tissues of rats with experimentally induced thyrotoxicosis model were evaluated on cellular connections and stem cell markers. Two experimental groups, thyrotoxicosis and control, each consisting of eight animals were used. Rats in the thyrotoxicosis group were injected intraperitoneally with 3,3′,5-triiodo-
Introduction
The incidence of thyroid diseases is very high in the countries where iodine deficiency is endemic in the world. 1 Hyperthyroidism or overactive thyroid is a condition that secretes excessive amount of the free thyroid hormones such as triiodothyronine (T3) and thyroxine (T4). Hyperthyroidism is one cause of thyrotoxicosis which is the hypermetabolic clinical syndrome. Various signs and symptoms related to different organs and systems can be determined in thyrotoxicosis due to hyperthyroidism. 2 Testes are one of the main organs affected in this regard. 3 Sertoli and Leydig cells of the testes include thyroid hormone receptors and are directly or indirectly affected by thyroid hormone level changes. Clinical studies conducted in the last 20 years revealed that thyroid hormones play an important role in the testicular development and function. Thyroid hormone receptors are present in the human and rat testes from neonatal period to adult life, and thyroid hormones have an important role in testicular development. 4 Fluctuations in thyroid hormone levels during early testis development may affect testicular maturation and reproduction over the ensuing years. 5 The testes are comprised of loose connective tissue involving steroidogenic Leydig cells and seminiferous tubules. 6 The seminiferous tubules are composed of germ cells and somatic Sertoli cells. The number of Sertoli cells is closely correlated with testicular size and sperm output in adulthood, thus with male fertility. 7 T3 was shown to regulate testicular maturation and growth and control proliferation and differentiation of Sertoli and Leydig cells during testicular development in rats and other mammalian species. 3,8 Although several factors have a role in proliferation and maturation of Sertoli cells, T3 generates major hormonal signals for Sertoli cell proliferation during testis development and Sertoli cell population establishment in adulthood. 6 Thyroid hormones are also known to affect functions of Leydig cells. Several animal studies displayed Leydig cell differentiation changes in parallel to alterations in serum thyroid hormone levels. 8
Spermatogenesis originates from a population of testicular stem cells (TSCs). The existence of TSCs offers clinically relevant options for preservation and reestablishment of male fertility. 9 TSCs generate spermatogonia that are precursor cells of spermatozoa. After being derived from epiblast layer, TSCs migrate toward gonadal crista and undergo mitosis here and increase in number. 10 Reserve TSC capacity in testes tissue and its response to endocrine problems such as thyrotoxicosis may be important. In thyrotoxicosis, determining expressions of embryonic and nonembryonic stem cell markers, such as Nanog, c-kit, and Thy-1, is suggestive but rare in literature. 11,12
Embryonic germ cells from neonatal mammalian testes are pluripotent and have differentiation potential similar to embryonic stem cells. 13 In addition to pluripotency capacity, cell junction complexes are also important for testicular architecture. Tight junctions, also known as occluding junctions or zonula occludens, are the closely associated areas of two cells whose membranes join together forming a virtually impermeable barrier to fluid. Occludin is an integral plasma membrane protein located at the tight junctions. Together with the claudin group of proteins, it is the main component of the tight junctions. Connexins, gap junction proteins, are also a family of structurally related transmembrane proteins that assemble to form vertebrate gap junctions. Connexin 43, the main gap junction protein, is predominantly localized in the sarcolemma. Gap junctional communication is important in the development and regulation of cell growth. 14 Cell protection involves activation of endogenous signaling, which can confer significant resistance to oxidant and other stresses associated with hypoxia/reoxygenation, thus promoting enhanced capacity for cell survival. 15 Tight junction and gap junction provide the most important support for the functional and morphological integrity of the testes. Through these junctional units, chemical and electrical stimulation between neighboring cells and intercellular transfer of solutions, ions, and water are ensured. In addition, apical/basolateral differentiation and polarization, contribution to epithelial barrier, and blood–testis barrier, which is formed mainly by Sertoli cells, are realized via junctional complexes. In this study, to determine the effect of thyrotoxicosis to testis architecture, expressions of connexin 43, a gap-junctional protein and occludin, a tight junction protein, are examined.
The objectives of this study are to present pluripotency capacity of testicular cells and how they interact with each other and to demonstrate the role of cell junction complexes, which are significant for cellular microenvironment and cellular behaviors, and finally the role of thyrotoxicosis on these parameters.
Materials and methods
Animals and experimental design
The protocol was approved by the Animal Experiment Local Ethical Committee of Ege University (November 26, 2010, number 2010-153). All animals were weighed weekly throughout the study. All experiments were performed in accordance with ethical guidelines. Tests were conducted on sexually mature Wistar albino rats (250–280 g in weight) obtained from Breeding Center of Experimental Animals in Ege University. Animals were housed in cages (two animals per cage) with solid plastic sides and stainless steel grid tops and floors (19 × 19 × 12 cm3). They were maintained in controlled laboratory conditions of 12-h dark/12-h light cycle, 21 ± 1°C temperature, and 45–75% humidity. Animals were orally fed daily with a normal diet in standard laboratory chow. Tap water was also available ad libitum.
The study was conducted on 16 male rats. After 10 days of acclimatization, the rats were assigned randomly to either the thyrotoxicosis group or the control group, each containing eight male rats. Animals of the control group were administered with physiological saline, while the animals of the thyrotoxicosis group were administered with 3,3′,5-triiodo-
Biochemical analysis
Plasma and serum of the intracardiac blood samples were separated, stored frozen at −80°C until assayed. Thyroid function tests were performed with enzyme-linked immunosorbent assay (ELISA) method (rat thyroid stimulating hormone (TSH) ELISA kit, Cusabio Biotech Co. Ltd, China; rat free T3 (fT3) ELISA kit, Cusabio Biotech Co. Ltd), according to the manufacturer’s instructions.
Tissue expression analysis
The expression of connexin 43 (CNX43) and occludin (OCLDN) genes in testes tissues was evaluated using real-time polymerase chain reaction (RT-PCR). Total RNAs of testes tissues (100 mg) were isolated using the TriPure Isolation reagent, (Roche, Switzerland, cat. no. 11 667 157 001) following the manufacturer’s instructions. First-strand complementary DNAs (cDNAs) were synthesized with Transcriptor First Strand cDNA Synthesis kit (Roche, cat. no. 04 379 012 001) using total RNA. Specific primers were purchased (Table 1). A total of 0.3 μl of each primer (0.5 μM) was mixed with 10.5 ml of LightCycler FastStart DNA Master SYBR Green I kit (Roche, cat. no. 03 003 230 001) in a final volume of 18 μl. The standard cycling conditions were 95°C for 10 min, followed by 40 cycles of 95°C for 15 s, 62°C for 8 s, and 72°C for 13 s. The comparative C t method (2−▵▵Ct method) was used to determine the expression level of analyzed genes. 18 The expression of cell junctional genes was normalized using the glucose 6 phosphate dehydrogenase (G6PDH) gene as a control housekeeping gene. Data were analyzed using the Student’s t test. Results were expressed as the mean ± standard deviation of the three different samples, and differences were considered statistically significant at p < 0.05.
Primer sequences (forward and reverse) used in the RT-PCR.

RT-PCR; real-time polymerase chain reaction.
Histochemical and immunohistochemical analysis
For light microscopic examination, tissue specimens were fixed by 4% paraformaldehyde (Merck KgaA, Darmstadt, Germany) for 24 h, at 4 C and washed off with phosphate-buffered saline (PBS, pH 7.4). After dehydration, tissues were processed for embedding in paraffin wax using routine protocols. After routine processing, paraffin sections of each tissue were cut into 5 µm thickness and stained with hematoxylin and eosin (H&E, Merck KGaA, Darmstadt, Germany).
For immunohistochemical analysis, sections were then dewaxed and rehydrated through a graded ethanol series using routine protocols. 19 Sections were then washed with distilled water and PBS for 10 min, then treated with 2% trypsin (Sigma Chemical Co.) in 50 mM Tris buffer (pH 7.5), at 37°C, for 15 min, and washed with PBS, and hydrogen peroxide was applied. Primary antibodies Nanog (Abbiotec, LLC, San Diego, California, USA), Thy-1 (Santa Cruz Biotechnology, Santa Cruz, California, USA), c-kit (Santa Cruz Biotechnology), connexin 43 (ABM Inc., Richmond, British Columbia, Canada) and occludin (Abcam, Burlingame, California, USA) were used in 1/100 ratios and incubated for 24 h. After washing with PBS, firstly biotinylated secondary antibody (Histostain Plus, Invitrogen, Camarillo, California, USA) and then streptavidin-labeled secondary antibody (Histostain Plus Invitrogen) were applied. After washing with PBS, diaminobenzidine (Invitrogen) was used and then stained with Mayer’s hematoxylin (Sigma Chemical Co.). After washing with distilled water, slides were dehydrated in an ascending alcohol series and finally coverslipped with mounting medium. Morphometric analysis was carried out with Olympus BX-51 light microscope and Olympus C-5050 digital camera (Tokyo, Japan).
Statistical methods
Among testes slides stained with hematoxylin and eosin (H&E), 10 slides from each group were randomly chosen and examined under light microscopy (40× magnification) for Johnsen testicular biopsy scoring. One hundred tubules per each group were assessed, and score was calculated. 20 In addition to this, among other testes slides stained with H&E, 20 slides from each group were randomly chosen and examined under light microscopy (40× magnification) for seminiferous tubule diameter measurements. One hundred seminiferous tubule diameters per each group were measured and with Image-Pro Express 4.5 program (Media Cybernetics, Inc., Bethesda, Maryland, USA), and average scores were calculated. Scoring system for testicular biopsies according to histological criteria is 10 (full spermatogenesis), 9 (slightly impaired spermatogenesis, many late spermatids, disorganized epithelium), 8 (less than five spermatozoa per tubule, few late spermatids), 7 (no spermatozoa, no late spermatids, many early spermatids), 6 (no spermatozoa, no late spermatids, few early spermatids), 5 (no spermatozoa or spermatids, many spermatocytes), 4 (no spermatozoa or spermatids, few spermatocytes), 3 (spermatogonia only), 2 (no germinal cells, Sertoli cells only (Sertoli cell-only syndrome)), and 1 (no seminiferous epithelium). In this study, for statistical evaluation, the Student’s t test was used for comparisons between the groups. Statistically, p < 0.05 was considered significant.
Results
Animals and their body weights
During the experiment, no animal died. Weight comparisons of the animals displayed 20–45 g decrease in the animals treated with 3,3′,5-triiodo-
Biochemical analysis
Thyroid function test results of the animals are shown in Table 2. While TSH levels were decreased statistically significantly in testes tissues of tyrotoxicosis group in comparison with control animals, fT3 levels were increased statistically significantly in testes tissues of tyrotoxicosis group in comparison with control animals (p < 0.01).
Thyroid function test results of control and thyrotoxicosis groups.

TSH: thyroid-stimulating hormone; FT3: free triiodothyronine.
a p < 0.01: statistically significant difference from control.
Tissue expression analysis
After normalized with the G6PDH gene, DC t values of the messenger RNAs (mRNAs) of cell junctional genes, CNX43 and OCLDN, are showed in Table 3. The expression levels of CNX43 and OCLDN were increased in testes tissues of tyrotoxicosis group in comparison with control animals but not statistically significant.
Tissue expression analysis results of control and thyrotoxicosis groups.

Histological results
Examination of testes histological cross sections of control group that were stained routinely with H&E displayed regular boundaries of seminiferous tubules, regular lined germ cells in the seminiferous tubules, and normal histological appearance of all the cells of the spermatogenic series such as spermatogonia, spermatocytes, spermatids, and spermatozoa in Sertoli cells and also in the tubules of different stages (Figure 1(a) to (d)).

In histological sections of the testes of control group, seminiferous tubules (t), cells of spermatogenic cells series, spermatogonia (sg), spermatocytes (sp), spermatides (st), and spermatozoa (sz) with Sertoli cells (Se) and interstitium (I) and Leydig cells (L) show normal histological structures. Hematoxylin and eosin staining; magnification (a) ×10, (b) ×20, (c) ×40, and (d) ×100.
In testes, histological cross sections of tyrotoxicosis group, regular boundaries of seminiferous tubules with increased diameters, irregularities in cells of the spermatogenic series of the basal and adluminal compartments of seminiferous tubules, and intercellular separations in spermatogenic cells and Sertoli cells have been observed compare with the control group. Also spermatids are localized in basal compartment in many of the seminiferous tubules. Leydig cells, vascular structures, endothelial cells, and myoepithelial cells had normal histological structures, but the interstitium is thinned compared with the control group (Figure 2(a) to (d)).

In histological section of the testes of thyrotoxicosis group, seminiferous tubules (t) are seen enlarged, irregularities and cellular spreads are detected in spermatogonia (sg), spermatocytes (sp), spermatides (st), spermatozoa (sz), and Sertoli cells (Se) and interstitial thinning (I) in basal compartments of seminiferous tubules (*) due to reverse migration of spermatides (st) and spermatozoa (sz); hematoxylin and eosin staining; magnification (a) ×10, (b) ×20, (c) ×40, and (d) ×100.
According to the Johnsen testicular score, histological sections obtained from male rats in both control and thyrotoxicosis groups were evaluated, and the mean scores are shown in Table 4. In this study, only scores 8–10 were observed in the testes histological sections. The scores of thyrotoxicosis group were significantly different than those of the control (p < 0.01). Seminiferous tubule diameter measurements are shown in Table 5. In comparison with control animal, there was significantly higher tubule diameter in the testes of tyrotoxicosis group.
Johansen testicular biopsy scores of control and thyrotoxicosis groups.

a p < 0.01: statistically significant difference from control.
Testes lumen diameters of control and thyrotoxicosis groups.

a p < 0.05: statistically significant difference from control.
Immunohistochemical results
c-kit Immunoreactivity
On testes sections of the control group, c-kit expressions were very strong (+++) in spermatogonia and spermatocytes, while no immunoreactions (−) in spermatids and somatic Sertoli cells and interstitial Leydig cells (Figure 3(a)). On testes sections of the thyrotoxicosis group, weak immunoreactions (+) were detected for c-kit immunohistochemical staining of spermatogonia and spermatocytes when compared with the control (Figure 3(b)). The decrease in the number of c-kit positive cells was higher for spermatogonia than spermatocytes in the thyrotoxicosis group.

c-kit Immunoreactivity is seen significantly decreased in the thyrotoxicosis group (b) compared with the control group (a). Nanog immunoreactivity is not seen in the control (c) and thyrotoxicosis (d) groups. Thy-1 immunoreactivity is seen significantly decreased in thyrotoxicosis (f) group compared with the control (e) group.
Nanog immunoreactivity
Immunohistochemical staining for Nanog on testes sections of the control (Figure 3(c)) and the thyrotoxicosis (Figure 3(d)) groups showed no Nanog expression in the cells of the spermatogenic series such as spermatogonia, spermatocytes, spermatids, and in Sertoli and Leydig cells.
Thy-1 immunoreactivity
A small number of spermatogonium, spermatocytes, and spermatids showed weak immunoexpression (+) in Thy-1 immunohistochemical staining of testes tissue sections of control (Figure 3(e)), whereas Sertoli cells and Leydig cells displayed no immunoreactivity. In thyrotoxicosis group (Figure 3(f)), spermatogonia showed decreased staining for Thy-1, while no staining in spermatocytes and spermatids. In addition, compared with the control group, Thy-1 positive cells (spermatogonium) were decreased in number.
Connexin 43 immunoreactivity
In the analysis of testes tissue sections of the control (Figure 4(a)), spermatogonia, spermatocytes, and spermatids and additionally Sertoli cells showed strong immunoreactivity, while Leydig cells showed no immunoreactivity for connexin 43. In the thyrotoxicosis group (Figure 4(b)), spermatogonia, spermatocytes, and Sertoli cells showed a significant decrease in connexin 43 expressions with weak immunoreactivity (+), whereas spermatids showed no expression. Compared with the control, connexin 43 positive cells were significantly decreased in number.
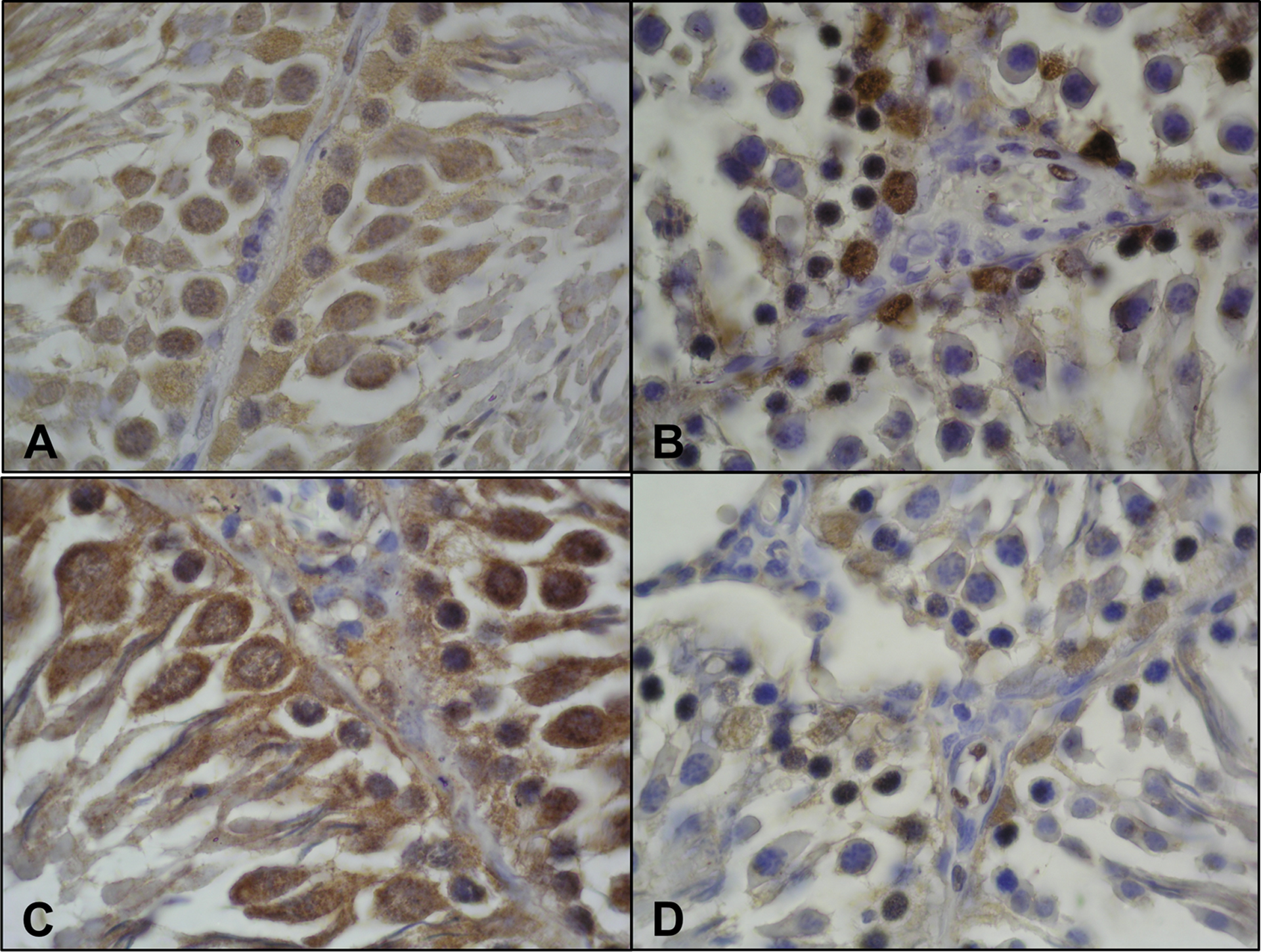
Connexin 43 immunoreactivity is seen significantly decreased in the thyrotoxicosis (b) group compared with the control (a) group. Occludin immunoreactivity is seen significantly decreased in the thyrotoxicosis (d) group compared with the control (c) group. Magnification ×100.
Occludin immunoreactivity
In the examination of testes tissue sections of control (Figure 4(c)), the cells of the spermatogenic series and Sertoli cells displayed significantly strong expression (+++) for occludin. A significant decrease in occludin expresions on spermatogonia and Sertoli cells was observed in testes sections of the thyrotoxicosis group (Figure 4(d)). Also significant decrease in the number of occludin positive cells was detected in addition to weak immunopositivity (+). However, spermatocytes and spermatids showed no immunoreactivity.
Discussion
Thyrotoxicosis is the clinical picture that occurs due to the elevation of thyroid hormones in the blood and may cause many organ disorders. Recent studies show that thyroid hormones have significant functions on the testis, in general, and Leydig cells, in particular. Some of these functions include triggering the Leydig stem cells to differentiate, producing increased numbers of Leydig cells during differentiation by causing proliferation of Leydig stem cells and progenitors, stimulation of the Leydig cell steroidogenic function, and cellular maintenance. 21 In this study, histological changes in testicular tissues of rats with experimentally induced thyrotoxicosis model were evaluated on cellular connections, embryonic, and nonembryonic stem cell markers. In the experimental thyrotoxicosis-induced testes of adult rats, decreases in the maturation of germ cells and increases in the germ cell apoptosis were reported previously. 22 Similarly, a decrease in the number of germ cells, loss in the spermatogenic cells and somatic Sertoli cells, and disorganization findings, such as increase the distance between the cells, were prominent in the histological examination of the testes in our study. Our findings also suggest that thyroid hormones may affect proliferation and function of Sertoli and Leydig cell as well as spermatogenesis and sperm motility. Starting from early postnatal period, TSCs are subject to dramatic functional and morphological changes throughout the entire process of spermatogenesis. 23 In two separate studies, c-kit was shown to act as a receptor for stem cell factor and as a marker to distinguish between types of spermatogonium. 24,25 In another study, it was determined that c-kit was expressed in late spermatocytes and round spermatids but not in spermatogonia and early spermatocytes. 26 In various available studies, truncated c-kit versions without extracellular and transmembrane domains are described in spermatids. 27 In our control group, in testes tissue sections, all spermatogenic cells showed strong positive immunoreaction against c-kit, except spermatids; on the other hand, Sertoli and Leydig cells did not exhibit positive staining. In thyrotoxicosis group, a significant decrease was detected in the number of c-kit positive cells and expressions for all spermatogenic cells except spermatids. Similar to control subjects, spermatids, Sertoli cells, and Leydig cells did not exhibit c-kit-positive staining. These findings indicated negative effects of thyrotoxicosis on the c-kit expression, which is a stem cell marker. Thy-1 is a surface marker for spermatogonial stem cells and precursors and still continues to be a unique marker for identification of spermatogonial stem cells in testes. 28,29 However, not only spermatogonia but all spermatogenic cells also showed weak Thy-1 immunoexprsessions in our control group. In our thyrotoxicosis group, spermatogonia showed similar weak immunostaining, while spermatocytes and spermatides exhibited no positive staining, which eventually indicated that thyrotoxicosis affected Thy-1 immunoexpression. Nanog is expressed in pluripotent embryonic stem and embryonic germ cells. 30,31 In vivo expression of Nanog is restricted to pluripotent cells of the inner cell mass, the epiblast, and primordial germ cells. 32 Despite the evidence for the expression of Nanog in the testes of several species and clear relationship between testicular function and pluripotency, little is known about the role of Nanog in the process of spermatogenesis. 22 It was stated that Nanog expression was not observed in mouse testes, although detected in inner cell mass, epiblast, and primordial germ cells. 30 Nanog was shown in human fetal gonocytes, testicular carcinoma in situ, and germ cell tumors, but not in normal adult testes. 11 Similarly, Nanog expression was detected in fetal gonocytes of marmoset, but not in adult testes. 33 Kuijk et al. demonstrated Nanog transcripts and protein in mouse, dog, pig, and human testes. 34 Expression was detected particularly in type A spermatogonia, spermatocytes, and spermatids. In a study of Conrad et al., spermatogonial stem cells obtained from adult human testes were isolated and cultured, and Nanog expression could not be determined. 35 Similar to the literature data, we could not detect Nanog expression in both the control group and the thyrotoxicosis group. The epithelium of the seminiferous tubules exhibit three intercellular junctions,namely cadherin-based adherence junction, occludin-based tight junctions, and connexin-based gap junctions. 36
Clear evidence suggested that protein components of gap junctions, tight junctions, and adherence junctions interact with each other and thus can control expressions and functions of each other. 37 Intercellular protein components by causing the formation and/or deterioration of testicular cell connections play a key role in spermatogenesis. Gap juntions in the testes and the most abundant gap junctional protein connexin 43 were suggested to play a crucial role in the development and maturation of spermatozoa. Although in vivo and in vitro comprehensive studies suggested connexin 43 may be critical for spermatogenesis and Sertoli cell function, the exact role of connexin 43 in Sertoli cells and seminiferous epithelium could not be determined. Among testicular connexin, only connexin 43 is predominant for spermatogenesis. 38 Connexin 43 deletion may lead to changes in the formation of blood–testes barrier and disintegration in Sertoli cell junctional complexes. Functions and numbers of gap junctions are correlated with thyroid hormone levels and thyroid functions. However, whether changes in thyroid hormones may change the expression of connexin is still not clarified. In the testes, blockage of connexin functions as well as decreased occludin and increased N-cadherin expressions were demonstrated by using panconnexin peptide, which can recognize all testicular connexins. Although there was no significant change in expression levels of CNX43 and OCLDN mRNAs in testes of tyrotoxicosis group in comparison with control animal, connexin 43 immunohistochemical staining of adult testes sections displayed a significant decrease in the cells of spermatogenic series and somatic Sertoli cells compared to the control group. A reduction in connexin 43 expression may explain intercellular spreading in seminiferous tubules, cellular loss, and intratubular derangements as well as reverse migration of spermatids and spermatozoa toward basal tubular compartment. These results may explain the key interaction between connexin 43 and other junctional complexes and spermatogenesis reported in the literature. Occludin is an integral membrane protein and mediates epithelial barrier functions depending on the tissue, and the epithelium of the seminiferous tubules is one of these tissues. 39 Occludin is also expressed in testicular vascular endothelium and retetestes epithelium of mice. Altay et al. reported occludin expression in germline cells of adult rat testes. 40 In our control group, we detected strong occludin expression in spermatogenic cells and somatic Sertoli cells, while a significant reduction in occludin expression was found in the spermatogonia and Sertoli cells in the thyrotoxicosis group.
Our study demonstrates that thyrotoxicosis may cause microenvironmental decay in addition to parenchymal organ damage in testes. Another striking outcome of our study is that thyrotoxicosis may have adverse effects on the cell junctional complexes, cell–cell interactions, and pluripotency capacity.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
The study is supported by Ege University, Faculty of Medicine (project number is 2011/TIP/042).
