Abstract
This study was conducted to investigate the protective effects of sodium p-aminosalicylic acid (PAS-Na) on learning and memory via increasing the number of basal forebrain choline acetyltransferase (ChAT) neurons in manganese (Mn)-exposed rats. Male Sprague Dawley rats were divided into following groups: the normal control I, II, and III groups, the model I, II, and III groups, low- and high-dose PAS-Na treatment (L- and H-PAS) group, PAS-Na prevention (PAS-P) group, and PAS-Na treatment (PAS-T) group. The model I, II, and III groups, L- and H-PAS, and PAS-T groups received intraperitoneal (i.p.) injection of 15 mg/kg manganese chloride tetrahydrate (MnCl2·4H2O) for 3 or 12 weeks, while the normal control I, II, and III groups received i.p. injection of an equal volume of saline; L- and H-PAS and PAS-T groups received back subcutaneous (s.c.) injection of PAS-Na (100 and 200 mg/kg) for the next 5 or 6 weeks, whereas model I and II group received back s.c. injection of an equal volume of saline. However, PAS-P group received back s.c. injection of 200 mg/kg PAS-Na + i.p. injection of 15 mg/kg MnCl2·4H2O for 12 weeks. Mn exposure significantly reduced the ability of spatial learning and memory capability, while PAS-Na prevention recovered it. Mn decreased the number of ChAT-positive neurons in vertical limb nucleus of the basal forebrain diagonal band/horizontal limb nucleus of the basal forebrain diagonal band and ChAT protein activity and treatment or prevention with PAS-Na restored those comparable with control. In brief, our results showed that PAS-Na may have protective effects on learning and memory against Mn via increasing the number of ChAT-positive neurons and activity of ChAT protein.
Keywords
Introduction
Manganese (Mn) is an essential trace element of an organism, and a coenzyme factor or activator which involves in metabolic reactions. However, excessive intake of Mn can damage learning and memory functions, 1,2 cause severe neurological dysfunction, including tremor, bradykinesia, and gait difficulties which are similar to Parkinsonian syndrome. Additionally, manganism patients also display neuropsychological difficulties including memory loss, apathy, and even psychosis. 3 Mn is mainly accumulated in the brain regions including basal ganglia striatum and globus pallidus, 4 which are important components of the basal forebrain. Magnetic resonance images of occupationally Mn-exposed workers exhibited high signal densities in the basal ganglia attributed to Mn. 5 –7 The cholinergic neurons of vertical limb nucleus of the basal forebrain diagonal band (vDB) send projection fibers to hippocampus and horizontal limb nucleus of the basal forebrain diagonal band (hDB) send projection fibers to the olfactory bulb. 8 In the above loop, cholinergic neurons synthetize and release acetylcholine (Ach) to maintain the normal spatial learning and memory function. Afferents of cholinergic is crucial for physiological movement, cognitive, emotional, and behavioral reactions, so that the loop damaged can lead to severe memory impairment. Mn is a chemical irritant in cholinergic neurons, and it can cause cell homeostasis disorder in the particular areas. The symptoms of manganism are apathy, cortical function, movement disorders, and other clinical symptoms that may be related to the effects of Mn on the cholinergic system. 9 Increased levels of Mn in the striatum, decreased ability of independent activities, and reduced levels of dopamine (DA) in striatum had been found in C57VL/6 mice that were exposed to Mn for 8 weeks by intragastric gavage. Histochemical staining with fluoro-Jade and cresyl fast violet showed that Mn exposure results in neuronal injury in the striatum and globus pallidus, particularly in the proximal part of the microvasculature. Terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate-biotin nick-end labeling (TUNEL) positive cells are expressed by neuronal nitric oxide synthase (NOS); enkephalin was observed almost exclusively in striatum and choline acetyltransferase (ChAT) positive neurons was observed in globus pallidus. 10 In addition, research showed that subchronic Mn exposure leads to a decrease in the number of vDB/hDB ChAT-positive neurons in basal forebrain. 11
In the 1970s, Tandon and Singh 12 first discovered that p-aminosalicylic acid (PAS) can promote excretion of Mn in Mn-exposed rats and rabbits. In 1992, Ky et al. 13 reported successful results after treatment with sodium p-aminosalicylic acid (PAS-Na) in two Mn-poisoned patients. Subsequently, we conducted a follow-up study on one of the patients for 17 years and confirmed its promising long-term prognosis. 3 Additionally, PAS reduced the concentration of Mn in blood and brain in Mn-exposed rats. Zheng et al. 14 used PAS (100 or 200 mg/kg/day) to treat Mn-exposed rats. They found that treatment with PAS for 3 weeks decreased the Mn content in striatum, choroid plexus, cerebrospinal fluid (CSF), heart, liver, testis, spleen, pancreas, and erythrocytes (RBCs) of subacute Mn-exposed rats; PAS treatment for 6 weeks reduced Mn content in striatum, hippocampus, thalamus, motor cortex, choroid plexus, and RBCs. In recent studies, we found that daily back subcutaneous (s.c.) injection of PAS-Na (100 or 200 mg/kg/day) for 3 or 6 weeks has antagonism effects on learning and memory dysfunction via improved hippocampus apoptosis, mitochondrial dysfunction, and the abnormal expression of neurogliocyte growth and differentiation in subacute Mn-exposed rats. 2 However, the exact mechanism of Mn and PAS-Na action on the learning and memory dysfunction remain unknown.
Therefore, we evaluated the therapeutic effects of PAS-Na on Mn-induced learning and memory impairment in rats. Furthermore, the underlying mechanism was investigated by immunohistochemical analysis and the activity of ChAT protein in the basal forebrain intend to explore the relationship between the number of ChAT-positive neurons in the basal forebrain and the ability of learning and memory. This research may provide new insight into the remedy of Mn-induced learning and memory impairment.
Materials and Methods
Chemicals
ChAT monoclonal mouse anti-rat antibody was obtained from Chemicon company (Temecula, California, USA), pikas Universal SuperPicTure-horseradish peroxidase (HRP) polymer detection kit was obtained from Tianjin veins Company (Tianjin, China), diaminobenzidine (DAB) chromogenic reagent kit and normal goat serum from Beijing Zhongshan Golden Bridge Biotechnology Co. Ltd (China), the organization ChAT assay kit and Coomassie blue protein assay kit from Nanjing Jiancheng Bioengineering Institute (China); manganese chloride tetrahydrate (MnCl2·4H2O) was obtained from Tianjin Bodi Chemical Co. Ltd (China); PAS-Na in powder form for injection was supplied by Liaoning times odd Pharmaceutical Co. Ltd (China). All reagents were of analytical grade, high-performance liquid chromatography grade, or the best available pharmaceutical grade. MnCl2·4H2O and PAS-Na salt were dissolved in sterile saline each day prior to administration.
Experimental animals
Male Sprague Dawley (SD) rats (weighing 180.2 ± 14.7 g and 80 ± 20 g), specific pathogen free, were obtained from the Experimental Animal Center of Guangxi Medical University [SCXKG 2009-0002]. All experimental procedures were conducted in conformity with institutional guidelines for the care and use of laboratory animals in Guangxi Medical University, Nanning, China, and conformed to the National Institutes of Health Guide for Care and Use of Laboratory Animals. The animals were housed under a temperature-controlled, 12-h/12-h light/dark cycle (temperature: 24 ± 1°C and humidity: 45–65%) room and were allowed to acclimatize in quarantine for 1 week prior to experimentation. Food and water were available ad libitum.
Experimental design
In the subacute experiment, for a period of 8 weeks, 32 male SD rats, weighing (180.2 ± 14.7 g) at the beginning of the experiment, were randomly divided into 4 groups with 8 rats in each group. To induce learning and memory impairment, the model I group, and low- and high-dose PAS-Na treatment (L- and H-PAS, representative of low and high dosage, respectively) group received intraperitoneal (i.p.) injection of 15 mg/kg MnCl2·4H2O, while the normal control I group received i.p. injection of physiological saline in the same volume, once a day, all at 5 days a week for 3 weeks. Then, L- and H-PAS group rats received back s.c. injection of PAS-Na (100 and 200 mg/kg). While the model I and normal control I groups received back s.c. injection of physiological saline in the same volume once a day, for 5 weeks.
In the subchronic experiment, for a period of 12 and 18 weeks, 48 male SD rats, weighing (80 ± 20 g) at the beginning of the experiment, were randomly assigned into 6 groups with 8 rats in each group. the model II and III groups, PAS-Na prevention (PAS-P) group, and PAS-Na treatment(PAS-T) group received i.p. injection of 15 mg/kg MnCl2·4H2O to induce learning and memory impairment, while the normal control II and III group received i.p. injection of physiological saline in the same volume once a day, 5 days a week for 12 weeks. In addition to MnCl2·4H2O administration, PAS-P group received back s.c. injection of 200 mg/kg PAS-Na, once a day, 5 days a week for 12 weeks. Then, PAS-T group rats received back s.c. injection of 200 mg/kg PAS-Na, while the model III group and the normal control III group received back s.c. injection of physiological saline in the same volume, once a day, which lasts for 6 weeks. The total duration of the experiment was 12 and 18 weeks.
Observational indexes
Morris water maze task
Learning and memory ability of rats was measured by Morris water maze test 6 days before the end of drug administration consecutively for 5 days, measured at the same time in each day. For memory training, each rat underwent four successive trials a day, and there were four different water-entering points in south, east, west, and north directions, but the location of the platform was constant. Latency to find the platform was measured up to a maximum of 90 s. The rat was left on locating the platform for 15 s prior to the next trial. If the rat failed to locate the platform within 90 s, it was guided to the platform and the escape latency was recorded for 90 s. The interval between the trials every day was 10 min to let rat recover physical capacity and to record escape latency and total distance swimming.
Immunohistochemical analysis detects the basal forebrain ChAT-positive neurons number
After Morris water maze experiment, rats were deeply anesthetized by i.p. injection of 3.5% chloral hydrate (10 ml/kg) and perfused with 150 ml of saline, followed by 500 ml of 4% paraformaldehyde in 0.1 M phosphate-buffered saline (PBS) with pH 7.4. The brain of rats were placed in the brain stereotaxic instrument, the anterior of bregma, which is 1.6 mm, and posterior fontanelle, which is approximately 0.3 mm, were removed, postfixed in the same fixative for 4 h, cryoprotected in 30% sucrose solution (4°C) for 12 h and 40-μm-thick serial coronal sections were cut on a freezing microtome (Leica Microsystems, Germany), to make ChAT immunohistochemical slice. Following permeabilization and washing with 0.01 M PBS for 15 min, cells were treated with 0.3% hydrogen peroxide to quench endogenous peroxidases and then blocked in normal horse serum for 25 min. Then, after 1 h at 37°C, rat anti-ChAT primary antibody (1:800) was added, and the sections were incubated overnight at 4°C. Then, the cells were rinsed for 15 min in PBS, SuperPicTure-HRP polymer was incubated at room temperature for 2 h, again rinsed for 15 min in PBS, and finally, nucleus staining was carried out with DAB dye. The tissue sections were dehydrated by alcohol, were rendered transparency by adding xylene, covered with neutral gum, and placed under an optical microscope To count the number of ChAT-positive neurons in the bilateral nuclear groups (cell body with claybank).
Assay of basal forebrain ChAT activity
After the Morris water maze test, the rats were euthanized by decapitation. Immediately after decapitation, the brain was rapidly excised and dissected on ice to obtain the basal forebrain. Brain samples were washed immediately with ice-cold saline to remove as much blood as possible, and the liquid in the brain tissue surface was absorbed using filter paper. All the tissue samples were stored at −80°C until analysis. Brain samples were homogenized in normal saline to give a 10% (w/v) brain tissue homogenate. The homogenates were then centrifuged at 3000 r/min for15 min at 4°C, and the supernatants were used immediately for the determination of ChAT activity. Activity of the basal forebrain ChAT was determined according to the instructions on the kit.
Statistical analysis
The statistical analysis was performed by Statistical Package for Social Sciences Version 16.0 software (SPSS Inc., Chicago, Illinois, USA), and all data were presented as mean ± SD. One-way analysis of variance was performed for each of the dependent variables measured in the experiment, and Tukey’s test was used in the post hoc multiple comparisons. The value of p < 0.05 was considered to be statistically significant.
Results
Results for Morris water maze test learning and memory abilities
Effects of PAS-Na on escape latency in Mn-exposed rats
The escaping latency for a period of 8 weeks obviously increased in the model I group when compared with the normal control I group (p < 0.05, Figure 1).
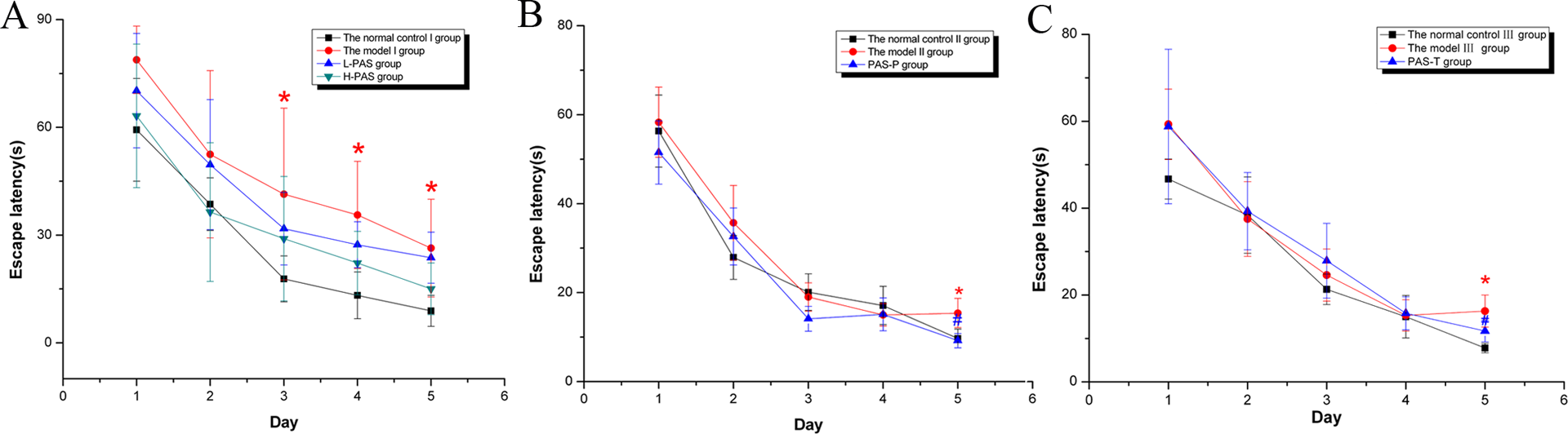
Effects of PAS-Na on escape latency in Mn-exposed rats. (A) Observation for 8 weeks; (B) observation for 12 weeks; (C) observation for 18 weeks. Note: Results were presented as the mean ± SE. *p < 0.05: significant as compared to the normal control group; # p < 0.05: significant as compared to the model group. PAS-Na: sodium p-aminosalicylic acid; Mn: manganese.
The escape latency of the model II and III groups for the period of 12 and 18 weeks were significantly longer than those of the normal control II and III groups, PAS-P, or PAS-T groups (p < 0.05, Figure 1).
Effects of PAS-Na on swimming distance in Mn-exposed rats
The swimming distance of the model I group for a period of 8 weeks was longer than that of the normal control I group (p < 0.05, Figure 2).

Effects of PAS-Na on swimming distance in Mn-exposed rats. (A) Observation for 8 weeks; (B) observation for 12 weeks; (C) observation for 18 weeks. Note: Results were presented as the mean ± SE. *p < 0.05: significant as compared to the normal control group; # p < 0.05: significant as compared to the model group. PAS-Na: sodium p-aminosalicylic acid; Mn: manganese.
The swimming distance of the model II and III groups for the period of 12 and 18 weeks were longer than those of the normal control II and III groups, and the swimming distance of PAS-P and PAS-T groups were shorter than those of the model II and III groups (p < 0.05, Figure 2).
The effects of PAS-Na on basal forebrain ChAT-positive neurons cell morphology and number
Morphological observation
ChAT immunoreactive products were stained brown, mainly in the cytoplasm of neurons and the proximal end of neuritis. ChAT-positive neurons were densely distributed in symmetrical left and right sides of basal forebrain (Figure 3).

The representative morphology of vDB/hDB ChAT-positive neurons (×400 magnification). (A) vDB ChAT-positive neurons are located in the central of the septal area, and their number in left and right sides are roughly equal. Both the side groups formed an inverted “V” shape, the dorsal part was close to the midline, and the ventral part was far away from the midline. Positive cells were mostly spindle shaped, oval or ovoid, axons were smooth and slender, and branch could be imaged. (B) The hDB ChAT-positive neurons were located in the ventral part and were densely arranged as the largest number of ChAT-positive neurons especially the medium- and small type of oval or fusiform cells. The arrows indicate vDB/hDB ChAT-positive neurons. vDB: vertical limb nucleus of the basal forebrain diagonal band; hDB: horizontal limb nucleus of the basal forebrain diagonal band; ChAT: choline acetyltransferase.
The numbers of ChAT-positive neurons in the normal control groups, L-PAS, H-PAS groups, PAS-P group, and PAS-T group were greater and evenly distributed in the vDB or hDB. The number of ChAT-positive neurons in the model group were significantly reduced (Figures 4 and 5).
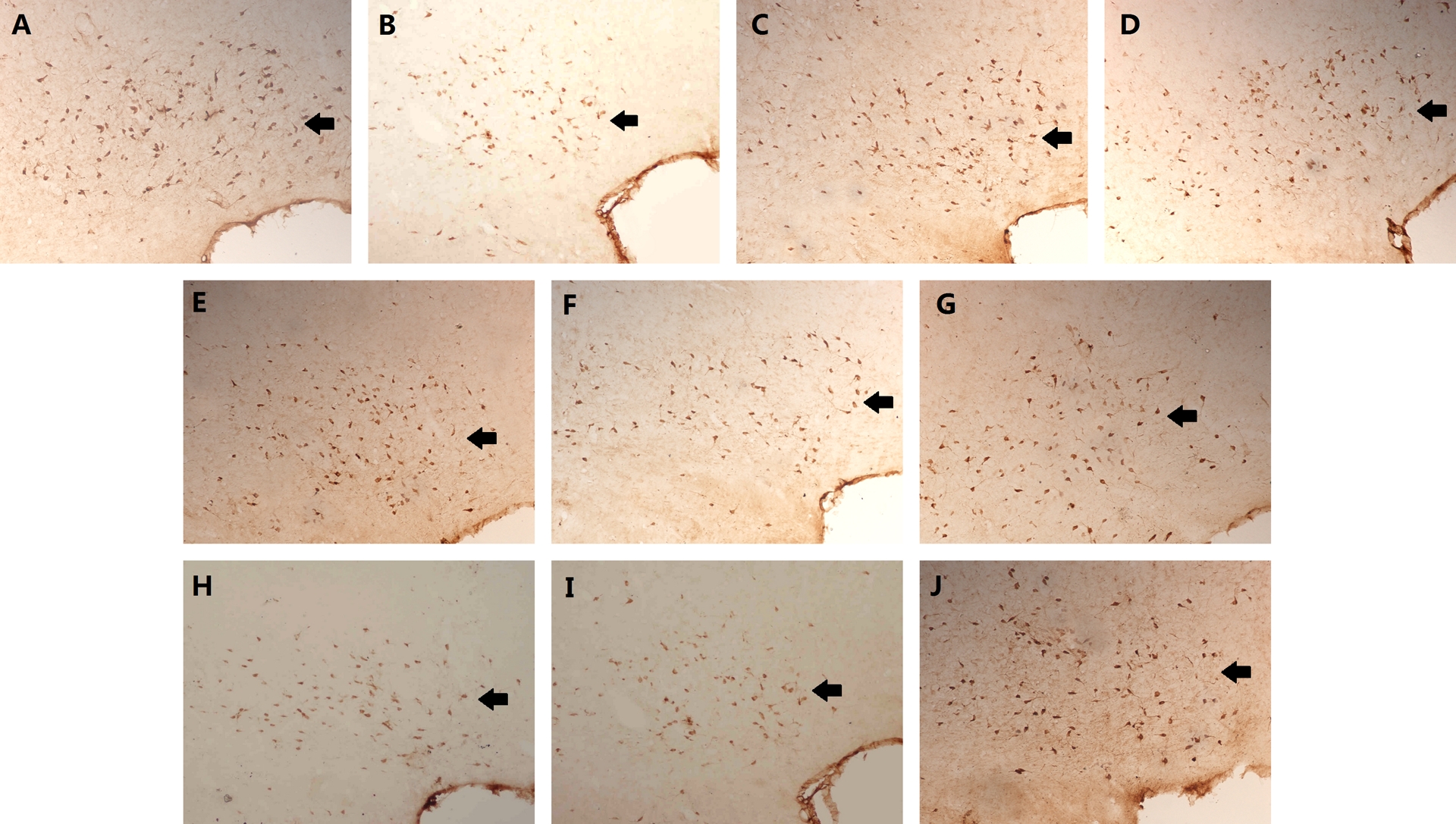
Effects of PAS-Na on positive neurons of vDB ChAT in Mn-exposed rats (×200 magnification). (A) the normal control I group; (B) the model I group; (C) L-PAS group; (D) H-PAS group; (E) the normal control II group; (F) the model II group; (G) PAS-P group; (H) the normal control III group; (I) the model III group; (J) PAS-T group. The arrows indicate vDB ChAT-positive neurons. PAS-Na: sodium p-aminosalicylic acid; Mn: manganese; vDB: vertical limb nucleus of the basal forebrain diagonal band; ChAT: choline acetyltransferase.

Effects of PAS-Na on positive neurons of hDB ChAT in Mn-exposed rats (×200 magnification). (A) the normal control I group; (B) the model I group; (C) L-PAS group; (D) H-PAS group; (E) the normal control II group; (F) the model II group; (G) PAS-P group; (H) the normal control III group; (I) the model III group; (J) PAS-T group. The arrows indicate hDB ChAT-positive neurons. PAS-Na: sodium p-aminosalicylic acid; Mn: manganese; hDB: horizontal limb nucleus of the basal forebrain diagonal band; ChAT: choline acetyltransferase.
Number of ChAT-positive neurons
The number of ChAT-positive neurons of hDB in the model I group for a period of 8 weeks was less than those of the normal control I group (p < 0.05, Figure 6).
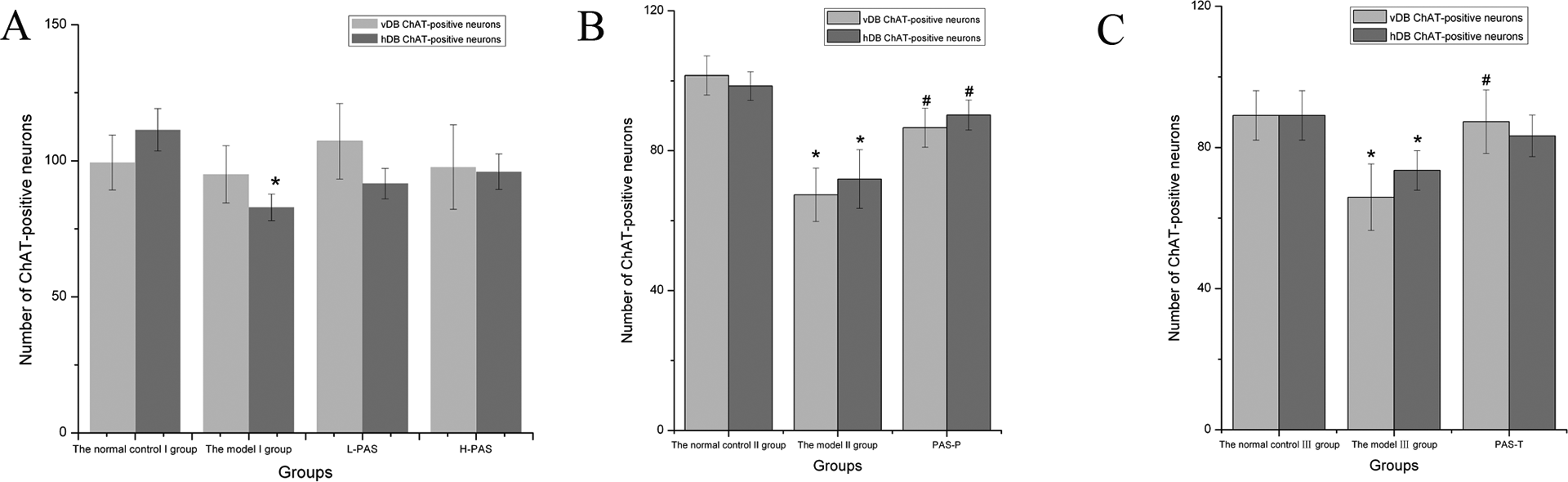
Effects of PAS-Na on the expression of vDB and hDB ChAT-positive neurons in Mn-exposed rats (A) Observation for 8 weeks; (B) observation for 12 weeks; (C) observation for 18 weeks. Note: *p < 0.05: significant as compared to the normal control group; # p < 0.05: significant as compared to the model group. PAS-Na: sodium p-aminosalicylic acid; Mn: manganese; vDB: vertical limb nucleus of the basal forebrain diagonal band; ChAT: choline acetyltransferase; hDB: horizontal limb nucleus of the basal forebrain diagonal band.
The number of ChAT-positive neurons of vDB/hDB in the model II group for a period of 12 weeks was less than those in the normal control II group or PAS-P group (P <0.05). For a period of 18 weeks, the number of ChAT-positive neurons of vDB/hDB in the model III group was less than those in the normal control III group, and the number of ChAT-positive neurons of vDB in PAS-T group was greater than those in the model II group (p < 0.05, Figure 6).
ChAT protein activity
For a period of 12 weeks, the ChAT protein activity in the model I group was less than that in the normal control I group or PAS-P group (p < 0.05, Figure 7).

Effects of PAS-Na on ChAT protein activity in Mn-exposed rats. (A) Observation for 12 weeks and (B) observation for 18 weeks. Note: *p < 0.05: significant as compared to the normal control group; # p < 0.05: significant as compared to the model group. PAS-Na: sodium p-aminosalicylic acid; Mn: manganese; ChAT: choline acetyltransferase.
Discussion
Morris water maze is not only a classic detective method of training learning and memory capability but also a reliability index to evaluate spatial learning and memory ability of rodent animals. The spatial memory impairment of rats induced by intranasal infection of MnCl2 (0.2 mg/kg/day and 0.8 mg/kg/day consecutively for 2 weeks) was related to the changes of 5-serotonin, norepinephrine, and DA neurotransmitter in brain. 15 On observation for 8 weeks, the escaping latency and swimming distance of the model I group obviously prolonged at the test days 3, 4, and 5, which confirmed that short-term Mn-exposure could reduce learning and memory ability. 1,15,16 On observation for 12 or 18 weeks, escaping latency and swimming distance of rats in the model groups increased, which suggested that subchronic Mn-exposure could cause spatial learning and memory ability impairment and supported the previous studies. 2,16,17 The learning and memory dysfunction are associated with dosage of Mn exposed. 1
Learning and memory is a very complicated and delicate process. The learning and memory impairment induced by Mn poisoning involves a series of changes in the nervous system, which are associated with multiple neurotransmitters changes. 18 ACh is the classical neurotransmitter in the CNS and plays an excitatory and inhibitory dual role, particularly in the learning and cognition. 19,20 Previous study found that Mn has inhibitory effects on acetylcholinesterase activity promoting increased levels of neuronal oxidative stress. 21 Additionally, CSF’s ChAT maintain a steady state level of Ach in the presence of physiological levels of fully active AChEs. 22 ChAT is the marking enzyme of central cholinergic neurons. However, vDB and hDB nuclei in the basal forebrain are rich of cholinergic neurons, and they can synthesize and secrete Ach. The present study suggested that numbers of vDB/hDB ChAT-positive neurons in the basal forebrain hDB in Mn-exposed groups were less than those of the normal control groups in the subacute and subchronic experiment supported by other studies, 23 which is related in parallel to the decline of learning and memory ability. Therefore, number of ChAT-positive neurons decreased may be one of the mechanisms of learning and memory ability impairment induced by Mn. 24
As it is well known, PAS-Na is a kind of anti-tuberculosis drugs, which can promote the exhaust of Mn and may also have neuroprotective effect. 25 In addition, PAS can reduce the brain inflammation and oxidative stress. 14,26,27 In the present study, escape latency and swimming distance in PAS-P and PAS-T groups were shortened in comparison with the Mn-exposed groups, which confirmed that PAS-Na has antagonistic effects on the spatial learning and memory impairment of rats induced by subchronic Mn exposure. 2,17 The mechanisms of PAS-Na improving the learning and memory may be related to trace elements, the expression of some genes in hippocampus, the expression of GFAP-positive cells, the expression of neurofilament protein, and the effects of Ach neurotransmitter in different brain regions, and so on. 1,2,16,17 This study found that PAS-Na has antagonistic effects on the decreasing number in vDB/hDB ChAT-positive neurons induced by Mn. However, the protective effects on the cholinergic neurons cell of PAS-Na preventive intervention were better than that of PAS-Na therapeutic intervention in the subchronic Mn-exposed rats, especially in hDB. This may be related to the mechanism of PAS-Na to promote excretion of Mn. PAS-Na can more easily chelate with Mn ion in blood or CSF under prophylactic administration, which is conducive to promoting the elimination of Mn. However, when treated with PAS-Na after subchronic Mn exposure, the Mn ion may have already entered the neurons and generated more stable compounds, which means that the capacity of PAS-Na in chelating with the Mn ion is limited or Mn has produced so serious damage in basal forebrain cholinergic neurons of rat that short-term repairs were unobvious. Additionally, it may be related to Mn exposure level, PAS-Na therapeutic level, and time and sample size, which remains to be elucidated.
Conclusion
In conclusion, results from the present study demonstrated that subacute or subchronic Mn exposure could decrease the number of cholinergic neurons in basal forebrain vDB/hDB ChAT-positive neurons of rats, which was related in parallel to the decline of learning and memory ability indicating the decreasing number of basal forebrain cholinergic neurons possibly relating to learning and memory ability impairment induced by Mn. PAS-Na may have intervention effect on the impairment of learning and memory induced by Mn via increasing the number of ChAT-positive neurons and activity of ChAT protein. Further studies to explicate the shielding effects of PAS-Na in Mn-induced learning and memory ability impairment is important and will provide additional evidence for the exploitation of broader remedial usage.
Footnotes
Acknowledgement
This study was supported by a grant from the National Natural Science Foundation of China (NSFC 30760210, 81072320).
Authors’ Note
The authors SJL, HYM, XFD, and XF contributed equally to this work.
