Abstract
Aim:
This study aimed to determine whether nuclear factor erythroid 2-related factor 2 antagonized the oxidative stress induced by di-
Methods:
Mouse TM3 testicular Leydig cells were treated with Nrf2 knockdown (KD) or overexpression in the presence and absence of DBP. Oxidative profiles were examined. Nrf2 target antioxidant genes were studied, and the effects of Nrf2 inducer sulphoraphane (SFN) were tested.
Results:
DBP induced intracellular oxidative stress to a similar extent with Nrf2 KD. Expression and protein levels of Nrf2 were increased together with its target genes, namely heme oxygenase 1, nicotinamide adenine dinucleotide phosphate quinone oxidoreductase 1 and peroxiredoxin 6, following DBP stimulation. Use of SFN not only restored the intracellular oxidative toxicity but also cell proliferation and testosterone secretion in response to DBP.
Conclusion:
Increased Nrf2 activity, for example, by SFN can effectively antagonize the oxidative stress in testicular Leydig cells caused by DBP.
Introduction
Pollution remains a major health problem by intracorporeal accumulation of toxins. Incidences of neonatal hypospadias, cryptorchidism and other birth defects have been increasing in the recent years.
1
Exposure to endocrine-disrupting chemicals (EDCs) has been proven to play an important role in the development of those deformities.
2
Amongst all, di-
Our previous studies using animal models indicate that DBP could induce hypospadias and cryptorchidism in progenies. 4 Amid all organs, DBP has the highest affinity for ovaries and testes and induced oxidative stress in those organs, causing impaired hormone production. 5 Exposure to high dose of DBP during embryonic or lactation period can bring about obvious genital toxicity and developmental disorder. 6 Further studies point out that EDCs target testicular Leydig cells resulting in oxidative injury and causal compromise in hormone production. 7
Transcriptional factor nuclear factor erythroid -related factor 2 (Nrf2) has been recognized as one of the most important antioxidative modulators. After translocation into the nucleus, Nrf2 binds the antioxidant response element (ARE) and can mediate the transcription of a handful of endogenous antioxidative genes including heme oxygenase 1 (HO-1), nicotinamide adenine dinucleotide phosphate quinone oxidoreductase 1 (NQO1), and so on to antagonize reactive oxygen species (ROS) injury. 8 In the current study, we hypothesized that Nrf2 was upregulated in the testicular Leydig cells exposed to DBP and application of exogenous Nrf2 or Nrf2 inducer could restore the oxidative toxicity of DBP.
Materials and methods
Cell culture
Mouse Leydig TM3 cell line was obtained from the cell bank of Chinese Academy of Science (Shanghai, China). TM3 cells were cultured in Dulbecco’s modified Eagle’s and Ham’s F12 (DME/F12 1:1, vol/vol) medium (PAA, Germany) and 2.5% foetal bovine serum (PAA). DBP (Sigma-Aldrich; St Louis, Missouri, USA) was applied at 100 mg/L in all single-dose assays.
Quantitative real-time reverse transcription polymerase chain reaction
Total RNA was extracted with RNAiso reagent (TaKaRa, Dalian, China). RNAs were then converted to complementary DNAs (cDNAs) with PrimeScript™ RT Reagent Kit (TaKaRa). Primers for mouse Nrf2, HO-1, NQO1, glutamate–cysteine ligase catalytic subunit (Gclc), glutamate–cysteine ligase regulatory subunit (Gclm), glutathione
Western blotting
Total protein of lysates was extracted and equal protein amount of 25 μg was loaded onto 10% sodium dodecyl sulphate polyacrylamide gel for electrophoresis. Gels were subsequently transferred to nitrocellulose membrane. The membranes were blockaded for 1 h with 5% non-fat milk. Primary antibodies of Nrf2, HO-1, NQO1, Gclc, Gclm, GSTM1, Prx6 and Tubulin were then added, and the membranes were kept at 4°C overnight. Corresponding secondary antibodies were applied followed by horseradish peroxidase application.
Measurements of 8-oxo-dGuo, GSH and ROS
Liquid chromatography–mass spectrometry was used to measure concentrations of 8-oxo-7,8-dihydro-2′-deoxyguanosine (8-oxo-dGuo) and glutathione (GSH). Total GSH and oxidized GSH (GSSG) concentrations were calculated from a standard curve based on GSSG (Cayman Chemical, Ann Arbor, Michigan, USA) according to the GSH assay kit and normalized to protein concentration. Net intracellular accumulation of ROS was measured with fluorescent probe species. 9 After 2 h of treatment with antioxidants, cells were processed with Hank’s balanced salt solution (HBSS) (Gibco, Carlsbad, California, USA) and loaded with dihydrodichlorofluorescein diacetate. After incubation, cells were washed twice with HBSS, suspended in complete medium and examined under a microscope. The number of dichlorofluorescein -stained cells was calculated.
Luciferase reporter assay
A detailed protocol was described previously by Tanigawa et al.
9
Briefly, the oligonucleotides corresponding to the ARE of the mouse
Generation of Nrf2-shRNA and Nrf2-lentivirus
Fifth passage TM3 cells were allowed to adhere for 24 h before transfection. Using Lipofectamine 2000, cells were transfected with plasmids-containing short hairpin RNA (shRNA) against mouse Nrf2 (Santa Cruz Biotechnology Inc., Santa Cruz, California, USA). Two shRNAs were generated together with a control shRNA to optimize efficiency. At 48 h post-infection, puromycin (1:5000) was added for selection. Cells were cultured for 2 weeks for generation of stable lines, and efficiencies were further assessed by Western blotting. Generation of Nrf2-bearing lentivirus was carried out according to well-established protocols. Briefly, the Nrf2 expression virus was cloned by digesting Nrf2 from percutaneous coronary intervention-Nrf2 and then half-blunt ligating it into the Lv control displacing control site. Production and titration of the virus were previously described by Hurttila et al. 10
CV assay
Leydig cells were seeded in 96-well plates at a density of 2500 cells/well and were treated with 10 μM of DBP and control respectively for 0, 24, 48 and 72 h. Crystal violet (CV) assay was applied at each time point. After medium was gently removed, cells were fixed with 10% formalin for 5 min and were then stained with 0.05% CV for 30 min. After two washes, straight methanol was applied and plates were read at absorbance of 540 nm. Normalization in panel (a) was made to absorbance of control at 0 h.
Measurement of testosterone level
Cells were treated with luteinizing hormone (LH), and testosterone content was detected by mouse testosterone enzyme-linked immunosorbent assay kit (Solarbio, Shanghai, China) according to the manufacturer’s protocol. Briefly, cells were stimulated by LH (100 ng/mL) for 12 h and media was collected. Samples were diluted serially, added to 96-well plates, and kept at room temperature for 12 min. After sequential applications of two working reagents, samples were treated with substrates and were kept in darkness for colour development. Optical densities were read at 450 nm of absorbance. Data were plotted as percentage of change compared with the control.
Statistical analysis
All assays were repeated at least three times and were presented as mean ± standard deviation. The two-tailed Student’s
Results
DBP and Nrf2-KD induces ROS in testicular Leydig cells
As DBP is an established toxin inducing oxidative stress, we compared ROS levels in Leydig cells treated either by DBP or by shRNA of Nrf2, a well-established transcription factor with antioxidative activity. The synthesized shRNAs as well as lentiviral vector for Nrf2 overexpression showed satisfactory efficacy in altering messenger RNA (mRNA) expression (Figure 1(a)) and protein levels (Figure 1(b)) in testicular Leydig cells compared with the control. DBP significantly increased ROS level in Leydig cells to a similar extent of Nrf2-KD (Figure 1(c)). Administration of TPA further increased ROS levels of control, DBP and Nrf2-KD groups (Figure 1(c)). Exposure to ROS inhibitor, carbon monoxide-releasing molecule (CORM) abolished the induction ROS by TPA (Figure 1(c)). ROS levels between cells treated with DBP and Nrf2-KD remained unchanged despite TPA or CORM exposures, indicating that loss of Nrf2 could be necessary to reduce ROS induced by DBP.

DBP induces intracellular ROS. Nrf2 shRNAs and lentiviral vectors showed satisfactory efficiency in mRNA (a) and protein level (b). Intracellular ROS levels were measured using DCF staining (c). Determination of the concentration of total GSH in cells treated and untreated with TPA (10 μM) for 24 h (d). Determination of the ratio of GSH/GSSG (e) and 8-oxo-dGuo levels in Leydig cells (f). Data are presented as mean ± SD (
In order to better profile ROS state, we then measured intracellular redox level in terms of the ratio of reduced GSH to GSSG. Total GSH was significantly increased by DPB and Nrf2-KD, and the level was further increased with the presence of TPA (Figure 2(d)). Nonetheless, the GSH/GSSG ratio was significantly reduced in cells treated with DBP and Nrf2-KD and was further decreased (Figure 2(d)). Consistently, effects between DBP and Nrf2-KD remained statistically insignificant. The redox profile indicated that DBP induced a similarly more oxidized intracellular milieu in Leydig cells to Nrf2-KD. We next looked at the level of 8-oxo-dGuo, a major product of DNA oxidation. Likewise, DBP and Nrf2-KD significantly increased 8-oxo-dGuo level, which was further elevated by TPA, with, still, insignificant changes between DBP and Nrf2-KD (Figure 1(f)). We thus concluded that DBP induced ROS in testicular Leydig cells to a similar extent of Nrf2-KD, which indicated that Nrf2 could play a critical role in the DBP-induced intracellular oxidative stress.

DBP induces increased Nrf2. Real-time qRT-PCR showing DBP-induced changes in Nrf2 and Nrf2-targeted antioxidant genes (a). Western blotting revealing Nrf2 protein change following DBP stimulation. (b) Relative NQO1 promoter reporter activity in Leydig cells with and without Nrf2-KD in response to DBP treatment over time (c) and at different doses (d). Protein level change of Nrf2 and its target genes with different Nrf2 status and DBP treatment (e). DBP: di-
DBP induced upregulation of Nrf2
We next hypothesized that endogenous Nrf2 could be increased in the presence of DBP to active downstream antioxidative responses. We first measured expressions of a series of Nrf2 responsive genes with qRT-PCR. We found that Nrf2 expression was significantly increased in response to DBP, together with upregulation of its target genes of HO-1, NQO-1 and Prx6 (Figure 2(a)). Protein levels of Nrf2 corresponded to expression change of mRNA (Figure 2(b)). In order to elucidate the role of Nrf2 in the antioxidative response to DBP, we constructed a luciferase reporter containing ARE derived from NQO1 promoter. As expected, ARE activity of Nrf2-KD Leydig cells was substantially decreased and responded to prolonged DBP stimulation to a much less fold change in comparison with the controls (Figure 2(c)). Using the same luciferase reporter, we also examined responses between Nrf-WT and Nrf-KD cells to different doses of DBP, which induced increased ARE promoter activity of Nrf2-KD cells to a much less extant compared with the controls (Figure 2(d)). The uncompromised ARE activity in the presence of both WT Nrf2 and DBP indicated that DBP did not impair the binding ability of Nrf2 to ARE elements.
We thus further investigated whether replenish or overexpression of Nrf2 in Leydig cells could implement target antioxidant genes in the presence of DBP. Using lentivirus delivered Nrf2 plasmid, we found that overexpression of Nrf2, in the presence of DBP, substantially upregulated HO-1, NQO1 and Prx6, which was in accordance with qRT-PCR results (Figure 2(e)). This effect could not be simulated by CORM, in line with results shown in Figures 1(c) to (f) and 2(e). We thus concluded that in order to keep the DBP-induced oxidative stress low, Nrf2 overexpression was an optimal modality surpassing the effect of ROS inhibitor.
Upregulation of Nrf2 by SFN restores the oxidative stress by DBP
Based on the results aforementioned, we hypothesized that oxidative stress by DBP could be restored by Nrf2 upregulation. We thus used SFN, a critical antioxidant that induced nuclear translocation of Nrf2 and ARE transactivation. Here we showed that SFN increased Nrf2 level comparable with lentiviral delivery (Figure 3(a)). We then examined intracellular ROS level and 8-oxo-dGuo. We found that SFN almost completely restored oxidative stress caused by DBP in Leydig cells (Figure 3(b) to (c)).
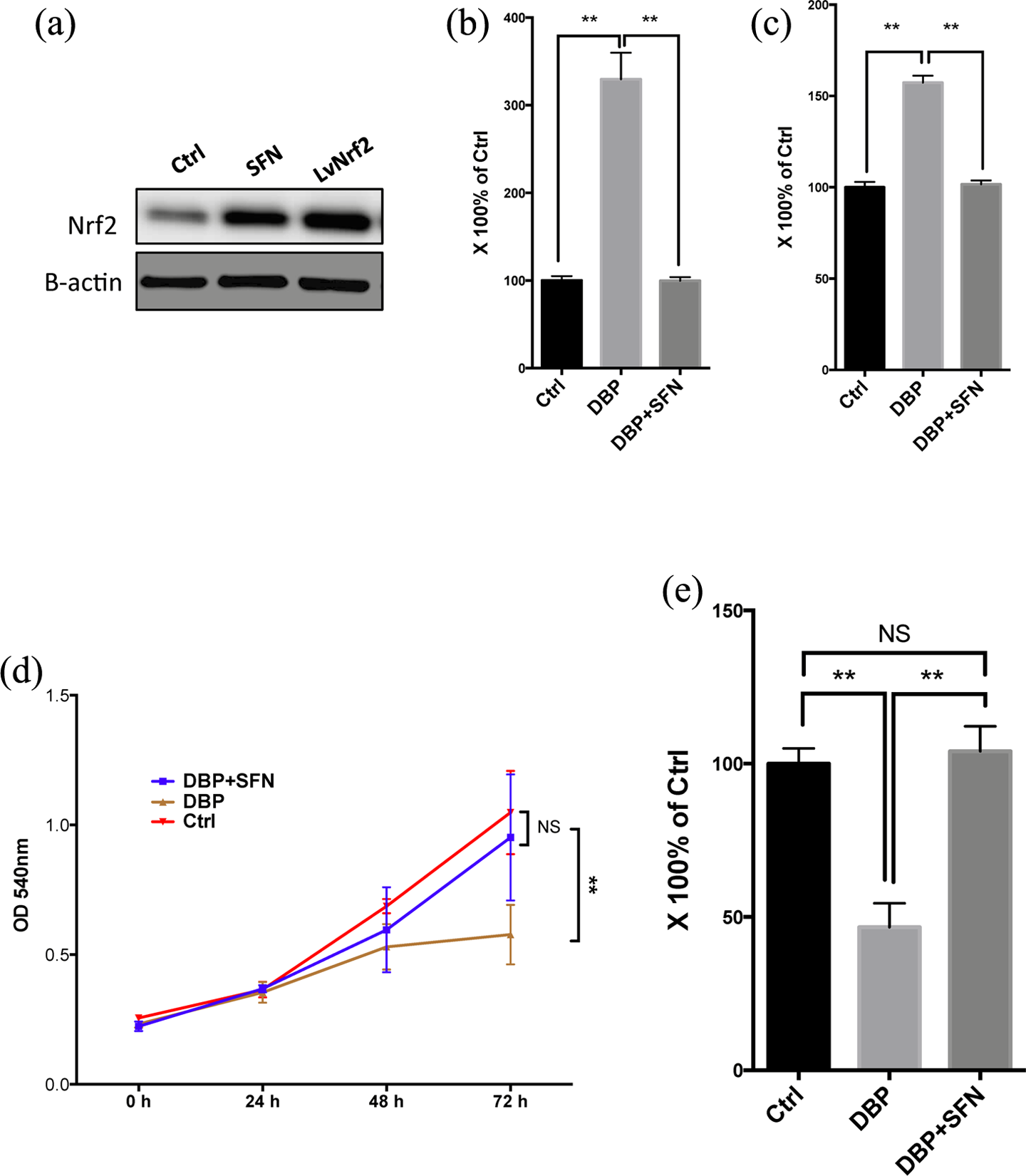
(a) Western blotting showing Nrf2 level treated by SFN and lentiviral overexpression. Determination of intracellular ROS (b) and 8-oxo-dGuo levels (c) in Leydig cells treated with DBP and DBP + SFN. Proliferation curves showing effects of DBP alone and with SFN on Leydig cells (d). ELISA detected testosterone levels in the culturing media of Leydig cells stimulated by LH between different treatment groups (e). Nrf2: nuclear factor erythroid 2-related factor 2; SFN: sulforaphane; 8-oxo-dGuo: 8-oxo-7,8-dihydro-2′-deoxyguanosine; DBP: di-
We next investigated whether salvage of ROS stress led to recovery of functions of Leydig cells. We first examined cell proliferative ability using CV assay and found that at 72 h of treatment, DBP significantly lowered proliferation compared with the control and DBP + SFN, indicating that SNF could recover cell proliferation declination induced by DBP (Figure 3(d)). One of the most critical biological activities of testicular Leydig cells was to produce testosterone in response to follicle-stimulating hormone (FSH) stimulation. We then measured testosterone level in the medium of cells stimulated by exogenous FSH. In comparison with the control, DBP induced significantly decreased hormone production, whilst SFN restored the impairment back to normal (Figure 3(e)). We therefore conclude that SFN, via induction of Nrf2 activity, antagonized the oxidative stress induced by DBP and restored cellular function.
Discussion
Orthophthalates are synthetic chemical esters derived from phthalic acid and serve various roles in many of the consumer products resulting in ubiquitous daily exposures in adults and children. 11 –13 Some phthalates are suspected EDCs, with well-described antiandrogenic properties in prenatally exposed rats. Phthalates have drawn both media attention and regulatory scrutiny because of their extensive prevalence in commercial products; measurable exposure in pregnant women, infants and children and potential hazard to normal human development. Amongst phthalate acid esters (PAEs), DBP has been extensively used in the production of plastics, paint, rubber, pesticides, cosmetics, fragrant and lubricants. As it detaches from organics easily, DBP is highly contaminant via air, water and soil. Nonetheless, the mechanism of cumulative DBP causing genital disorders remains unclear. We have previously revealed that maternal exposure to DBP and other PAEs induced hypospadias and cryptorchidism in progenies. Also, DBP caused oxidative injuries in a variety of organs like brain, lung, liver, ovaries and testes, amongst which testes and ovaries are most sensitive to the toxicity, indicating the major toxicity of DBP in genitourinary system. 4,5 As embryonic period is essential for organ development and DBP can pass through placenta, maternal accumulation can easily induce disorders in multiple foetal organs. Testicular Leydig cells are where testosterone is synthesized and are major target of PAEs. Exposure to PAEs renders Leydig cells incapable of secreting sufficient hormone sustaining the genital tubercle for further development. 7 Therefore, we conclude that DBP may impair testicular Leydig cells via oxidative stress like in other organs. 14 Importantly, we have previously screened a series of proteomic profiles in the DBP-exposed murine testes and penis and have found a series of increased antioxidant proteins levels. 15,16
The Nrf2 is encoded by the
In the current study, we not only reveal that DBP induces oxidative stress in testicular Leydig cells but also demonstrate that Nrf2 expression is increased in response to DBP stimulation. However, the response is not sufficient to further restore the intracellular ROS-associated injury nor can the ROS inhibitor CORM recover the damage. We find that direct upregulation of Nef2 is capable of restoring the ROS stress, which supports the use of SFN restore DBP-induced Leydig cell injury. Future study is thus warranted to recapitulate our finding in vivo.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was supported in part by the General Project of National Natural Science Foundation of China (No. 81370781), the Natural Science Foundation of Jiangsu Province (No. BK2011860) and the College Students’ Practice Innovation Training Project for Higher Education of Jiangsu Province (No. 201310312001).
