Abstract
Mitomycin C (MMC) is one of the most effective chemotherapeutic drugs. However, the dose of MMC is greatly limited by its toxicity in normal tissues. Recombinant human erythropoietin (rhEPO), an erythropoietic hormone, has also been shown to exert tissue protective effects. The purpose of this study was to explore the protective effect of rhEPO against MMC-induced heart, liver, and renal dysfunction. Adult male Wistar rats were divided into six groups (with six animals each), namely control, rhEPO alone group, MMC alone group, and rhEPO + MMC group (pre-, co-, and posttreatment conditions). The results showed that MMC induced a marked cardiac, renal, and liver failure characterized by a significant decrease in body weight, organs weight, and organs ratio and a significant increase in creatinine, blood urea nitrogen, alanine aminotransferase, aspartate aminotransferase, γ-glutamyl transferase, and conjugated and total bilirubin levels in serum. Histological examination showed that MMC caused liver alterations. rhEPO treatment restored body weight, organs weight, and organs ratio as well as serum biochemical parameters and histological damage caused by MMC exposure.
Keywords
Introduction
Mitomycin C (MMC), an antitumor antibiotic isolated from Streptomyces caespitosus, is currently one of the most important cytostatic agents in treatment of a wide range of solid tumors. However, the clinical usefulness of this drug is restricted due to some adverse side effects, such as cardiotoxicity, nephrotoxicity, and hepatotoxicity. It has been shown that MMC may cause cardiomyopathy, 1,2 which seems to be related to the cumulative dose of the drug. 1 –3 The most serious cardiac failure of MMC therapy is congestive heart failure (CHF), the clinical presentation of which was similar to CHF of other etiologies. 2 Moreover, several data showed that MMC induced glomerular and tubular renal damage as a part of the hemolytic uremic syndrome. 4 Hepatotoxicity caused by MMC has been rarely characterized and is less studied. However, it is known that MMC is significantly taken up and metabolized in human liver, and at cumulative dose of the drug hepatotoxicity can occur. 5 Until now, a large number of studies have been focused on the ways for prevention of MMC side effects. Nevertheless, the use of recombinant human erythropoietin (rhEPO) has not been well underlined.
EPO, which is clinically used as rhEPO, is a cytokine originally used for its effects on erythropoiesis. Recently, many studies have shown that the biological effects of rhEPO are not limited to the hematopoietic system. In fact, rhEPO was found to be a pleiotropic cytokine that exerts broad tissue-protective effects in diverse nonhematopoietic organs. 6 rhEPO has been found to protect the brain and the spinal cord from ischemic injury, the peripheral nerve from diabetic damage, and the heart from acute ischemic/reperfusion injury. 7 –10 Given that rhEPO receptors are expressed on cardiac cells, renal tubular epithelial cells, and hepatic cells, 11 –13 it is possible that the systemic administration of rhEPO may also provide protection against acute heart, renal, and liver damages caused by MMC.
In this study, we looked for the protective effect of rhEPO on MMC-induced nephrotoxicity and hepatotoxicity in Wistar rats. Using histological and biochemical analysis, we investigated whether rhEPO avoids MMC-induced kidney and liver failure when it was administered simultaneously, before or after MMC administration.
Materials and methods
Chemicals
MMC (cis-diamminedichloroplatinum II) was purchased from Sigma–Aldrich (France). Commercially available preparation of rhEPO (Hemax®, Biosidus S.A., Argentina) was used.
Animal treatments
Experiments were performed on male Wistar rats weighing approximately 120–140 g; the rats were kept at controlled environmental conditions at room temperature 22 ± 2°C and 12-h light/12-h dark cycles and allowed free access to food and water but fasted overnight before treatment. The experimental procedures were carried out according to the American College of Toxicology Statement on the Use of Animals in Toxicology and approved by the local ethics committee. For the time course experiment, rats were divided at random into six groups, with six animals in each group. All injections were administered by intraperitoneal (i.p.) route. The control group received a single injection of saline solution at 7.5 mg/kg body weight (b.w.) In each type of treatment, rhEPO and MMC were used at 3000 IU/kg b.w. and 3 mg/kg b.w, respectively. The rhEPO group received only rhEPO, and the MMC group received only a single injection of MMC. To test the effect of rhEPO on MMC-induced cardiotoxicity, nephrotoxicity, and hepatotoxicity, three treatment conditions (co-, pre-, and posttreatment) were tested. In co-treatment group, a single dose of rhEPO was administered simultaneously with MMC. In pretreatment group, a single dose of rhEPO was injected 1 day before MMC treatment. In posttreatment group, a single dose of rhEPO was injected 5 days after MMC exposure. Table 1 presents the detailed experimental design.
Animal groups and treatments in the experimental design of this study.a

rhEPO: recombinant human erythropoietin; MMC: mitomycin C; b.w.: body weight.
aRats were exposed to MMC and rhEPO in different treatment conditions, namely MMC alone, rhEPO alone, and rhEPO with MMC (co-, pre-, and posttreatment conditions.).
After killing the animals, blood samples were collected for measurement of serum creatinine, blood urea nitrogen (BUN), alanine aminotransferase (ALT), aspartate aminotransferase (AST), γ-glutamyl transferase (GGT), alkaline phosphatase (ALP), conjugated bilirubin (DBIL), and total bilirubin (TBIL) analysis. Then, the animals were euthanized, and kidneys and liver were immediately removed and weighed for histological examinations.
Animal weight
Animal weight was measured and evaluated between the first day of MMC/rhEPO administration and the final day of treatment.
Kidney and liver ratio
Heart, kidney, and liver were removed and weighed immediately. Heart, kidney, and liver ratios were calculated using following formula:
Serum biochemical analysis
Serum creatinine concentration was measured by the picric acid colorimetric method. 14 BUN was measured using colorimetric assay kit according to the manufacturer’s instructions (Stanbio Laboratory, Boerne, Texas, USA). The activity of ALT, AST, GGT, and ALP enzymes in serum and concentration of DBIL and TBIL in serum were determined with the biochemical parameter counter (CX99 Pro analyzer, Beckman Coulter Inc., Brea, California, USA).
Histopathological examination in heart, kidney, and liver tissues
Histological evaluation of the heart, kidney, and liver was performed on separate groups of rats. Briefly, heart, kidney, and liver were removed from rats at the end of the experimental period and were cut in a sagittal section into two halves, which were fixed in immersion in 10% formaldehyde at room temperature for 24 h. After dehydration using graded ethanol, pieces of organs were embedded in paraffin. The sections were stained with hematoxylin and eosin. For histopathological examination in heart, kidney, and liver tissues, we examined, for each organ, 36 slides per group (6 slides per animal). Light microscopy was used to evaluate different organ lesions and was graded as follows: Mild (+): only single vacuoles in myocardial and renal tubules and slight lymphocytic inflammatory infiltrate periportal and intralobular. Moderate (++): moderate vacuoles in myocardial cells and renal tubules and moderate lymphocytic inflammatory infiltrate in liver tissue. Severe (+++): extensive vacuoles in myocardial cells and in renal tubules and marked lymphocytic inflammatory infiltrate.
Statistical analysis
Data are expressed as means ± SD Statistical comparison between different groups were carried out using one-way analysis of variance, followed by Tukey’s post hoc test, to detect the difference between various groups. Value of p < 0.05 was considered to be significant: * indicates significant difference from control; # indicates significant difference from MMC-treated rats; a indicates significant difference from co-treatment group; and b indicate significant difference from posttreatment group.
Results
Effect of rhEPO on MMC-induced body weight loss and changes in organs weights and organs ratio
The results showed that final body weight in MMC-treated group decreased significantly when compared with the control group (Table 2). Thus, animal weight loss passed from 8.2 ± 1.07 g in the control group to 35.4 ± 5.28 g in the MMC group. rhEPO + MMC treatments (co-, pre-, and posttreatment), especially in pretreatment condition, restored body weight loss as compared to the MMC-treated group (Table 2). The animal weight loss passed from 35.4 ± 5.28 g in the MMC group to 12.6 ± 1.56 g in the pretreatment group.
Effects of rhEPO on MMC-induced body weight loss, organ weight, and organ ratio in Wistar rats.

rhEPO: recombinant human erythropoietin; MMC: mitomycin C; b.w.: body weight.
Values are expressed as means ± SD. Superscript characters indicate a significant difference at p ≤ 0.05.
In the second set of experiments, we examined the effects of MMC (3 mg/kg b.w. i.p.) on heart, kidney, and liver weights (Table 2). Our results clearly demonstrated that organ (heart, kidney, and liver) weights decreased significantly as compared to the control group. Thus, heart weight passed from 1.93 ± 0.24 g to 1.06 ± 0.18 g, kidney weight passed from 1.13 ± 0.13 g to 0.62 ± 0.17 g and liver weight passed from 4.35 ± 0.47 g to 3.32 ± 0.41 g in the control and the MMC group, respectively. This decrease in organ weights was significantly ameliorated with rhEPO administration especially in the pretreatment condition. Kidney weight passed from 0.62 ± 0.17 g in the MMC group to 0.95 ± 0.14 g in the pretreatment group. Furthermore, MMC exposure induced significant change in organ ratio (heart, liver, and kidney) as compared to the control group (Table 2).
Effect of rhEPO on MMC induced kidney dysfunction
As shown in Figure 1, MMC treatment caused a marked elevation of serum BUN and creatinine concentration in comparison with the control group (85.17 ± 10.19 mmol/l vs. 5.4 ± 2.07 mmol/l and 161.33 ± 23.69 μmol/l vs. 24.33 ± 3.06 μmol/l, respectively). rhEPO treatments (co-, pre-, and posttreatment) resulted in a significant reduction in BUN and serum creatinine levels. The results showed that rhEPO administration 24 h before MMC promoted the best protection against MMC nephrotoxicity, that is, 21.6 ± 2.03 versus 85.17 ± 10.19 and 32 ± 9 versus 161.33 ± 23.69 in BUN and serum creatinine levels, respectively.

rhEPO ameliorates renal functional impairment following MMC injury. Values are expressed as means ± S.D. Superscript characters indicate a significant difference at p ≤ 0.05.
Effect of rhEPO on MMC induced liver dysfunction
The results of serum biochemical analysis revealed that treatment with a single dose of MMC increased significantly (p < 0.005) serum levels of ALT, AST, DBIL, TBIL, ALP, and GGT, indicating liver toxicity, as compared to the control group (Figures 2(a) and (b), 3(a) and (b), 4, and 5, respectively). For example, serum AST level passed from 130 ± 8.72 IU/l in the control group to 221.67 ± 15.37 IU/l in the MMC-treated group. Simultaneous administration of rhEPO 24 h before or 5 days after MMC exposure ameliorated the biochemical parameters as compared to the MMC group. AST serum level passed from 221.67 ± 15.37 IU/l in MMC-treated group to 154 ± 9.17, 117 ± 9.17, and 140 ± 9.85 IU/l in co-, pre-, and posttreatment conditions, respectively. The result showed clearly that rhEPO administration in the pretreatment condition promoted the best protective effect against MMC-induced liver toxicity.

rhEPO ameliorates (a) serum ALT and (b) AST level concentrations following MMC exposure. Values are expressed as means ± SD. Superscript characters indicate a significant difference at p ≤ 0.05. rhEPO: recombinant human erythropoietin; MMC: mitomycin C; AST: aspartate aminotransferase; ALT: alanine aminotransferase.

rhEPO changes serum (a) DBIL and (b) TBIL level concentrations following MMC treatment. Values are expressed as means ± SD. Superscript characters indicate a significant difference at p ≤ 0.05. rhEPO: recombinant human erythropoietin; MMC: mitomycin C; DBIL: conjugated bilirubin; TBIL: total bilirubin.
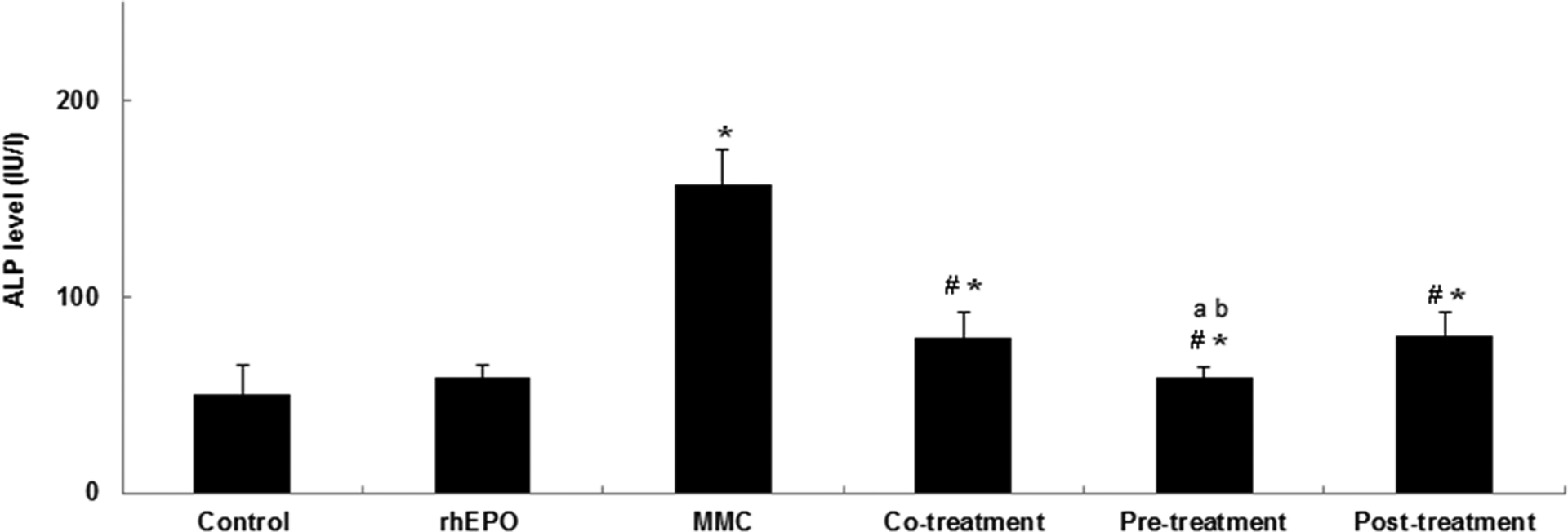
rhEPO ameliorates serum ALP level following MMC injury. Values are expressed as means ± SD. Superscript characters indicate a significant difference at p ≤ 0.05. rhEPO: recombinant human erythropoietin; MMC: mitomycin C; ALP: alkaline phosphatase.
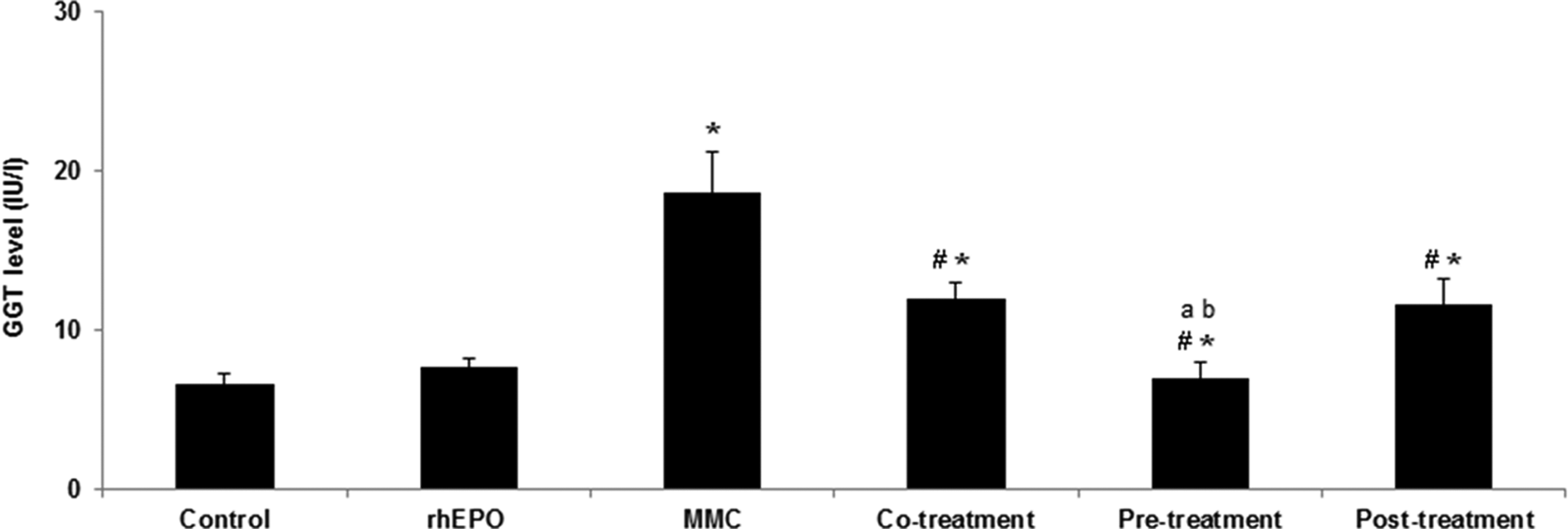
Effect of rhEPO on MMC induced change in GGT level. Values are expressed as means ± SD. Superscript characters indicate a significant difference at p ≤ 0.05. rhEPO: recombinant human erythropoietin; MMC: mitomycin C; GGT: γ-glutamyl transferase.
Effect of rhEPO on histological changes caused by MMC in heart, kidney, and liver
The histological changes in kidneys, heart, and liver were evaluated as described in ‘Materials and Methods’ section. Results are presented in Table 3. The histopathology of heart is illustrated in Figure 6. The results showed the presence of extensive vacuoles in myocardial cells of MMC-treated heart (+++; Figure 6(c)). Treatment with rhEPO (co-, pre-, and posttreatment) decreased the MMC-induced myocardial cell damage (+; Figure 6(d) to (f)). The histopathology of kidney is illustrated in Figure 7. The results showed the presence of marked number of vacuoles in renal tubules of MMC-treated kidneys (+++; Figure 7(c)). MMC + rhEPO administrations (co-, pre-, and posttreatment) improved the histological appearance and reduced the tubular cell damage caused by MMC exposure (+; Figure 7(d) to (f)). Similarly, histopathology of liver demonstrated that MMC administration (Figure 8(c)) caused an extensive histological damage in liver as compared to the control group (+++). In liver tissue, a lymphocytic inflammatory infiltrate in periportal and intralobular zones was observed. rhEPO administration in any treatment conditions decreased liver failure caused by MMC treatment (+; Figure 8(d) to (f)).
Semi-quantification of histological damages in rat heart, kidney, and liver tissues.

rhEPO: recombinant human erythropoietin; MMC: mitomycin C; b.w.: body weight; +++: severe; +: mild.

Histopathological changes in heart after rhEPO and MMC treatment conditions. Transverse section of control rat heart (a), rhEPO-treated heart (b), MMC-treated heart (c), co-treatment condition (d), pretreatment condition (e), and posttreatment condition (f). Arrow indicates vacuoles in myocardial cells. rhEPO: recombinant human erythropoietin; MMC: mitomycin C.

Histopathological changes in kidney after rhEPO and MMC treatment conditions. Transverse section of control rat kidney (a), rhEPO-treated kidney (b), MMC-treated kidney (c), co-treatment condition (d), pretreatment condition (e), and posttreatment condition (f). Arrow indicates vacuoles in tubular cells. rhEPO: recombinant human erythropoietin; MMC: mitomycin C.

Histopathological changes in liver of rat after rhEPO and MMC treatment conditions. Transverse section of control rat liver (a), rhEPO-treated liver (b), MMC-treated liver (c), co-treatment condition (d), pretreatment condition (e), and posttreatment condition (f). Arrow indicates lymphocytic inflammatory infiltrate in periportal and intralobular zones. rhEPO: recombinant human erythropoietin; MMC: mitomycin C.
Discussion
MMC is one of the most potent chemotherapeutic anticancer drugs for the treatment of various cancers. 15 In spite of its significant anticancer activity, the clinical use of MMC was limited due to several side effects including cardiotoxicity, nephrotoxicity, and hepatoxicity. 2,5,16,17 It has been shown that this drug has quinone and aziridine moieties, and tumor cell cytotoxicity has been attributed to DNA alkylation and cross-linking. However, Silva et al. 18 suggested that MMC caused oxidative stress in nondividing cells by different mechanisms.
Many efforts have been made to improve the therapeutic index of MMC using pharmacological strategies, such as the administration of melatonin, curcumin, or flavonoid compound like quercitin. 18 –20 rhEPO, apart its hematopoietic role, has been shown to prevent several injuries. 21,22 In fact, it has been demonstrated that rhEPO receptors are expressed in many tissues, including heart, kidney, and liver. Several studies demonstrated that rhEPO administration decreased tissue damage by inhibition of apoptosis, reduction of inflammatory cytokines, maintenance of vascular flow through, increase of nitric oxide synthesis, and decrease of lipid peroxidation. 23,24 In this study, we demonstrated that rhEPO has a preventive effect against MMC-induced heart, kidneys, and liver injuries in rats.
Heart failure induced by MMC exposure was evaluated by weight loss, heart weight loss, and heart ratio. Moreover, histological examination of heart tissue treated by MMC alone showed significant myocardial cells vacuolization in the MMC-treated group. rhEPO administration especially in pretreatment condition ameliorated cardiotoxicity caused by MMC. Our results were in agreement with those of Shan et al. 25 who demonstrated that rhEPO ameliorates cardiotoxicity in ischemia/reperfusion injury.
Nephrotoxicity caused by MMC in this study was gauged by body weight loss, kidney weight loss, kidney ratio, and biochemical parameter changes (creatinine and BUN in serum). Furthermore, the results clearly demonstrated that the administration of MMC to rats caused an extensive vacuolization in renal tubules. These results are in agreement with Wahid et al. 26 and Verweij et al. 27 showing that a single dose of MMC induced severe renal failure in rats characterized by an enhanced serum BUN and creatinine levels and obvious renal tissue damage. Groups treated by rhEPO, either simultaneously, before or after MMC administration, showed a lower level of serum BUN and creatinine than group receiving the MMC alone. Furthermore, our findings demonstrated that the pretreatment condition was the most efficient condition in preventing renal dysfunction caused by MMC. The histological examination of the kidney showed that rhEPO administration in co-, pre-, and posttreatment condition improved renal lesions significantly as compared to MMC-treated group. This is in accord with Esposito et al. 28 who demonstrated that rhEPO preconditioning is effective in protecting the kidney against ischemia/reperfusion injury by reducing both tubular cell injury and interstitial infiltration.
The measure of liver weight demonstrated that MMC caused a loss in liver weight as compared to the control group, and an increase in serum levels of ALT, ALT, GGT, ALP, DBIL, and TBIL indicates the hepatotoxicity. Histological examination of liver showed that MMC exposure caused significant lymphocytic inflammatory infiltrate in periportal and intralobular zone as compared to the control group. rhEPO administration, especially in the pretreatment condition, restored the loss of liver weight and decreased significantly the level of biochemical parameters changed by MMC. Moreover, rhEPO treatment (co-, pre-, and posttreatment), especially, administrated 24 h before MMC exposure protects liver against histological damages caused by the anticancer drug.
In conclusion, this study provided evidence that in addition to its well-known erythropoietic effects, rhEPO administration in co-, pre-, and posttreatment conditions protected against MMC-induced heart, kidneys, and liver toxicity in rats. Furthermore, we demonstrated that the protective action of rhEPO was more pronounced when it was administrated to rats 24 h before MMC exposure.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research was supported by the “Ministère Tunisien de l’Enseignement Supérieur et de la Recherche Scientifique et de la Technologie (Laboratoire de Recherche sur les Substances Biologiquement Compatibles (LRSBC)).”
