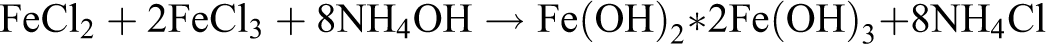Abstract
Aqueous suspensions of 10 nm, 50 nm, or 1 μm Fe3O4 particles were injected intraperitoneally (ip) to rats at a dose of 500 mg/kg in 4 mL of sterile deionized water 3 times a week for 5 weeks. Following exposure, functional and biochemical indices and histopathological examinations of spleen and liver tissues of exposed rats were evaluated for signs of toxicity. The iron content of the blood was measured photometrically, and that of the liver and the spleen by atomic adsorption spectroscopy (AAS) and electron paramagnetic resonance (EPR) methods. It was found that, given equal mass doses, Fe3O4 nanoparticles possess considerably higher systemic toxicity than microparticles, but within the nanometric range the relationship between particle size and resorptive toxicity is intricate and nonunique. The latter fact may be attributed to differences in different nanoparticles' toxicokinetics, which are controlled by both more or less substantial direct penetration of nanoparticles through biological barriers and their unequal solubility.
Introduction
According to theoretically sound perception 1 –3 and some experimental data reviewed by us recently, 4 nanoparticles (NPs) can be much more toxic as compared with particles of the same substance in the micrometric range. However, “this common perception of greater NP toxicity is based on a limited number of studies,” 5 and even less is really known about the comparative toxicity of chemically identical but having different size particles within the generally accepted nanometric range.
In our previous study, 4 it was demonstrated that the intratracheal instillation of magnetite (Fe3O4) NPs caused a higher increase in the alveolar macrophages and neutrophil leucocyte count in the bronchoalveolar lavage fluid than the instillation of 1 μm size particles. It was also shown that the cytotoxicity of 10 nm particles was higher than that of 50 nm particles, whereas both were much more cytotoxic than the 1-μm particles of the same substance.
Pulmonary clearance of NPs may establish conditions conducive to toxicity in other organs related to the dissolution and resorption, or the direct penetration of the NPs into the blood. Nanoparticles not cleared out very soon after aerosol exposure and thus chronically retained in the lung tissue may also have more potential for systemic toxicity than an identical mass-based exposure to microparticles. It suggests that chronic exposure to nano-aerosols creates more prerequisites for the systemic toxicity than similar exposure to micro-aerosols of the same chemical substance. In this connection, it is especially important to know whether such toxicity is identical for NPs of different size.
As the object of this study, we selected an iron oxide because of the low systemic toxicity and cytotoxicity of micrometer particles of iron and its oxides. We viewed this circumstance as essential in light of the important—from the standpoint of occupational and environmental medicine—question of whether the toxicity of substances in nano-disperse state is enhanced so much as to render hazardous even a substance that is biologically inert as a bulk material.
By way of considering these problems, we carried out experiments with the same, as in our previous study, 4 3 batches of magnetite (Fe3O4) particles having different primary sizes: 10, 50, and 1000 nm (1 μm). However, for studying the comparative subchronic toxicity of these samples, we decided to evade the issue of the role of the intricate dependencies on different physiological and other mechanisms underlying the kinetics of particle elimination from or retention in the lung tissue. These dependencies would have substantially complicated the interpretation of effects at organism level. Meantime, we wanted to reveal those differences in comparative systemic toxicity that are associated with the mechanisms of toxicokinetics specific for NPs (such as active dissolution and resorption from the primary depot; transfer of not only dissolved material but also NPs as such to remote organs by blood; secondary retention of migrating NPs in these organs, first of all, in those rich with reticuloendothelial system, (RES) cells; their possible dissolution in these secondary depots). Therefore, it was desirable to have an experimental model in which the mass of the material in the primary depot would be strictly set and equal for substances compared. From this standpoint, a sufficiently adequate experimental model for subchronic toxic exposure to particles of different size compared seems to be repeated intraperitoneal (ip) injections of their suspensions in sublethal doses during a period which is long enough relative to rat’s life span.
Certainly, it should be borne in mind that barriers through which particles penetrate into the blood from the lungs and from the peritoneal cavity are different anatomically and functionally, and these differences can be reflected on NPs' toxicokinetics. We may assume, however, that this fact does not create any substantial bias for comparative estimation of the capacity of particles of different size for such penetration and migration to remote organs. It should be noted that intraperitoneal administration was used for studying the resorptive toxicity and bioaccumulation of some NPs by other authors as well. 6,7
Materials and Methods
Synthesis of Nano-and Microparticles of Fe3O4
To obtain magnetic NPs with given dimensions, we used synthesis in “nano-reactors"/micelles. 8 Strict dosing of iron salts makes it possible to vary NP size and obtain NPs with a sufficiently narrow particle size distribution.
The synthesis was conducted by the following reaction:
The mixture was heated to a temperature of 60°C on a water bath with intensive stirring. Particles were settled by slow, “drop-by-drop” addition of microemulsion 2 to microemulsion 1. The reaction lasted 4 hours at the temperature of 60°C. The particles thus obtained were deposited by centrifugation at 8000 rpm and roasted at a temperature of 600°C. The 50 nm particles were obtained by the same process using cetyl trimethyl ammonium bromide (CTAB) as SAA.
The NP size and phase distribution was checked by electronic microscopy (FEI MORGAGNI 286 (D) (US) and Philips CM 300 (the Netherland) electronic microscopes). As can be seen from the histograms (Figure 1), in both cases the dominant particle size by diameter really falls within the nominal value, that is 10 nm or 50 nm, with the particle size distribution in a narrow range around this value. Note, in particular, that in the nominally 10 nm material there were only 8.9% of 50 nm or larger particles, and in the nominally 50 nm material only 4.4% of particles under 20 nm in diameter. The magnetic properties of the NPs were confirmed by the Faraday balance method, and the magnetization amounted to 82 emu/g.
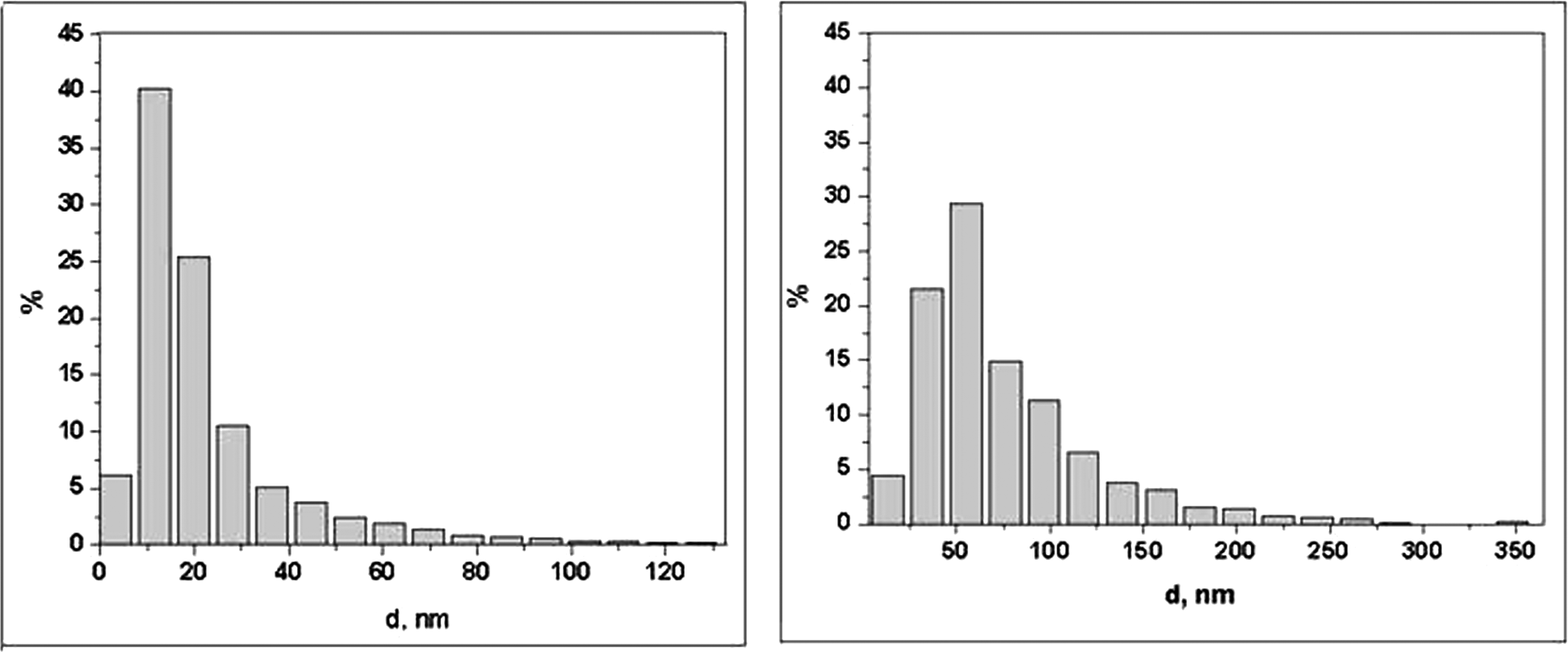
Particle size distribution by diameter in the electronic microphotography of nano-magnetite with the nominal diameters of 10 nm (A) and 50 nm (B). The total number of particles measured was 3332 for (A) and 3301 for (B).
Magnetite microparticles were obtained by mechanically grinding for 2 minutes the agglomerates obtained by storing 50 nm NPs for 30 days. Then the 1-μm class of particles was separated with the help of a set of sieves. Particle size was checked by optical microscopy. The phase distribution of the microparticles was checked by x-ray phase analysis (a “Dron-2.0” diffractometer, Cu Kα radiation), and the magnetization by the Faraday balance method.
The method for preparing suspensions (sols) of NPs for toxicological experiments was designed based on the results of research into the kinetics of aggregate formation in micrometric and nanometric particle suspensions in distilled water and in normal saline. This research was carried out by the method of dynamic light scattering on a Brookhaven ZetaPlus universal analyzer of suspensions. It was established that nano-sols of Fe3O4 are not stable, and the kinetics of the process of aggregation of a disperse phase depends strongly on the dispersion medium. Water-based sols proved more stable; and for 3 to 5 minutes after stopping their exposure on a VCX-750 ultrasonic processor (Sonics and Materials, Inc., Newtown, CT, USA), the NP aggregation process in theme was less dynamic than in sols on the basis of normal saline. In particular, 3 minutes after ultrasonication (the first time point for reading the analyzer) of 10 nm particles in normal saline, the weighted average diameter of an aggregate was equal to 150 nm, whereas in distilled water it was 96 nm. When our toxicological study had been completed, we repeated the tests only with aqueous suspensions and found that the weighted average diameter of aggregates in the nominally 10 nm suspension was 133 nm while in the nominally 50 nm oneit was 240 nm (with another analyzer, Malvern Zetasizer Nano-ZS these values were even approximately 2 times lower
Design of the Animal Experiment
This experiment was carried out on outbred white female rats (from our own breeding colony) with the initial body weight of 150 to 220 g, 12 animals in each exposed and control group. All rats were housed in conventional conditions, breathed unfiltered air, and were fed standard balanced food and clean bottled water. The study was planned and implemented in accordance with the “International guiding principles for biomedical research involving animals” developed by the Council for International Organizations of Medical Sciences (1985).
The NPs studied were administered ip as suspensions in sterile deionized water because, as indicated in the prior discussion, in normal saline suspensions the rate of agglomeration is too high. The suspensions were administered to rats after preliminary ultrasonication for 20 minutes in 10-second periods at 10-second intervals between theme under continuous cooling of the suspension to keep the temperature under 60°C. Then ultrasonication of the suspension was continued in the same regime, being interrupted only for drawing 4 mL of the suspension into a syringe for ip injection. Preliminary training of experimenters ensured the speed and coordination of all manipulations such that the time from the short interruption of the ultrasonication process to the end of injecting was 20 to 44 seconds (28 seconds on average).
Preliminary tests showed that a single ip injection at a dose of up to 2000 mg/kg of body mass of either nano- or microparticles of magnetite provoked no death in rats within 14 days of observation, though after an injection of NPs at a dose of 1000 or 2000 mg/kg, we observed statistically significant shifts (in comparison with control values) in some of the indices that were subsequently used for estimating subchronic toxicity. Meanwhile, when a single dose was decreased to 500 mg/kg, it caused no such shifts in response to even as many as 10 repeat injections of any of the substances compared (3 injections a week). We chose just this dose, easily tolerated by animals in the context of a short-term exposure, for carrying out our principal experiment in which the aqueous suspensions containing 10, 50, or 1000 nm (1 μm) particles of magnetite were administered to rats ip at a dose of 500 mg/kg, 4 mL, 3 times a week for 5 weeks, and control animals were administered 4 mL of sterile deionized water in the same manner.
After the exposure period, the following procedures were performed for all rats: weighing; estimation of central nervous system (CNS) ability to the temporal summation of subthreshold impulses—a variant of withdrawal reflex and its facilitation by repeated electrical stimulations in intact, conscious rat
9
; recording of the number of head dips into holes of a hole board, which is frequently used for studying behavioral effects of toxicants and drugs (eg ref 10,11); collection of daily urine for analysis of its density, urine output, coproporphyrin, δ-aminolevulinic acid (δ-ALA), and creatinine contents; sampling of capillary blood from a notch on the tail for examining the hemogram, hemoglobin content, and for cytochemical determination of succinate dehydrogenase (SDH) activity in lymphocytes (by the reduction of nitrotetrazolium violet to formazane, the number of granules which is counted under immersion microscopy).
Then rats were killed by decapitation and blood was collected by exsanguination. Biochemical indices determined from serum included total protein, albumin, globulin, bilirubin, ceruloplasmin, malonyldialdehyde (MDA), total iron, alkaline phosphatase, alanine and aspartate transaminases (ALT and AST), catalase, γ-glutamine transferase. For 4 rats in each treatment and control group, liver and spleen tissue sections were prepared for histologic examination by staining with hematoxylin−eosin and by Perl iron stain (based on the so-called Prussian blue reaction). Livers and spleens of the other animals were homogenized for the measurement of iron contents of tissues by atomic adsorption spectroscopy (AAS) using SOLAAR M6 atomic absorption spectrograph, Thermo Scientific (Waltham, MA, USA), and by electron paramagnetic resonance (EPR) using EMX Plus spectrometer, Bruker (Germany). The iron content of the blood was measured photometrically. Histologic examinations and tissue iron content determinations were conducted in semi-blind fashion. Samples were grouped, and groups were coded without disclosure of group treatment.
Numerical data processing
In all experiments, the statistical significance of the difference between arithmetic mean values characterizing the groups under comparison was estimated using Student t test and Mann-Whitney test. However, these 2 estimates were mutually corroborating for the majority of indices; we, therefore, present in the tables as a rule only those based on Student t test. The standard error of the percentage (p) of different liver cells was first estimated as
Results
Table 1 shows treatment and control groups status summary for indices that showed statistically significant (P < .05) differences between any 2 groups. Body mass showed no significant increase over 5 weeks of treatment, indicating that doses were generally well tolerated.
Some Indices to the Status of the Organism of Rats Under Subchronic Intraperitoneal Exposure to Magnetite Particles of Different Size (x ± sx)
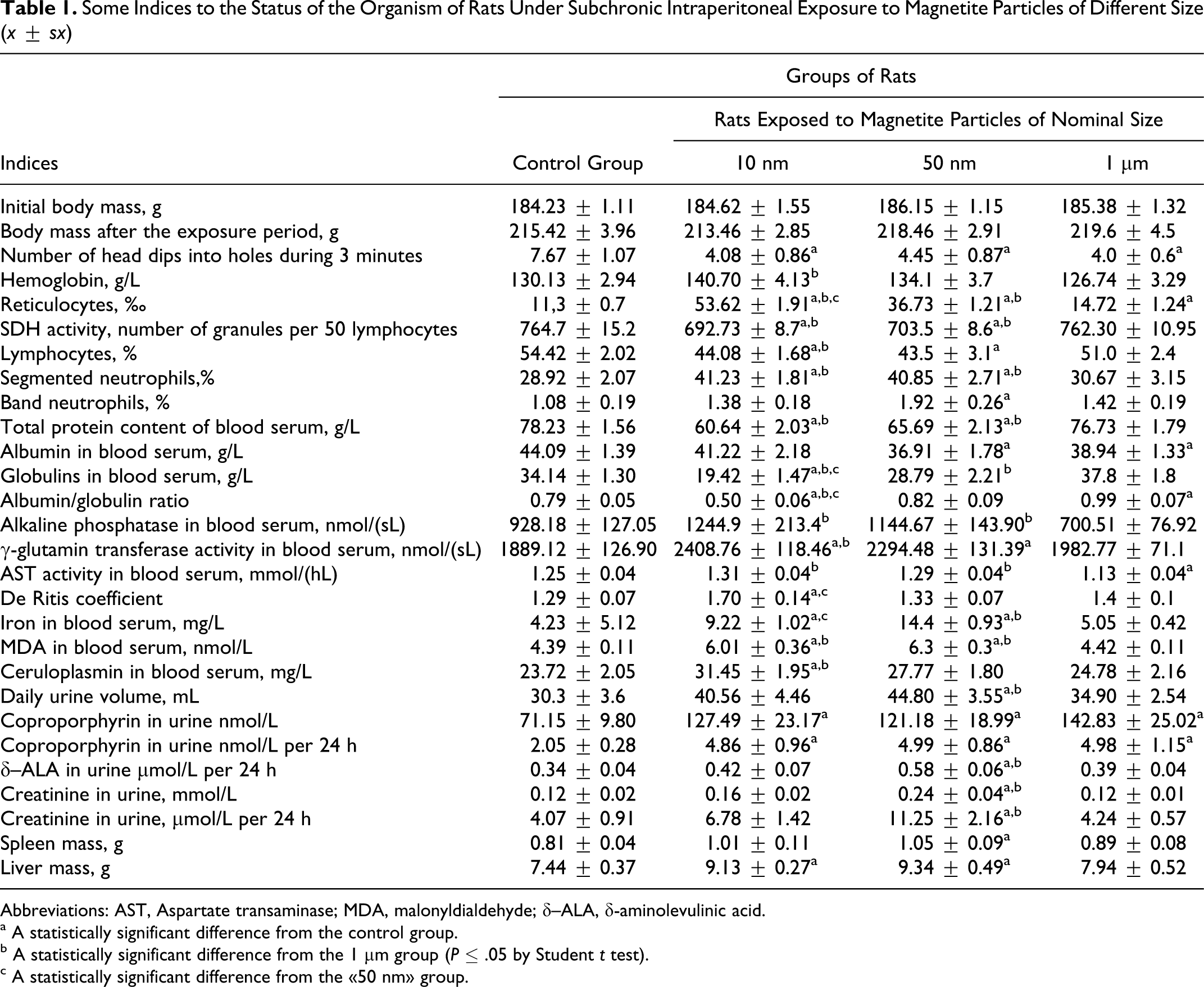
Abbreviations: AST, Aspartate transaminase; MDA, malonyldialdehyde; δ–ALA, δ-aminolevulinic acid.
a A statistically significant difference from the control group.
b A statistically significant difference from the 1 μm group (P ≤ .05 by Student t test).
c A statistically significant difference from the 50 nm group.
Estimated iron contents of the liver and spleen homogenates by AAS and EPR are shown graphically in Figures 2 and 3.
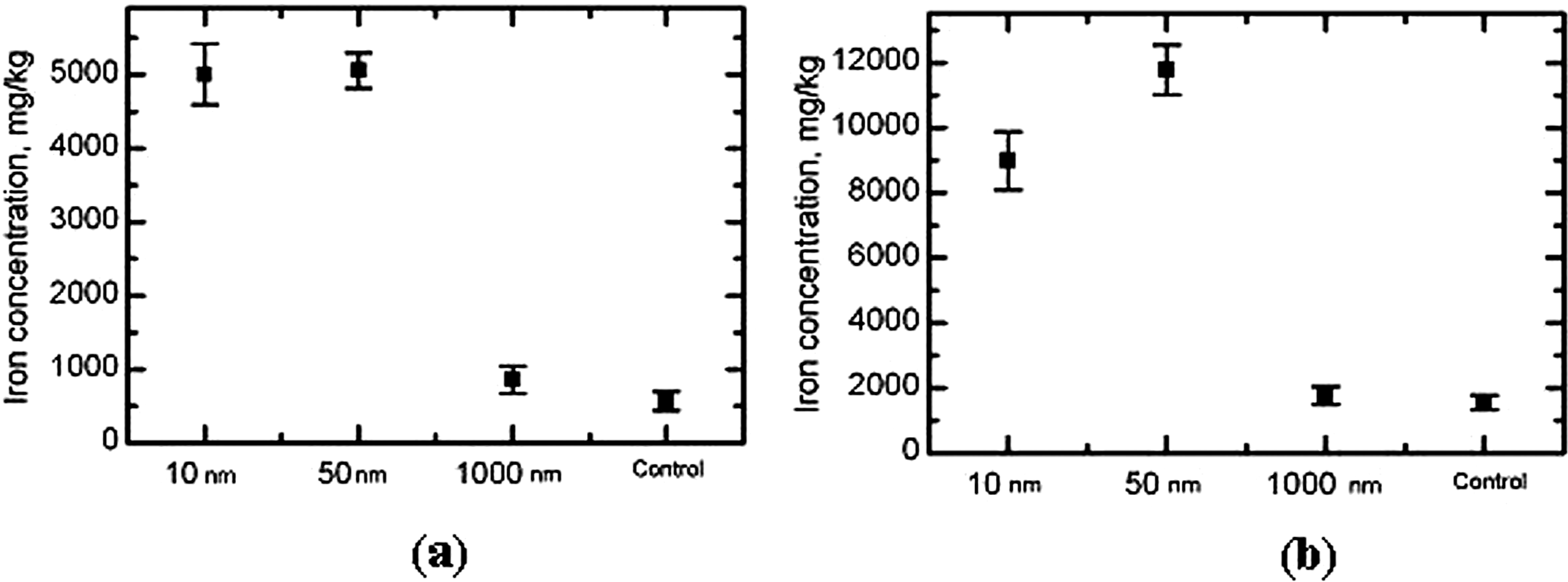
Mean value (±SD) of total iron concentration in the rat tissues of (A) liver and (B) spleen by groups of rats exposed to magnetite particles of different size. AAS method. AAS indicates atomic adsorption spectroscopy; SD, standard deviation.
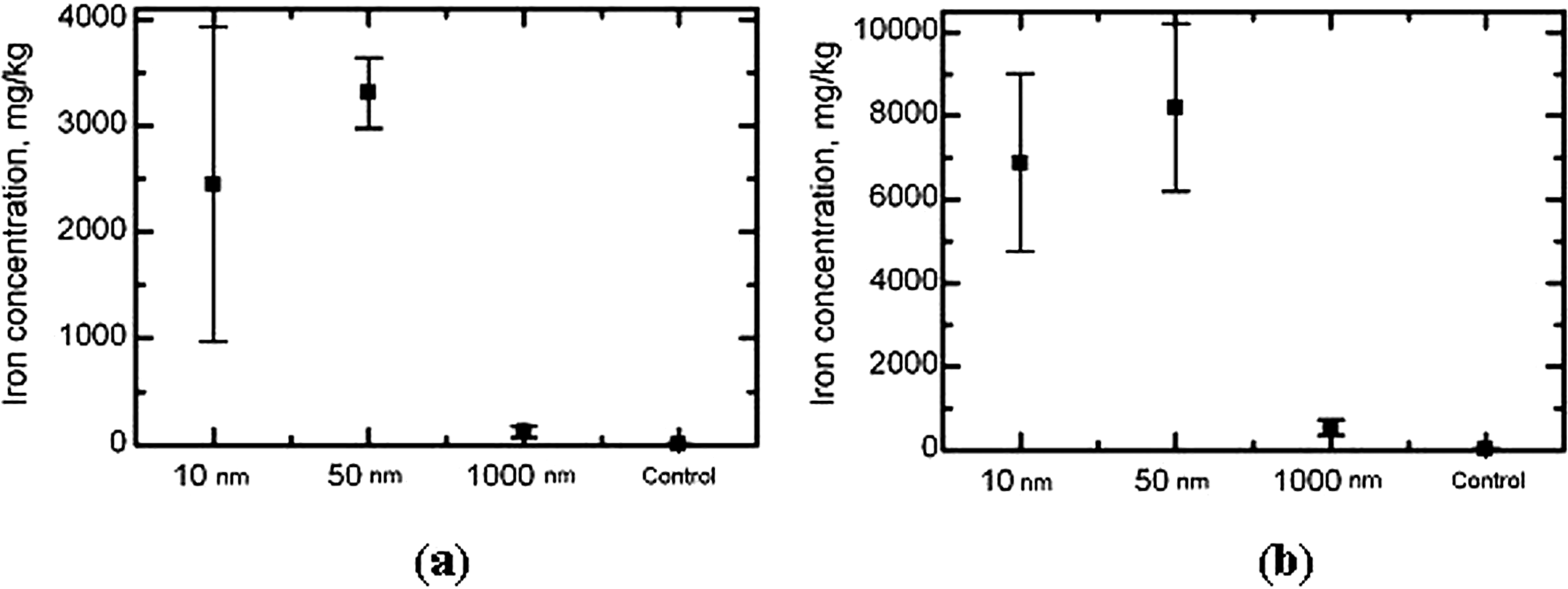
Mean value (±SD) of iron concentration in the form of magnetite in the rat tissues of (A) liver and (B) spleen by groups of rats exposed to magnetite particles of different size. EPR method. EPR indicates electron paramagnetic resonance; SD, standard deviation.
Histological examination showed that in the livers of the rats exposed to NPs the structure of the lobules was damaged; there was discomplexation of hepatic tubules due to the deposition of a large amount of iron-containing pigment as conglomerates in periportal zones, intralobularly and, occasionally, centrilobularly, and as separate particles in sinusoids. The iron-containing pigment was also detected in the cytoplasm of Kupffer cells. Hepatocytes were in a state of profound vacuolar degeneration, and their cytolysis was observable in some places. The degree and prevalence of pathological changes in response to 50 nm magnetite was a little higher than in response to 10 nm magnetite.
Changes in the liver of the rats given 1000 nm (1 μm) magnetite were minimal. The tubular structure of the hepatic lobules was not damaged, and only some of the hepatocytes were observed to have hyaline drop-like degeneration; the portal paths were intact. The sinusoid spaces were free, and solitary small granules of iron-containing pigment were observable in Kupffer cells only.
The spleen shows considerably more marked changes in response to NPs of both sizes (extensive deposits of lumps of iron-containing pigment in the red pulp, and occasionally in compressed follicles of the white pulp, in which light-colored reactive centers can be seen) in comparison with the effect of micrometric ones, for which the histological picture of this organ is little different from that in the control group. No marked differences in the response of this organ to the effect of NPs of the 2 sizes tested were observed.
The morphometric estimates are presented in Table 2 . The qualitative description of histological changes given in the preceding section is illustrated by microphotographs (Figures 4–6), comparing corresponding pictures of liver and spleen in control rats and in those given 1 μm or 50 nm magnetite (keeping in mind that there were no visually evident distinction between histological pictures in the latter rats and in those administered 1 nm magnetite).
Morphometric Characteristics of Rat Livers
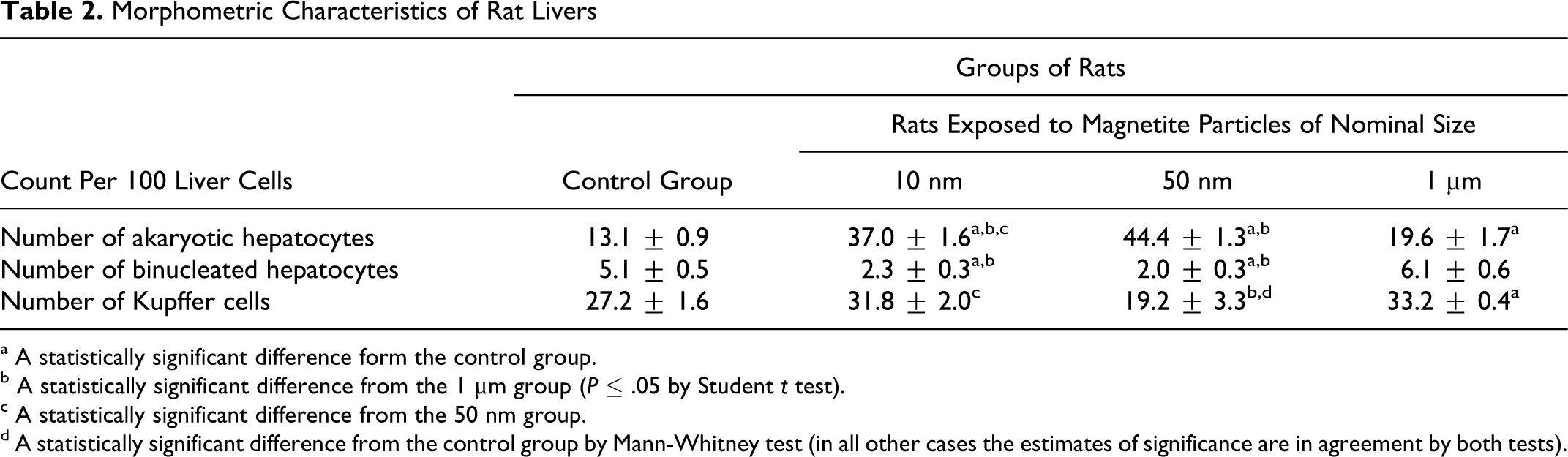
a A statistically significant difference form the control group.
b A statistically significant difference from the 1 μm group (P ≤ .05 by Student t test).
c A statistically significant difference from the 50 nm group.
d A statistically significant difference from the control group by Mann-Whitney test (in all other cases the estimates of significance are in agreement by both tests).
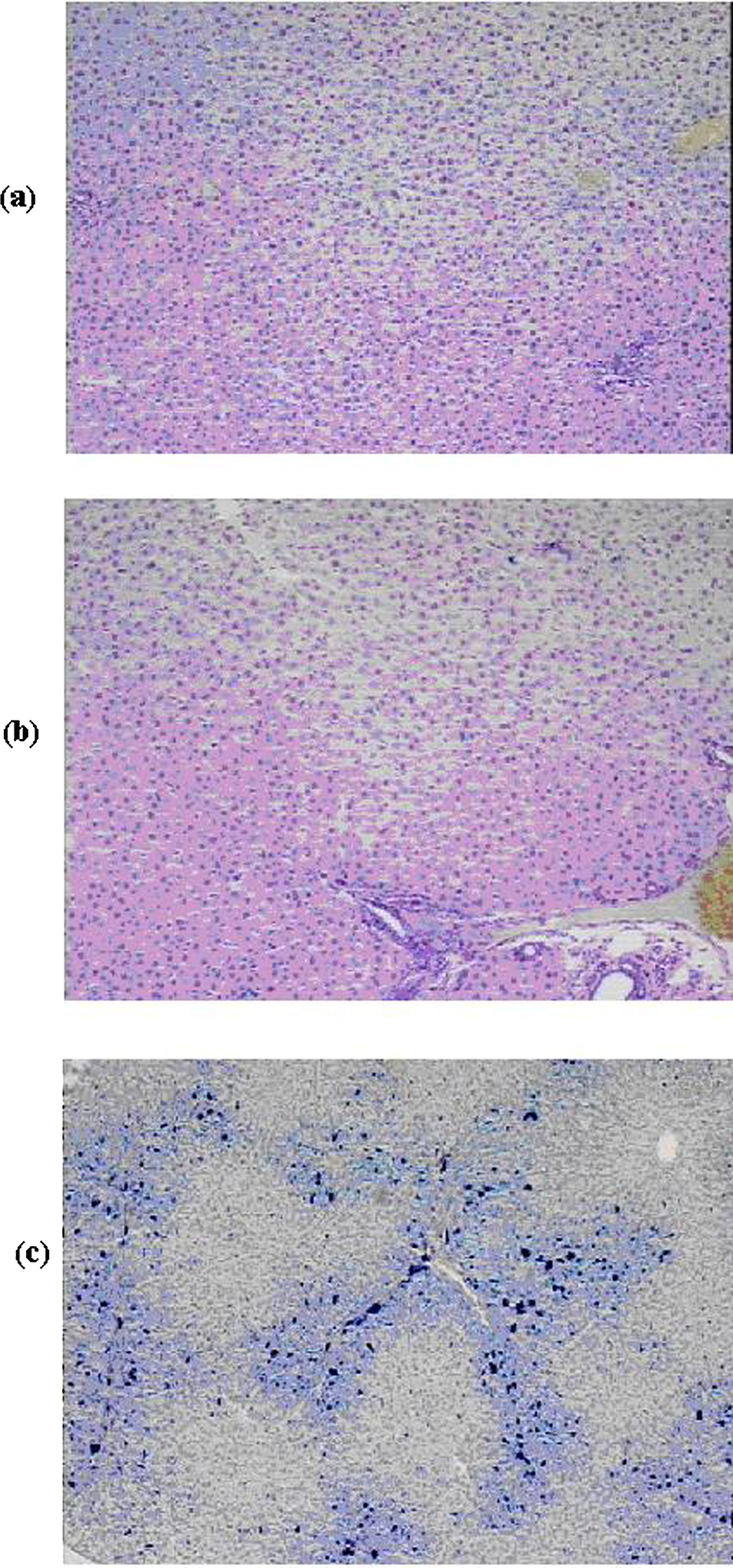
Livers of the rats in groups: (A) control, (B) administered 1 μm magnetite, (C) administered 50 nm magnetite. Perl iron stain, nuclei counterstained with hematoxylin. Magnification ×100.
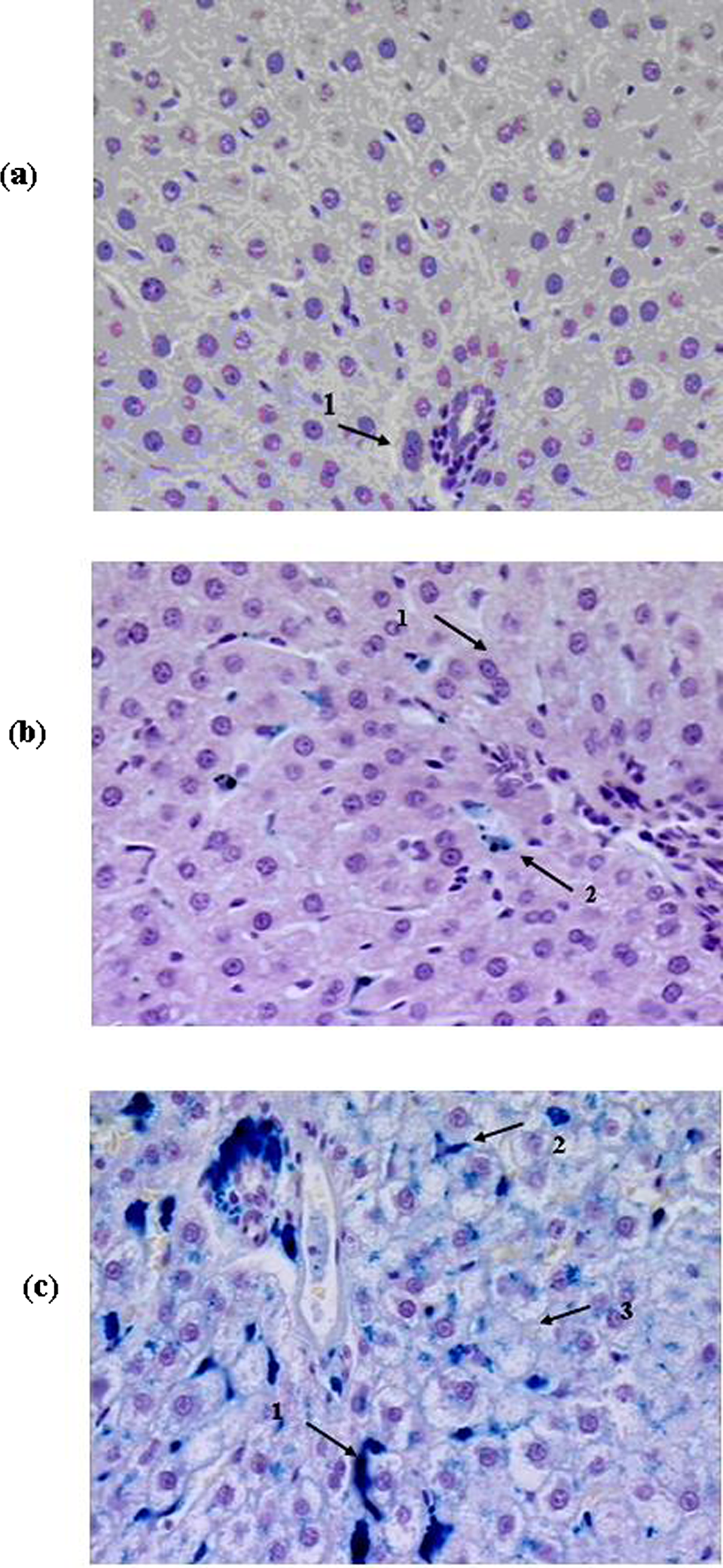
Livers of the rats in groups: (A) controls (1 = binucleated hepatocytes); (B) administered 1 µm magnetite (1 = binucleated hepatocytes; granules of iron-containing pigment in some Kupfer cells); and (C) administered 50 nm magnetite (1 = deposits of iron-containing pigment periportally and within sinusoids; 2 = the same in Kupfer cells, 3 = vacuolar degeneration and karyolysis of hepatocytes). Perl’s iron stain, nuclei counterstained with hematoxylin. Magnification ×400.
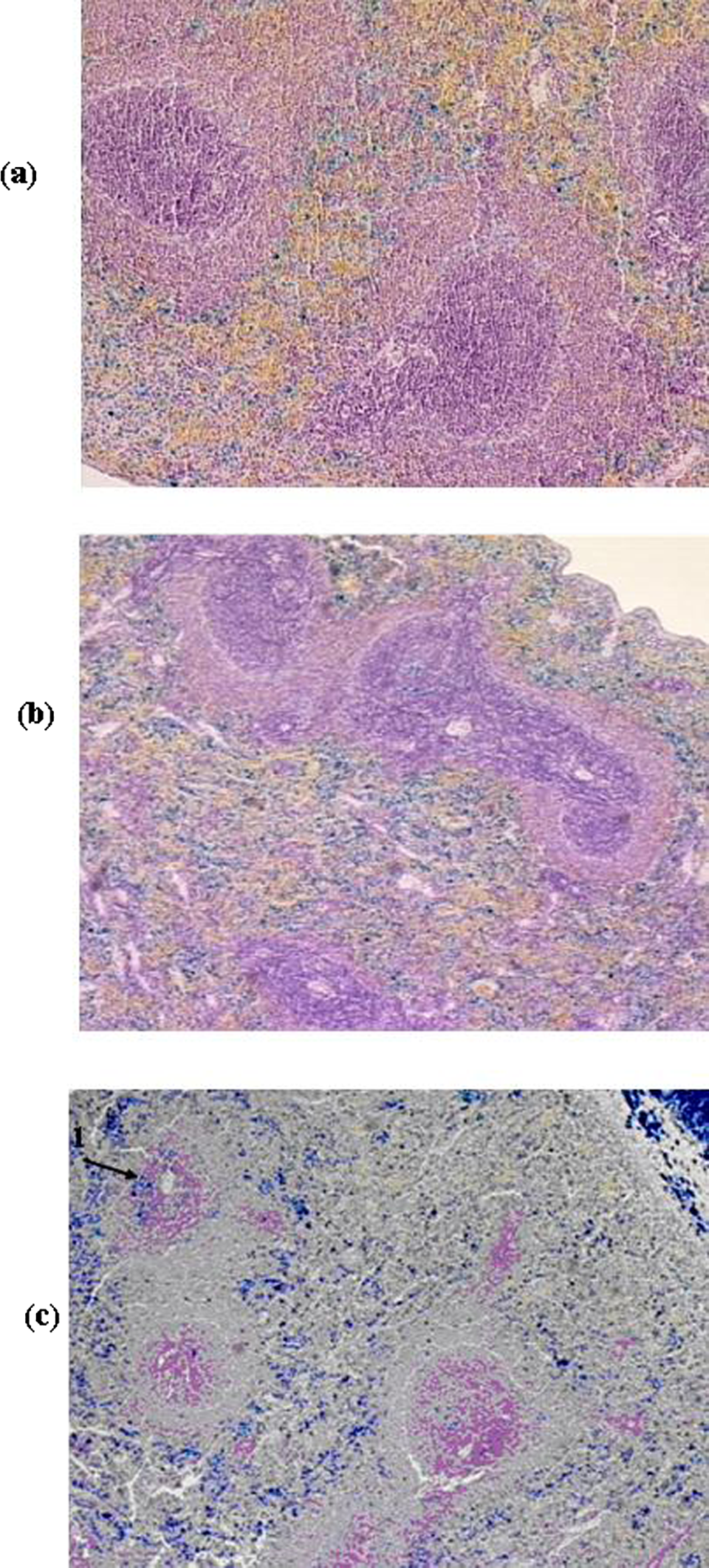
Spleens of the rats in groups: (A) controls; (B) administered 1 µm magnetite; and (C) administered 50 nm magnetite (conglomerates of the iron-containing pigment in the red pulp, hypoplasia of lymphoid follicles, 1 = intrafollicular deposits of the iron-containing pigment). Perl’s iron stain, nuclei counterstained with hematoxylin. Magnification ×100.
Discussion
In the majority of published works, any route of dosing with any NPs for the purpose of studying their systemic toxicity and bioaccumulation, whether intraperitoneal, 6,7 intravenous, 6,13 intragastric, 14 –17 or inhalational 18 –20 usually implies a single exposure (acute toxicity) or repeat exposures over a period from several days to, less often, 2 to 4 weeks (subacute toxicity). A 4-week exposure was used in only 2 of the studies mentioned here. 14,20 Such short-term studies of toxicity are necessary and sufficient in cases where the researcher is interested, first of all, in estimating the hazard of medical or diagnostic use of a specific nanomaterial for the patient exposed to it. However, where the problem under research is some general patterns in the development of adverse effects of a chronic exposure to NPs, it is desirable to make the exposure as long term as it may be possible without causing any appreciable harm to the animal by repeat manipulations themselves. Our long-standing experience showed that repeat ip injections, if administered to rats as frequently as up to 3 times a week, might be given for 5 to 7 weeks. That was why we decided to inject rats ip with different magnetite suspensions 15 times during 5 weeks. The results of this study seem to have confirmed adequacy of such experimental model of a relatively moderate subchronic intoxication.
Judging from the majority of the indices shown in Table 1, the 50-nm magnetite particles cause more marked statistically significant negative shifts as compared with control than 1 μm particles do. These shifts were, as a rule, even greater in response to 10 nm magnetite. The exception constitutes shifts in iron content of the blood serum, daily urine output, creatinine in urine, liver mass, and spleen mass, for which the 50 nm group is the leader.
The majority of changes are nonspecific in character, being rather a reflection of many sided disturbance of homeostasis on organism level—what Nikolay Lazarev the eminent Russian toxicologist and pharmacologist 21 called integral indices of intoxication. At the same time, the response of the red blood might be presumably associated with the specific role of iron in hemopoiesis. Note that the administration of magnetite NPs increased not only the hemoglobin content as shown in Table 1 but also the number of erythrocytes. In the case of 10 nm magnetite, this increase (5.76 ± 0.14 × 1012 against 5.37 ± 0.12 × 1012 in controls), though not high, was statistically significant by the Mann-Whitney test. A sharp increase in the number of reticulocytes provides evidence of erythropoeisis stimulation, which in the case of the 10-nm particles seems to have reached a degree, resulting in a significant increase in total erythrocytes count.
However, this quasi-beneficial effect had developed against the background of other, undoubtedly unfavorable ones. They include not only some changes in the indices shown in Table 1 (the inhibition of the natural “exploratory” behavior; decrease in the protein content of the blood serum due, mainly, to globulins; suppression of lymphocyte SDH and enhanced lipid peroxidation; neutrophil leukocytosis; changes in the activity of some serum enzymes; increased creatinine excretion with urine at increased diuresis; increase in liver and spleen mass), but also pathological changes in the histological picture of the liver and spleen.
We have not included enhanced renal excretion of δ-ALA and coproporphyrin into this list of certainly adverse effects because we cannot rule out that it is merely a side effect of enhanced heme synthesis. However, these shifts, observable in many a pathological process, are no less likely to be associated with some disturbance of porphyrin metabolism possibly connected with the liver damage. For instance, it is assumed that the well-known alcohol porphyria is metabolically connected with damage to this organ, 22 which is also damaged under effect of magnetite NPs. In this context, of some significance may be the fact that the most pronounce increase in the δ-ALA excretion was observed in rats administered 50 nm magnetite which, as will be discussed in the following paragraphs, induced the most marked pathological changes in the liver morphology.
We might suggest that enhanced use of iron for boosting heme synthesis explained the fact that its concentration in the serum, increased in comparison with control in response to the administration 10 nm magnetite, is however significantly less than in response to the administration of 50 nm magnetite. The fact that 1 μm magnetite yielded the least and, moreover, a statistically insignificant increase in this index agrees well with the lowest solubility and penetrating ability of this material.
Indeed, there can be a transfer from the primary depot into the blood and through it into parenchymatous organs not only of the iron resorbed in ionic-molecular form but also of undissolved iron oxide NPs; this ability of direct penetration, characteristic of all NPs, being higher, the finer they are. However, having been retained from the blood in the organs rich in reticuloendothelial cells, NPs presumably continue dissolving in these secondary depots. This process should be a lot more intensive for the finest NPs by virtue of their greater total surface area and so may more markedly reduce total retention of iron in the tissue of a given organ.
Measurements of iron concentrations in liver and spleen tissues agree with these hypotheses quite well. As can be seen from Figure 2, the concentration of total iron in the liver and the spleen following ip injections of magnetite NPs as measured by AAS is much higher than in the case of micrometric particles, where it is only slightly and statistically insignificantly higher than in the organs of control rats. In both organs (but in the spleen especially) it is a little lower in response to 10 nm in comparison with 50 nm particles. At the same time, as follows from Figure 3, in the organs of both control rats and those injected 1 μm magnetite, there is virtually no iron, as measured by EPR, in the initial chemical form of the Fe3O4 (which might occur in the tissue only if transported to it while nondissolved). Meantime, in response to injection of NPs of both sizes, its contents are very high in the liver and, especially, in the spleen, in both organs being a little lower in the case of 10 μm NPs as compared with 50 nm NPs.
We chose these 2 organs as indicative for estimating the dependence of iron bioaccumulation on particle size, taking into consideration their importance for the reticuloendothelial system and some literature data on the biodistribution of other nanomaterials Thus, Kumar et al 23 who studied biodistribution in nontumored nude mice of organically modified silica NPs with diameters 20 to 25 nm conjugated with near-infrared fluorophores and radiolabeled with 124I, administered by gavage, found that their accumulation in liver, spleen, and stomach was greater than in kidney, heart, and lungs. According to a review, 24 the primary site of gold and silver NPs' bioaccumulation “has been consistently demonstrated to be the liver.” Yokel et al 13 found that 1 hour and 20 hours after intravenous administration of aqueous suspension of cerium oxide NPs (approximately 31 ± 4 nm) Ce concentrations decreased in the sequence spleen > liver > blood > brain. In our study, the iron content of the blood of the rats exposedto NPs is also much lower than in the liver and the spleen, which, thus, act as secondary depots. Furthermore, we revealed higher bioconcentration in the spleen than in the liver for both total iron and iron in magnetite form for any NP size (Figures 2 and 3). It may be that the dominant role of the spleen in the accumulation of iron is associated with its well-known special role in the normal metabolism of this essential element.
That such accumulation of iron in the tissues may be excessive and result in its well-known pathological consequences (hypersiderosis) is supported in our experiment not only by the increase in the mass of both organs as shown in Table 1 (particularly for exposure to 50 nm magnetite) but also by the histopathological changes in theme as described in the results section. The degree and prevalence of histopathological changes under the effect of 50 nm magnetite are a little higher than under the effect of 10 nm magnetite. Thus, the higher hepatotoxic effect of the former agrees quite well with the higher accumulation of these NPs in the organ. As for changes in the liver of the rats administered 1 μm magnetite, they were minimal, which may be linked to both insignificant transfer of this material into the liver and its lower cell and tissue toxicity.
The increase in the number of akaryotic hepatocytes beyond doubt, and the reduction in the number of Kupffer cells presumably are indices to liver damage, whereas the reduction in the number of binucleated hepatocytes may be regarded an index to the inhibition of reparative proliferation of these cells. As can be seen from Table 2, these changes are most marked in response to 50 nm NPs and least marked for micrometric particles.
The spleen also demonstrated considerably more marked changes in response to NPs of both sizes in comparison with micrometric particles for which the histological picture of this organ was little different from the controls. However, no obvious differences in the degree of these changes under the effect of NPs of different size were observable.
Conclusions
The results of our study show that subchronic resorptive toxicity of iron oxide Fe3O4, which proved negligible under the action of even finest particles in the micrometer range, is substantial for NPs.
It is more difficult to give a single unqualified answer to the question whether the toxicity of this substance increases in the same manner with a decrease in the size of its particles within the nanometric range. Our results suggest that this dependence on NP’s size (for a given mass dose) is different for essentially different effects and is not unique due to the complex and often opposing relationships between the intrinsic bioaggressivity of NPs, on one hand, and complex mechanisms that control their biokinetics, on the other hand.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Financial support provided by Russian Federal Agency of Science and Innovations.