Abstract
As the skin is the primary barrier to infection, the importance of wound healing has been understood since ancient times. This article provides a synopsis on the symposium presentations focusing on how wounds were traditionally treated, what models and pathology endpoints exist to study wound healing, special considerations for wound healing studies, an overview of regulatory aspects of new pharmaceutical and medical device development, and the clinical relevance of such models. The clinical treatment of small and large wounds is also considered.
As the skin is the primary barrier to infection, the importance of wound healing has been understood since ancient times. This symposium focused on how wounds were traditionally treated, what models and end points exist to study wound healing, and the clinical relevance of such models. Pathology end points, special considerations for dermal studies, and an overview of regulatory aspects of new pharmaceutical and medical device development were also discussed in addition to the clinical aspects of treating small and large wounds.
The first talk entitled “A Brief History of Wound Healing” by Dr. David Hobson provided an overview of wound classification by depth of involvement, a brief history and review of wound healing knowledge and technology from ancient times to the present state of the art. This talk also introduced the science behind the development of wound healing therapeutics and the in vitro and in vivo models used for the assessment of product safety. Some highlights from his talk are below.
Some important characteristics, considerations, and terminology of skin and wounds important to understanding wound healing in modern context include the following:
Skin Largest organ of the body, adult human ∼1.8 m2. Has multiple, essential functions. Weight: 3 to 4 kg (7-9 lb); ∼6% of the total body weight. pH range: 4.7 to 7.4. Composed of a wide variety of cell types. Chemical signaling is rich, complex, and dynamic
Wounds Include damaged skin and underlying tissues. Partial, full, or complex thickness (depth). Wound healing includes acute or chronic biologic processes (each is different). Chemical signaling to promote healing is often disrupted in chronic wounds. The biology for a complete healing response may or may not be intact. “Tissue management” is the 21st century medical and market concept for wound healing.
The ancients were remarkably knowledgeable of treatments that could promote wound healing, even though they did not understand the science behind them. Healing was often attributed to the actions of ancient gods and the application of remedies developed over time by trial and error. The Egyptian goddess Sekhmet, daughter of Ra, was the ancient Egyptian goddess of healing. The Ebers papyrus
1
(circa ∼1600
The Mayans (circa 2000
Wound healing practice began to change in the late 1800s and early 1900s, with scientific advances in infection control and bandaging materials that advanced throughout the century. These advances then developed into an understanding of wound healing at the biochemical and molecular biological level. With these advances, we observed the mean human life expectancy of the most advanced civilizations go from ∼35 years in ancient Egyptian times to ∼47 years at the beginning of the 1900s but increase to ∼78 years by the end of that century. Due to many advances in medicine, to include wound healing, by 2015, mean life expectance was ∼80 years, and it is anticipated to exceed 100 years by 2050. The history of wound healing clearly shows that control of infection in wounds, as well as advancements in material science and medical practice, is critical to advance healing, survival, and human longevity. Thus, current advances in the development of new means to eliminate infectious agents, the promotion of moist wound healing, improved conditions for optimum cellular signaling, and improved molecular targeting of wound healing signal processes are critical to future advancement of wound-healing science.
The wound healing process can be described temporally as a series of phases, starting with hemostasis and progressing through inflammation, proliferation, and remodeling, as shown in Table 1. The pathophysiologic events in the wound healing process relative to time after injury and time to heal are shown in Figure 1. 4 -6
Phases of Wound Healing and Biophysiologic Events That Differentiate Each Phase.
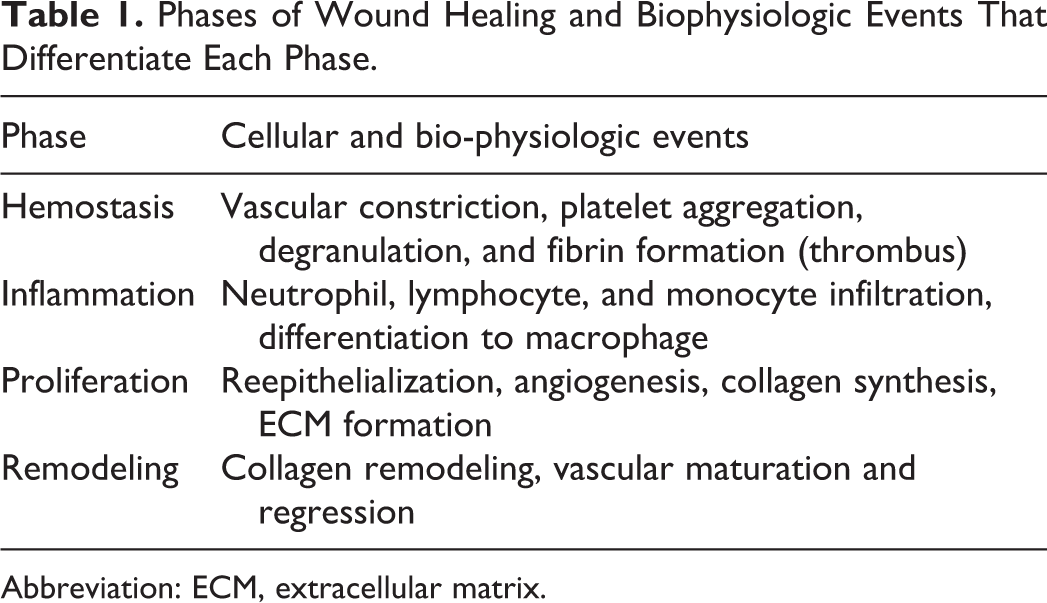
Abbreviation: ECM, extracellular matrix.
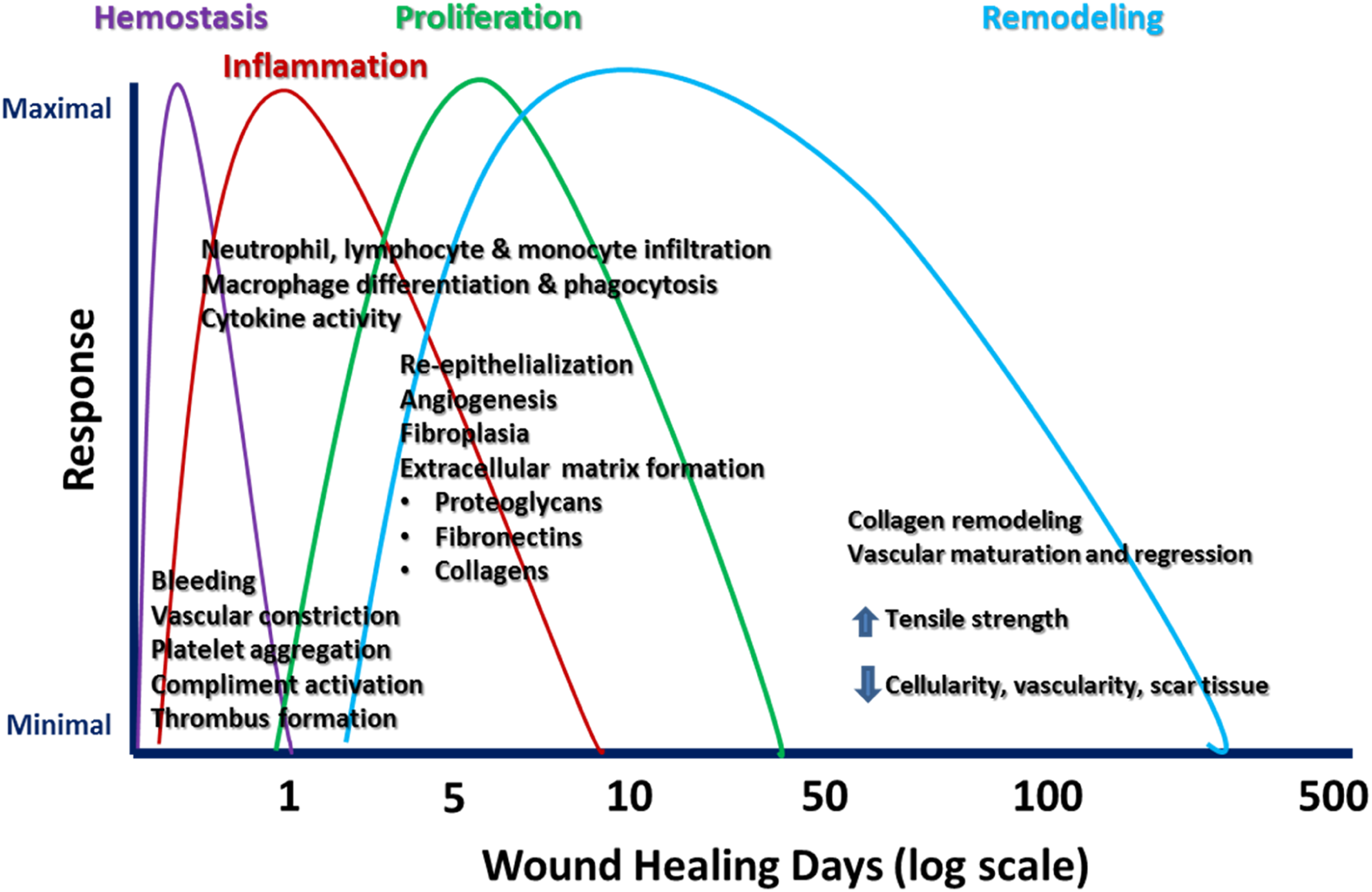
Phases of wound healing with respect to time after injury and time to healing. 4
Wounds besides being classified as first, second, and tertiary intention in terms of their closure characteristics are now generally classified as acute healing wounds and chronic nonhealing wounds, which may have a number of etiologies as well as being infected or not.
5
-7
For example, wounds arising from pressure ulcers are described as “stages” in their etiology where the following stages are identified: stage I: tissue redness and warmth, stage II: visible shallow ulcer with distinct edges, stage III: full-thickness loss of skin, stage IV: involvement of fascia, connective tissue, muscle, and bone, and stage V: area covered with black eschar (also known as scab).
Wounds are also classified with respect to whether they are infected with the following features: clean wound—operative incisional wounds that follow nonpenetrating (blunt) trauma, clean/contaminated wound—uninfected wounds in which no inflammation is encountered, but the respiratory, gastrointestinal, genital, and/or urinary tract have been entered; contaminated wound—open, traumatic wounds or surgical wounds involving a major break in sterile technique that show evidence of inflammation; and infected wound—old, traumatic wounds containing dead tissue and wounds with evidence of a clinical infection (eg, purulent drainage).
A general but useful scheme of classification for the toxicologist is to describe wounds in terms of their depth or “thickness” using the following terms relative to the skin surface: superficial—epidermis only, partial—epidermis with small amount of dermis, full—entire epidermis and dermis with small amount of subcutaneous fat, and complex—entire epidermis, dermis, subcutaneous fat as well as underlying muscle, and possible bone and connective tissue.
So today, there are formal wound healing classification systems that have been developed and are continuously evolving to serve as aids to the wound healing scientist and practitioner in describing the science of wound healing and determining the best course of treatment.
The science behind modern wound treatments that incorporate modern concepts and the development of treatments that incorporate moist wound healing and wound exudate management was summarized, and the following desirable characteristics of an “ideal” wound dressing were identified: reduces, eliminates, prevents infection; manages or absorbs exudate; requires infrequent or no changing; aids in removal of necrotic tissue; promotes moist wound healing; accelerates the rate of healing; manages wound odors effectively; and has environmentally favorable biodegradation characteristics.
The need for toxicological evaluation of novel wound-healing technologies and associated products was discussed. Currently, this is largely driven by an increasing demand for more clinically and cost-effective products based on practical and market-driven factors such as the following: worldwide growth in aging populations with increasing prevalence of chronic, infected, and diabetic wounds; skin aging and solar damage (cosmetic effects, fine lines, wrinkles, solar damage); medical advances in wound healing science and technology;
surgical/burn—rate of closure/scar elimination, keloid resolution, advanced materials and therapeutics (eg, nanotechnology, biopharmaceuticals, drug-device combinations), new/novel end points and molecular factors (cytokines, chemokines, growth factors, structural factors, matrix metalloproteinases [MMP-9], etc); Improvement and efficiency of nonclinical models to evaluate product efficacy and safety (reduction of time, cost, animal usage, etc).
Toxicological concerns that have eliminated some therapeutics in the past and are now evaluated when developing current therapeutics involve both localized reactions and systemic effects.
Localized reactions include significant degrees of skin irritation that exacerbate wound healing and cause significant discomfort relative to alternative, but equally effective, treatments. Irritant effects can be caused by a wide variety of wound healing product constituents including presence of chemical irritants, chemical inflammatory agents to include pyrogens, surfactant effects causing cellular lysis of wound bed tissues and imbalances in ionic strength, as well as with tissue water and salt content. Other types of localized reactions can also include various forms of skin sensitization that can be caused by a variety of factors such as sensitizing formulation contaminants, preservative agents, unreacted polymers, and degradation products.
Systemic toxicity can also be caused by wound healing products, especially those that are applied into open wounds or burns involving large surface areas. Substances such as polyethylene glycol that can be used topically on undamaged skin can be fatally toxic to burn patients, for example. 8 With the increasing advancement of new polymeric and nanomaterials being incorporated into novel wound treatments, the toxicologist must be constantly vigilant to the potential for systemic effects with treatments to be used on compromised and wounded skin.
JoAnn Schuh followed with “Pathologic Evaluation of Wound Healing Models and Relevance to Human Tissue Healing,” which included an overview of animal models of wound healing based on comparative skin anatomy, pathophysiology, and immunology and optimization of study design for appropriate histopathology outcomes. The skin of rodents and other small mammal has distinct anatomical and physiological differences from human skin that frequently makes large animals, particularly the pig, the preferred species for testing therapeutic interventions in wound healing models.
These species differences, including between human and pig, need to be considered in selection of the most appropriate test species. Naturally glabrous (hairless) skin of human and pig, which also shows epidermal rete pegs or epidermal extensions into the dermis (Figure 2A and B, respectively), contrasts with the moderately haired nonhuman primate skin (Figure 2C) and heavily haired species of dog (Figure 2D), rabbit (Figure 2E), guinea pig (Figure 2F), rat (Figure 2G), mouse (Figure 2H), and nude mouse (Figure 2I). There are numerous small and large animal models of wound healing, and careful consideration of the pros and cons of each model or induction method needs to be understood to select the best wounding model for investigative or therapeutic studies. 9,10 Once the model or models are selected, the study must be designed carefully to ensure that preparation of the test sites, induction and maintenance of the wounded site(s), interim biopsies, and dressings do not affect the study end points. At study termination, adequate size, types, and numbers of skin sections need to be appropriately preserved, processed, sectioned, and stained (routine or specialized stains) for histopathology evaluation. Specialized imaging techniques, electron microscopy, immunohistochemistry, molecular pathology, and image analysis are also frequently used adjunct techniques for investigation of animal models of wound healing.
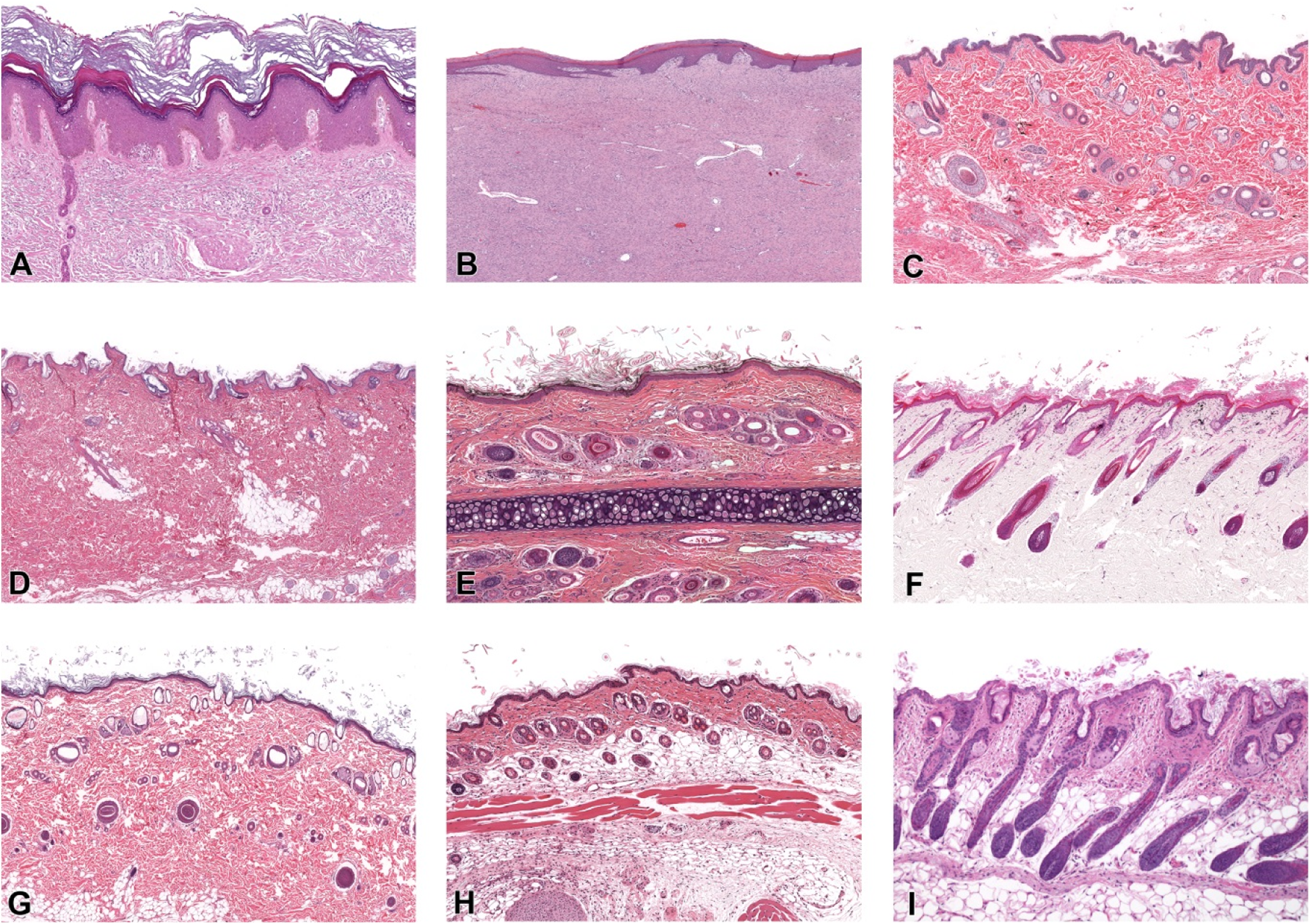
Comparative histology of skin from human (A), pig (B), nonhuman primate (C), dog (D), rabbit (E), guinea pig (F), rat (G), mouse (H), nude mouse(I). Note the generally glabrous (hairless) skin of human (A) and pig (B) versus nude mouse (I) and moderately haired nonhuman primate (C) versus heavily haired dog (D), rabbit (E), guinea pig (F), rat (G), and mouse (H). Compared to normal hair follicles (F) or hairless skin (A and B), hair shafts in nude mouse skin (H) are abnormal and hair development is aborted. Epidermal thickness is greatest and rete ridges (epidermal extensions) are only present in human (A) and pig (B) compared to other species. Sections are from the body except for the image of rabbit hair from an ear section (cartilage in the middle of section) and unknown location for the human skin. Magnification ×10.
Daniel V. Zurawski presented “Acinetobacter baumannii: A Bacterial Pathogen That Causes Wound Infections in Wounded Warriors and Civilians.” From 2004 to 2009, the US Armed Forces, besides dealing with the military conflicts in Iraq and Afghanistan, was fighting another war, an infectious diseases war behind the scenes against antibiotic resistant bacteria infecting its wounded warriors. More than 10% of US wounded soldiers and their caregivers were specifically battling against the bacterial species, Acinetobacter baumannii, which was responsible for more than 3,300 infections over this time period. 11 The loss of limbs due to infection and subsequent amputation revisions along with the lack of treatment options for this multidrug-resistant (MDR) pathogen became a common and repeated narrative. 12 During this time and the years following, MDR A baumannii also spread within civilian hospitals, became responsible for 10% of all hospital-acquired infections, was responsible for numerous outbreaks, and was a leading cause of ventilator-associated pneumonia associated with high mortality rates among this patient population. 13,14 The problem of antibiotic resistance has only worsened as pandrug-resistant strains have now emerged. 15 More alarming, strains of increased virulence have been identified and responsible for deaths even stemming from wound infection. 16,17 In order to combat the growing threat of A baumannii to wounded soldiers and civilians, the Wound Infection Department (WID) at Walter Reed Army Institute of Research focused on a remediation strategy that included the Sun Tzu-like approach of “knowing thy enemy” because how A baumannii actually causes an infection was understudied. Over the last 5 years, the WID made an effort to better understand the virulence of A baumannii and what A baumannii proteins were responsible for its pathogenesis. Specifically, a diversity set of A baumannii isolates was studied and a more virulent isolate, AB5075 was discovered. 18 The use of this strain facilitated genetic studies and the development of animal models because attributes of pathogenesis became more pronounced and often occurred at earlier time points when compared to less virulent isolates. For example, AB5075 could enter the bloodstream more rapidly and colonize organs (Figure 3) in a murine pulmonary model of infection. Separately, WID also developed a full-thickness, murine wound puncture model, which displayed many of the same characteristics of wound infection that were being observed in wounded warriors such as necrotic tissue and dissemination into deeper tissues. 19 With these models available, WID was able to identify virulence factors on the surface of the bacterium. A better understanding of this bacterial enemy allowed for the development of antibodies that target these key protein factors and interfere with infection by the organism. In fact, one of WID’s antibodies can protect mice from both pulmonary and wound infection caused by A baumannii when injected prophylactically. In the future, WID plans on generating more monoclonal antibodies against virulence factors on the bacterial surface of not just A baumannii but also other nosocomial, opportunistic MDR-bacterial species such as Klebsiella pneumoniae and Pseudomonas aeruginosa that are also responsible for wound infections in wounded warriors and civilians.
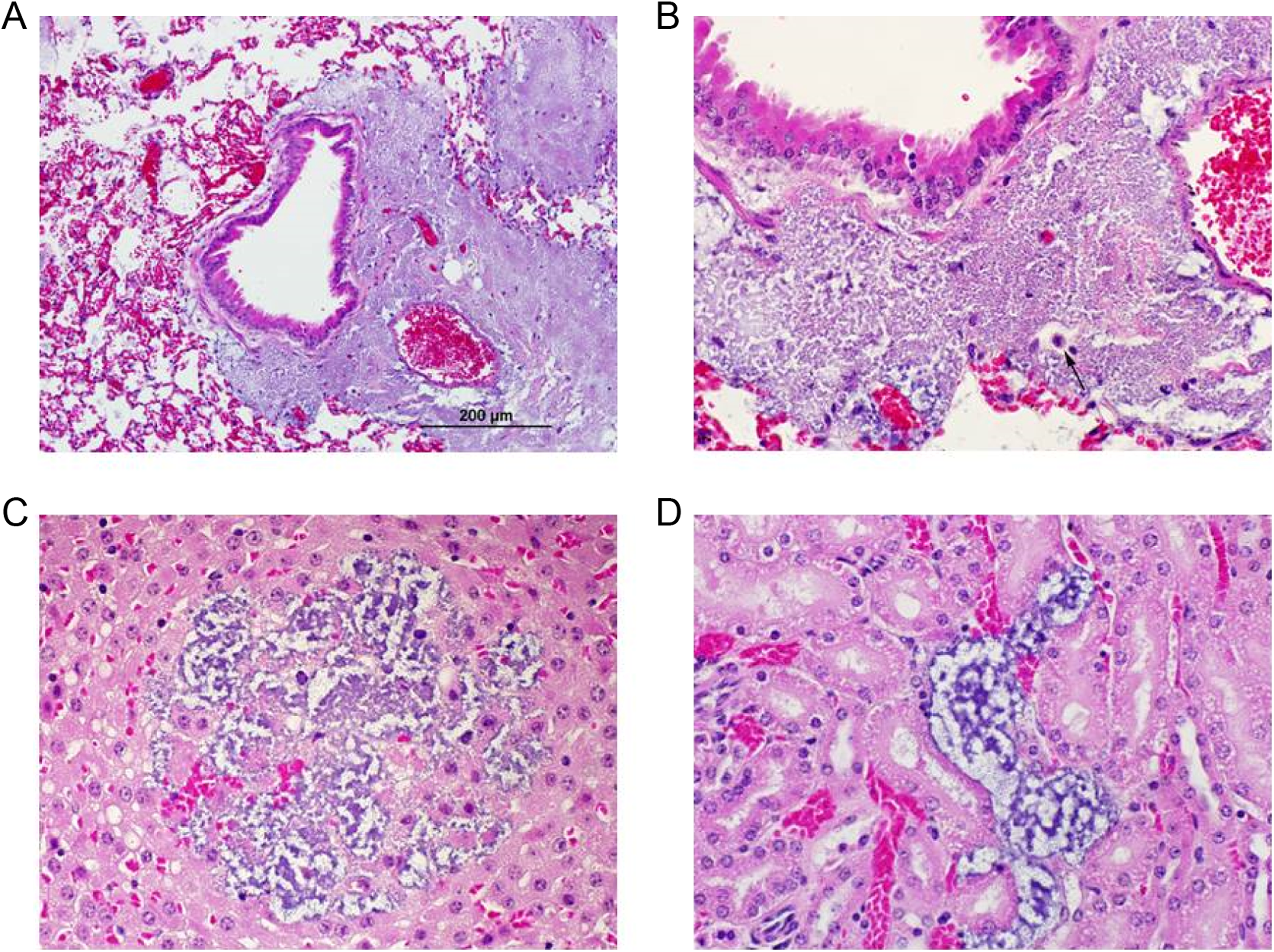
A, Photomicrograph of the murine lung 2 days after an AB5075 infection demonstrating bacteria filling several adjacent alveolar lumen in the absence of any remarkable inflammatory response (×200 magnification). B, An increased magnification (×600) of the infected murine lung showing the occasional alveolar lumen containing macrophage with phagocytized bacteria (small black arrow). C, Photomicrograph of the adrenal gland demonstrating bacterial colony within the cortex following dissemination into the bloodstream from the original site of infection in the lungs. Again, there is a noticeable absence of remarkable inflammatory response (×600 magnification). D, Photomicrograph of the kidney demonstrating bacterial colonies within the renal interstitium. Again, this demonstrates organ colonization after dissemination into the bloodstream and an absence of an inflammatory response (×600 magnification).
Jianyong Wang presented “Nonclinical Regulatory Considerations of New Drug Development for Wound Healing.” Wound healing drug products are developed to treat many different indications, including but not limited to diabetic ulcers, venous ulcers, pressure ulcers, burn wounds, surgical wounds, scarring, radiation dermatitis, and oral mucositis. 20 -24 Drug candidates can be very diverse (small molecules, proteins, peptides, oligonucleotides, botanicals) with a variety of modes of action, such as improving circulation, anti-inflammation, vascular protection, stimulating cellular growth, angiogenesis, antisense activity, scar reduction, and debridement.
Nonclinical studies needed to support the development of wound healing drug products usually include animal wound-healing studies, dermal irritation studies, dermal sensitization studies, dermal phototoxicity studies, ocular irritation studies, pharmacology studies, pharmacokinetic/toxicokinetic studies, repeat-dose toxicology studies, genetic toxicology studies, reproductive and developmental toxicology studies, carcinogenicity studies, and special toxicology studies. Nonclinical requirements may vary from case to case, and the needed studies to support different phases of clinical development should be scientifically justified.
Animal wound healing studies should be properly designed to support the specific clinical use. Relevant species and models should be used, and appropriate end points should be evaluated. Relevant end points may include rate of complete wound closure, time to complete wound closure, incidence of wound infection, and evaluation of reepithelialization, skin histopathology, wound appearance/scar formation, and tensile strength. Repeat-dose toxicology studies in appropriate rodent and mammalian nonrodent species with appropriate length and design should be conducted to support different phases of clinical trials. The studies should include complete clinical pathology, histopathology, and toxicokinetic analysis. In addition to dermal toxicology studies, oral or parenteral administration is usually needed to achieve adequate levels of systemic exposure. The high dose for repeat-dose toxicology studies is usually either limited by toxicity (maximum tolerated dose) or by administration method (maximum feasible dose, usually seen in dermal toxicology studies). Chronic toxicology studies and carcinogenicity studies are generally required for chronic indications. The evaluation of safety pharmacology and dermal irritation end points may be incorporated into repeat-dose toxicology studies to avoid stand-alone studies. A dermal sensitization study in guinea pigs to evaluate dermal sensitization potential and a bovine corneal opacity and permeability assay to evaluate ocular irritation potential are recommended. Dermal phototoxicity studies may be waived if there is no sun exposure under clinical use conditions or no light absorption in the 290- to 700-nm wavelength range. Certain systemic toxicology studies may be waived if systemic exposure under clinical maximum use conditions is negligible.
And, finally, Sam Arbabi put the entire symposium into perspective with his talk “Clinical Considerations of Wound Healing.” Large wounds and burn injuries are significant causes of military and civilian mortality and morbidity. Postinjury secondary damage to organs and wound complications are common without early intervention. Release of inflammatory mediators from burn wounds results in systemic inflammatory response syndrome (SIRS), multisystem organ dysfunction syndrome (MODS), and death. Moreover, patients surviving deep burns and/or large wounds have the highest incidence of debilitating fibroproliferative hypertrophic scar formation. 25 Hypertrophic scar may cause significant morbidity, disabling loss of function, and extended difficult recovery times, dramatically affecting the patient’s quality of life physically, psychologically, and economically. 26,27 Worldwide efforts to develop new biological therapeutics for large wounds and severe injuries have met with disappointing results. The ideal therapeutic agent that can improve wound healing and scar formation remains elusive, in part, due to our incomplete understanding of the functional integration of the complex host response and identification of appropriate patients and therapeutic targets. Another major limitation is the lack of appropriate animal models that resemble the human condition. Murine wound models have failed to produce scarring analogous to the human fibroproliferative scar. 28 The female red Duroc pig has been validated as a model of fibroproliferative scarring that best simulates human wound healing and hypertrophic scar formation. 29,30
The desired characteristics for treatment agents and wound dressings depend on the wound type and timing of therapy. Initially, the dressing should stop bleeding and loss of moisture or heat. In general, the desired aims for a biological wound dressing include improving wound healing and closure, avoiding infectious complications, attenuating pain or pruritus, inhibiting overstimulation of the inflammatory response and subsequent development of SIRS and MODS, decreasing future scar formation and contraction, maintaining function, and improving future cosmetic results. The ideal dressing should be easy to apply and economically viable. The p38MAPK inhibitors have shown significant promise in animal burn and wound models as a potential early topical treatment and may be a viable future therapeutic option. 31 -35
Footnotes
Authors’ Note
Images of human skin and nude mouse provided by Wendy Rosebury-Smith and Mark Hoenerhoff, respectively, University of Michigan. Images prepared for publication by Emily Singletary, EPL, Inc. All animal research was conducted in compliance with the Animal Welfare Act and other federal statutes and regulations relating to animals and experiments involving animals and adheres to principles stated in the Guide for the Care and Use of Laboratory Animals, NRC Publication, 2011 edition. The findings, opinions and information in this presentation are those of the authors and do not represent the views and/or official policies of the US Food and Drug Administration and of the WRAIR, the Department of the US Army, the Department of Defense, or the US Government.
Acknowledgments
Daniel V. Zurawski would like to acknowledge his support by the Military Infectious Diseases Research Program (MIDRP) and the Defense Medical Research and Development Program (DMRDP).
Author Contributions
David W. Hobson contributed to conception and design and contributed to acquisition and interpretation. JoAnn C. L. Schuh contributed to conception and design and contributed to acquisition, analysis, and interpretation. Daniel V. Zurawski contributed to conception and design and contributed to acquisition, analysis, and interpretation. Jianyong Wang contributed to conception and design and contributed to analysis and interpretation. Sam Arbabi contributed to conception and design and contributed to acquisition, analysis, and interpretation. Maralee McVean contributed to conception. Kathleen A. Funk contributed to conception. All authors drafted the manuscript, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
