Abstract
Long-acting injectable formulations of antipsychotics have been an important treatment option to increase the compliance of the patient with schizophrenia by monitoring drug administration and identifying medication noncompliance and to improve the long-term management of schizophrenia. Risperidone, a serotoninergic 5-HT2 and dopaminergic D2 receptor antagonist, was developed to be a long-acting sustained-release formulation for the treatment of schizophrenia. In this study, 12-week subchronic toxicity study of risperidone-loaded microspheres (RMs) in rats by intramuscular injection with an 8-week recovery phase was carried out to investigate the potential subchronic toxicity of a novel long-acting sustained-release formulation. The results indicated that the dosage of 10–90 mg/kg of RM for 2 weeks did not cause treatment-related mortality. The main drug-related findings were contributed to the dopamine D2 receptor and α1-adrenoceptor antagonism of risperidone such as elevation of serum and pituitary prolactin levels and ptosis and changes in reproductive system (uterus, ovary, vagina, mammary gland, testis, seminal vesicle, epididymis, and prostate). In addition, foreign body granuloma in muscle at injection sites caused by poly-lactide-co-glycolide was observed. At the end of the recovery phase, these changes mostly returned to normal. The results indicated that RM had a good safety profile in rats.
Keywords
Introduction
Schizophrenia is a chronic psychiatric disorder associated with substantially reduced quality of life and significant impairment in psychosocial functioning. Early and maintenance treatment of schizophrenia with antipsychotics has been shown to be essential and generally efficacious and it also decreases hospitalization and reduces economic burden. 1 However, noncompliance and high discontinuation rates are common problems in the management of schizophrenia. 2 More than 40% of first-episode psychosis patients discontinue medication during the first 9 months of treatment and the chances of relapse are concurrently increased dramatically. 3 Even up to 74% of schizophrenic patients discontinued their medicinal treatments within 18 months. 4 Long-acting injectable antipsychotics have been an important treatment option to increase the compliance of the patient with schizophrenia and to improve the long-term management of schizophrenia because they may provide sustained drug delivery and regular treatment regimen. 2 At present, six different long-acting antipsychotics are available for individualized treatment of each case. 1,5 Long-acting risperidone is used to treat schizophrenia and bipolar I disorder. The recommended dosage of long-acting risperidone is 12.5–50 mg for every 2 weeks, with oral supplementation for the first 3 weeks of treatment. The accumulating data demonstrate that long-acting risperidone has been shown to increase the patient compliance, reduce hospital time and relapse rates, and increase time spent in rehabilitation. 6,7 Long-acting risperidone is a selective monoaminergic antagonist with a high affinity for serotoninergic 5-HT2 and dopaminergic D2 receptors. 8 Compared with oral equivalent of risperidone in clinical trials, long-acting risperidone caused fewer side effects like prolactin elevation and extrapyramidal side effects while it produced equivalent efficacy. 9,10 However, a disadvantage associated with long-acting risperidone is that oral supplementation of antipsychotic medications is needed, which results in inconvenience for the patients and doctors.
Risperidone-loaded microsphere (RM) is an investigational drug product of risperidone formulated as extended-release microspheres for intramuscular (i.m.) injection, which is developed for the treatment of schizophrenia by Shandong Luye Pharmaceutical Co. Ltd (Luye Pharma, Shandong, China). The drug carrier, poly-lactide-co-glycolide (PLGA), of RM is approved by US Food and Drug Administration as biodegradable materials used in medicine. RM is expected to be injected i.m. once in every 2 weeks and release the drug after injections without lag time. This would simplify the dosage and administration by eliminating the oral antipsychotic medication in the first 3 weeks of the therapy.
In the present study, the main objective is to establish the nonclinical safety profile of RM, a new long-acting injectable risperidone formulation without lag time. A 12-week subchronic toxicity study with an 8-week recovery phase was conducted in rats to determine the reversibility, persistence, or delayed occurrence of target organ toxicity.
Materials and methods
Test substance
RM was supplied by Luye Pharma. 11 Risperidone, the active drug of RM, was microencapsulated in PLGA at a concentration of 410–500 mg of risperidone per gram of microspheres (drug content is 41.0–50.0%). The mean diameter of microspheres was 60–110 μm. Placebo microspheres (without active drug) and vehicle (1% carboxymethylcellulose sodium, SCMC) were also provided by Luye Pharma. RM and placebo microspheres were suspended in SCMC to the desired concentration. The concentration, homogeneity, and 24-h stability of suspensions were analyzed after preparation to ensure proper administration of the intended doses.
Animals and management
Sprague Dawley rats (190 rats/sex) were obtained from Shanghai SLAC Laboratory Animal Co. Ltd (Shanghai, China). Rats were quarantined and adapted to the new environment for at least 5 days before the start of the experiment. When grouping, the rats were 5–6 weeks old. Mean body weight of females was 189 g and male was 210 g. All rats were housed in polycarbonate cages, that is, <5 animals/cage. All animals had free access to diet and water. Rat diet was provided by Shanghai SLAC Laboratory Animal Co. Ltd. Drinking water for laboratory animals was supplied ad libitum. The animal room was maintained at a temperature of 20–25°C, relative humidity 55 ± 15%, and ventilation frequency at 10–20 times/h with 100% fresh air with 12-h light–dark alternation.
This nonclinical study has been conducted in compliance with the Testing Guidelines for Safety Evaluation of Drugs (Notification [H], GPT2-1 issued by China's State Food and Drug Administration on March 2005) in WestChina-Frontier PharmaTech Co. (WCFP) and National Chengdu Center for Safety Evaluation of Drugs (NCCSED), Sichuan, China. Animal experiments complied with the relevant regulations in Institutional Animal Care and Use Committee in WCFP and NCCSED.
Study design
In the main subchronic toxicity study, the range of female and male body weight was 162.4–216.2 g and 192.8–239.2 g, respectively. Fifteen rats per sex per /group were randomly assigned to five groups by body weight. Animals in different groups were injected i.m. with corresponding vehicle, placebo microspheres (105.7 mg/kg equal to the microspheres content of 90 mg/kg RM), and active drug concentrations at 10, 30, or 90 mg/kg of RM at the same volume of every 2 weeks for 12 weeks (total 6 doses per animal). In our single-dose pharmacokinetics study, after a small initial release on day 1, drug was continuously released and absorbed from day 3 to day 21. In our pharmacologic studies, RM had antischizophrenic potency at 5–20 mg/kg following single i.m. administration for at least 2 weeks. According to pharmacokinetics and pharmacologic studies, dose regimen was designed as once for every 2 weeks. Ten rats/sex/group were autopsied at the end of treatment phase, while 5 rats/sex/group were killed at the end of a 8-week recovery phase.
In the toxicokinetics (TKs) study, another 110 rats were used to evaluate the relationship between toxic reactions and systemic exposure. The number of rats in vehicle group and placebo microspheres group were reduced to 5 rats/sex/group, totaling to 10 rats per group. The number of rats in each RM-treated groups was 15 rats/sex/group. The dose designed for TKs study was the same as that in the main subchronic toxicity study.
Clinical observations, body weight, and food consumption
Clinical signs were assessed and recorded at least once a day throughout the study, including animal’s signs, injection site, behavior, mental status, respiration, hairs, stool property, genitals, mortality, and other toxic manifestations. Body weight was measured twice a week in the first 4 weeks of treatment phase and once a week thereafter. Food consumption was measured once a week during the treatment and recovery phases.
Laboratory test
After anesthetization, blood samples were collected from abdominal aorta with or without anticoagulation for hematological or clinical chemistry measurements at the end of the treatment phase (day 85) and recovery phase (day 141). The hematology and clinical chemistry parameters were analyzed using auto hematology analyzer (advia2120, SIEMENS, Eschborn, Germany), auto blood hemagglutination analyzer (CA-7000, SYSMEX, Kobe, Japan), or auto biochemistry analyzer (cobas C501, ROCHE, Rotkreuz, Switzerland). Clinical chemistry parameters included chloride, sodium, calcium, potassium, phosphate, glucose, amylase, lipase, aspartate amino transferase, alanine aminotransferase, alkaline phosphate, total bilirubin, creatinine, urea, albumin, globulin, albumin/globulin ration, cholesterol, total protein, triglycerides, and creatine kinase. Hematology parameters included red blood cells, hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, white blood cells; neutrophil, lymphocyte, monocyte, eosinophil and basophil counts and percentage, platelet count, mean platelet volume, prothrombin time, activated partial thromboplastin time, and reticulocyte count.
Pathology
Gross pathology and organ weights
At the end of the treatment phase (day 85), 10 rats/sex/group animals were killed in each group. At the end of the recovery phase (day 141), the remaining animals in each group were killed. All animals were fasted for at least 12 h and weighed before necropsy, and then anesthetized with pentobarbital sodium injected intraperitoneally at 60 mg/kg prior to exsanguination, followed by complete necropsy. The organs and tissues including brain, heart, liver, spleen, pituitary gland, lung, kidney, adrenal gland, thymus, prostate, testes, epididymis, ovaries, and uterus were weighed. Paired organs were weighed together and the relative weights (organ-to-body weight ratio) were calculated.
Histopathology
The following organs and tissues were collected for histopathologic examination: adrenal gland, aorta, bone (femur and knee joint), brain (cerebrum, cerebellum, and brain stem), cecum, colon, duodenum, epididymides, esophagus, gross lesions (if present), heart, ileum, jejunum, kidney, liver, lungs, lymph nodes (mesenteric and inguinal), mammary gland, muscle of injection site, ovaries, pancreas, parathyroid gland, pituitary, prostate (including coagulating gland), rectum, sciatic nerve, seminal vesicle, spinal cord (cervical, thoracic, and lumbar), spleen, stomach, submandibular gland, testes, thymus, thyroid gland, trachea, urinary bladder, uterus (including cervix), and vagina. Testes and epididymides were fixed in modified Davidson’s solution and all other tissues were preserved in 10% of neutral-buffered formalin. The samples were then paraffin embedded, sectioned, and hematoxylin–eosin stained. Finally, microscopic examination was performed.
Bone marrow smear
At the end of the treatment phase (day 85) and recovery phase (day 141), 10 rats/sex/group and 5 rats/sex/group rats were selected for bone marrow smear, respectively. Bone marrow was taken from sternum 12 and then smeared followed by Wright-Giemsa staining. Smears were air-dried and examined under a light microscope. The granulocytes, erythroid cells, lymphocytes, monocytes, and megakaryocytes were counted under a light microscope and the percentage was calculated.
Prolactin
Prolactin levels in serum and pituitary tissue were determined in this study.
Serum prolactin
Blood samples were collected from abdominal aorta without anticoagulation on days 85 and 141for the determination of serum prolactin. Then serum prolactin was determined using Luminex 200 System and Milliplex MAP Rat Pituitary Magnetic Bead Panel Immunoassay kit (Merck Millipore (Shanghai, China), cat. no.: RPTMAG-86K-01; lot no.: 2014021).
Pituitary prolactin
Pituitary prolactin was detected by immunohistochemistry at the end of the treatment phase (day 85) and trecovery phase (day 141). Briefly, the formalin fixed, paraffin-embedded pituitary glands were sectioned and placed on the slides. After deparaffinization and rehydration, endogenous peroxidase activity was quenched with 3% of hydrogen peroxide. The slides were rinsed three times with phosphate buffer solution followed by blocking with 3% albumin from bovine serum. After overnight incubation with antiprolactin antibody (1:4000) at 4°C, sections were incubated in Polymer Helper. They were subsequently incubated in goat antirabbit IgG per horseradish peroxidase. Finally, the slides were stained with 3,3′-diaminobenzidine tetrahydrochloride substrate kit (Beyotime Institute of Biotechnology, Jiangsu, China) and examined under a light microscope.
TKs
About 0.5 mL blood were collected from the orbital venous plexus of rats followed by anesthesia with carbon dioxide and oxygen in the ratio of 7:3 prior to dosing, 1 h, 1 day, 3 days, 5 days, 7 days, 9 days, 11 days, 14 days, and 21 days after the first, third, and the last dose from animals in RM groups. Blood samples were anticoagulated with heparin sodium and stored temporarily in ice before centrifugation. Then the samples were centrifuged at 1200g for 10 min at 4°C to separate plasma. Finally, 100 μL of plasma were transferred into a new tube and the rest in another one and were stored at −20°C. Concentration of risperidone and 9-hydroxyrisperidone in plasma was analyzed and qualified by liquid chromatography–tandem mass spectrometry method, with the lower limit of quantitation of 0.2 ng/mL.
Statistical analysis
Quantitative data such as body weight, food consumption, hematology, clinical chemistry, hormone related indexes, percentage of bone marrow cells and megakaryocyte count, and organ weights and ratios were presented as mean ± SD. Levene’s test was used to analyze the variance homogeneity. In the case of heterogeneity of variances (p < 0.05), Kruskal–Wallis (K-W) H test was used to analyze the quantitative data, and if there was significant difference (p < 0.05) then Mann–Whitney (M-W) U test would be adopted to compare the differences between groups. Otherwise in case of homogeneity of variance (p > 0.05), quantitative data were evaluated using one-way analysis of variance (ANOVA). If the ANOVA was significant (p < 0.05), Fisher's least significant difference test was used for pairwise comparisons between RM-treated groups, placebo microspheres group, and vehicle group.
Qualitative data of nucleate cell proliferation on the bone marrow slides (binomial categories data, unordered multicategory data, and ordinal data) were described using frequency. Unordered categories variable and dichotomous variable were assessed using Fisher’s exact probabilities test (Fisher Exact) in whole groups. Ordered categories data and ranked data were evaluated using K-W nonparametric tests followed by M-W U tests.
The Statistical Package for the Social Sciences (SPSS; SPSS 13.0; SPSS Inc., Chicago, Illinois, USA) was used for all statistical analysis.
Results
Clinical signs
Ptosis was observed at all doses of RM with dose-dependent severity, incidence, and onset time from week 2 to the end of treatment phase. This sign gradually disappeared with dosing times in 10 mg/kg group. From day 73 to the end of treatment phase, nodules were observed at injection sites in placebo microsphere group and each RM group. Brownish red eye secretion of several animals was observed in RM groups.
The above abnormalities disappeared gradually in RM groups after drug withdrawal. Till the end of recovery phase, all changes in RM groups were back to normal.
Body weight and food consumption
The effects of RM on the body weight and food consumption were differed by gender (Figures 1 and 2). Compared with females in the vehicle group, 10, 30, and 90 mg/kg RM increased the body weight from 3 to 11 weeks, 1.5 to 11 weeks, and 1 to 11 weeks, respectively. No obvious toxicological significance was observed in food consumption of females compared with the historical data of control animals, though some increase or decrease in food consumption was occasionally observed at individual time points. While compared with males in vehicle group, at the dosage of 90 mg/kg RM, body weight was decreased from 2.5 to 11 weeks and food consumption was reduced at 2.5, 4.5, 5.5, 8.5, 9.5, 10.5, and 11.5 weeks.

Mean body weights of female rats (a) and male rats (a) of RM group by intramuscular injection for 12 weeks. (a) Compared with females in vehicle group (–О–), 10 (–Δ–), 30 (–∇–), and 90 (–♦–) mg/kg RM increased the body weight from 3 to 11 weeks, 1.5 to 11 weeks and 1 to 11 weeks, respectively. (b) Dosage of 10 and 30 mg/kg RM did not affect the male body weight. However, body weight was decreased by 90 mg/kg RM from 2.5 to 11 weeks and was also reduced on 2.5, 4.5, 5.5, 8.5, 9.5, 10.5, and 11.5 weeks. RM: risperidone-loaded microsphere.

Food consumption curves for female (a) and male (b) rats treated with different doses of RM. (a) Compared with females in vehicle group (–О–), increased food consumption was observed on 0.5 week at 10 mg/kg (–Δ–), 3.5, 6.5, and 9.5 weeks at 30 mg/kg (–∇–), and 0.5 week at 90 mg/kg (–♦–), decreased food consumption was noted at 10 weeks at 90 mg/kg. (b) Compared with males in vehicle group, decreased food consumption was observed on 11.5 weeks at 30 mg/kg and on 2.5, 4.5, 5.5, 8.5, 9.5, 10.5, and 11.5 weeks at 90 mg/kg. RM: risperidone-loaded microsphere.
Hematology
The results are shown in Table 1. In females, dose-dependent increase in neutrophil granulocyte percentage and decrease in lymphocyte percentage were observed at the end of treatment phase. Changes in neutrophil granulocyte or lymphocyte count were consistent with their percentage changes. The above changes were back to normal after drug discontinuation. So these changes were considered to be related with RM. In males, decreased lymphocyte count was found in 90 mg/kg RM group at the end of treatment phase. This change was dose dependent and considered to be RM related.
Selected hematological parameters.

aCompared with vehicle group, significant difference is found at p < 0.05).
bCompared with placebo microsphere group, significant difference is found at p < 0.05.
Clinical chemistry
As shown in Table 2, potassium concentration was apt to decrease and was significantly reduced in 90 mg/kg male group. It recovered at the end of recovery phase. This change was considered to be caused by RM. Other changes such as decrease in urea, sodium, or chloride were due to experimental fluctuation. An increase in lipase was also observed in 90 mg/kg male group at the end of treatment phase and recovery phase. However, no lipase-related histopathologic finding (pancreatic or gastrointestinal changes) was noted. The increase in lipase had no biological significance.
Selected clinical chemistry parameters.

aCompared with vehicle group, significant difference is found at p < 0.05).
bCompared with placebo microspheres group, significant difference is found at p < 0.05.
Bone marrow smear
No significant changes in the measured bone marrow parameters except a decrease in basophilic granulocyte percentage in 90 mg/kg group was observed compared with those in vehicle group at the end of treatment phase and recovery phase (data not shown).
Organ weight
Selected organ weight data are shown in Tables 3 and 4. The main findings were changes in the reproductive system. In females, absolute and relative weight of uterus decreased in all RM-treated groups at the end of treatment phase. Absolute and relative weight of adrenal gland decreased at 90 mg/kg. In males, decreases in absolute and relative weight of kidney were observed in all RM-treated groups. Increases in absolute and relative weight of adrenal gland were observed in 30 and 90 mg/kg groups. Other small differences that existed were attributed to the fluctuations in absolute weight of organs or body weight.
Selected absolute organ weight in rats (g).

aCompared with vehicle group, significant difference is found at p < 0.05.
bCompared with placebo microspheres group, significant difference is found at p < 0.05.
Selected organ to body weight ratio (g/100 g body weight).
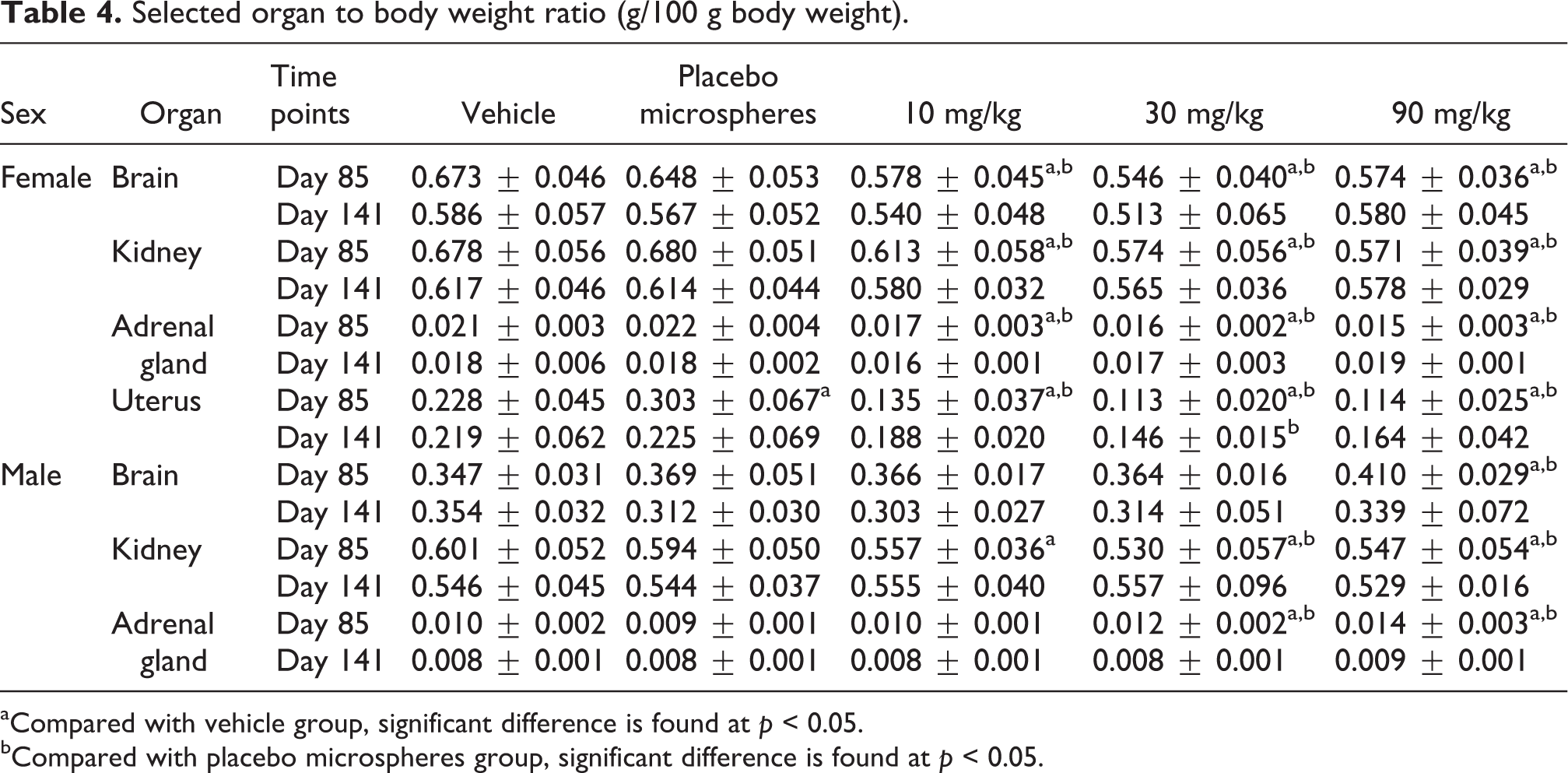
aCompared with vehicle group, significant difference is found at p < 0.05.
bCompared with placebo microspheres group, significant difference is found at p < 0.05.
Gross pathology
At the end of treatment phase, gray nodules were observed in the muscle at the injection site of almost all the animals in placebo microspheres and in RM groups. Brownish yellow nodules could still be found in the injection site except 10 mg/kg group at the end of recovery phase. With the exception of changes in the injection site, no test article–related abnormality was observed in brain, heart, liver, spleen, kidney, and reproductive systems.
Histopathology
Treatment-related histopathologic findings are shown in Tables 5 and 6 and Figures 3 to 7. At the end of treatment phase, minimal to marked foreign body granuloma in the muscle at the injection site and around the sciatic nerve was found in placebo microspheres and in all RM groups, and the incidence and severity were dose related to microspheres (content of PLGA). Dose-related hypertrophy/hyperplasia (minimal to moderate) in interstitial glands of ovary, slight to minimal atrophy of uterus (including cervix) and vagina and minimal to moderate mucinous metaplasia of epithelial cells were found in all RM female groups. Minimal to moderate atrophy of seminal vesicle and increased incidence of prostate inflammation were found in males at 30 mg/kg and above. Minimal to moderate hyperplasia of mammary glands and minimal to marked lactation were found in all RM groups. Dose-related increase (minimal to slight) of cell secretory granules in the intermediate lobe of pituitary gland was found in all RM groups. Increased red pulp macrophages of spleen were observed in all RM groups. In addition, an obvious increase in bone marrow adipocytes of femur was found in 90 mg/kg group.

H&E-stained histologic sections of muscle at the injection site (40×). Muscle sections were from vehicle group (a), placebo microspheres group (b), and 90 mg/kg RM group (c). In (a), the morphology of normal skeletal muscle is confirmed with the H&E stain at the injection site in the vehicle group. Arrows in (b) and (c) showed a large number of extracellular empty vesicles at the injection site in the muscle in placebo and RM groups. H&E: hematoxylin and eosin; RM: risperidone-loaded microsphere.

H&E-stained histologic sections of ovary ((a) to (d), 100×) and vagina ((e) to (h), 400×). Ovary and vagina sections were from vehicle group (a), 10 mg/kg (b), 30 mg/kg (c), and 90 mg/kg (d) RM groups. Solid arrows in (b), (c), and (d) showed hyperplasia in interstitial glands of ovary. In (e), the vagina was lined by keratinized stratified squamous epithelium (solid arrow). The lamina propria was constituted of dense connective tissue, rich in cells and blood vessels (dotted arrow). Solid arrows in (f), (g), and (h) showed atrophic lamina propria of vagina compared with (e). Dotted arrows in (f), (g), and (h) showed mucinous metaplasia of epithelial cells. H&E: hematoxylin and eosin; RM: risperidone-loaded microsphere. H&E: hematoxylin and eosin; RM: risperidone-loaded microsphere.

H&E-stained histologic sections of mammary gland (100×). Mammary gland sections were from vehicle group (a), 10 mg/kg (b), 30 mg/kg (c), and 90 mg/kg (d) RM groups. In (a), mammary gland tissue consists of ducts that was separated by adipose and fibrous connective tissue stroma. Solid arrows in (b), (c), and (d) showed minimal to moderate lobular hyperplasia of mammary glands. H&E: hematoxylin and eosin; RM: risperidone-loaded microsphere.

H&E-stained histologic sections of pituitary (400×). Pituitary sections were from vehicle group (a), 10 mg/kg (b), 30 mg/kg (c), and 90 mg/kg (d) RM groups. In (a), normal pituitary gland cells were observed in the vehicle group. Arrows in (b), (c), and (d) showed increase in secretory granules in the intermediate lobe of pituitary gland. H&E: hematoxylin and eosin; RM: risperidone-loaded microsphere.

H&E-stained histologic sections of spleen (400×). Spleen sections were from vehicle group (a), 10 mg/kg (b), 30 mg/kg (c), and 90 mg/kg (d) RM groups. In (a), a few macrophages were found in the red pulp (arrow). In (b), (c), and (d), arrows showed much more macrophages in red pulp of spleen. No dose relationship was found in RM-treated groups. H&E: hematoxylin and eosin; RM: risperidone-loaded microsphere.
Histopathologic findings in rats (day 85).a

aGrades: 0 = normal, 1 = minimal, 2 = slight, 3 = moderate, 4 = marked, and 5 = severe.
Histopathologic findings in rats (day 141).

Grade: 0 = normal, 1 = minimal, 2 = slight, 3 = moderate, 4 = marked, 5 = severe.
Some spontaneous lesions were observed in the study. Minimal hepatocellular vacuolation was sporadically found in vehicle or treatment group. Pituitary cyst was found in one male rat in vehicle group and one male rat in placebo microsphere group. Chronic progressive nephrosis was found in one male rat in placebo group. Red blood cell cast was observed in one female rat in vehicle group. All the incidence rates were very low and no dose relation was observed. Therefore, above changes were categorized as spontaneous lesions. Color changes in pancreas and liver and changes in weight of brain, kidney, and adrenal gland were observed in the study. However, no changes were observed in the corresponding tissues by histopathologic examination. These changes might be not related with RM.
At the end of recovery phase, all the above findings returned to normal except foreign body granuloma observed in the muscle at the injection site in placebo microsphere groups (30 and 90 mg/kg).
Prolactin
At the end of treatment phase, serum prolactin level was elevated in 90 mg/kg male group as shown in Table 7. The result of immunohistochemical detection suggested that the pituitary prolactin levels in 30 and 90 mg/kg groups were obviously higher than those in vehicle and placebo microsphere groups at the end of treatment phase as shown in Figure 8.

Immunostaining for prolactin in the pituitary (200×). Prolactin sections were from vehicle group (a), placebo microsphere group (b), 10 mg/kg (c), 30 mg/kg (d), and 90 mg/kg (e) RM groups. Arrows showed that the prolactin positive cells were stained yellow. The sections were weakly stained by prolactin antibody in vehicle and placebo groups ((a) and (b)). Pituitary prolactin staining in 30 mg/kg (d) and 90 mg/kg (e) RM groups was obviously stronger than those in vehicle and placebo microsphere groups. RM: risperidone-loaded microsphere.
Serum prolactin level (nanogram per milliliter).
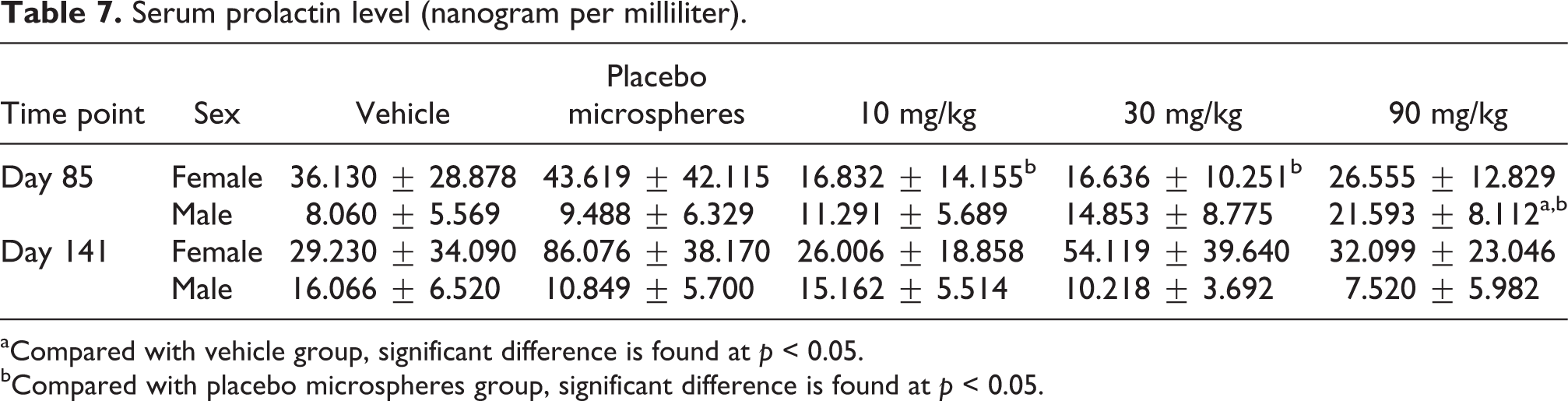
aCompared with vehicle group, significant difference is found at p < 0.05.
bCompared with placebo microspheres group, significant difference is found at p < 0.05.
TKs
The TK parameters are presented in Table 8 and Figure 9. The TK study results revealed that RM provided an initial immediate release and then a sustained release from 1 day to 21 days after injection. The drug exposure levels were estimated using area under the curve (AUC) method and the maximum concentration (C max) of the active moiety increased in a dose proportional manner for 10, 30, and 90 mg/kg groups following the first, third, and sixth doses of RM. With the advancement of treatment period, the AUC for both sexes presented with a trend of increase. The ratios of AUC(0–t) and C max between the third or sixth dosing and the first dosing were larger than 1 (2.4–8.1). There was no significant gender difference in active moiety exposure for rats. Mean time to reach peak concentrations (T max) was shortened with an increase in dosing times.

Mean plasma concentration–time profiles of active moiety in SD rats. Rats were treated with 10 (–♦–), 30 (–, –), and 90 (–Δ–) mg/kg RM. (a), (c), and (e) The first, third, and sixth dose in males. (b), (d), and (f) The first, third, and sixth dose in females. Initial immediate release and then a sustained release from 1 day to 21 days after injection were observed. C max of the active moiety increased in a dose proportional manner for 10, 30, and 90 mg/kg groups. No significant gender difference was observed in active moiety exposure for rats. SD: Sprague Dawley; RM: risperidone-loaded microsphere; C max: maximum concentration of active moiety.
TK parameters in rats (risperidone + 9-OH-risperidone).

TK: toxicokinetic; AUC: area under the curve; C max: maximum concentration of active moiety; T max: mean time to reach peak concentration.
Discussion
Long-acting injectable antipsychotics were developed with characterization by increased compliance and reduced side effects over oral antipsychotics. 13–15 Long-acting risperidone has been launched to treat schizophrenia and bipolar I disorder. However, the long-acting risperidone needs oral supplementation of antipsychotic medications. This results in inconvenience for the patients and doctors. In the present study, another long-acting injectable formulation of RM provided an initial immediate release and then a sustained release from 1 day to 21 days after injection. This would simplify the dosage regimen by eliminating the oral antipsychotic medication in the first 3 weeks of the therapy. In the present study, subchronic toxicity of RM was also evaluated in rats.
Ptosis was observed at all doses of RM with dose-dependent severity, incidence, and onset time. It was already reported that antipsychotic medication such as clozapine, quetiapine, and risperidone could induce palpebral ptosis. 16 This sign was related to α-adrenergic blockade of risperidone. 17,18 An increase in body weight was found in the females treated with RM in this study. It was commonly reported that antipsychotics resulted in body weight gain. 19–21 In this study, decrease in body weight and food consumption of males at high doses induced by RM was also observed. The weight change had no relationship with dopamine D2 receptors. 22 Many atypical antipsychotic drugs cause weight change. 23,24 Most evidence indicates that antipsychotics induced weight gain include investigations of the histamine H1, 25 serotonin 5-HT2A, 26 and 5-HT2C 27 receptors. However, no clear mechanism is the cause of weight gain liability with risperidone. It must be a multifactorial phenomenon.
Brownish red eye secretion of RM-treated rats gradually disappeared after drug discontinuation. Moreover, no secretion was found in the animals that received vehicle and placebo microspheres. Therefore, the sign was considered to be related with RM.
RM could elevate serum and pituitary prolactin levels by prolactin detection and immunohistochemical detection. These changes were attributed to the antagonism of dopamine D2 receptors. 28,29 The elevated prolactin induced by risperidone resulted in the mammary lobular hyperplasia and secretion and reproductive system changes including uterus and vagina atrophy together with epithelium mucification, endocrine, seminal vesicle atrophy changes, and so on. Apart from these, the incidence and severity of inflammation of prostate were obviously increased in RM-treated groups except 10 mg/kg group. The possibility that this was RM related could not be excluded.
Lymphopenia induced by risperidone and paliperidone was found in only a few case reports. 30 The incidences were seemed to be very low. In the present study, the decrease in lymphocytes recovered after drug discontinuation. No corresponding compensation was found in bone marrow parameters. This indicated that lymphopenia was minor. Neutropenia has been sporadically reported in humans. 30,31 No increased neutrophil count or percentage induced by risperidone was reported as far as we can determine. The mechanism of neutrophil changes needed more research. At the end of treatment phase, a slight decrease in the concentration of potassium for animals in high-dose group was measured. Hypokalemia is a well-known toxic effect of risperidone. 32 There is a risk to induce QT interval prolongation in patients, which is associated with paroxysmal ventricular arrhythmia. Decreases in potassium, urea, and sodium suggested that urine volume and specific gravity were affected. Though urinalysis was not carried out in this study, the previous toxicity study indicated that risperidone could slightly increase urine volume and decrease specific gravity. 33 It might be a result of the integrated effect of decreases in urea, potassium, and sodium for the impairment of the countercurrent multiplier system. 34 Most of the solute-free water reabsorbed by the kidney is dependent on the concentration of urea. 35 The decreased urea and potassium result in increased urine volume.
At the end of treatment phase, changes in absolute or relative weight of kidney and adrenal gland were recorded in all RM-treated male groups. The changes were reversible. Although we could not exclude the possibility that the changes were RM related, no histopathologic finding in kidney and no change in renal function parameters were found. Similarly, no abnormality was observed in adrenal gland by histopathologic examination.
Compared with vehicle group, nodules and foreign body granuloma were observed in placebo microspheres group. These changes must be caused by the material of microspheres, PLGA. PLGA is considered to be safe and used in many drugs such as long-acting risperidone, naltrexone injection, and triptorelin. PLGA is rapidly accumulated in reticuloendothelial system, including the macrophages of the spleen and bone marrow. 36 In the present study, nodules were observed in the muscle at the injection site of animals in RM and placebo microsphere groups. Histopathologic examination showed that foreign body granuloma in the muscle at the injection site and around the sciatic nerve was found in placebo microspheres and in all RM groups. These suggested the microspheres deposited in the muscle for a long time and formed the granuloma. Multinuclear macrophage accumulation at the periphery of granuloma was generated by stimulus of chronic inflammation, which might be related to the degradation of microspheres. At the end of recovery phase, the severity of granuloma was relieved, suggesting the microspheres could be degraded and eliminated gradually in the muscle. Minimal to slight increase in macrophages in red pulp of spleen was found in vehicle, placebo microsphere, and RM groups. Compared to vehicle and placebo microsphere groups, an obvious increase in incidence and severity of the macrophage accumulation in red pulp was detected in all RM groups. We could not exclude the possibility that RM could aggravate the pathological change in spleen. In the present study, the safety of RM was well established in the subchronic toxicity study in rats.
Footnotes
Acknowledgments
The authors thank the staff of CMC Department in Luye Pharma for providing the RM and help.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by the National Natural Science Foundation of China (no. 81001661), the Shandong Province Young and Middle-Aged Scientists Research Awards Fund (no. BS2009SW012), Taishan Scholar Project, and the National Basic Research Program of China (no. 2012CB724003).
