Abstract
Aim: In cyclosporin-A (CsA)-induced toxicity, oxidative stress has been implicated as a potential responsible mechanism. Therefore, we aimed to investigate the protective role of erdosteine against CsA-induced nephrotoxicity in terms of tissue oxidant/antioxidant parameters and light microscopy in rats. Materials and methods: Wistar albino rats were randomly separated into four groups. Group 1 rats treated with sodium chloride served as the control, group 2 rats were treated with CsA, group 3 with CsA plus erdosteine, and group 4 with erdosteine alone. Animals were killed and blood samples were analyzed for blood urea nitrogen (BUN), serum creatinine (Cr), uric acid (UA), total protein (TP), and albumin (ALB) levels. Kidney sections were analyzed for malondialdehyde (MDA) and nitric oxide (NO) levels and superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px) activities, as well as for histopathological changes. Results: In the CsA group, MDA, GSH-Px, BUN, and Cr levels were increased. The TP and ALB levels were decreased. These changes had been improved by erdosteine administration. Other biochemical parameters did not show any significant change. Conclusion: These results indicate that erdosteine produces a protective mechanism against CsA-induced nephrotoxicity and suggest a role of oxidative stress in pathogenesis.
Introduction
Cyclosporin A (CsA), a potent immunosuppressant, is widely used after organ transplantation, in the treatment of several autoimmune diseases and malignancy1–4 such as Behcet’s uveitis 5 and renal transplantation. 6 Nephrotoxicity is the most prevalent adverse effect induced by CsA 7 and is the major dose-limiting side effect of the drug.7–9 Although the underlying mechanism of the nephrotoxicity is as yet unknown, reactive oxygen species (ROS) have been implicated extensively in the toxic effect. 9 The administrations of various types of antioxidants attenuate CsA-induced acute and chronic nephrotoxic effects. 10
Erdosteine (N-[caboxy methyl thio acetyl]-homocysteine thiolactone), which is a thiol derivative, has been developed as a mucolytic agent. It is used in the treatment of patients with chronic obstructive lung diseases and mucopurulent disease of respiratory tract. 11 Pharmacological and experimental studies also demonstrated that erdosteine was able to wipe out oxygen free radicals.11–15 Erdosteine prevented toxicities in kidney such as induced by cisplatin 14 and doxorubicin. 16 It carries out free-radical-eliminating activity via its active metabolites. Therefore, erdosteine seems to be a promising drug for the prevention of free-radical-induced damage in many pathologic conditions.
In the present study, the contribution of oxidative stress to CsA-induced nephrotoxicity and the protective role of erdosteine were investigated in a rat model.
Materials and methods
Animals and experimental procedures
Adult male Wistar albino rats (200–220 g in weight) were used in this experiment. The animals were housed in quiet rooms with 12-hour light–dark cycles (7 a.m. to 7 p.m.), and the experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals (National Academy Press, Washington, DC, 1996). Rats were randomly assigned to four groups, each comprising seven rats: rats treated with erdosteine only (7 days with an oral dose of 10 mg/kg/day), CsA (7 days with an intraperitoneal dose of 20 mg/kg/day), erdosteine plus CsA (7 days with an oral dose of 10 mg/kg/day), and NaCl as the control group. In some studies, erdosteine was given to rats in various doses (10 mg/kg, 150–300 mg/kg).17–19 The erdosteine capsules were obtained from Ilsan-Iltas Pharmaceuticals, Istanbul, Turkey. It was prepared from sodium bicarbonate and administered orally via gavage. CsA ampoules were obtained from Novartis (East Hanover, NJ, USA) and administered intraperitoneally. On the 8th day of CsA therapy, the animals of all groups were anesthetized with ketamine hydrochloride (75 mg/kg) and xylazine (8 mg/kg), venous blood samples were taken, and serums were separated. Then, kidneys were rapidly excised and right kidneys were used for microscopic examination and left ones for biochemical analyses.
Biochemical analyses
Oxidative parameters
The kidney tissue samples were stored at −70°C until assayed for kidney tissue malondialdehyde (MDA) levels, catalase (CAT), superoxide dismutase (SOD), and glutathione peroxidase (GSH-Px) activities. Kidney tissues were homogenized (for 2 min at 5000 rpm) in four volumes of ice-cold Tris-HCl buffer (50 mM, pH 7.4) using a glass Teflon homogenizer (Ultra Turrax IKA T10 Basic, Germany). From the homogenate, the MDA and protein levels were measured. Then the homogenate was centrifuged at 5000g for 60 min to remove the debris. The supernatant fluid was collected, CAT and GSH-Px activities and protein concentration were measured. The supernatants used for the assay were mixed with an equal volume of ethanol/chloroform mixture (5/3, volume per volume [v/v]). After centrifugation at 5000g for 30 min, the clear upper layer (the ethanol phase) was collected and used for the analysis of SOD activity and protein assays. All preparation procedures were carried out at +4°C.
Determination of MDA levels
The thiobarbituric acid reactive substance (TBARS) levels were determined by a method based 20 on the reaction with TBA at 90–100°C. In the TBA test reaction, MDA or MDA-like substances (i.e. the by-product of lipid peroxidation process of the polyunsaturated fatty acids [PUFAs]) and TBA react together for the production of a pink-colored pigment having a maximum absorption at 532 nm. The reaction was performed at pH 2–3 at 90°C for 15 min. The sample was mixed with two volumes of 10% cold (weight per volume [w/v]) trichloroacetic acid to precipitate protein. The precipitate was pelleted by centrifugation and an aliquot of the supernatant was reacted with an equal volume of 0.67% (w/v) TBA in a boiling water bath for 10 min. After cooling, the absorbance was read at 532 nm (Ultra spec Plus, Pharmacia LKB Biochrom Ltd, England). Results were expressed as nmol/g wet tissue, by reference to a standard curve prepared from measurements made with a standard solution (1,1,3,3-tetramethoxypropane).
Determination of CAT activity
CAT (EC 1.11.1.6) activity was measured according to the method of Aebi. 21 The principle of the assay is based on the determination of the rate constant k (dimension: s−1, k) of hydrogen peroxide (H2O2) decomposition. By measuring the absorbance changes per minute, the rate constant of the enzyme was determined. Activities were expressed as k (rate constant) per gram protein.
Determination of GSH-Px activity
GSH-Px (EC 1.6.4.2) activity was measured using the method of Paglia and Valentine. 22 The enzymatic reaction was initiated by the addition of H2O2 to the reaction mixture containing GSH, NADPH, and glutathione reductase. The change in the absorbance at 340 nm was monitored by a spectrophotometer. Activity was expressed as U/g protein.
Determination of SOD activity
The principle of the total SOD (EC 1.15.1.1) activity method is based, briefly, on the inhibition of nitro blue tetrazolium (NBT) reduction by the generated xanthine/xanthine oxidase system. 23 Activity was assessed in the ethanol phase of the serum after the addition of 1.0 ml of ethanol/chloroform mixture (5/3, v/v) to the same volume of the serum and centrifuged. One unit of SOD was defined as the enzyme amount causing 50% inhibition in the NBT reduction rate. The SOD activity was expressed as U/g protein.
Determination of NO activity
Nitric oxide (NO) has very short half-life. The oxidation products of NO, nitrite (NO2 −), and subsequently nitrate (NO3 −) serve as an index of NO production. The method for measuring plasma nitrite and nitrate levels was based on the Griess reaction. 24 Samples were initially deproteinized with Somogyi reagent. Total nitrite (nitrite + nitrate) was measured by spectrophotometry at 545 nm after conversion of nitrate into nitrite by copperized cadmium granules. A standard curve was established from nitrite standards to analyze unknown sample concentrations. Results were expressed as micromoles per gram wet tissue (μmol/g wet tissue).
Renal impairment was assessed by serum creatinine (Cr), blood urea nitrogen (BUN), uric acid (UA), total protein (TP), and albumin (ALB) levels as well as by renal histology. The BUN and Cr levels were determined by an autoanalyzer (Syncron LX 20, Ireland) using commercial Beckman Coulter diagnostic kits.
Histological evaluation
For light microscopic examination, right kidneys of each rat were removed and tissue pieces were fixed in 10% neutral buffered formalin solution, embedded in paraffin, sectioned at 5 μm and then stained with hematoxylin and eosin (H&E). Sections were examined and photographed with Olympus CX-41 photomicroscope for histopathological changes. In order to obtain a histological score, histopathological changes were graded according to cortical involvement and the severity of the lesions modified from Karahan et al. 25
Statistical analysis
Data were analyzed using a commercially available statistics software package (SPSS® for Windows). Distributions of the groups were tested using the one-sample Kolmogrov-Smirnov test. 26 Measured parameters for all groups were normally distributed, and parametric statistical methods were used to analyze the data. Quantitative data are expressed as mean ± SEM. Comparisons between groups were performed using the Kruskal-Wallis analysis of variance for unpaired comparisons followed by the Mann-Whitney U test. 26 Significant level was fixed as 0.5 in statistical analyses performed with SPSS 11.0 for Windows software.
Results
Observation of the rats
During the experiment, decreased water and feed consumption, weight loss, decreased mobility, shedding fur, and color changes in the skin were observed in CsA group. These findings were found to be more serious than all other groups. Similar to erdosteine + CsA group, decreased water and feed consumption and decreased mobility were observed except shedding fur and color changes in the skin. There were no pathological changes in the control and erdosteine groups. On the contrary, these groups had significant weight gain. Weights of rats before and after the experiment are shown in Table 1 .
Weight changes in rats before and after the experiment

C: control group (G), E: erdosteine group, CsA-E: CsA plus erdosteine group, CsA: CsA group, N: normal, M: moderate, D: decreased.
Biochemical results
Oxidants and antioxidants
The SOD, CAT, and GSH-Px activities of the renal tissues with MDA and NO levels were presented in Table 2 . The activities of GSH-Px were increased significantly in the CsA groups in comparison with the control groups (p < 0.0001). No significant differences were noted between CAT activity and NO levels. In addition, MDA levels, which are considered as an important parameter for oxidative damage and a marker for lipid peroxidation of the tissue, were significantly higher in the CsA-administered group than in the control group (p < 0.0001). Rats exposed to CsA plus erdosteine administration had unchanged NO, SOD, and CAT enzyme levels. The rats had increased GSH-Px and decreased MDA levels compared to the CsA-administered group (p < 0.0001; Table 2).
MDA, NO, SOD, CAT, and GSH-Px values in the kidney of the three groups of rats (n = 7 for each group) a

CAT: catalase, CsA: cyclosporin A, GSH-Px: glutathione peroxidase, MDA: malondialdehyde, NO: nitric oxide, NS: non-significant, SOD: superoxide dismutase.
a The mean difference is significant at the 0.05 level. Values are expressed as mean ± SD.
Serum albumin, creatinine, total protein, and blood urea nitrogen values
There were significant increases in BUN and serum Cr levels in the CsA group compared to the control group (p < 0.05). Treatment of rats with erdosteine alone did not cause any significant change in BUN, TP, ALB, or serum Cr levels. The rats exposed to CsA plus erdosteine had increased ALB and decreased Cr levels compared to CsA-administered group (p < 0.05; Table 3 ).
Blood urea nitrogen (BUN), serum uric acid (UA), creatinine (Cr), albumin (ALB), and total protein (TP) levels
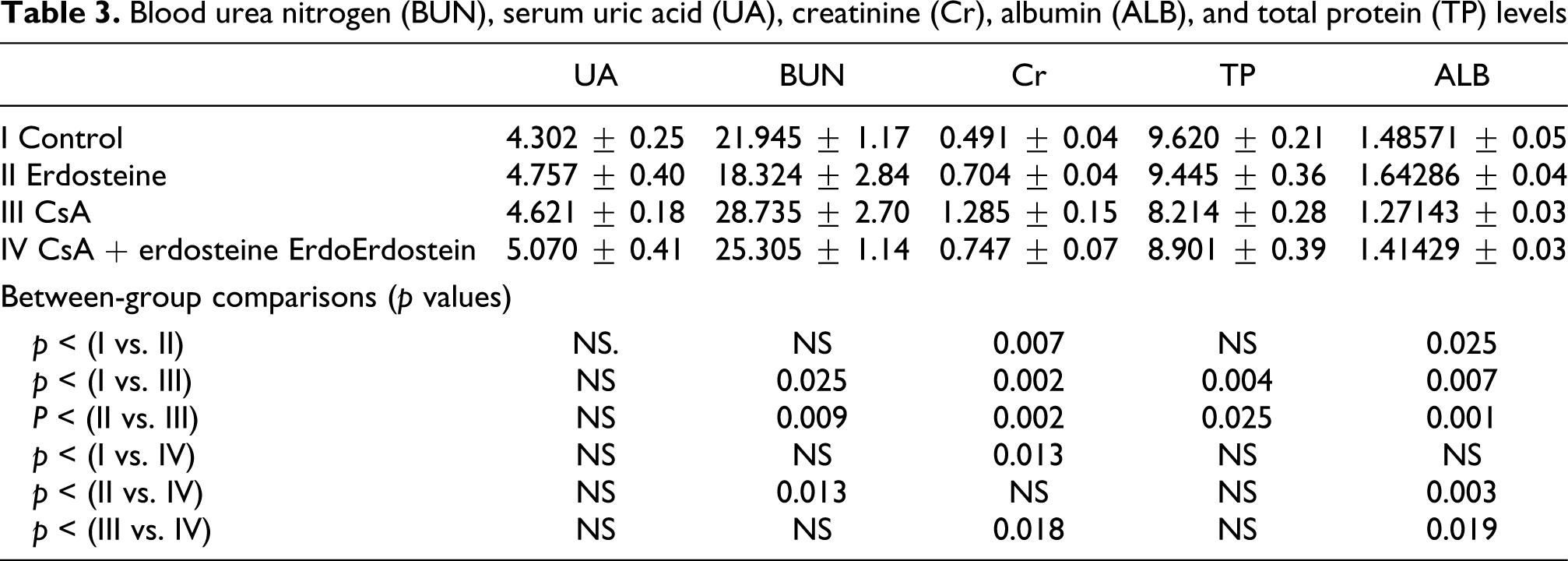
Histopathological results
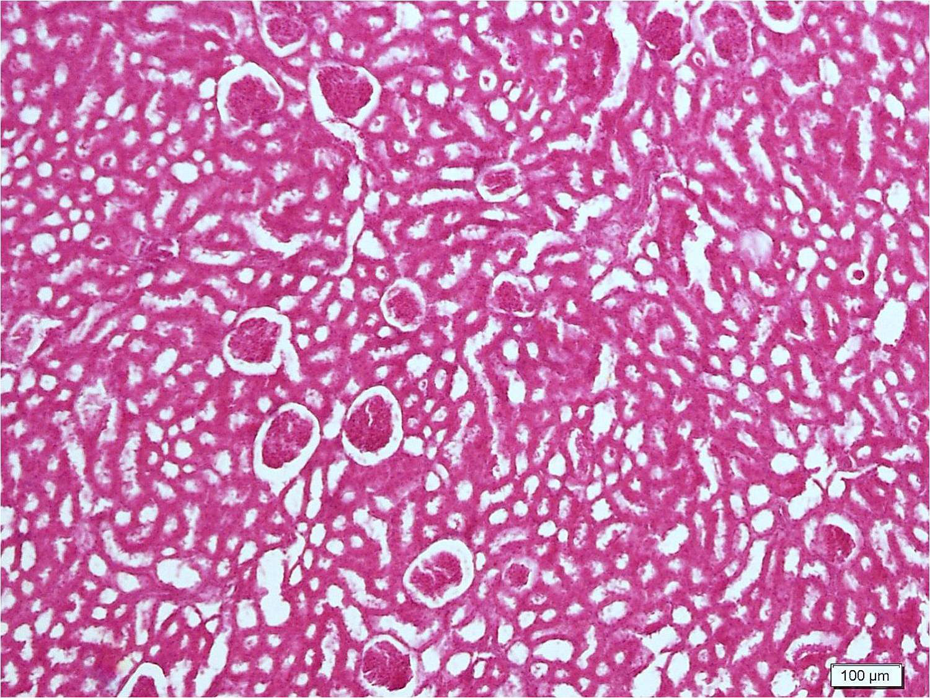
Histopathology scores are shown in Table 4 . Light microscopic examination of control sections revealed normal cortical morphology (Figure 1). Erdosteine caused no significant changes compared to the control group. CsA significantly damaged renal morphology compared to control and erdosteine groups. CsA-treated group showed extensive degeneration throughout the cortex (Figure 2). Tubular atrophy, hemorrhage, renal tubules with vacuolated cytoplasm, and brush border loss were observed (Figure 3). Degeneration was mostly evident in proximal tubules. In addition to atrophic glomerulus, the renal corpuscles contained shrunken glomeruli with widening of their Bowman’s spaces (Figure 4). Inflammatory cellular infiltrate, increase in the collagen fibers, and tissue necrosis were observed between the renal tubules (Figure 5). Erdosteine treatment significantly improved kidney histology in CsA group compared to CsA-alone group. In the group treated with CsA + erdosteine, the structure of renal tubules and corpuscles improved compared to CsA group (Figures 6 and 7). However, necrotic areas with degenerated tubules and glomerulus persisted in some areas (Figure 8). In addition, tissue damage was significantly evident in CSA + erdosteine group compared to control and erdosteine groups.
Histopathological changes with grade

Normal renal morphology.

Extensive degeneration throughout the cortex in the group treated with cyclosporin A (CsA). Note atrophic and degenerated glomerulus (*).

Tubular atrophy (*), hemorrhage, renal tubules with vacuolated cytoplasm (-->), and brush border loss in the group treated with cyclosporin A (CsA).

Shrunken glomeruli with widening of their Bowman’s spaces (*) in cyclosporin A (CsA) group. Note inflammatory cell infiltrate (-->) in tubules.

Extensive tissue necrosis (*) in the group treated with cyclosporin A (CsA).
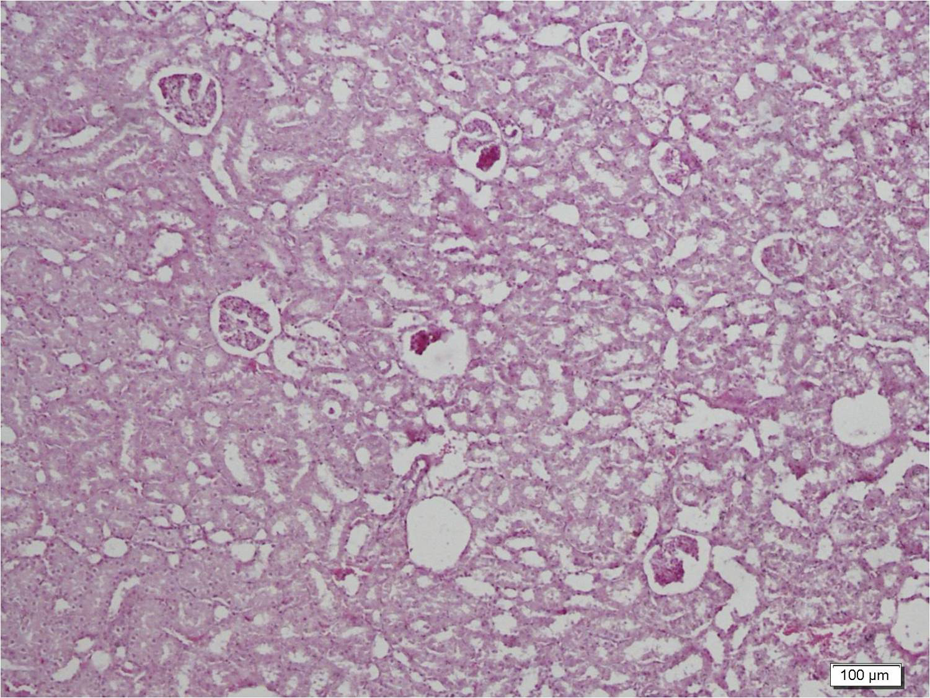
Improved general structure in the group treated with cyclosporin A (CsA) + erdosteine.

Well-preserved tubules with no vacuoles and brush border loss (-->) in the group treated with cyclosporin A (CsA) + erdosteine.
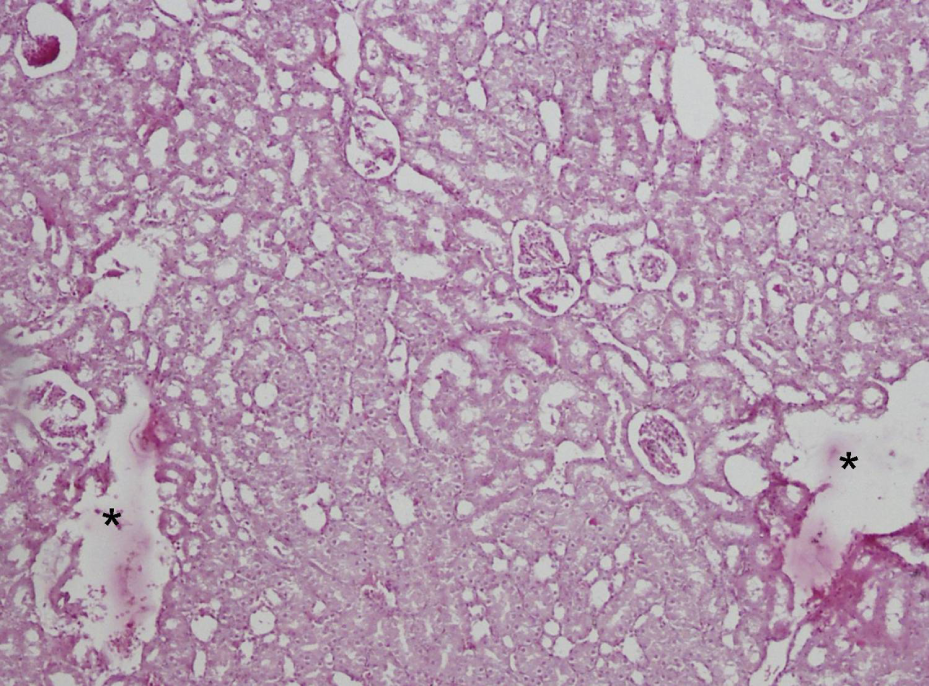
Necrotic areas (*) with degenerated tubular and glomerular structures in the group treated with cyclosporin A (CsA) + erdosteine.
Discussion
CsA, a potent immunomodulator, has been used in the treatment of various diseases. However, nephrotoxicity, the major side effect of CsA treatment, limits the use of this drug in high doses. The exact mechanism of CsA-induced nephrotoxicity remains obscure, but some studies suggest that oxidative stress is involved in this pathway 11 and CsA increases ROS. 27 On the other side, CsA had a great number of side effects on renal, hepatic, cardiac, alimentary tract, and nervous systems.28,29 ROS have been blamed extensively in the toxic effect. 9 According to our literature search, this is the first study reporting that erdosteine attenuates the detrimental effects of CsA in kidney tissues as shown by histologic and biochemical data.
The detrimental effect of CsA was shown morphologically in several studies particularly in kidney transplant recipients.30,31 Eşrefoglu et al. reported similar results in kidneys of CsA-treated rats, such as tubular atrophy, vacuolization, and tubulointerstitial fibrosis. 28 However, their results were not graded and evaluated statistically. Different from their study, we evaluated the glomerular structure in addition to the tubules and graded according to the extent of tissue injury. We found significant changes in CsA group compared to the control and CsA + erdosteine groups as also supported by Kim et al. 32 who reported vacuolization in all segments of the proximal tubules, tubular inclusion bodies, and peritubular capillary congestion.
Increased serum BUN and Cr levels, markers of renal functional impairment were previously reported in several studies.1,10 Duru et al. 1 reported significant increases in BUN and serum Cr levels in the CsA group as compared with the control. Similarly Tariq et al. showed that treatment of rats with CsA produced a significant increase in serum BUN and Cr levels. 10 We also found that plasma Cr and BUN levels were significantly higher after CsA administrations when compared to the control group. But Duru et al. 1 reported a higher increase in serum levels of BUN and Cr than ours. This difference could be due to the higher total dose of CsA administered to the rats. Different from similar studies, we also evaluated TP and ALB levels and found that both decreased significantly in CsA group.
Lipid peroxidation is initiated as a result of ROS-induced abstraction of hydrogen from PUFAs of cellular membrane, which results in the formation of relatively stable compounds such as MDA. The lipid peroxidation radical results from subsequent interaction with molecular oxygen and hydrogen abstraction from an adjacent lipid hydroperoxide, which are accompanied by cellular degeneration. Increased levels of lipid peroxides and conjugated dienes were shown previously in the renal tissue of CsA-treated rats. 12 Similarly, in our study the MDA levels of the CsA-administered group were significantly higher than those of the controls. This showed that CsA caused lipid peroxidation in renal tissue, thus leading to oxidative damage.
Degradation of H2O2 is catalyzed by the CAT enzyme. Gokce et al. 33 reported a significant decrease in the levels of CAT in renal tissue in a study where they investigated the protective effect of caffeic acid phenethyl ester on CsA-induced nephrotoxicity in rats. In our study, the CAT activities were also diminished in the CsA group, but it was not statistically significant. But the total dose of CsA in the study of Gokce et al. 33 was higher than that in our study.
Increasing evidence suggests that NO has an important role in the modulation of oxidative stresses and tissue damage. Peresleni et al. 34 demonstrated that oxidative stress on the epithelial cells cause an increase in inducible NO synthase, which results in the elevation of NO release, NO2 − production, and decreased viability. In the present study, there was no significant difference between NO levels in all groups.
Endogenous antioxidative defense system prevents the production of free radicals. The antioxidative enzymes SOD and GSH-Px build the major defense against ROS-induced oxidative damage. SOD constitutes the first line of defense against the harmful effects of free oxygen radicals in the cells. SOD activity after CsA administration was reported to decline in the renal tissue in some other studies 35 and treatment with the SOD mimetic tempol prevented CsA-induced renal dysfunction. 36 The present study revealed no significant difference between SOD and GSH-Px levels in all groups.
Erdosteine, a thiol derivative, has been developed as a mucolytic agent. It is used in the therapy of patients with chronic obstructive lung diseases and mucopurulent disease of respiratory tract. 11 Pharmacological and experimental studies demonstrated that erdosteine could wipe out free oxygen radicals.11,12 Erdosteine has been efficiently used as a preventive agent against various toxic agents in both animals and humans.12–15 Erdosteine prevented doxorubicin- 16 and cisplatin-induced 14 toxicities in kidney. It performs free radical-scavenging activity via its active metabolites. Therefore, erdosteine appears to be a promising drug to prevent free radical-induced damage in many pathologic conditions.12,15
The protective effects of erdosteine were shown in different ways previously in the literature. Yıldırım et al. 15 showed that erdosteine attenuated cisplatin-induced renal injury histologically. In another study by Selcoki et al., 37 protective effect of erdosteine on cyclosporin-induced cardiotoxicity was shown morphologically. With some differences, our study also revealed that erdosteine significantly regressed CsA-induced nephrotoxicity to some extent. However, tissue degeneration remained evident in CsA + erdosteine group compared to control and erdosteine groups. From the histological point of view, this study is the first to evaluate the protective effect of erdosteine on CsA-induced nephrotoxicity with a detailed grading system including both glomerulus and tubules. In the current study, erdosteine significantly reversed the CsA-induced depletion of MDA in renal tissue consistent with Gokce et al. 33 Furthermore, CAT levels were also reversed via the administration of erdosteine with CsA, but it was not statistically significant.
Erdosteine produced a significant decrease in BUN and serum Cr levels in CsA group in the current study. There are similar studies reporting the protective role of erdosteine in nephrotoxicity induced by various toxic agents apart from CsA.13–15,28 In addition to serum BUN and Cr levels, we also investigated the role of erdosteine on ALB and TP levels and found that erdosteine administration improved TP levels significantly and ALB levels insignificantly in CsA + erdosteine group compared to CsA-alone group.
In conclusion, erdosteine prevented CsA-induced nephrotoxicity as shown by our biochemical and histological findings, suggesting that CsA-induced nephrotoxicity may be secondary to free oxygen radicals, and erdosteine’s protective role may arise from its antioxidative and radical-scavenging effects. Thus, the concomitant use of erdosteine-like antioxidants may help prevent CsA-induced nephrotoxicity.
Footnotes
Acknowledgements
Before the experiment, ethical approval was obtained from Mustafa Kemal University Animal Ethics Committee. This study was supported by Mustafa Kemal University scientific Research Project Board with project number 1102M0108.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
