Abstract
This study investigated the hypothesis that administration of tilapia fish oil diet would attenuate warm liver ischemia/reperfusion injury (IRI) and whether fish oil modulates prooxidant/antioxidant status. Male Wistar rats were subjected to 30 min of approximately 70% hepatic ischemia followed by 1, 12, and 24 h reperfusion. Rats were randomly divided into three groups: sham-operated group (SO), control–warm hepatic ischemia (WI) group, and Oil–WI group given tilapia oil for 3 weeks followed by liver IRI. Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels were measured in the plasma. Levels of thiobarbituric acid reactive substances (TBARS) and antioxidant enzymes as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) activities were measured in liver fractions.
In the sham group, there was no enzymatic or histological change. I/R caused significant increase in serum AST, ALT, and tissue TBARS levels. As compared to the control group, animals treated with tilapia oil experienced a significant decrease (p < 0.05) in AST and ALT levels in reperfusion periods. Tissue TBARS levels in Oil–WI group were significantly (p < 0.05) reduced as compared to control group at 60 min after reperfusion. After ischemia, 1, 12, and 24 h of reperfusion, CAT, SOD, and GPx values were the lowest in the Oil–WI group and highest in the control group and were statistically significant (p < 0.05). Histological analysis also revealed that fish oil provided some protection compared with the control group.
Tilapia oil exerts a protective effect during the early phase of reperfusion, and it modulates prooxidant/antioxidant status of rat liver subjected to warm IRI.
Introduction
Warm hepatic ischemia–reperfusion (WI/R) injury is a serious complication of many clinical situations such as major liver resection surgery (Pringle maneuver), hemorrhagic and septic shock, and liver transplantation.
1,2
However, the precise mechanisms of hepatic damage is still not clearly demonstrated; it is believed that several mediators, such as reactive oxygen species (ROS), proinflammatory cytokines, chemokines, adhesion molecules, and excess nitric oxide (NO) contribute to this injury.
1,3–5
The development of hepatic I/R injury (IRI) can be divided into initial and late phases.
6
The initial phase (<2 h after reperfusion) is characterized by oxidative stress, where production and release of ROS appear to directly result in hepatocellular injury.
7
There was a significant increase in
In histologically normal liver tissues, a burst of ROS occurs during the reperfusion, and it is not clear whether the process followed after warm or cold ischemia. 9,10 ROS cause cell injury directly by attacking a great variety of cellular molecules, including lipids, proteins, and nucleic acids, 11,12 and indirectly by promoting other damaging mechanisms, such as the activation of transcription factors leading to the synthesis of proinflammatory cytokines and adhesion molecules. 12 ROS produced on reperfusion plays a critical role in the injury caused by I/R. 13–15 Accumulation of purine derivatives due to adenosine triphosphate degradation and reduction of mitochondrial ubiquinone produce superoxide radicals on reoxygenation. 14–16 In addition, macrophages and neutrophils in previously ischemic tissue are activated and produce oxygen radicals via nicotinamide adenine dinucleotide phosphate oxidase. 17 Indeed, superoxide production by Kupffer cells and neutrophil accumulation increases over fivefold after hepatic I/R. 18 Moreover, gadolinium chloride, a drug that selectively destroys and/or inactivates Kupffer cells, minimizes liver injury after ischemia reoxygenation. 19
Several studies have indicated the preoperative nutritional state to be an important factor predicting postoperative complications because the nutritional state greatly affects the postoperative metabolism, organ function, and inflammatory responses. 20–22 The oxidant stress may activate signaling mechanisms provoking more toxic events and eventually cause tissue damage. Therefore, treatments with antioxidants, free radical scavengers, and their mimetics represent potential therapeutic options to correct the redox imbalance. 7 Production of cytokines and arachidonic acid metabolites by Kupffer cells can be influenced by diets supplemented with fish oils. 23,24
Fish and marine mammals are by far the richest sources of long-chain n−3 polyunsaturated fatty acid (PUFA) in nature. Fish oil-supplemented diets have been shown to alleviate IRI in myocardial muscles, 25 small intestines, 26 and skeletal muscles. 27 In hepatic tissue, fish oil reduced the liver injury in a low flow, reflow perfusion model in the rat. 28 A controversy is raised from the study of Lo et al. which stated that dietary supplementation of rats with fish oil does not attenuate hepatic injury after WI/R. 29 However, the ability of tilapia fish oil to protect the liver from WI/R has not been investigated.
Based on the pathophysiology of hepatic IRI, the current study particularly focused on a nutritional approach to preoperative preparation. The effect of administration of tilapia fish oil by gavage on antioxidant enzyme activities (catalase (CAT), superoxide dismutase (SOD), and glutathione peroxidase (GPx)), lipid peroxidation, and liver damage after hepatic WI/R was investigated in the rats.
Materials and methods
Fatty acid composition
Wet fish was stored at −20°C before freeze-drying. After freeze–drying, fish were transferred to tight packages and stored at −80°C. Freeze-dried fish were ground and sifted through a 1.18-mm analytical sieve for removing coarse particles such as fish scales and bones, and the powder was then stored at −80°C until use.
Total lipids for fatty acid analysis were extracted from aliquot samples of thigh according to Folch et al. 30 Fatty acid methyl esters (FAMEs) were prepared according to the method described by Wang et al. 31 and measured by gas chromatography in a Chrompack CP 900 apparatus (Raritan, New Jersey, USA) fitted with a flame ionization detector (FID). Analytical gas chromatography was carried out on a Hewlett–Packard 6890 gas chromatograph series II (Agilent Technologies, Palo Alto, California, USA) equipped with HP Innowax (30 m × 0.25 mm, 0.25 mL film thickness) capillary column. Each sample was injected with a split ratio of 1:100 and a continuous flow rate of 1.5 mL min−1 of chromatographic grade helium was used. The oven temperature was initially held for 20 min at 165°C, ramped at 5°C min−1 up to 240°C, and held isothermal for 25 min. Injector and FID temperature were held at 250°C. FAMEs were identified by comparison of their retention time with respect to pure standard purchased from Sigma and analyzed under the same conditions. FAMEs were quantified according to their percentage area, obtained by integration of the peaks. The results were expressed as a percentage of individual fatty acids in the lipid fraction as described by Pordomingo et al. 32
Animal preparation and feeding
For the purpose of this study, adult male Wistar rats, weighing 200–250 g, were obtained from the Central Pharmacy, Tunisia. The animals were divided into three groups (sham, control, and fish-oil supplemented) and kept in an environmentally controlled breeding room (temperature: 20 ± 2°C, humidity: 60 ± 5%, 12-h dark/12-h light cycle).
The animals were allowed to acclimatize to the laboratory conditions and after 3 weeks, animals in the sham and control groups received phosphate-buffered saline (PBS) by gavage, and the fish oil-supplemented group received isovolumetric amounts of tilapia fish oil rich in unsaturated fatty acids.
The amount of tilapia fish oil given for rats (0.4% of body weight) 33 was recalculated daily with increases of body weight. This amount of oil was high enough to show effects without causing diarrhea. The administration of oils or saline was performed daily between 7:00 p.m. and 8:00 p.m. The rats were handled every day, and therefore, they accepted the treatment well without anesthesia. The handling of the animals was approved by the local ethical committee for the care and use of laboratory animals.
Liver I/R model
Three different groups of animals (n = 7–9 per group) were subjected to sham operation (SO) or I/R proceeded by vehicle (Control-IR) or tilapia oil (Oil-IR) pretreatment. To induce ischemia, rats were anesthetized with pentobarbital (50 mg kg−1 intraperitoneally). The visceral organs were then exposed by a midline abdominal incision. Ischemia was induced to the left and median lobes of the liver by occluding the left branches of the portal vein, hepatic artery, and bile duct for 30 min using a microvascular clamp. The blood flow to the right lobe and the two small caudate lobes was kept intact during the ischemic time. This model causes partial ischemia (approximately 70%) without intestinal congestion. During ischemia, the body temperature of the animals was closely monitored and maintained at 37°C by a heating lamp. After 30 min of in vivo partial ischemia, the clamp was removed to allow blood to the ischemic lobes, and the blood flow to the nonischemic lobes was stopped by ligating vessels that supply blood to the right and caudate lobes. The ligation of nonischemic lobes during reperfusion prevents stealing of blood flow by nonischemic lobes from the ischemic lobes during the reperfusion period. 34,35 The reperfusion was allowed for 1, 12, and 24 h in all the groups.
Serum parameters
Serum samples were obtained by the centrifugation of blood at 4000 r min−1 for 15 min at 4°C and were then divided into Eppendorf tubes and stored at −30°C until they were used for the analyses. The level of serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were determined using commercial kits supplied by Sigma (Munich, Germany).
Determination of lipid peroxidation markers in liver tissue
As markers of lipid peroxidation, we used the quantification of thiobarbituric acid reactive substances (TBARS) as described by Esterbauer. 36 Briefly, 125 µL of supernatant from tissue homogenate were mixed with 125 µL 20% trichloroacetic acid containing 1% butylated hydroxytoluene in order to precipitate proteins. After centrifugation (1000g, 10 min at 4°C), 200 µL of supernatant were mixed with 40 µL 0.6 N hydrochloric acid and 160 µL of 120 mM TBA in 25 mM Tris buffer (pH 7.4). The mixture was heated at 80°C for 10 min, and the optical density of the solution was measured at 530 nm. TBARS concentration was calculated using an extinction coefficient of 156 mM−1 cm−1.
Determination of CAT, GPx, and SOD in liver tissue
CAT activity was determined at 25°C according to the method described by Aebi 37 by measuring the decrease of H2O2 absorbance at 240 nm for 1 min. CAT activity was calculated using an extinction coefficient of 0.043 mM−1 cm−1 and expressed in micromoles of H2O2 destroyed per minute per milligram of protein.
Total (copper–zinc and manganese (Mn)) SOD activity was determined at 25°C by measuring its ability to inhibit the photoreduction of nitroblue tetrazolium (NBT) into blue formazan. 38,39 The assay was performed in 50 mM PBS (pH 7.4) containing 13 mM methionine, 0.1 mM ethylenediaminetetraacetic acid, 2 l M riboflavin, and 75 l M NBT. SOD activity was expressed in units per milligram protein, and one unit of SOD is the amount inhibiting the photoreduction of NBT by 50%.
GPx activity was assayed at 25°C according to Flohe and Gunzler with some modifications. 9 GPx activity was measured in 250 µL tissue extract mixed with glutathione (GSH; final concentration: 0.35 mmol L−1). Reaction started with the addition of H2O2 (0.2 mmol L−1). After the reaction stopped, 5,5′-dithiobis-2-nitrobenzoic acid was added, and the absorbance was recorded at 412 nm. The activity was expressed in micromoles of GSH oxidized per minute per gram protein.
Histology
Formalin (10%)-fixed liver samples were embedded in paraffin, and sections of 6 μm thickness were prepared and stained with hematoxylin and eosin for histological evaluation.
Data analysis
Data are presented as means ± SE. Statistical significance between two groups of parametric data was examined by one-way analysis of variance followed by Fisher’s test (StatView, SAS Version 5.0). The value of p < 0.05 was considered to be statistically significant.
Results
The food intake of all rats was always around 20 g per rat. The average of body weight gain was not different among the groups.
Fatty acid composition
The composition of fatty acids (% total fatty acids) of the oils is presented in Table 1. The intensively cultured Nile tilapia contained amounts of saturated fatty acids (SFA), monounsaturated fatty acids (MUFA), and PUFA. However, SFA and MUFA were the dominant category of fatty acids in the intensively cultured Nile tilapia. A higher fraction of MUFA in intensively farmed fish has also been reported for other fish species including Atlantic salmon (Salmo salar) 40 and turbot (Scophthalmus maximus). 41 This was largely due to the increased presence of 18:1n−9 in tissues of farmed fish, which is consistent with our findings. The important amounts of 18:2n−6 in tilapia fish oil originated from the high inclusion of grains and/or plant seed oils in the commercial diets and is a common feature in cultured fish.
Fatty acid composition of tilapia oils (% total fatty acids).

SFA: saturated fatty acid; UFA; unsaturated fatty acid; MUFAs: monounsaturated fatty acids; PUFAs: polyunsaturated fatty acids.
Serum markers of cell damages
AST and ALT were released into the blood when certain organs or tissues, particularly liver and heart, are injured and was the most sensitive parameters to I/R in this study. Changes in serum AST and ALT concentrations after operation in each group are shown in Figure 1. Serum ALT and AST levels increased significantly after reperfusion in I/R group compared with those of the sham group. Pretreatment with tilapia oil significantly decreased AST and ALT levels compared with those of control–WI group.
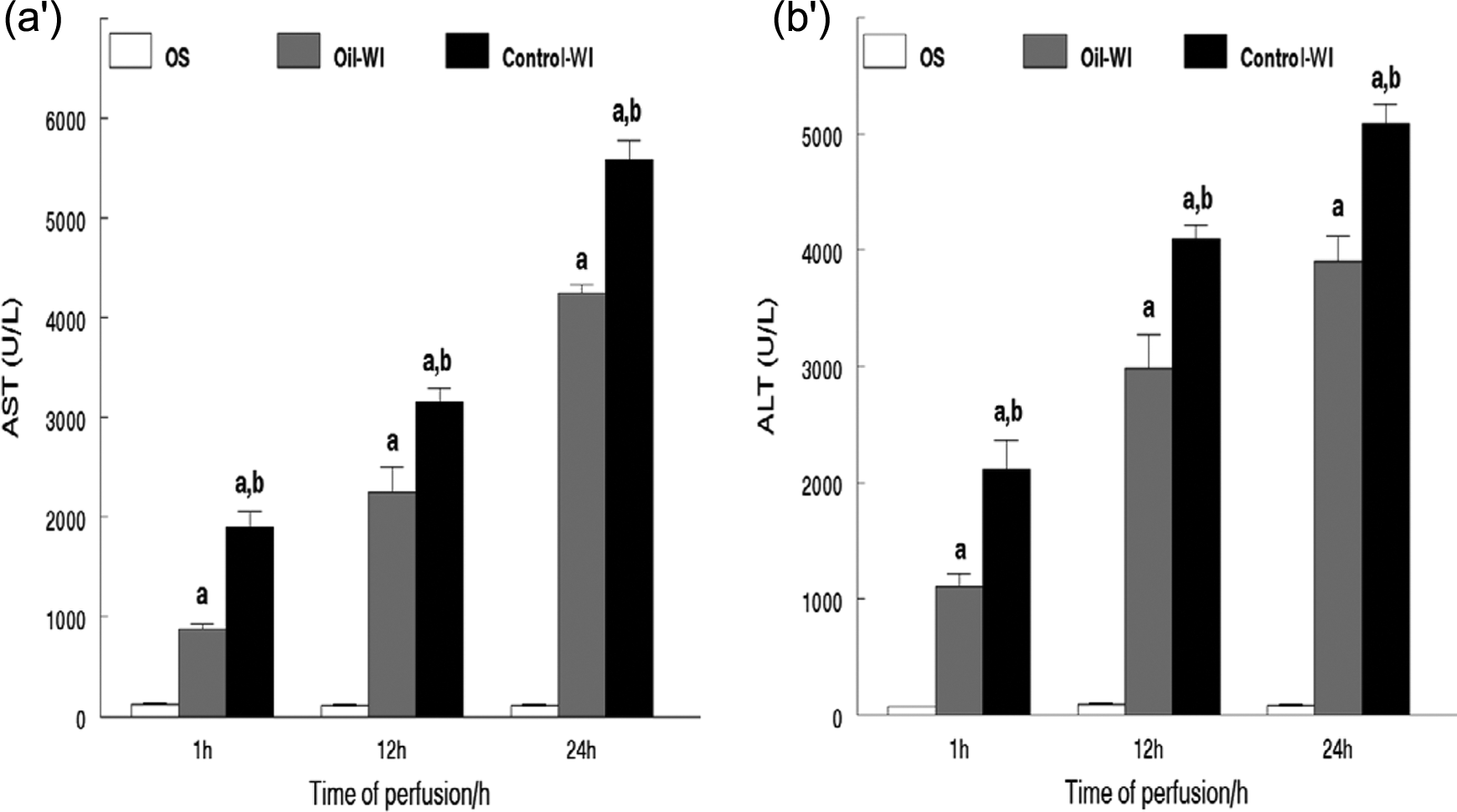
Significantly increased serum concentrations of AST (a′) and ALT (b′) after reperfusion as compared to the sham group. a p < 0.05 versus sham group; b p < 0.05 versus oil–WI group. Values are represented as means ± SE (all n = 6). AST: aspartate aminotransferase; ALT: alanine aminotransferase; WI: warm hepatic ischemia.
Concentrations of lipid peroxidation markers in liver tissue
The concentrations of lipid peroxidation markers in the liver are presented in Figure 2. The TBARS content as an index of lipid peroxidation was significantly higher after reperfusion in I/R group than in the sham group. Pretreatment with tilapia oil decreased TBARS content during the early phase of reperfusion.

The results of TBARS content in liver tissue. Tilapia oil pretreatment do not reduce MDA content significantly as compared to control–WI group after 12 and 24 h of reperfusion. a p < 0.05 versus sham group. Values are represented as means ± SE (all n = 5). b p < 0.05 versus oil–WI group. eTBARS: thiobarbituric acid reactive substances; MDA: malondialdehyde; WI: warm hepatic ischemia.
Antioxidant enzyme activities
Compared with the sham group, the hepatic activities of CAT, SOD, and GPx significantly decreased in control–WI and oil–WI groups (p < 0.01) during reperfusion, decreased more obviously in control–WI group, as shown in Figure 3.

The results of CAT (a′), SOD (b′), and GPx (c′) activities in liver tissue. Tilapia oil pretreatment significantly increased CAT, SOD, and GPx activities as compared to I/R group. a p < 0.05 versus sham group; b p < 0.05 versus oil–WI group. Values are represented as means ± SE (all n = 5). CAT: catalase; SOD: superoxide dismutase; GPx: glutathione peroxidase; I/R: ischemia/reperfusion; WI: warm hepatic ischemia.
Liver histology
Photomicrographs of the representative rat livers, stained with hematoxylin–eosin, are presented in Figure 4. As expected, the control livers had normal lobular and cellular structures (Figure 4(a)). However, IR resulted in substantial injury (Figure 4(b)), presented as cytoplasmic vacuolation of hepatocyte that were randomly distributed throughout the parenchyma, infiltration of polymorphonuclear cell, and leukocyte adhesion to venular endothelium (Figure 4(b)) in the control–WI group. The areas of cytoplasmic vacuolation were slightly less frequent in the Oil–WI group (Figure 4(c)).
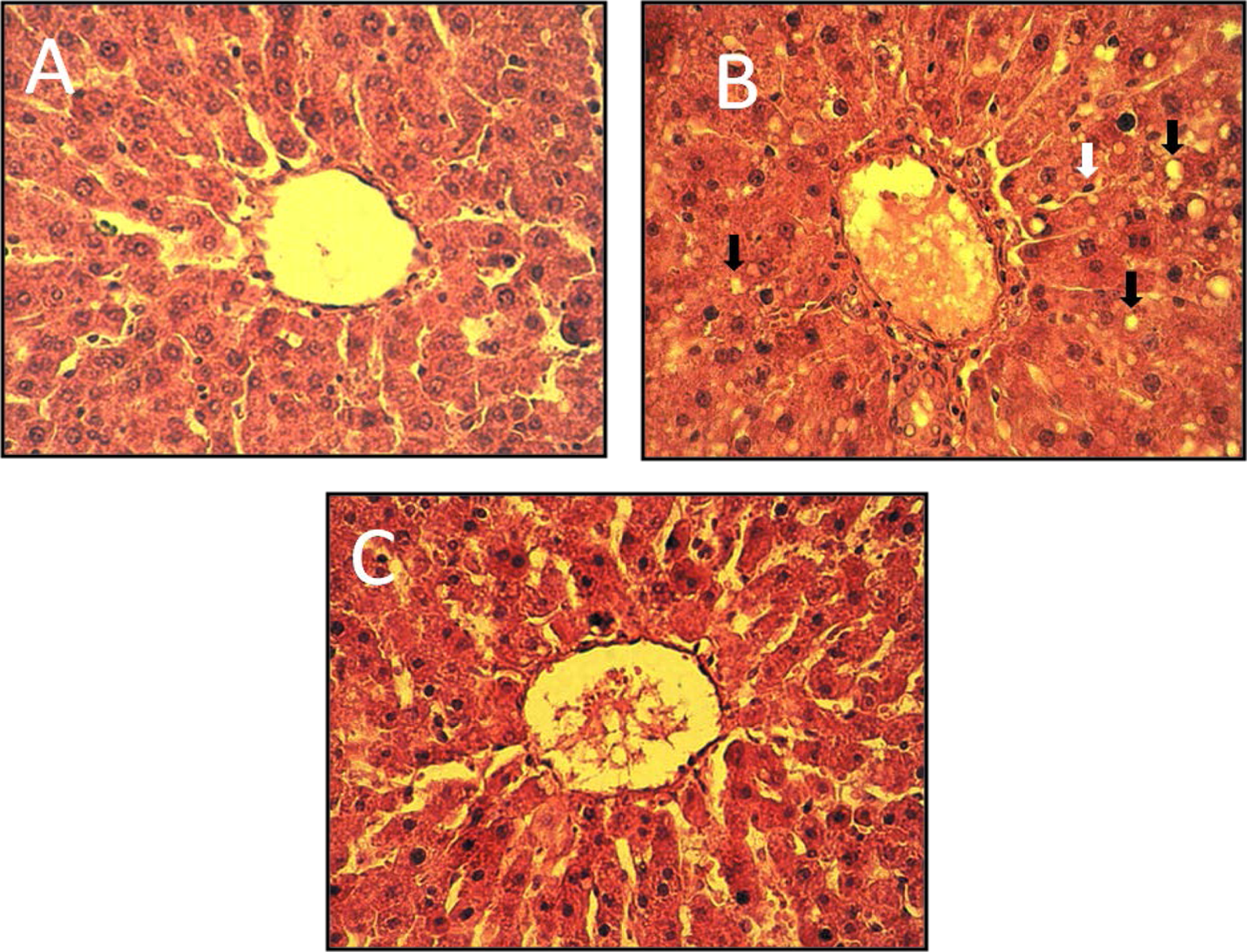
Hematoxylin–eosin-stained photomicrographs of representative rat livers from the control-IR (b), SO (a), and oil-IR (c) rats. Black arrows indicate cytoplasm vacuolation white arrows show PMNL infiltration. PMNL: polymorphonuclear; IR: ischemia–reperfusion; SO: sham operation.
Discussion
We investigated the temporal changes in three different antioxidant enzymes induced by tilapia fish oil on hepatic warm IRI. Study has revealed that administration of tilapia oil may be beneficial in the amelioration of antioxidants enzymes activities during hepatic ischemic injury. The postischemic hepatoprotection in tilapia oil treatment groups was accompanied by decreases in AST and ALT activities (Figure 1) and increased SOD, CAT, and GPx activities compared with the control group (Figure 3). Daily administration of tilapia oil for 3 weeks reduced the TBARS production during early phase of reperfusion (Figure 2).
Interruption of blood supply to the warm liver causes ischemia, which results in a decline in the energy supply of the metabolically active organ and severe damage. Paradoxically, the reinstatement of blood flow after ischemia induces additional damage due to production of ROS, leading to inflammation, cell death, and organ failure. 42,43 Mitochondria are accepted to be the main intracellular source of ROS within the hepatocytes. 44 During reperfusion, the mitochondrial ROS generation increases dramatically, 8,45 because electrons released by the respiratory chain can be directly donated to the newly supplied oxygen. 46 In this regard, much higher lipid peroxidation in the liver, measured during postischemic reperfusion, was not surprising and could be useful to estimate the extent of oxidative damage in tissues. 47 In the present study, it was demonstrated that I/R led to an increase in TBARS level both in liver control–WI group and Oil–WI, with a compensatory decrease in the SOD, CAT, and GPx activities. Possible consequences of elevated oxidative stress are overproduction of lipid peroxidation products such as TBARS. 48 Fish oil provides large amounts of PUFAs and thus could increase susceptibility to lipid peroxidation. 28 However, in this study, TBARS release in the hepatic tissue was not significantly different in ischemic groups during the late phase of reperfusion. Moreover, increased lipid peroxidation would be expected to increase cell injury, and in this study, tilapia fish oil was protective during the early phase of reperfusion.
To reduce the damaging effects of ROS, besides diminishing their production, organisms have developed their own antioxidant mechanisms, including various antioxidant enzymes. In the present study, antioxidant enzyme activities were decreased in ischemic groups during reperfusion, which was in agreement with some studies 49,50 and disagreement with other studies 51,52 . Normally, there is a balance between the ROS production and the endogenous scavenging system which detoxifies the ROS. 53 Dysfunction of CAT, SOD, and GPx may result in loss of protective activity exerted by these enzymes, and this may be the reason why we observed a reduction in CAT, SOD, and GPx levels after reperfusion in ischemic groups. If so, an antioxidant treatment should prevent the loss of antioxidants activities by effectively scavenging the excess ROS. In the present study, the reduction in the CAT, SOD, and GPx activity was limited by tilapia fish oil administration. Increases of the hepatic antioxidant activities and messenger RNA expression of those in mice fed on a diet rich in fish oil and the efficacy of SOD on IRI of the liver have also been reported. 51,54–59 CAT, with GPx, metabolizes peroxides including H2O2 and protects the cellular membranes from lipid peroxidation. 60 The hepatoprotective effect observed in the Oil–WI group may have existed in part because of sustained activities of CAT, SOD, and GPx compared with the control–WI group. It might also be attributable to higher activity of Mn-SOD after ischemia. Previous studies have reported that Mn-SOD plays a role against superoxide anions, which may directly affect mitochondrial function by initiating a free radical-mediated chain reaction. 61,62 The results from previous studies suggest that H2O2 inactivates SOD through a modification in histidine residue located in the active site of the enzyme. 63,64
Vacuolization of the liver hepatocyte after I/R is well studied. 65,66 The appearance of vacuoles in the cytoplasm of hepatocytes has already been reported to be induced by anoxia, as described in the morphogenesis of postmortem hepatocyte vacuolation. 67 Histological alterations and the extent of parenchymal vacuolation, in particular, reflected the severity of hepatocellular damage induced by WI and reperfusion. We observed that vacuolation of the hepatocyte cytoplasm was associated with I/R, but tilapia oil pretreatment could reduce the degree of cytoplasm vacuolation.
In a low flow, reflow perfusion model in the rat, fish oil reduced hepatic reperfusion injury. Following reperfusion, the rise of portal pressure was markedly minimized with significant reduction in trypan blue distribution time indicating improved microcirculation. 28 A controversy is raised from the study of Lo et al. which stated that dietary supplementation of rats with fish oil does not attenuate hepatic injury after WI/R. 29 However, the beneficial effects of fish oil in other studies have been attributed to the high concentrations of ω-3 fatty acid and tilapia oil contains amount of ω-3 fatty acid (C18:3n−3). Both n−6 and n−3 PUFAs are cellular lipids and substrates for the synthesis of physiological mediators. Linolenic acid (C18:3n−3) is the predominant dietary n−3 PUFA and the precursor for the long-chain n−3 PUFAs eicosapentaenoic acid (C20:5n−3; EPA) and docosahexaenoic acid (C22:6n−3; DHA). Both EPA and DHA have anti-inflammatory and tissue protective effects, 68 which may involve a direct antioxidant action due to their high susceptibility to free radical reactions. 69 Changes in membrane levels of EPA and DHA similar to the changes in plasma fatty acids of rats have been shown to inhibit the production of prostaglandin PGE2, 23,70 alter platelet adhesiveness, 71 and neutrophil adhesion to endothelial cells. 27,72 Therefore, one possible explanation is the fact that a fish oil-supplemented diet will decrease the production of endogenous protective prostaglandin PGE2 and PGE1 in the liver. Both PGE2 and PGE1 have been shown to be important in preventing liver injury following I/R. 73,74 Hence, fish oil-supplemented diets could enhance liver injury by decreasing membrane arachidonic acid content that leads to decreased production of protective prostaglandins.
Another explanation for the protective effect of tilapia fish oil on liver IRI may be related to the fact that dose of fish oil supplementation and the time of feeding used in our study may have been adequate to protect the liver from 30 min of I/R. It is possible that greater amount of fish oil and/or the longer periods of feeding could confer some protection on the liver to I/R. 29 In addition, the severity of our liver I/R rat model may be adequate to detect a difference between the diets used in our study.
The findings presented here indicate a clear effect of the tilapia fish oil-supplemented diet on the antioxidant enzyme activities of the liver after IRI but did not reduce the TBARS content during the late phase of reperfusion compared with those of the control group; however, the mechanism involved remains to be investigated. Histological examination as the end points when compared with the control–WI group show that tilapia fish oil pretreatment reduce the degree of cytoplasm vacuolation. In fact, pharmaceutical strategies have focused on the use of multifactorial biologic modulation cocktail to reduce the liver ischemic reperfusion injury and may be the administration of selected biologic reagents combined with fish oil targeting various processes helps to control self-perpetuating IRI.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
