Abstract
In this experimental design, we explored the neuroprotective potential of zingiberene (ZGB), a monocyclic sesquiterpene, in hydrogen peroxide (H2O2)-induced toxicity in newborn rat cerebral cortex cell cultures for the first time. The rats were exposed to H2O2 for 6 h to determine the oxidative stress levels. To evaluate cell viability, both 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyltetrazolium bromide and lactate dehydrogenase assays were carried out. Total antioxidant capacity (TAC) and total oxidative stress (TOS) parameters were used to evaluate oxidative changes. Besides determining 8-hydroxy-2-deoxyguanosine (8-OH-dG) levels in vitro, single-cell gel electrophoresis was also performed to measure the resistance of neuronal DNA to H2O2- exposed rats. Our results showed that survival and TAC levels of the cells decreased, while TOS, 8-OH-dG levels and the mean values of the total scores of cells showing DNA damage increased in the H2O2 alone-treated cultures. But pretreatment of ZGB suppressed the cytotoxicity, genotoxicity and oxidative stress that were increased by H2O2. Based on these observations, it is suggested that the sesquiterpene ZGB can be used as a novel and natural potential therapeutic in counteracting oxidative damages in the field of neurodegenerative disorders.
Introduction
Reactive oxygen species (ROS) and reactive nitrogen species (RNS) such as singlet oxygen, superoxide, peroxyl radicals, hydroxyl radicals, hydrogen peroxide (H2O2) and nitric oxide are produced normally in living tissues at relatively low steady state levels. In these levels, ROS and RNS may actually play important roles for survival of most living organisms including intracellular signal transduction and gene expression. However, their uncontrolled and excessive increases in the steady state concentrations and exogenous exposure cause free radical-mediated chain reactions that indiscriminately target proteins, lipids, polysaccharides, RNA and DNA. 1–3 Under extreme oxidative conditions, or if the antioxidant protective mechanisms of cells are compromised, cellular injury and death may occur. 4,5 In fact, oxidative stress has been implicated under various pathological conditions involving cardiovascular disease, cancer, neurological disorders, diabetes, ischaemia/reperfusion, other diseases and ageing. 6–8
Central nervous system cells were considered to be more vulnerable to ROS and RNS toxicity due to their inherent higher oxidative metabolism and less antioxidant enzymes such as catalase and superoxide dismutase as well as higher content of membranous fatty acids. 9,10 Accumulated evidence showed that the most common neurodegenerative disorders are Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, multiple sclerosis, stroke and brain trauma in which elevated levels of ROS might induce severe cell damage through oxidative stress. 11,12 Consequently, due to the efficacy of antioxidant-natured compounds in preventing oxidative damage in either cultured neuronal cells or in the brains of animals treated with various neurotoxic agents, it was suggested that antioxidants have important potential therapeutic value as neuroprotective drugs in treatment of many neurodegenerative disorders. 13–20
Zingiberene (ZGB) is a monocyclic sesquiterpene which is the predominant constituent of ginger with oil content (Zingiber officinale) from which it gets its name (Figure 1). It can contribute up to 30% of the essential oils in ginger rhizomes. 21 The limited numbers of recent investigations have revealed that ZGB exhibits many important biological activities, including antioxidant activity, 22 anticarcinogenic activity, 23 antiulcer activity, 24 antibacterial activity 25 and antiviral activity. 26 However, whether ZGB exerts protective effects against oxidative cytotoxicity in neuronal models as a result of its antioxidant property has not been investigated. Therefore, the aim of the present study was to firstly evaluate the cytotoxic (3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and LDH assays), cytogenetic (comet and 8-hydroxy-2-deoxyguanosine (8-OH-dG) assays) and oxidative effects (total antioxidant capacity (TAC) and total oxidative stress (TOS) analyses) of ZGB against H2O2-induced neuronal damage using newborn rat cerebral cortex cell cultures to explore their neuroprotective potentials.

Chemical structure of ZGB. ZGB: zingiberene.
Materials and methods
Chemicals and reagents
ZGB (CAS no. 495-60-3, C15H24, purity: ≥95%) was purchased from Guide Chem® (China). Dulbecco’s modified Eagle’s medium, Hank’s balanced salt solution (HBSS), neurobasal medium (NBM), sodium phosphate, potassium phosphate monobasic, ethylenediaminetetraacetic acid (EDTA), phosphate-buffered solution (PBS), dimethyl sulphoxide (DMSO), Triton-X-100, DNase type 1, Tris, low-melting point agarose, normal melting point agarose and ethidium bromide were purchased from Sigma-Aldrich® (Steinheim, Germany). H2O2 was purchased from Merck® (Germany). Fetal calf serum (FCS) and trypsin-EDTA were purchased from Biol Ind® Inc. (Wheeling, West Virginia, USA). All other chemicals were of analytical grade.
Cell culture
Primary rat cerebral cortex neuron cultures were prepared using rat foetuses as described previously.
27
Briefly, a total of nine newborn Sprague Dawley rats were used in the study. The rats were decapitated by making a cervical fracture in the cervical midline, and the cerebral cortex was dissected and removed. The cerebral cortex was placed into 5 ml of HBSS, which had already been placed in a sterile Petri dish, and macromerotomy was performed with two lancets. This composition was pulled into a syringe and treated at 37°C for 25–30 min as 5 ml HBSS + 2 ml trypsin-EDTA (0.25% trypsin-0.02% EDTA) and chemical decomposition was achieved. To this solution, 8 µl of DNase type 1 (120 U/ml) was added, treated for 1–2 min and centrifuged at 800 r/min for 3 min. After having thrown away the supernatant, 31.5 ml of NBM and 3.5 ml FCS were added to the residue. The single cell that was obtained after physical and chemical decompositions was divided into 3.5 ml samples in each of 10 flasks coated with poly-
Treatments
Cytoprotective activities of ZGB on 0.5 mM H2O2-induced cell injury were investigated by MTT, LDH, TAC, TOS and comet assays and 8-OH-dG analysis. For determining cytoprotectivity, the cells were seeded into a 48-well plate at a density of 5 × 104 cells/well for 16 h and then exposed to medium in the presence of different concentrations of ZGBs for 0.5 h before exposure to 0.5 mM H2O2 for 6 h. The cytotoxicity and genotoxicity of ZGB were also investigated. ZGB was dissolved in ethanol, and ethanol was evaporated to dryness at ambient temperature. ZGB was applied into cultures at concentrations of 6.25, 12.5, 25, 50 and 100 µg/ml for 24 h. The doses were selected according to the works of Togar 23 and Si et al. 12 Cells incubated without ZGB and H2O2 were considered as control group. The cell viability, oxidative alterations and DNA damage analyses were carried out in four totally independent experiments
Evaluation of cell viability
MTT assay
Viability of cells was assessed by measuring the formation of a formazan from MTT spectrophotometrically via commercial kits (Cayman Chemical®, Ann Arbor, Michigan, USA). At the end of the experiment, the neurons were incubated with 0.7 mg/ml MTT for 30 min at 37°C. After washing, blue formazan was extracted from cells with isopropanol/formic acid (95:5) and was photometrically determined at 560 nm. 28 The density of formazan formed in control cells was taken as 100% viability. 29
LDH assay
LDH released from damaged cells in culture medium was quantified using LDH assay kit (Cayman Chemical). A total of 100 μl of cell medium was used for LDH analysis. Released LDH catalyzed the oxidation of lactate to pyruvate with simultaneous reduction of nicotinamide adenine dinucleotide (NAD+) to NADH. The rate of NAD+ reduction was measured as an increase in absorbance at 490 nm. The rate of NAD+ reduction was directly proportional to LDH activity in the cell medium.
TAC and TOS analyses
The major advantage of TAC test is to measure the antioxidant capacity of all antioxidants in a biological sample and not just the antioxidant capacity of a single compound. In this test, antioxidants in the sample reduce dark bluish green coloured 2,2′-azino-bis (ABTS) radical to colourless reduced ABTS form. The change of absorbance at 660 nm is related with total antioxidant level of the sample. The assay is calibrated with a stable antioxidant standard solution which is traditionally named as Trolox equivalent that is a vitamin E analogue. Since measuring different oxidant molecules separately is not practical and their oxidant effects are additive, the TOS of a sample is measured and this is named total peroxide, serum oxidation activity, reactive oxygen metabolites or some other synonyms. In TOS assay performed here, oxidants present in the sample oxidize the ferrous ion–chelator complex to ferric ion. The oxidation reaction is prolonged by enhancer molecules, which are abundantly present in the reaction medium. The ferric ion makes a coloured complex with chromogen in an acidic medium. The colour intensity, which can be measured spectrophotometrically, is related to the total amount of oxidant molecules present in the sample. The assay is calibrated with H2O2, and the results are expressed in terms of micromolar H2O2 equivalent per litre. After cells were exposed to H2O2 for 6 h, the cultures were washed with ice-cold PBS and homogenized with 0.9% normal saline. Following homogenization, intracellular levels of TAC and TOS were determined by commercially available kits (Rel Assay Diagnostics®, Gaziantep, Turkey). 30
Evaluation of DNA damage
The DNA damage evaluation was performed by single-cell gel electrophoresis (also known as comet test) assay. After the application of coverslips, the slides were allowed to gel at 4°C for 30–60 min. The slides were immersed in freshly prepared cold lysing solution (2.5 M sodium chloride, 100 mM disodium EDTA, 10 mM Tris, 1% sodium sarcosinate, pH 10.0) with 1% Triton X-100 and 10% DMSO added just before use for a minimum of 1 h at 4°C and refrigerated overnight followed by alkali treatment, electrophoresis (at 1.6 V/cm for 20 min, 300 mA) and neutralization (0.4 M Tris, pH 7.5). The dried slides were then stained using ethidium bromide (20 μg/ml) after appropriate fixing for 10 min. 31 The whole procedure was carried out in dim light to minimize artefacts. DNA damage analysis was performed at a magnification of 100× using a fluorescence microscope (Nikon Eclips E6600, Japan) after coding the slides by one observer (Togar B). A total of 100 cells were screened per slide. A total damage score for each slide was derived by multiplying the number of cells assigned to each grade of damage by the numeric value of the grade and summing over all grades (giving a maximum possible score of 500, corresponding to 100 cells at grade 5). The scoring criteria for determining damage levels in cultured neurons treated with the compounds in our comet analysis are shown in Figure 2.

Viability of rat primary cortical neurons after 24 h exposure to 0–100 µg/ml ZGB. The results were presented as percentage of the control group (n = 4). Data are expressed as mean ± SD. Values with the same superscript letters are not significantly different from each other at the level of p < 0.05. ZGB: zingiberene; SD: standard deviation.
Nucleic acid oxidation
8-Hydroxy-2′-deoxyguanosine assay kits were purchased from Cayman Chemical for determining 8-OH-dG levels in the cultures. Since it is a competitive assay that can be used for the quantification of 8-OHdG in homogenates and recognizes both free 8-OHdG and DNA-incorporated 8-OH-dG, many lines of research are being performed to use this protocol. This assay depends on the competition between 8-OHdG and 8-OHdG-acetylcholinesterase conjugate (8-OHdGTracer) for a limited amount of 8-OHdG monoclonal antibody. 32 All procedures were carried out in accordance with the provider manual.
Data analysis
The data are expressed as the mean ± standard derivation of four repetitions. One-way analysis of variance was used to determine the significant differences between the groups followed by a Dunnett’s t test for multiple comparisons. The value of p < 0.05 was considered as significant. All analyses were performed using Statistical Package for Social Sciences version 15.0 (SPSS Inc®, Chicago, Illinois, USA).
Results
The cultured cortical neurons exposed to 6.25, 12.5, 25, 50 and 100 µg/ml concentrations of ZGB did not show any significant changes in cell viability during 24 h as determined by MTT and LDH assays (Figures 2 and 3). When cerebral cortical neurons were exposed to 0.5 mM H2O2, MTT absorbance was 42.4 ± 1.9% of that of untreated controls, indicating that H2O2 caused to neuronal cell death. ZGB applications significantly inhibited the decrease of MTT reduction by 0.5 mM H2O2. In fact, in cultures treated with ZGB (12.5, 25 and 50 µg/ml), H2O2-induced neuronal death was significantly reduced (MTT, 9.43, 31.8 and 6.60%, respectively) (Figure 4).
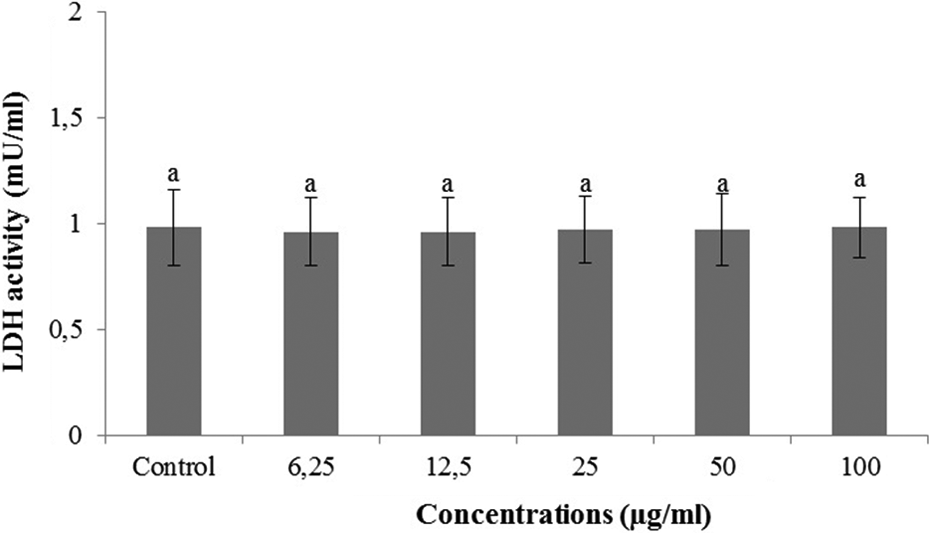
Extracellular level of LDH in cultured rat cortical neurons maintained in the presence of ZGB for 24 h. Data are expressed as mean ± SD. Values with the same superscript letters are not significantly different from each other at the level of p < 0.05. LDH: lactate dehydrogenase; ZGB: zingiberene.

The effects of ZGB applications on cell viability against H2O2-induced cell death. The cells were pretreated with different ZGB concentrations for 30 min before exposure to 0.5 mM H2O2 for 6 h. Then, cell viability of ZGB treated cultures was determined via MTT assay. Data are expressed as mean ± SD. Values with the same superscript letters are not significantly different from each other at the level of p < 0.05. ZGB: zingiberene; H2O2: hydrogen peroxide; MTT: 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyltetrazolium bromide.
H2O2-induced neuronal cell death was clearly evidenced by fivefold increase (from 0.98 to 4.76 mU/ml) in the activity of LDH compared with the observations of untreated controls. In contrast, 12.5, 25 and 50 µg/ml of ZGB (LDH, 22.3, 28.6 and 41.2%, respectively) significantly blocked the H2O2-induced elevation of intracellular LDH release (Figure 5).

The effects of ZGB applications on H2O2-induced LDH. The cells were pretreated with different ZGB concentrations for 30 min before exposure to 0.5 mM H2O2 for 6 h. After, intracellular levels of LDH were measured in ZGB-treated cultures. Data are expressed as mean ± SD. Values with the same superscript letters are not significantly different from each other at the level of p < 0.05. LDH: lactate dehydrogenase; ZGB: zingiberene; H2O2: hydrogen peroxide.
Table 1 reflects the oxidant–antioxidant profile of ZGB on cultured primary rat neurons. As seen from the table, the applications with the lowest concentrations (6.25 µg/ml) of all ZGB compounds did not lead any alterations in TAC levels, whilst their higher concentrations below 50 µg/ml (12.5 and 25 µg/ml) significantly ( p < 0.05) increased TAC levels compared with the control value. In fact, after ZGB applications at 12.5 and 25 µg/ml for 24 h, TAC levels reached maximum of the observed levels as 23.6, 33.2 and 25.5%, respectively. ZGB treatment at concentrations of 50 and 100 µg/ml did not lead to increase in TAC levels. Results reveal that exposure to H2O2 for 6 h resulted in significant decreases in the TAC levels as compared to those of the untreated group. Also, the addition of H2O2 for 6 h led to a significant elevation in the level of TOS as compared to that of the control group (Table 2). Results of the present study demonstrate that level of TAC was significantly increased in cortical neurons pretreated with ZGB as compared to those of H2O2-intoxicated cultures. Also, pre-exposure to ZGB led to a significant augmentation in the levels of TOS as compared to those of H2O2-intoxicated cultures (Table 2).
In vitro levels of TAC (as mmol Trolox equiv./l) and TOS (as mmol H2O2 equiv./l) in cultured rat cortical neurons maintained in the presence of ZGB for 24 h.a

TAC: total antioxidant capacity; TOS: total oxidative stress; ZGB: zingiberene.
aData are expressed as mean ± SD. Values with the same superscript letters are not significantly different from each other at the level of p < 0.05.
The effects of ZGB applications on oxidative alterations by H2O2 in vitro.a

TAC: total antioxidant capacity; TOS: total oxidative stress; ZGB: zingiberene; H2O2: hydrogen peroxide.
aThe cells were pretreated with different ZGB concentrations for 30 min before exposure to 0.5 mM H2O2 for 6 h. After exposure, oxidative effects of ZGB-treated cultures were determined via TAC and TOS assays. Data are expressed as mean ± SD. Values with the same superscript letters are not significantly different from each other at the level of p < 0.05.
In the comet assay, no significant difference in the induction of DNA damage was observed between the groups treated with ZGB concentrations of 6.25, 12.5, 25, 50 and 100 μg/ml and the negative control for 24 h (Table 3). However, DNA damage was significantly increased by H2O2 intoxication at 0.5 mM for 6 h when compared with the untreated group. Furthermore, ZGB pretreatment significantly reduced DNA damage in cultures treated with the two concentrations of the compound (12.5 and 25 μg/ml) + H2O2 when compared to treatment with H2O2 alone. The highest percentage reductions in DNA damage for ZGB treatment was 19.7% (Table 4).
Total DNA damage score (comet assay) and the levels of 8-OH-dG adducts (as pg/ml) in cultured rat cortical neurons maintained in the presence of different ZGB concentrations for 24 h.a

ZGB: zingiberene; 8-OH-dG: 8-hydroxy-2-deoxyguanosine.
aData are expressed as mean ± SD. Values with the same superscript letters are not significantly different from each other at the level of p < 0.05.
The effect of ZGB pretreatments on DNA damage and 8-OH-dG levels (as pg/ml) generated by H2O2 treatment.a

ZGB: zingiberene; 8-OH-dG: 8-hydroxy-2-deoxyguanosine; H2O2: hydrogen peroxide.
aData are expressed as mean ± SD. Values with the same superscript letters are not significantly different from each other at the level of p < 0.05.
The levels of 8-OH-dG, a hallmark of oxidative stress–DNA base damage, were measured using an 8-OH-dG detection kit. There were no significant differences between the intracellular levels of 8-OH-dG in the control and all ZGB-treated groups. In contrast, the intracellular level of 8-OH-dG was significantly higher in H2O2-treated cultures in comparison with untreated cultures. But pretreatment of ZGB decreased the 8-OH-dG formations which were increased by H2O2. The percentage decreases in the adduct formation ranged from 4.65% to 16.27% (Table 4).
Discussion
Oxidative stress is known to contribute to the pathogenesis of many neurodegenerative diseases such as Parkinson’s and Alzheimer’s diseases. 33,34 ROS, by-product of oxidative phosphorylation, inflicted oxidative insults by attacking nucleic acids, proteins and lipids. 35 Neurons have been known to be more susceptible to oxidative damage than other cells due to their high oxygen consumption, low activity of antioxidant enzymes, elevated concentration of polyunsaturated fatty acids in the cell membrane, high number of mitochondria, unfavourable space/volume ratio and vicinity of microglia cells which are likely to produce increased amounts of superoxide radical. 36 Based on this, searching for neuroprotective drugs of natural origin against oxidative stress-induced neuronal death has thereby attracted increasing research interests. In particular, our investigation has been focused on cortex cells since they were evaluated as vulnerable to Alzheimer’s and Parkinson’s pathologies via oxidative stress. 37,38 In comparison to more mature neurons in culture, immature neurons are particularly susceptible to cell damage induced by oxidative stress, for example, by H2O2. 39 Thus, primary cultured cortical neurons are commonly used as a suitable in vitro model system for protection against H2O2-induced cellular damages.
ZGB, isolated from Z. officinale, showed antioxidant properties. 22 However, perhaps for the first time, here different ZGB applications were shown to have protective effects against H2O2-induced cell death in primary cultured cortical neurons. H2O2, the freely diffusible form of ROS, is generated by different intracellular reactions. Extracellular H2O2 might cross membranes, thereby directly altering their intracellular concentrations. Hence, neuronal overload with ROS initiated a chain of deleterious cellular responses via forming oxidative stress. 40 Both LDH leakage and MTT assays were performed for in vitro cytotoxicity testing since they were considered as sensitive, accurate and rapid methods. 41 In this study, the cultured primary rat neuron cells exposed to 6.25, 12.5, 25, 50 and100 µg/ml concentrations of ZGB did not show any significant changes in cell viability for 24 h as determined by MTT and LDH assays (Figures 2 and 3). Besides, the cytotoxic effects of H2O2 on cultured rat cortical neuron cells were demonstrated by its strong inhibition on cell viability (Figure 4) and elevated LDH leakage (Figure 5). Our findings also revealed that ZGB pretreatments reduced the cytotoxicity. These results suggest that ZGB is capable of reducing H2O2-induced cytotoxicity and lipid peroxidation. Similar to our findings, α-coat proteins showed hepatoprotective activity by reducing the malondialdehyde levels in liver homogenates from Wistar rats. 42 Moreover, Türkez et al. 30 found that farnesene (α-FNS and β-FNS), natural sesquiterpenic compounds, were capable of protecting against H2O2-induced cytotoxicity and oxidative DNA damage in cultured rat primary cortical neuron cells. Since high levels of exogenously generated H2O2 induced cell death leading to extensive DNA damage 43 and comet formations with almost all DNA in the tail often referred to as ‘hedgehog’ comets and widely assumed to represent apoptotic cells, 44 we further determined high rates of total damage scores indicating increases of DNA strand breaks, alkali-labile sites and incomplete excision repair in cells after H2O2 intoxication. Here, we also presented evidence that ZGB pretreatment possessed protective effect against H2O2-induced DNA damage in cultured cortical neurons.
Consistent with the protective effect on cytotoxicity caused by H2O2, ZGB displayed a significant protective capability against H2O2-induced DNA damage (Table 4). ROS-induced oxidative DNA damage has been implicated in mutagenesis and carcinogenesis and has attracted much attention in last few years since ROS might attack DNA readily, generating a variety of DNA lesions, such as oxidized bases and strand breaks. 45 Then, ROS-induced DNA damage is believed to contribute to carcinogenesis, ageing and neurodegeneration. 46 Recent reports revealed that both mitochondrial oxidative damage and oxidative DNA damage played important roles in the pathogenesis of many neurodegenerative diseases like Alzheimer’s and Parkinson’s. 47 In addition to comet test, oxidative DNA damage was also evaluated in cell cultures by measuring the 8-OH-dG level, since, in particular, this marker is most frequently measured as an indicator of oxidative DNA damage in neuronal degeneration models both in vitro and in vivo. 48 Significant elevation in 8-OH-dG release from neurons was observed after treatment with H2O2; in contrast, there was a significant decrease when we pretreated with ZGB concentrations (Table 4). Here, in this study, to the best of our knowledge, we present the first evidence that ZGB supplementation possesses protective capability against H2O2-induced oxidative DNA damage.
The neuroprotection of ZGB against H2O2-induced oxidative cell death in cultured cortical neurons was further thought to be likely associated with alleviation of free radical production, although little is known on the antioxidative property of ZGB and its mechanisms have not been elucidated yet. To further explore the mechanism of the in vitro protective effect of ZGB on H2O2-induced oxidative damage in cortical neurons, TAS and TOS levels were also analyzed. The results in Table 1 show that ZGB applications increased TAC levels of the cells without any alterations in TOS levels as compared to untreated cultures. The data further show that ZGB-treated cells displayed modulating effect on H2O2-induced abnormalities in oxidative metabolism (Table 2). Then, our results suggest that ZGB is capable of suppressing intracellular H2O2 formation. Although other mechanisms are possible, the above observations involving neuroprotective actions might be explained by suppressing intracellular H2O2 formation via pretreatment with ZGB. However, recent studies suggested several mechanisms on not for the tested ZGB sesquiterpene compound but for other natural antioxidant-mediated neuroprotection against H2O2 intoxication attributed to be via modulation of activity of tyrosine kinase of Trk receptors,
49
activation of the PI3K/Akt signalling pathway,
50
regulation of apoptosis by c-Myv Bcl-2 and Bax gene expressions,
51
alpha 2-adrenergic receptor signalling pathway
52
inhibition of n-methyl-
In summary, the present findings suggest that ZGB, a natural sesquiterpenic compound, is capable of protecting against H2O2-induced cytotoxicity and oxidative DNA damage in cultured rat primary cortical neuron cells. Taken together, present data suggest that ZGB compound may prevent neurodegeneration via the antioxidative and antigenotoxicity mechanisms and have potential therapeutic values for Alzheimer’s- and Parkinson’s-like neurodegenerative disorders of central nervous system. Further studies will be needed to clarify the mechanisms involved.
Footnotes
Acknowledgements
We are grateful to our lab specialists for their help and efforts in experiments done in the medical genetics and pharmacology laboratories and animal housing.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
