Abstract
The role of oxygen radicals are known for the pathogenesis of kidney damage. The aim of the present study was to investigate the antioxidative effects of melatonin, quercetin, and resveratrol on streptozotocin (STZ)-induced diabetic nephropathy in rats. A total of 35 male Wistar rats were divided into 5 groups as follows: control, diabetes mellitus (DM), DM + melatonin, DM + quercetin, and DM + resveratrol. All the injections started on the same day of single-dose STZ injection and continued for 30 days. At the end of this period, kidneys were removed and processed for routine histological procedures. Biochemical parameters and morphological changes were examined. In DM group, blood glucose levels were significantly increased, whereas body weights were decreased compared with the control group. Significant increases in blood urea nitrogen and tissue malondialdehyde (MDA) levels and decreases in superoxide dismutase and catalase activities were detected in DM group. Administration of melatonin, quercetin, and resveratrol significantly reduced these values. Melatonin was more efficient in reducing MDA levels than other antioxidants (
Introduction
Diabetes mellitus (DM) is a metabolic disease that develops in case of insulin release from β cells is insufficient or absent. 1 Many of the complications of DM including renal failure, cardiomyopathy, vascular damage, and vision loss have been attributed to chronic hyperglycemia. 2 Oxidative stress also plays an important role in the etiology of diabetic complications. 3,4 Oxidative stress is caused by the overproduction of reactive oxygen radicals that are highly toxic to all the components of the cells, particularly to the cellular membranes in which they interact with the lipid bilayer and produce lipid peroxides. 5 Hyperglycemia is suggested to promote oxidative stress through both nonenzymatic and enzymatic mechanisms. 4
Streptozotocin (STZ), an antibiotic produced by
Antioxidant treatment has been shown to be an important therapeutic option for reducing tissue damage and preventing complications in DM.
7
Melatonin (
Many studies emphasize the central role of transforming growth factor-β (TGF-β) produced by renal mesangial and tubular cells in the initiation and progression of diabetic renal disease. 12,13 TGF-β stimulates these cells to synthesize collagen and other matrix components. Hyperglycemia increases the production and activation of TGF-β in the kidney. 14,15
In the present study, we aimed to evaluate the effects of melatonin, quercetin, and resveratrol administration on STZ-induced diabetes via investigating histopathological and biochemical alterations in kidneys.
Materials and methods
Experimental design
A total of 35 male Wistar albino rats (Inonu University Animal Research Center, Malatya, Turkey), weighing between 300 and 350 g, were housed in individual cages for 30 days in a well-ventilated room with a 12-h light:12-h dark cycle at 21°C. Animals were fed with standard rat chow and tap water ad libitum. After 12-h starvation, the animals were induced diabetes by a single intraperitoneal (i.p.) injection of STZ freshly dissolved in 0.9% saline at a dose of 45 mg/kg (Sigma, St Louis, Missouri, USA). Three days after STZ injection, blood glucose levels of all groups were measured using reagent strips (Accu-Check Active Glucose test strips, Roche, Germany) with a glucometer (Accu-Check Active, Roche) in samples obtained from the tail vein. Animals having 270 mg/dL or higher blood glucose levels were considered to be diabetic. Rats were observed daily for mortality or morbidity. Body weights of all animals were recorded.
The animals were randomly divided into 5 groups each consisting of 7 rats. Group 1—control group (C): nondiabetic rats received 4% ethanol by i.p. injections for 30 days; group 2—diabetes mellitus group (DM): rats received single i.p. injection of STZ dissolved in 0.9% saline at a dose of 45 mg/kg (diabetic rats); group 3—diabetes + melatonin group (DM + M): diabetic rats treated with melatonin (i.p.; Apollo Scientific Limited, UK, product no: BIM0105), dissolved in 4% ethanol, at a dose of 10 mg/kg/day; group 4—diabetes + quercetin group (DM + Q): diabetic rats treated with quercetin (i.p.; Alfa Aesar, Ward Hill, Massachusetts, USA, lot no: 10159068), dissolved in 4% ethanol at a dose of 25 mg/kg/day; and group 5—diabetes + resveratrol group (DM + R): diabetic rats treated with resveratrol (i.p.; Molekula, UK, product no: M57735199), dissolved in 4% ethanol at a dose of 10 mg/kg/day. All the injections started on the same day of single dose STZ injection and continued for 30 days.
The experiments were performed in accordance with the guide for the care and use of laboratory animals of Inonu University’s Animal Ethics Committee.
Histopathological evaluations
After 30 days of dosage administration, the rats were anesthetized by ketamine. Blood samples were collected from tail vein of the rats and then kidneys were removed by laparotomy. The right kidneys from each rat were weighed. Tissue samples were divided into two portions. The first part of the samples was processed for light microscopic examination, while the second part was prepared for biochemical evaluation. The first part of the samples were placed in 10% formalin for 24 h and prepared for routine paraffin embedding. Paraffin blocks were cut at 5 μm thickness, mounted on slides, and stained with hematoxylin–eosin (H-E), periodic acid Schiff (PAS), and toluidine blue (TB) staining methods. Under 20× magnification, tubular changes including hydropic degeneration (swelling/vacuolization), desquamation, brush border loss, and peritubular infiltration were semiquantitatively graded as follows: 0: (
Immunohistochemical analysis
For immunohistochemical analysis, sections were mounted on polylysine-coated slides. After rehydrating, samples were transferred to citrate buffer (pH 7.6) and heated in a microwave oven for 20 min. After cooling for 20 min at room temperature, the sections were washed with phosphate-buffered saline (PBS), then kept in 0.3% hydrogen peroxide (H2O2) for 7 min and afterward washed with PBS. Sections were incubated with a primary rabbit polyclonal anti-transforming growth factor-β1 (TGF-β1) antibody (sc-146, lot no: C0211; Santa Cruz Biotechnology, Santa Cruz, California, USA) for 2 h. They were rinsed in PBS and incubated with biotinylated goat anti-polyvalent for 10 min and streptavidin peroxidase for 10 min at room temperature. Staining was completed with chromogen + substrate for 15 min, and slides were counterstained with Mayer’s hematoxylin for 1 min, rinsed in tap water, and dehydrated. Anti-TGF-β1 antibody was used according to the manufacturer’s instructions. Brownish yellow granules in cytoplasm were recognized as positive staining for TGF-β1. Under 20× magnification, 100 tubules were selected randomly from each section and the relative intensity of immunostaining within cells was also scored as: 0 =
All sections were examined using a Leica DFC280 light microscope and a Leica Q Win and Image Analysis system (Leica Micros Imaging Solutions Ltd, Cambridge, UK).
Biochemical evaluations
Preparation of tissue homogenates
Tissues were homogenized (PCV Kinematica Status Homogenizator) in ice-cold PBS (pH 7.4). The homogenate was sonified with an ultrasonifier (Bronson sonifier 450) by 3 cycles (20-s sonications and 40-s pause on ice) and then was centrifuged (15,000
MDA levels
The analysis of lipid peroxidation was carried out as previously described by Buege and Aust with a minor modification.
16
The reaction mixture was prepared by adding 250 μL homogenate into 2 mL reaction solution (15% trichloroacetic acid:0.375% thiobarbituric acid:0.25 N hydrochloric acid, 1:1:1, w/v) and heated at 100°C for 15 min. The mixture was cooled to room temperature, centrifuged (10,000
CAT activity
Catalase (CAT) activity was measured for the supernatants by the method followed by Luck. 17 The decomposition of the substrate H2O2 was monitored spectrophotometrically at 240 nm. Specific activity was defined as micromole substrate decomposed per minute per milligram of protein. CAT levels were expressed as micromole per milligram of protein.
SOD activity
Superoxide dismutase (SOD; copper, zinc-SOD) activity was measured in the supernatant fraction using xanthine oxidase/cytochrome
tGSH levels
The formation of 5-thio-2-nitrobenzoate is followed spectrophotometrically at 412 nm. 19 The amount of glutathione GSH in the extract was determined as nanomoles per milligram protein utilizing a commercial GSH as the standard.
Determination of protein
Protein levels of the tissue samples were measured by the Bradford method. 20 The absorbance measurement was taken at 595 nm using an ultraviolet–visible spectrophotometer. Bovine serum albumin was used as protein standard.
BUN and creatinine levels
Blood samples for the measurement of blood urea nitrogen (BUN) and creatinine levels were drawn into ethylenediaminetetraacetic acid-containing tubes and immediately placed on ice. All tubes were centrifuged within several minutes of collection and stored at −70°C until assay. Serum samples were assayed for BUN and creatinine. Analyses were performed on an Abbott Architect c 1600 automatic analyzer (Abbott Park, Illinois, USA).
Statistical analysis
Statistical analysis was carried out using the Statistical Package for Social Sciences for Windows version 13.0 (SPSS Inc., Chicago, Illinois, USA) and MedCalc (SBB Consulting 10, UK) statistical programs. All data are expressed as arithmetic mean ± standard error. Normality for continued variables in groups was determined by the Shapiro–Wilk test. The variables didn’t show normal distribution (
Results
Body and kidney weights
The initial body weights were similar in control and the other groups (
The mean BWs and kidney weights of all groups (in grams).a

DM: diabetes mellitus; C: control; M: melatonin; Q: quercetin; R: resveratrol; BW: body weight.
aData are expressed as arithmetic mean ± SE of seven animals.
b
c
d
Blood glucose levels
Blood glucose levels of all groups were calculated to be from 94.85 ± 2.93 mg/dL to 115.71 ± 1.42 mg/dL at the beginning of the experiment. At 72 h following STZ injection, blood glucose levels were significantly increased compared with the control group (

The mean blood glucose levels of all groups (in milligram per deciliter). There were no significant differences among all groups in initial blood glucose levels (
Histopathological findings
Glomerulus and tubules were normal in histological appearance (Figure 2). DM group showed severe glomerular and tubular alterations. Tubular changes including epithelial desquamation, cellular swelling, intracytoplasmic vacuolization, and peritubular infiltration were obvious (Figure 3). In PAS-stained sections, brush border loss from proximal tubules, glycogen granules in the tubular cells, thickened capillary and tubular basal membranes, and sclerotic changes were detected (Figure 4). In TB-stained sections, numerous granules were observed in the proximal tubule cells (Figure 5(a)). The mean histopathological damage score (MHDS) of this group was 5.85 ± 0.34. Significant increase in MHDS was detected in DM group in comparison with the control group (
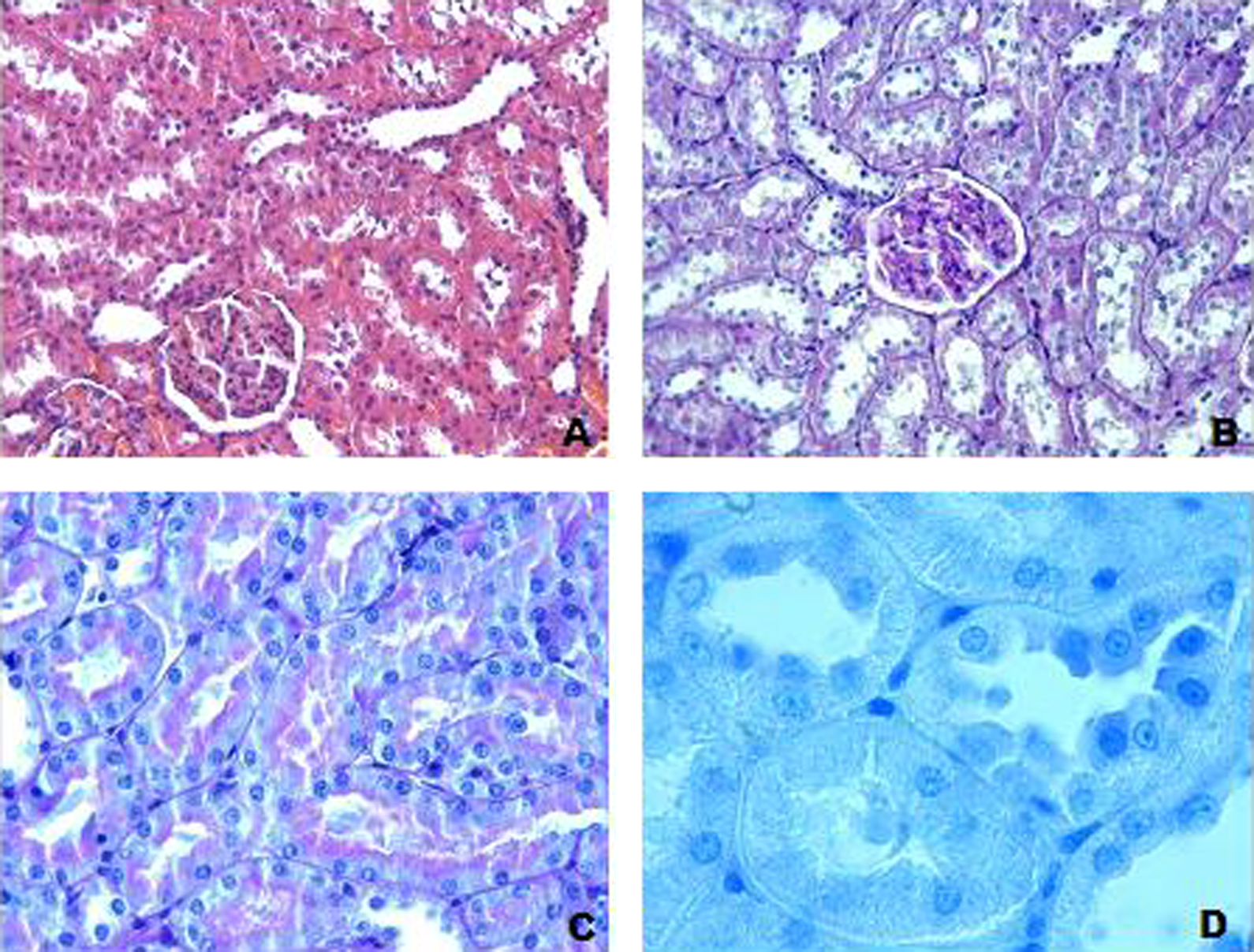
Control group. Histological appearance was normal. (a) H-E; ×20; (b) PAS; ×20; (c) PAS; ×40; (d) TB; ×100. H-E: hematoxylin–eosin; PAS: periodic acid Schiff; TB: toluidine blue.

We detected different histopathological changes in H-E-stained sections in DM group. (a) Epithelial desquamation in tubules (arrows), H-E: ×20; (b) common infiltration (stars) and hemorrhagic areas (arrows), H-E: ×20; (c) swelling of tubular cells (arrows), H-E: ×40; and (d) extensive vacuolization in proximal tubular cells (arrows), H-E: ×100. H-E: hematoxylin–eosin; DM: diabetes mellitus.
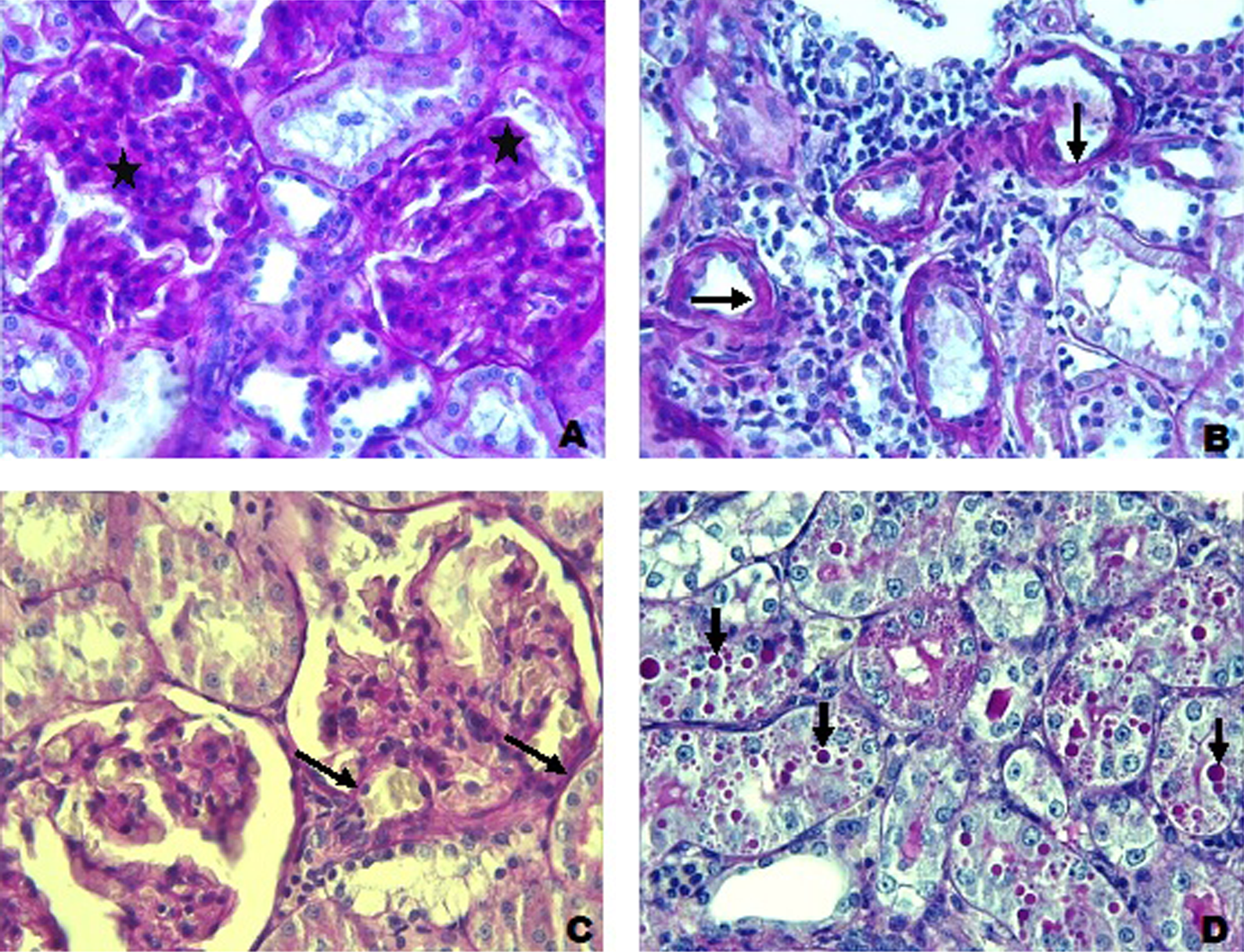
Histopathological changes in PAS-stained sections were detected in DM group. (a) Glomerulosclerosis (stars), PAS: ×40; (b) thickening of tubular basal membrane (arrows) and loss of microvilli in proximal tubules, PAS: ×40; (d) thickening of capillary basal membrane in glomerulus (arrows), PAS: ×40; and (e) in tubules, a great number of PAS (+)-stained granules in different sizes are shown, PAS: ×40. PAS: periodic acid Schiff; DM: diabetes mellitus.

(a) DM group: blue-stained, large and small granules in tubular epithelial cells (arows) were observed in this group; TB: ×100. (b) DM + M group: blue-stained granules decreased (arrows); TB: ×100. (c) DM + Q group: blue-stained granules were detected infrequently (arrow); TB: ×100. (d) DM + R group: blue-stained granules were observed rarely (arrow) in proximal tubule; TB: ×100. DM: diabetes mellitus; TB: toluidine blue; M: melatonin; Q: quercetin; R: resveratrol.

Histopathological alterations markedly diminished in treatment groups. (a) DM + M group: mild epithelial desquamation in tubules and hydropic degeneration in some tubular cells, H-E: ×20. (b) DM + M group: tubular basal membrane and microvilli in proximal tubules were partly in good condition, PAS: ×40. (c) DM + Q group: moderate epithelial desquamation in tubules (arrows), H-E: ×20. (d) DM + Q group: glomerular capillary lumen was observed clearly, PAS: ×40. (e) DM + R group: epithelial desquamation and hydropic degeneration in tubules, H-E: ×20. (f) DM + R group: loss of microvilli in some proximal tubules (thin arrow) and partly thickened basal membrane in glomerulus (thick arrow), PAS: ×40. DM: diabetes mellitus; H-E: hematoxylin–eosin; PAS: periodic acid Schiff; M: melatonin; Q: quercetin; R: resveratrol.
MHDS of all groups.a
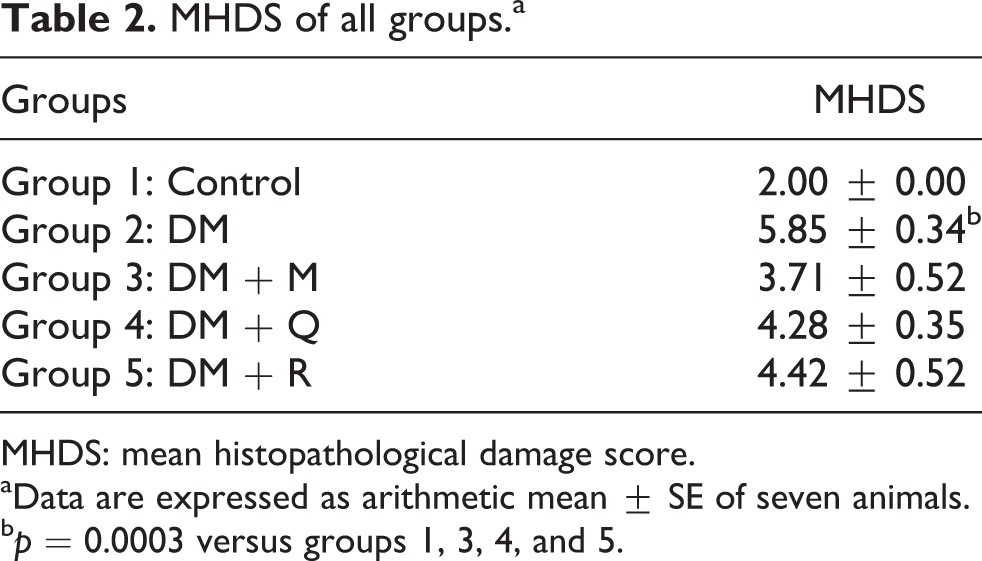
MHDS: mean histopathological damage score.
aData are expressed as arithmetic mean ± SE of seven animals.
b
The mean glomerulosclerotic index of all groups.a

DM: diabetes mellitus; M: melatonin; Q: quercetin; R: resveratrol.
aData are expressed as arithmetic mean ± SE of seven animals.
b
c
d
e
f
g
Immunohistochemical findings
TGF-β1 intensity was not obvious in control group. In DM group, TGF-β1 expression was more prominent in comparison with that of the control group (

(a) Control group: glomerulus (G) and tubules (T) were normal; TGF-β1; ×40. (b) DM group: severe immunopositivity in tubules; TGF-β1; ×20. (c) DM group: slight immunopositivity in glomerulus; TGF-β1; ×40. (d) DM + M group: immunopositivity in tubules was markedly reduced; TGF-β1: ×40. (e) DM + Q group: slight immunopositivity in tubules was detected; TGF-β1: ×40. (f) DM + R group: slight immunopositivity in tubules was observed; TGF-β1; ×40. TGF-β1: transforming growth factor-β1; DM: diabetes mellitus; M: melatonin; Q: quercetin; R: resveratrol.
The number of TGF-β1 immunostaining positive tubules of all groups.a

TGF-β1: transforming growth factor-β1; DM: diabetes mellitus; M: melatonin; Q: quercetin; R: resveratrol.
aData are expressed as arithmetic mean ± SE of seven animals.
b
c
d
Biochemical findings
Mean tissue MDA level of DM group was significantly increased when compared with the control group (
Mean tissue SOD and CAT activities of DM group was significantly decreased in comparison with the control group (

The mean tissue MDA, tGSH levels and SOD, CAT activities of all groups. a
BUN and creatinine levels
Mean BUN level of DM group was significantly increased in DM group when compared with the control group (

The mean serum BUN and creatinine level of all groups. a
Discussion
Oxidative stress has an important role in the pathogenesis of diabetes-induced nephropathy. 21 The aim of the present study was to investigate the antioxidant effects of melatonin, quercetin, and resveratrol against diabetic renal damage primarily by microscopic and biochemical methods. Previous studies on antioxidant administration to diabetic rats emphasized their hypoglycemic effects in STZ-induced diabetes. 22 –25 In our study, we also detected reducing potency of antioxidants on blood glucose. Our results confirm those of some other studies stating that melatonin, quercetin, and resveratrol are beneficial on stabilizing blood glucose values. 26 –29
Body weight loss is commonly observed during short- and long-term experimental diabetes studies 9,30 We detected significant weight loss in diabetes group in comparison with the control group. Interestingly, Guneli et al. and Andallu and Varadacharyulu have reported that natural and chemical agents having free radical-scavenging properties prevented weight loss in diabetic rats. 31,32 In the present study, antioxidant-administered diabetic rats lost body weights lesser than the nontreated rats. Body weight loss in diabetic rats has been found to be related with muscle loss due to over catabolism of tissue proteins as a result of hyperglycemia. 32 Thus, the antioxidants we administered maintain the body weight by controlling blood glucose level thereby preventing protein catabolism.
In the experimental diabetes studies, results on kidney weights are contradictory. Coldiron et al. reported decreases in kidney weights, whereas Garman et al. reported significant increases in kidney weights. 9,23 Teoh et al. found no significant difference in kidney weights 33 as we did.
The most common histopathological alterations including capillary basal membrane thickening and diffused or nodular glomerulosclerosis on diabetic nephropathy occur in glomerulus. 34 Diffused glomerulosclerosis is characterized by mesangial cell proliferation and expansion of mesangial matrix. 35,36 We observed mesangial matrix expansion within glomerulus, capillary, and tubular basal membrane thickening in diabetic groups. Eddy suggested that the cause of glomerular and tubular basal membrane thickening is similar. 37 In vitro studies have proved that at high concentrations of glucose, proteoglycans, decorin, fibronectin, type-4 collagen, and laminin levels as well as TGF-β expression are increased. TGF-β inhibits collagenase production but stimulates metalloproteinase inhibitor production in tissues thus causing accumulation of extracellular matrix (ECM) proteins in glomerular and tubular basal membranes by preventing degradation. 38
We detected some diabetes-related alterations including loss of brush border, epithelial swelling or desquamation, accumulation of glycogen granules, and peritubular inflammation. Some previous studies reported similar histopathological alterations on rats. 22,26,39,40 Glomerular and tubular changes were reduced in melatonin-, quercetin-, and resveratrol-administered groups. The therapeutic effects of resveratrol is attributed to its hypoglycemic, antioxidative, anti-inflammatory, and adenosine monophosphate active protein kinase regulatory properties. 29 Chang et al. reported that resveratrol treatment improved hyperglycemia and hyperglycemia-related renal dysfunction. 29 We found quercetin beneficial in reducing diabetes-related alterations. Quercetin, a free radical scavenger and superoxide radical inhibitor, is known to be a strong antioxidant. 27,41 Mahesh and Menon and Edremitlioglu et al. reported its antioxidant power in the course of diabetes. 28,42 Beneficial effects of quercetin are attributed to its antioxidant effects as well as protective effects on β cell integrity. As a matter of fact, Vessal et al. reported its increasing effect on the number of islets of Langerhans in pancreas. 27 By this way, there is a significant decrease in plasma glucose level. The third agent we used, melatonin, is a powerful free radical scavenger and antioxidant enzyme stimulator. 43,44 Chronic melatonin application has been reported to reduce renal damage on STZ-induced diabetes in rats. 45 In tissues like kidney where glucose input is not related with insulin, blood glucose concentration is increased when aldose reductase is activated resulting in sorbitol production. Sorbitol not capable of transmitting through cell membranes but accumulates within the cytoplasm and eventually causes disintegration of the cell membranes. Additionally by its osmotic effect, it causes cell swelling. 46 Melatonin stabilize cell membranes by its high lipophilic property thus preventing the cells from hydrophobic changes.
Another interesting result of the present study was the presence of numerous granules within the cytoplasm of tubule cells in TB-stained sections. Yakan and Esrefoglu and Vardi et al. suggested that these large, heterogeneous, dense granules are in fact lysosomes increased for eliminating excess amount of intracytoplasmic glycogen. On the other hand, hyperglycemia causes glycosylation of protein and peptides resulting in the formation of glycosylation end products. 22,47 Gugliucci reported that advanced glycation end products (AGE) peptides that are filtered by glomerulus are metabolized by endolysosomal system at proximal tubules. 46 Cathepsin and metalloproteinase significantly reduce renal lysosomal enzyme activities. Both endocytosis of AGE peptides at proximal tubules and decreased lysosomal activity can result in increase of lysosome number. 48,49
TGF-β is an important growth factor stimulating ECM synthesis by mesangial and epithelial cells. 50,51 In the present study, TGF-β1 immune positivity was strong in proximal tubules whereas was weak on glomerulus in diabetic group. Rocco et al. reported increased TGF-β expression at proximal tubule epithelial cells under the effect of high glucose concentration. 52 However, in another study, a weak positivity in tubules and glomerulus was observed in the course of acute phase of diabetes in rats. 51 It is known that reactive oxygen species has a key role in increased TGF-β synthesis in diabetes. 36 In our study, we detected that melatonin, quercetin, and resveratrol administrations reduced TGF-β1 expression at proximal tubules of diabetic rats. Ha et al. reported that melatonin reduced TGF-β messenger RNA level in the kidney 1.4 times in diabetic rats. 53
Serum BUN and creatinine levels are the most important indicators of kidney functions. Lu et al.reported that BUN and creatinine levels were increased in diabetes.
24
On the other hand, Choi et al. reported no significant increase in BUN and creatinine levels in diabetic group when compared with the control group.
40
In the present study, BUN levels were increased, whereas creatinine levels were unchanged in diabetes. In experimental diabetes, some of the antioxidants including
In the present study, oxidative stress was evaluated by means of tissue MDA and tGSH levels and SOD and CAT activities. In DM group, tissue MDA levels were increased, whereas tissue SOD and CAT activities were decreased. Similar results were reported by Obrosova et al. and Kataya and Hamza. 30,54 Glucose is oxidized to reactive ketoaldehyde and superoxide radicals. If it is not decomposed by CAT or GSH peroxidase it causes production of reactive hydroxyl radicals. Excess amounts of free radicals damage cellular proteins and nucleic acids by attaching to them. 55 They cause lipid peroxidation by increasing low-density lipoprotein. MDA is the most commonly used indicator of lipid peroxidation. Increase in tissue MDA level and decreases in cellular antioxidant enzymes emphasize oxidative stress. 56 We suggest that melatonin, quercetin, and resveratrol prevent from oxidative stress by reducing lipid peroxidation and increasing SOD and CAT activities. 57 Cam et al. reported that melatonin treatment decreased lipid peroxidation, inhibited glomerular basal membrane thickening, and mesangial matrix expansion. 45 Edremitlioglu et al. reported that quercetin treatment significantly decreased diabetes-related oxidative damage in various organs by decreasing MDA levels, but increasing antioxidant enzyme activities. 42 Lu-Jia and Li-Jing reported that resveratrol decreased MDA content and increased the SOD activity in the kidneys of diabetic rats. 58
In present study, we found that mean tissue tGSH levels of all the groups were similar. There was no significant difference among groups. On the other hand, previous experimental studies have shown that GSH enzyme activities reduced in STZ-induced diabetic nephropathy by antioxidative agents. 31,54
As a conclusion we suggest that melatonin, quercetin, and resveratrol are beneficial in improving diabetic nephropathy by preventing oxidative stress.
Footnotes
Acknowledgment
The authors extend their sincere gratitude to the Department of Histology and Embryology, Faculty of Medicine, Inonu University, Malatya, Turkey, for their support.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was financially supported by a grant from Scientific Research Fund of Inonu University (Grant number: 2011/63).
