Abstract
Long-acting formulations of antipsychotics are important treatment options to increase the compliance of schizophrenic patients. Risperidone, a 5-HT2 and dopaminergic D2 receptor antagonist, was developed as long-acting sustained-release microspheres with poly(lactide-co-glycolide) (PLGA) as a drug carrier for the treatment of schizophrenia. In the present study, the main objective is to determine the nonclinical safety profile of risperidone-loaded microspheres (RM) in Beagle dogs after intramuscular administration for 3 months, once in 2 weeks, followed by 8-week recovery phase. No animal death was found and no special toxicological findings were observed. The findings, such as hypoactivity, ptosis, increased heart rate, and elevated serum and pituitary prolactin levels, were observed and related to the pharmacological effects of risperidone. The changes in the reproductive system (uterus, ovary, vagina, cervix, and mammary gland) were considered secondary to the prolactin elevation, and the congestion of spleen was related to risperidone. The foreign body granulomas at injection sites might be caused by PLGA. At the end of recovery phase, the above changes mostly recovered to normal, and on administering 3 mg/kg dose level once in 2 weeks on Beagle dogs showed no observed adverse effect. Taken together, RM had exhibited the acceptable safety.
Keywords
Introduction
Schizophrenia is a chronic psychiatric disorder associated with substantially reduced quality of life and significant impairment in psychosocial functioning. 1 Atypical antipsychotics (second-generation antipsychotics) are a major step forward in this respect, which have beneficial effects against positive symptoms, negative symptoms, and cognitive deficits of schizophrenic patient, with low extrapyramidal side effects and tardive dyskinesia. 2 However, the noncompliance and high discontinuation rates are the common problems in the management of schizophrenia, 3 in which nearly half of the patients with oral antipsychotic medication showed noncompliance or partial compliance. 4 Medication noncompliance is often associated with negative consequences, including symptom exacerbation, rehospitalizations, and relapse. 5 –7 Therefore, improving the medication compliance is critically important in the treatment of schizophrenia.
Risperidone, a representative drug of atypical antipsychotics, is a serotonin type 2 (5-HT2) and dopaminergic D2 receptor antagonist. 8 It also acts as a moderate antagonist at histaminergic H1 and adrenergic α1 and α2 receptors. 9 Risperidone is quickly transformed into 9-hydroxyrisperidone by cytochrome P450 2D6 enzyme in liver, 10 which were together contributed to the antipsychotic activity with risperidone. 11 Risperdal® Consta® (Consta) is a long-acting injectable formulation of risperidone by intramuscular administration and has been shown to increase patient compliance, reduce hospital time and relapse rates, and increase time spent in rehabilitation. 12 –15 While there is a 3-week lag of drug release after the first injection of Consta, which makes it necessary for oral supplementation of antischizophrenic drug during this time 10 and results in the inconvenience for the patients and physicians. Therefore, it is essential and significant to develop a more convenient long-acting sustained-release formulation of risperidone for the maintenance treatment of schizophrenia.
Risperidone-loaded microspheres (RM) is developed by Luye Pharmaceutical Co. Ltd (Luye Pharma, Yantai, Shandong, China) with poly (lactide-co-glycolide) (PLGA) as the drug carrier. RM is designed to be used for the treatment of schizophrenia (the same indication as Consta) with the maximum recommended single dosage of 50 mg per person once in 2 weeks in clinic by intramuscular injection. Almost no lag time is observed in the single-dose pharmacokinetics study of intramuscular RM in dogs (unpublished data), which is different from Consta.
In the present study, the main objective is to determine the nonclinical safety profile of RM in Beagle dogs during 12-week intramuscular administration and the reversibility, persistence, or delayed occurrence of target organ toxicity.
Materials and methods
Test articles
RM, which occurs as white powdery particles containing 50 mg active drug (risperidone) per vial, is supplied by Luye Pharma. The risperidone is microencapsulated in PLGA at a concentration of 470 mg of risperidone per gram of microspheres in RM. The content of risperidone is 47% and the placebo microsphere is 53% in RM. The control articles, placebo microspheres without active drug (125.3 mg per vial), and vehicle (1% carboxymethylcellulose sodium, SCMC) were also provided by Luye Pharma. RM and placebo microspheres were suspended in the vehicle at the designed concentrations. The concentration, homogeneity, and 24-h stability of the prepared suspensions were analyzed to ensure the accurate scheduled dosages.
According to the non-disclosure agreement (NDA) review documents for Consta (NDA 021-346), in a 12-month toxicity study, the Beagle dogs in the high-dose group were intramuscularly injected with 5 mg/kg of Consta at the dosage 1 and 2, 10 mg/kg at the dosage 3 and 4, and 20 mg/kg at the dosage 5–26, respectively, once in 2 weeks for a total of 26 times. The prolactin-related pathological changes in the reproductive systems were observed. So, 30 mg/kg was selected as a toxic dosage in this study, which was 36 times higher than the recommended clinical dose. The medium and low doses were determined as 10 mg/kg and 3 mg/kg, respectively.
Animals
Conventional 30 male and 30 female Beagle dogs were supplied by Beijing Marshall Biotechnology Co. Ltd (Beijing, China, production license No.: SCXK (Jing) 2011-0003). The body weight range and age of Beagle dogs at grouping assignment were 5.35–7.15 kg and 6–7 months, respectively. Animals were quarantined and accommodated in a new environment for 27 days and were separately housed in stainless steel cages, with free access to sterilized food and water. The animal room was maintained with 12-h light/12-h dark cycle at the temperature of 21 ± 5°C and relative humidity of 55 ± 15%.
The contents and procedures related to the animal experiments in this study were complied with the regulations in the Institutional Animal Care and Use Committee (IACUC) in WestChina-Frontier PharmaTech Co. Ltd. (WCFP) and National Chengdu Center for Safety Evaluation of Drugs (NCCSED).
Experimental design
Beagle dogs were randomly assigned into five groups: vehicle group (1% SCMC), placebo microspheres group (35.2 mg/kg, equivalent to the microspheres content in RM 30 mg/kg), and RM 3, 10 and 30 mg/kg groups. Each group contained 12 animals (6 animals in each sex). The animals were intramuscularly injected once in 2 weeks for 3 months (i.e., a total of 6 doses), using a dosing volume of 1 mL/kg, at gluteus medius muscle, gluteus biceps femoris, arm triceps muscle, or other appropriate sites of hind limb. For each dose, the drug was only injected in unilateral muscle, alternating between the left side and the right side. The recovery phase was 8 weeks. The day of the first dose was designated as day 1. Then, 14 days after the 6th dose was the end of treatment phase, and the next day (day 85) was designated as the first day of recovery phase. At the end of treatment phase (day 84) and the end of recovery phases (day 140), four animals in each sex per group and two animals in each sex per group were necropsied, respectively.
Clinical observation
The following clinical signs such as animal’s signs, hair, injection site, behavior, mental status, food consumption, respiration, feces, genitals, mortality, and other toxic manifestations were evaluated daily at least once.
Body weight and food consumption
During the treatment and recovery phases, for all surviving animals, the body weights were measured once prior to dosing and, thereafter, once a week. The food consumptions were measured once a week.
Blood pressure, body temperature, and electrocardiogram
On days 13, 27, 55, 83, and 139, for all surviving animals, the arterial blood pressures in the forelimb were measured once by wrist sphygmomanometer, the rectal temperatures were measured once by thermometer, and II-lead electrocardiogram (ECG), parameters included heart rate (ventricular rate), RR interval, P duration, PR interval, QRS duration, QT interval, and QTc interval, were recorded by a four-channel polygraph (RM6240, Chengdu Instrument Factory, Sichuan, China).
Hematology, clinical chemistry, urinalysis, and feces
Blood samples were collected from cephalic veins in the forelimbs of all surviving animals for hematology and clinical chemistry detections on days 28, 84, and 140. Hematology was examined by an auto hematology analyzer (Advia2120, SIEMENS, Germany) and an automatic reticulocyte (RET) analyzer (R-520, Sysmex, Japan) using ethylenediaminetetraacetic acid anticoagulated blood samples. Clotting time test was performed by auto blood hemagglutination analyzer (CA-7000, SYSMEX, Japan) using the plasma separated from sodium citrate anticoagulated blood samples. Clinical chemistry was examined by an auto biochemistry analyzer (Cobas C501, ROCHE, Switzerland) using the serum separated from unanticoagulated blood sample.
Urinalysis was performed by an auto urine chemistry analyzer (Clinitek Status, SIEMENS, Germany) using the samples collected through bladder puncture on days 84 and 140.
The detected parameters in the above tests (hematology, clinical chemistry, and urinalysis) are listed in Table 1.
Parameters detected in hematology, clinical chemistry, and urinalysis.

RBC: red blood cell; HGB: hemoglobin; HCT: hematocrit; MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin; MCHC: mean corpuscular hemoglobin concentration; WBC: white blood cell; NEU: neutrophil granulocyte; LYM: lymphocyte; MONO: mononuclear cell; EOS: eosinophilic granulocyte; BASO: basophil; PLT: platelet; MPV: mean platelet volume; PT: prothrombin time; APTT: activated partial thromboplastin time; RET: reticulocyte; Cl−: chloride ion; Na+: sodium ion; K+: potassium ion; Ca2+: calcium ion; P: phosphate; GLU: glucose; BIL: bilirubin; KET: ketone body; AMY-P: amylopsin; SG: specific gravity; LIP: lipase; BLO: occult blood; AST: aspartate amino transferase; ALT: alanine aminotransferase; PRO: protein; GGT: γ-glutamyl transferase; UBO: urobilinogen; ALP: alkaline phosphate; NIT: nitrite; TBIL: total bilirubin; ALB: albumin; A/G: albumin/globulin ratio; CHOL: cholesterol; TP: total protein; TG: triglyceride.
Feces samples were collected within 4 days before the necropsy in both treatment and recovery phases. Feces parameters including color, character, parasite, red blood cells, white blood cells (WBC), epithelial cell, and fat droplet were examined by routine analysis method. Occult blood was detected by ortho
Ophthalmoscopy
After being mydriasised by tropicamide eye drops (Santen Pharmaceutical, Japan), animals were examined once by binocular indirect ophthalmoscope (YZ25A, 66 Vision-Tech, China) on days 84 and 140. The examined parameters included cornea, iris, lens, ocular fundus, anterior chamber, and posterior chamber.
Prolactin in serum and pituitary
Serum prolactin was detected prior to dosing and on days 28, 56, 84, and 140. The serum samples were taken from those for clinical chemistry analysis or were separated from unanticoagulated blood samples on the scheduled days. Luminex® 200™ System and Milliplex MAP Canine Pituitary Magnetic Bead Panel Immunoassay Kit (Millipore Corporation, Billerica, Massachusetts, USA) were employed for determining the concentration of serum prolactin.
The samples of pituitary were collected from randomly selected four dogs from each group (two in each sex) on days 84 and 140. The level of prolactin expression in pituitary was detected by immunohistochemical method.
Bone marrow smear
Bone marrow was collected from the anesthetized animal (pentobarbital sodium, intravenous injection, 30 mg/kg) by puncturing through the posterior superior iliac spine with a myeloid puncture needle on days 84 and 140. Bone marrow smears were prepared and stained by Wright–Giemsa stain and observed under a light microscope to examine the parameters such as granulocytic series, erythrocytic series, lymphocytes, monocytes, megakaryocytes, and myeloid erythroid ratio.
Gross pathology and histopathology
Eight animals (four in each sex per group) and four animals (two in each sex per group) in each group were necropsied on days 84 and 140, respectively. The animals were anesthetized with pentobarbital sodium (intravenous injection, 30 mg/kg) followed by complete necropsy. The organs for gross pathology, listed in Table 2, were weighed and paired organs were kept together. Organ/body weight ratios and organ/brain weight ratios were calculated according to the organ and body weights.
Organs selected for gross pathology and histopathology examination.

The organs for histopathology examination were listed in Table 2. Testes and epididymis were fixed in modified Davidson’s solution, eyeballs were fixed in glutaraldehyde solution, and all the other tissues were preserved in 10% neutral phosphate-buffered formalin. Then, dissecting, paraffin embedding, sectioning and hematoxylin–eosin staining were performed according to standard histopathological technical operation, followed by microscopic examination.
Toxicokinetics study
Prior to dosing (0 h) and 1 h, 1, 3, 5, 7, 9, 11, and 14 days after the 1st, 3rd, and 6th doses, heparin sodium anticoagulated blood was taken from the animals’ cephalic vein of forelimb in RM groups. The plasma was separated by centrifugation and was stored at −80°C. The above work was completed at WCFP, then the toxicokinetics (TK) determinations, including drug concentration detection and method validation, were conducted by the Research and Development Center of Luye Pharma.
Statistical analysis
Quantitative data such as body weight, food consumption, body temperature, ECG, hematology, clinical chemistry, prolactin, blood pressure, percentage of bone marrow cells and megakaryocyte count, organ weights and ratios were presented as group mean ± SD, which were analyzed by one-way analysis of variance followed least significant difference method. If after analysis, Levene’s homogeneity test of variance suggested heterogeneity of variance (p < 0.05), then Kruskal–Wallis H rank-sum test was used for statistical analysis; when intergroup difference was statistically significant (p < 0.05), Mann–Whitney U rank-sum test was used for intragroup comparison. Qualitative parameters were described using frequency in urinalysis, feces, and bone marrow smear examinations. Fisher’s exact probability test (Fisher EXACT) was utilized to analyze binomial categories data and unordered multicategories data. Ordered categories data were evaluated using Kruskal–Wallis nonparametric tests followed by Mann–Whitney U nonparametric tests. All the statistical analyses were performed using Statistical Package for the Social Sciences software (v 13.0) (International Business Machines Corporation, USA) at two-tailed probability level (α = 0.05).
Results
Clinical signs
All the animals survived the scheduled necropsy. Almost all the animals in RM groups were observed with hypoactivity and/or ptosis, which were dose related in symptom level, incidence, and onset time. The above-mentioned signs were gradually recovered to normal at the end of the recovery phase. No obvious abnormality in clinical signs was observed for animals in vehicle and placebo microspheres groups during the study.
Body weight and food consumption
The significant decreases in body weight were recorded in 30 mg/kg group in weeks 3, 6, and 7 of recovery phase compared with that in the vehicle group, and no significant changes were recorded in all the groups at other time points (Figure 1(a)). The significant decreases in food consumption were found in 3 and 10 mg/kg group in week 5 compared with that in the vehicle group, and no significant changes were recorded in all the groups at other time points (Figure 1(b)).

The curves of body weight (a) and food consumption (b) in Beagle dogs treated with different doses of risperidone-loaded microspheres.
These changes were observed only at individual group or time point and had no dose dependency. So, they were considered to be normal, without obvious toxicological significance.
Body temperature and blood pressure
No abnormal changes related to RM in body temperature and blood pressure were noted in all the groups during the treatment and recovery phases compared with those in the vehicle group (data not shown).
ECG analysis
Slight increases in the heart rate were recorded in 10 and 30 mg/kg groups after the 4th dosing compared with that in the vehicle group (Table 3). No abnormality was found in other parameters such as QRS wave, P wave, and QT interval (data not shown). At the end of the recovery phase, no abnormal changes were recorded in all the ECG parameters.
Effects of RM on heart rate and R-R interval in Beagle dogs.a

RM: risperidone-loaded microspheres.
aData represent mean ± SD.
b p < 0.05 compared with vehicle group.
Hematology
Slight decrease in WBC counts (neutrophilic granulocyte and lymphocyte), decreases in RET counts/percentages and platelet (PLT) counts were noted in each RM group after the second or/and sixth dosing (Table 4). The above changes returned to normal after the drug discontinuation. No abnormal changes related to RM in all the groups were noted in other hematological parameters during the study (data not shown).
Selected hematological parameters in Beagle dogs.a

RM: risperidone-loaded microspheres; WBC: white blood cell; NEU: neutrophil granulocyte; LYM: lymphocyte; RET: reticulocyte.
aData represent mean ± SD.
b p < 0.05 compared with vehicle group.
Clinical chemistry
Slight increase in cholesterol (CHOL) was recorded in 30 mg/kg group after the 2nd and 6th dosing (Table 5). A decrease in the concentration of potassium ion (K+) was found in each RM group after the second and sixth dosing (Table 5). No abnormal changes related to RM were found in other clinical chemical parameters during the study. The changes in total protein, albumin, and glucose (GLU) were small (within 15%) and have no dose dependency. The decreases in parameters of triglycerides, alkaline phosphate, γ-glutamyl transferase, and amylopsin were considered to have no clinical significance. The slight changes in urea, albumin/globulin ratio, sodium, phosphate, calcium, and chloride ions were likely to be normal fluctuation from the data.
Selected clinical chemical parameters in Beagle dogs.a

RM: risperidone-loaded microspheres; CHOL: cholesterol; K+: potassium ion.
aData represent mean ± SD.
b p < 0.05 compared with vehicle group.
Urinalysis and feces
No abnormal changes related to RM in urinalysis parameters, such as GLU, bilirubin, ketone body, specific gravity, occult blood (BLO), pH, protein, urobilinogen, nitrite, WBC were observed, and no BLO in feces were found in all the groups at the end of treatment and recovery phases (data not shown).
Ophthalmoscopy
No abnormal changes related to RM in dioptric media, fundus vascular, optic papilla, and retina were observed at the end of treatment and recovery phases (data not shown).
Organ weights
At the end of the treatment phase, dose-dependent increases in the weight and organ/body (brain) weight ratios of spleen and decreases in the weight or organ/body (brain) weight ratios of uterus were observed in each RM group. The decreases in the weight and organ/body (brain) weight ratios of testis and epididymis were observed for males in 10 and/or 30 mg/kg groups (Table 6). At the end of the recovery phase, the above-mentioned changes in the above organs had almost recovered to normal, except that the weight and organ/body (brain) weight ratios of uterus in RM groups were obviously lower than that in control groups.
Effects of RM on organ weights, organ/body (brain) weight ratios in Beagle dogs (in grams).a
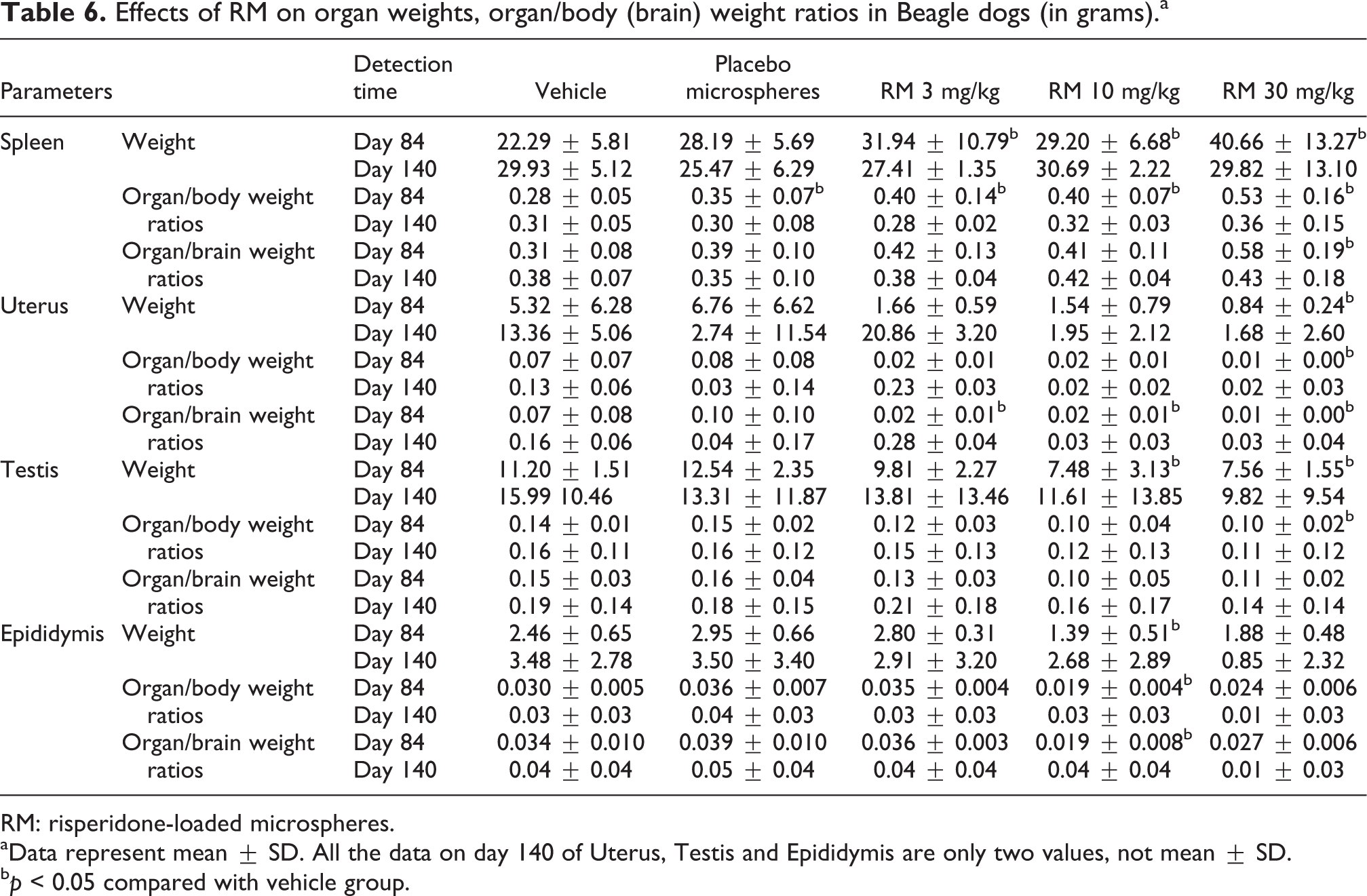
RM: risperidone-loaded microspheres.
aData represent mean ± SD. All the data on day 140 of Uterus, Testis and Epididymis are only two values, not mean ± SD.
b p < 0.05 compared with vehicle group.
During the study, no abnormal changes related to RM in each group were found in weights and organ/body (brain) weight ratios of heart, lung, brain, kidney, adrenal gland, and liver (data not shown).
Bone marrow smear
Slight decreases in neutrophil percentage and ratios of granulocytes to erythroid in 30 mg/kg group and increase in megakaryocyte in each RM group were noted at the end of the treatment phase (Table 7). No abnormal changes related to RM were found in other parameters such as erythron, lymphocyte, and mononuclear cells (data not shown).
Selected parameters of bone marrow in Beagle dogs.a

RM: risperidone-loaded microspheres; NEU: neutrophil; G/E: granulocytes/erythrocytes.
aData represent mean ± SD.
b p < 0.05 compared with vehicle group.
Prolactin in serum and pituitary
Prior to dosing, on 14 days after the 2nd, 4th, and 6th dosing, and at the end of the recovery phase, the concentrations of serum prolactin for most dogs in each group were lower than the detectable limits of the kits. The ratios of detected animals to total animals in each group were listed as follows: 3 of 52 in vehicle group, 1 of 52 in placebo microspheres group, 5 of 52 in 3 mg/kg group, 6 of 52 in 10 mg/kg group, and 14 of 52 in 30 mg/kg group.
The expression levels of pituitary prolactin in 10 and 30 mg/kg groups were higher than those in vehicle group at the end of the treatment phase (Table 8 and Figure 2(a) to (c)). No statistical difference in the levels of pituitary prolactin between vehicle group and RM groups were observed at the end of the recovery phase.

Histological sections of pituitary by immunohistochemical staining and histological sections of muscle at the injection site and spleen by hematoxylin–eosin staining. (a) to (c) Immunohistochemical staining of pituitary sections (×100) for examination of prolactin level, + and +++ present weak positive and intensively positive, respectively; (d) to (f) hematoxylin–eosin staining of muscle sections (×40); (g) to (i) hematoxylin–eosin staining of spleen sections (×40).
Immunohistochemical findings for prolactin expression in pituitary.

aGrade 1: weak positive (+), grade 2: moderate positive (++), and grade 3: intensively positive (+++).
Gross pathology
At the end of the treatment phase, gray–white nodules were observed in muscles at the injection sites in placebo microspheres and RM groups. Increases in volume of bilateral mammary glands were observed in one female in 10 mg/kg group. Decreases in volume of uterus and dark red cyst in unilateral ovary were observed in one female in 30 mg/kg group. No abnormal changes related to RM were found in brain, heart, liver, lung, and kidney. At the end of the recovery phase, no abnormal changes related to RM were found in all the organs.
Histopathology
At the end of the treatment phase, minimal to marked foreign body granuloma in muscles at the injection sites and peripheral connective tissue of sciatic nerve were observed in placebo microspheres and/or RM groups, with microspheres dose related in severity and incidence (Figure 2(d) to (f)). Minimal to marked congestions in spleen were found in each RM group, with RM dose related in severity and incidence (Figure 2(g) to (i)). Minimal atrophy of thymus was observed in 10 and 30 mg/kg groups (Figure 3(a) and (d)). Atrophy/hypoplasia of uterus and ovary were noted in 10 and/or 30 mg/kg groups (Figure 3(b), (c) to (f)). Atrophy/hypoplasia of vagina and cervix were noted in each RM group (Figure 4(a), (b), (d) and (e)). All the above-mentioned changes were dose related in incidence. Minimal to moderate alveoli secretion of mammary glands was found in 10 and 30 mg/kg groups (Figure 4(c) and (f)).
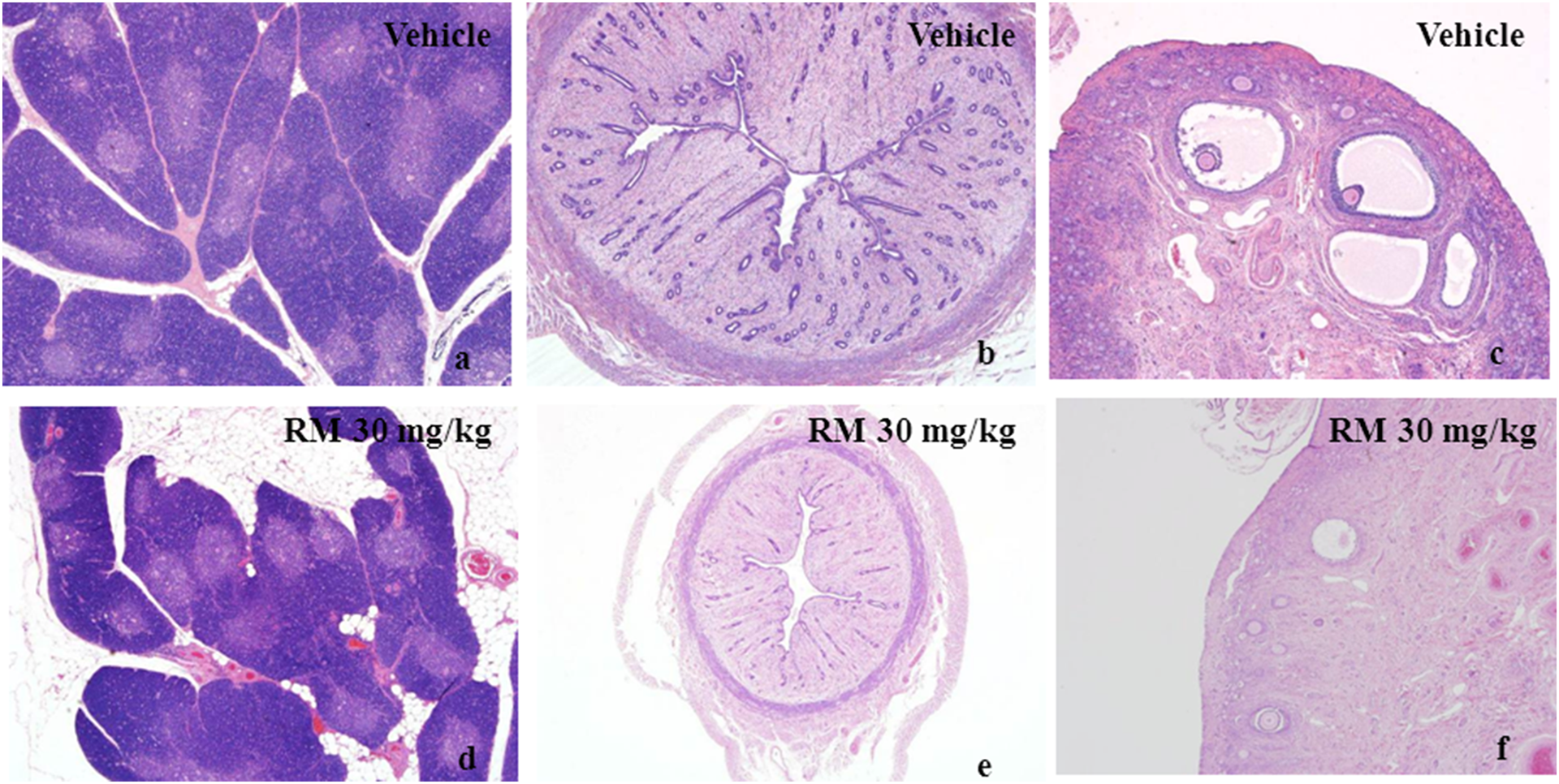
Histological sections of thymus, uterus, and ovary by hematoxylin–eosin staining. (a,d) thymus sections (×40), (b,e) uterus sections(×40), and (c.f) ovary sections (×40).

Histological sections of vagina, cervix, and mammary glands by hematoxylin–eosin staining. (a,d) vagina sections (200×), (b,e) cervix sections (200×), and (c,f) mammary glands sections (400×).
At the end of the recovery phase, minimal foreign body granuloma in muscles at the injection sites were noted in placebo microspheres and 30 mg/kg groups. Atrophy/hypoplasia of uterus, ovary, vagina, and cervix were noted in 10 and 30 mg/kg groups. Except for these, other changes were returned to normal or to a certain degree at the end of the recovery phase.
The incidence and grade of histopathological findings related with RM/placebo microspheres are shown in Table 9.
Incidence and grade of histopathological findings related with RM/microspheres.

RM: risperidone-loaded microspheres.
a0: normal, 1: minimal, 2: slight, 3: moderate, 4: marked, and 5: severe.
TK study
The TK parameters are presented in Table 10. After the first, third, and sixth doses of RM, the increases in the area under concentration–time curve (AUC(0– 336 h)) and maximum concentration (C max) of active moiety (risperidone + 9-hydroxyrisperidone) were dose proportional in the range of 3–30 mg/kg. With the advancement in the administration times, a trend of elevation in exposure level was observed in Beagle dogs. The ratios of AUC(0– 336 h) and C max for both three and six doses to those of one dose of RM were in the range of 1.5–3.1. The AUC(0– 336 h)and C max for three doses were comparable to those following six doses of RM, without obvious difference. There was no significant gender difference in AUC(0– 336 h) and C max for dogs.
TK parameters of active moiety in Beagle dogs after single- and repeated-dose injection of RM.

RM: risperidone-loaded microspheres; TK: toxicokinetics; C max: maximum concentration; AUC: area under concentration–time curve.
Discussion
Second-generation antipsychotics, such as risperidone, were a major step forward in improving the treatment for schizophrenic patients. However, important unmet needs such as a better solution for non- and partial compliance with therapy are still remaining. 16 It is recognized that long-acting antipsychotics can improve treatment compliance and simplify the medication process, 3,17 which may help to prevent relapse and improve functional outcomes in schizophrenic patients. 18 Long-acting antipsychotics also produce the lower steady state peak concentrations and less fluctuation in plasma levels, thereby further reducing the risk of adverse effects. 19,20
The first depot formulation of atypical antipsychotic on the market is long-acting risperidone injection (Consta). However, a disadvantage associated with Consta was the required 3-week supplementation with oral antipsychotics after the first dosing. RM is developed by Luye Pharma with PLGA as a drug carrier. The pharmacokinetics showed that RM can release the drug after injections with almost no lag time, which would simplify the medication by eliminating the oral supplementation of antipsychotics. In the present study, a 12-week intramuscular toxicity study of RM was evaluated in Beagle dogs.
After administration, hypoactivity and/or ptosis were observed in all RM groups with dose-related severity, incidence, and onset time. These changes were related to the pharmacologic actions of RM antagonism on dopamine D2 receptor or adrenergic α1 receptor. 11
Increase in the heart rate was recorded in RM groups, which might be related with RM. There are reports of similar adverse effects in the clinical use of Consta and analogous drugs, with undefined mechanisms, but likely involve the blockade of adrenergic receptors and human Ether-á-go-go Related Gene (hERG) channels. 10,21
The decreases in neutrophil percentage of bone marrow smear and the reductions in neutrophil granulocyte, lymphocyte, and RET of peripheral blood were observed in RM groups, which could be drug-related changes. Furthermore, a decrease in PLT of peripheral blood and an increase in megakaryocyte of bone marrow were also recorded in RM groups. Congestions in spleen and organ weight and organ/body weight ratios of spleen were noted. Therefore, the enlarged spleen could be the compensation to the reduction of blood-producing function in bone marrow.
Compared with the vehicle, all the changes in clinical chemical parameters had no toxicological significance except increase of CHOL in 30 mg/kg groups and decrease of K+ in each RM group, which were likely to be RM related. An increased plasma level of CHOL has been reported in clinical trial on long-acting injectable risperidone. 22
The nodules observed in muscles at the injection sites in placebo microspheres and each RM group were presented as foreign body granuloma in histopathological examination. The incidence and grade of nodules/foreign body granuloma were basically identical in each group, which indicated the microspheres, drug carrier of RM, could deposit in muscles for a longtime and form the granuloma. The above changes were considered to be microspheres related. At the end of the recovery phase, no obvious nodules were detected and the pathological grades for foreign body granuloma were apparently relieved. These suggested that the microspheres could be gradually degraded and eliminated in local muscles. PLGA, the main component of microspheres, are neither toxic nor carcinogenic or mutagenic, with many beneficial features, such as biodegradability, biocompatibility, and bioabsorbability. It has been approved as a pharmaceutical excipient by Food and Drug Administration. Many extended-release preparations using PLGA have been approved for marketing, such as Zoladex®.
Pathological changes in the reproductive systems including atrophy of thymus, atrophy/hypoplasia of uterus, cervix, ovary, and vagina, and alveoli secretion in mammary glands were observed to be dose related in RM groups. Abnormal changes in organ weight or organ/body (brain) weight ratios of uterus, prostate, testis, and spleen were recorded in the RM groups. The expression levels of prolactin in pituitary and serum were noted to be obviously elevated for animals at 10 and 30 mg/kg, which were related with the pharmacologic action of RM on dopamine D2 receptor. 23 Based on the documents and reports on clinical adverse effects, risperidone can induce the elevation of prolactin levels, which result in the endocrine alteration and further cause menoxenia, amenorrhea, lactation, and mammary hyperplasia in female patients, and disorders of mammary gland development and reproduction in male patients. 24 –26 Therefore, the pathological changes in the reproductive systems in this study were considered to be secondary to elevated prolactin. At the end of the recovery phase, atrophy/hypoplasia in uterus, cervix, ovary, and vagina were still observed in 10 and 30 mg/kg groups. Except for these, other changes returned to normal or to a certain degree. These aforementioned findings were substantially consistent with those recorded in the chronic toxicity studies of Consta in dogs. 27
In this study, the toxicological effects profile for RM has been well established. However, there are still some problems to be further studied or be improved, such as the mechanisms and consequence of the decreased K+, the detection method of serum prolactin, and the reason for the congestion of spleen.
In conclusion, in this 12-week intramuscular toxicity study in Beagle dogs, RM showed a linear kinetics characteristics in the dose range of 3–30 mg/kg. Almost all the histopathological findings were related to the prolactin elevation or the known pharmacological side effects of risperidone. Risperidone-related histopathological changes of spleen and foreign body removal reaction induced by PLGA were also noted. Based on the absence of treatment-related toxicological changes, no observed adverse effect level was established at 3 mg/kg dosage. Taken together, RM has exhibited the acceptable safety on Beagle dogs and could be a potential long-acting sustained-release formulation.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Acknowledgments
We gratefully acknowledge Hongbo Wang, Xin Yu, Qiuju Dong, Fangxi Zhang, Xiaoyin Zhu, and Wei Zhu for assistance with data analysis and helpful comments and advice.
Funding
This work was supported by the National Natural Science Foundation of China [grant numbers 81001661],the Shandong Province Young and Middle-Aged Scientists Research Awards Fund [grant numbers BS2009SW012], the National Basic Research Program of China [grant numbers 2012CB724003], and the Taishan Scholar Project.
