Abstract
There is considerable evidence indicating an increase in neurodegenerative disorders in industrialized countries. The clinical symptoms and the possible mutagenic effects produced by acute poisoning and by chronic exposure to metals are of major interest. This study is a review of the data found concerning the genotoxic potential of three metals: aluminum (Al), iron (Fe) and manganese (Mn), with emphasis on their action on human cells.
Introduction
Metals are among the oldest toxic agents known by humans. 1 In an industrialized world, there are thousands of types of metals in use, and humans are exposed to them at work, or as a result of contamination of food, water and environment. One feature of the normal human diet is the simultaneous presence of both essential and toxic metals. 2 There is also abundant evidence indicating an increase of neurodegenerative disorders in industrialized countries.3–5 Thus, the clinical symptoms and the possible mutagenic effects produced by acute poisoning and chronic exposure to metals are of considerable interest.3,6
In this review, we compare the potential genotoxic effects of exposure to Al, Fe and Mn from different sources, both in in vitro and in vivo assays. Although there are numerous studies in the literature on the toxicological effects of exposure to these metals, investigations on direct genotoxicity/mutagenicity are scarce. Much remains to be established regarding the genotoxic effects of metals.
Literature review
General information
Aluminum
Aluminum (Al) is the most widely distributed metal in the environment and is extensively used in daily life that provides easy exposure to human beings. The exposure to this toxic metal occurs through air, food, water and it is also present in medical, cosmetic and environmental products. Aluminum chloride (AlCl3) is an important coagulant used in water treatment and purification. This wide distribution of the element clearly facilitates the potential for human exposure and for causing harm.7–12
No biological function of the element has been identified, whereas some aspects of its toxicity have been described.7,13–16 Daily consumed dose of Al by food and beverages is 2.5 to 13 mg, where drinking water can contribute to 0.2 to 0.4 mg of Al daily. Drugs can contribute with increased levels of Al. Antiacid drugs (2 tablets) can contribute up to 500 mg of Al. 17
It has been suggested that there might be a relationship between high levels of Al and increased risk of a number of pathogenic disorders, such as microcytic anemia, osteomalacia and, possibly, neurodegenerative disorders including dialysis encephalopathy, Alzheimer’s disease and Parkinson’s disease.6,7,12,14,18–21
Iron
Populations are exposed to iron mainly through foods and beverages. 22 Iron (Fe) is an essential trace element used by almost all living organisms, being often incorporated into the heme complex, which mediate redox reactions, and of oxygen transport proteins, such as hemoglobin in red cells and myoglobin in muscle cells. It can also be found in the bone marrow, liver and spleen and is required for the immune system response and for the production of energy.23,24 Also, daily recommended doses of Fe varies among age. For children up to 3 months, 1.7 mg/kg/daily are recommended, whereas for adults this is 10 times more (18 mg/kg/daily). 25
Although the intake of iron is regulated, when it is ingested in large amounts it can cause excessive blood iron levels, which in turn can cause damage to the cells of the gastrointestinal tract, preventing them from regulating its absorption. 26 The corrosive nature of iron seems to further increase its absorption, leading to poisoning. In human beings, several alterations have been related to high iron intake, especially in the pulmonary tract, leading to cancer caused by inhalation of iron oxide (FeO), skin rashes by inhalation of iron salts, heart, kidney, liver and gastrointestinal tract alterations and also diabetes mainly because of the ingestion of high concentrations of iron sulfate (FeSO4; 0.5–2.5 g), found currently in drugs. 27
Iron is moreover toxic to neural tissue, leading to neurodegenerative disorders.28,29 Advanced neuroimaging techniques and pathological studies have demonstrated increased brain iron with aging, and increased iron deposition has also been observed in patients with a constellation of neurological diseases, including Alzheimer's disease, Parkinson's disease and stroke. 30
Manganese
Manganese (Mn) is an essential element for humans, animals and plants and is required for growth, development, and maintenance of health. In plants, Mn participates in the respiratory process, where its deficiency can affect agriculture worldwide. 31 In animals, Mn acts in physiological processes such as the regulation of reproduction, formation of connective tissues and bone marrow, lipid and carbohydrate metabolism and in the maintenance of the brain. Mn is present in most tissues of all living organisms and is present naturally in rocks, soil, water and food.32–34 Daily recommended doses of Mn for children is 0.3 mg/kg/daily, being 3 times more for adults (10 mg/kg/daily). 35
This metal is also an important cofactor for numerous enzymes involved in the biosynthesis of DNA and neurotransmitters and in transduction signals.33,36 Despite the fact that the absorption of Mn is of only 3%–5%, food is the primary source of the metal ingested by human beings and animals. 31,36 Other sources of Mn exposure are related to working conditions, where Mn particles can be inhaled or absorbed.31,35
Mn deficiency during the initial phases of development can induce skeletal muscle abnormalities and irreversible ataxia, in addition to fertility problems. 32 Poisoning by high levels of Mn can lead to some alterations in the organism, where the lungs and the central nervous system (CNS) are the main target organs. 31
Moreover, exposure to Mn doses 5 to 6 times higher than the required daily amount together with trace element Mn was reported in parenteral nutrition and can cause neurotoxicity. The regulatory mechanisms of Mn homeostasis are bypassed via the parenteral route, consequently elimination via the hepatobiliary system is impaired resulting in tissue Mn accumulation. 37
Exposures to high levels of manganese by ingestion or inhalation can damage the central nervous system. 38 High doses (1800–2250 mg/kg/day as manganese (II) sulfate) in mice induce hyperplasia, erosion and inflammation in the stomach. 39 Chronic exposure to high levels of Mn can also induce a syndrome known as manganism, characterized by extrapyramidal dysfunction (bradykinesia, rigidity and dystonia) and neuropsychiatric symptoms that resemble idiopathic Parkinson’s disease.31,40,41
Metal genotoxicity
Genotoxicity of aluminum
There are only few studies in the literature about the genotoxic activities of Al. Its mutagenic potential has been studied by micronucleus assay, sister chromatid exchange, Ames and chromosomal aberration analysis.
Moreno et al. showed the induction in vitro of chromosomal aberrations, mostly numeric (anaphasic), in the Balb c 3T3 cell line exposed to atmospheric dust (20–80 mg/mL) from the city of Mexicali, Mexico, a mixture of particles of potassium aluminum silicates (98%) and sodium dioxide (2%).
42
Dovgaliuk et al. studied the cytogenetic effects of toxic metal salts including aluminum (Al[NO3]3; 0.01 mM–1 mM) using meristematic cells from
Yi et al. investigated the genotoxic potential of AlCl3 using
On the other hand, other studies have demonstrated the mutagenic potential of Al in human cells. For example, genotoxicity of the dust derived from an electrolytic Al plant was evaluated using the Ames assay, unscheduled DNA synthesis test, sister chromatid exchange and micronuclei frequencies in human lymphocytes. The results of these four experiments indicated a high genotoxicity of the dust organic extract. 48 There was also observed chromosome breaks in V79-4 Chinese hamster cells irradiated with low-energy aluminum ions. 49
The mutagenic activity of waste material originated from an Al products factory was determined by the
Our research group recently published a study on the genotoxic, clastogenic and cytotoxic effects of AlCl3 (5, 10, 15 and 25 μM) in different phases of the cell cycle using in vitro temporary cultures of human lymphocytes. We analyzed the mitotic index (MI), chromosomal aberrations (CAs) and DNA damage index as detected by the comet assay. That study indicated that AlCl3 produces DNA damage and is cytotoxic during all phases of the cell cycle, and the treatment of the cells at phase G1 resulted in polyploidy and endoreduplication, consistent with AlCl3 interacting with the mitotic spindle apparatus. 52 Also was reported that iron- and aluminum-sulfate together, at nanomolar concentrations, trigger the production of reactive oxygen species (ROS) in cultures of human brain cells, up-regulating pro-inflammatory and pro-apoptotic genes that redirect cellular fate toward cytoplasmic dysfunction, nuclear DNA fragmentation and cell death. 53
Taking together, our results and other studies reported in the literature indicate that AlCl3 is genotoxic and should be used with caution.
Genotoxicity of Fe
Several studies have been conducted to demonstrate the potential induction of DNA aberrations by Fe and also by drugs and compounds containing this metal. However, the results are inconclusive, and the mutagenic effect of Fe has yet to be elucidated.
Free iron catalyzes the conversion of superoxide and hydrogen peroxide into hydroxyl radicals, which promote oxidative stress by the Fenton reaction. 54 In this way, organic Fe may increase the genotoxic effects of other compounds when they are combined. For example, the mutagenic activity of doxorubicin is significantly increased by this metal, as evaluated by the Ames test. 55 In addition, it was demonstrated in Jurkat cells that simultaneous treatment with desferrioxamine (Fe chelator) and hydrogen peroxide inhibited significantly the DNA damage induced by hydrogen peroxide, indicating that intracellular Fe, which is a redox-active metal, plays a role in the induction of DNA breaks induced by hydrogen peroxide. 56
High levels of chromosome and chromatid aberrations were found in human lymphocytes and TK6 lymphoblast cells exposed to high-energy iron ions (56Fe).57–59 Significant DNA damage was detected, microgel electrophoresis, in differentiated human colon tumor cells (HT29 clone 19A) incubated with ferric-nitrilotriacetate (Fe-NTA; 250–1000 μM).
60
Mutagenic activity was also found in elemental and salt forms of Fe, evaluated by mutagenicity tests in
Its also reported that little or no DNA damage (detected by the comet assay) occurred after treatment of human lymphocytes with the iron compounds ferric chloride (FeCl3) and ferrous chloride (FeCl2).65,66 However, at high concentrations of ferrous sulfate, significant DNA damage was observed, probably as a consequence of chemical contamination of the metal salt. Low concentrations of either Fe2+ or Fe3+ (1.25; 2.5 and 5 μg/mL) were not mutagenic in Chinese hamster ovary cells (CHO-9) treated in vitro, and the mitotic indices were also unaffected when compared to negative control cultures. 67
George et al. related that high-energy iron ions (LET = 151 keV/microm) induces chromosomal aberrations (measured using the fluorescence whole-chromosome painting technique) in normal and repair-deficient human fibroblasts cell lines. 68
Mutagenic potential of metallic agents used in dietary supplementation, including iron sulfate, was investigated by means of the comet assay. The authors reported a genotoxic effect of this metal in mouse blood cells after 24 h of treatment, at all concentrations used. 69 Genotoxic effects of Fe were also reported by Garry et al. in rats treated with iron oxide (Fe2O5; .75 mg) for 24 h; they observed that this metal only showed mutagenic potential when the animals were simultaneously treated with benzopyrene. 70
Furthermore, Hasan et al. reported that ferritin, a ubiquitously distributed iron storage protein, interacts with microtubules in vitro.
71
In a study conducted by Maenosono et al. the bacterial reverse mutation assay using
In our experiments, we used iron sulfate alone to determine at which concentration this metal begins to exert its genotoxic effects. An in vitro study aiming to investigate the genotoxic, clastogenic and cytotoxic effects of FeSO4 (4.5, 9.0 and 18 μM) were performed in different phases of the cell cycle, using short-term cultures of human lymphocytes. The bioactivity parameters tested were the mitotic index, chromosomal aberrations and DNA damage index as detected by the comet assay. Our results showed that, despite the doses, Fe induces alterations and inhibition of DNA synthesis (in a dose-dependent manner). 74
Furthermore, ROS interacts with a variety of molecules, including in saturated fatty acids, proteins and DNA leading to subsequent cell death/apoptosis, especially in the CNS tissue, where the antioxidant defenses are rare.75–77 Taking all together, all those mechanisms may explain all those effects and the concomitance occurrence of mutagenicity and cytotoxicity.
Genotoxicity of Mn
The association of Mn with the risk of developing neurodegenerative processes can be related to DNA damage. Relatively high doses of Mn can disrupt DNA integrity and DNA replication.78–80 Also, neurotoxic effect of Mn can be due to its interaction with detoxification enzymes that protects the cells, and/or its interaction with the redox system. In this way, Mn2+ (essential to the brain) can be oxidized to Mn3+, a toxic compound that enhances the oxidation of dopamine leading to the generation of several neurotoxic products and thus genotoxicity.81,82
There are few studies in the literature on the genotoxic action of Mn. Its mutagenic potential has been studied by in vitro tests in bacteria and by in vivo/in vitro tests in insect and mammalian cells, showing that some chemical forms of this metal have mutagenic potential. Gerber et al. demonstrated that high doses (0.05 M) of various Mn compounds can affect DNA replication and repair in bacteria.
31
As for mammalian cells, high doses of Mn (compared to the Mn doses recommended for daily consumption) can affect fertilization and are toxic to the embryo and fetus, demonstrating the teratogenic potential of this metal. Manganese chloride (MnCl2) was also subjected to the wing spot test of
Brega et al. demonstrated that farm workers exposed to pesticides containing Mn, even at a low levels, revealed an increased mutagenic potential of those pesticides, as evidenced by an increased number of CAs. It is possible that chronic exposure to low doses of Mn induces CAs over the years, since they are not acutely cytotoxic. 84
It is also possible that, at low doses (around 0.19−1.39 mg/m3 for 1−45 years), Mn has genotoxic effects only with long-term exposure, and this may be the reason why Timchenko et al. did not find CAs in the nasal mucosa of mammals exposed to Mn dioxide aerosol (40–12,000 Hz, 80–100 dB). 85 In contrast, Dutta et al. related manganese dioxide as an established genotoxicant and clastogen which could cause induction of DNA strand break, chromosomal aberration and micronucleus in human peripheral lymphocytes. 86
Manganese sulfate (MnSO4) did not show mutagenic potential in different strains of
De Meo et al. evaluated the genotoxicity of potassium permanganate (KMnO4), MnSO4 and MnCl2 using the Ames test with the tester strains TA97, TA98, TA100 and TA102, with and without metabolic activation. The presence of direct-acting mutagens was detected in all the Mn samples with the tester strain TA102 without metabolic activation. Only MnCl2 induced DNA damage in human lymphocytes with a dose-response relationship, as determined by the comet assay. The mutagenic potential was 2.4 revertant/nmol. 80 According to WHO data, other chemical forms of Mn have mutagenic potential, both in vitro and in vivo. More studies are necessary in order to elucidate the probable mutagenicity of Mn and its chemical forms and their effects on human health. 35
Our research group also conducted a study in vitro on the genotoxic, clastogenic and cytotoxic effects of MnCl2-4H2O (15, 20 and 25 μM; one of the most common forms of Mn) in different phases of the cell cycle, using short-term cultures of human lymphocytes. These effects were determined by the mitotic index (MI), chromosomal aberrations (CAs) and DNA damage index as detected by the comet assay. MnCl2-4H2O shows strong cytotoxicity in all phases of the cell cycle. The genotoxicity observed at phase G2 and in the comet assay may be related to the lack of time for the cellular repair system to act. The absence of CAs in the other phases of the cell cycle suggests that Mn-mediated damage may be repaired in vitro. 87
Conclusion
When we compare the mutagenic/genotoxic effects of the three metals under study based on the data previously published by our research team, we can see that Al is the one that induces the greatest amount of chromosomal aberrations and DNA damage, as observed by the comet test, suggesting that this metal has a direct interaction with the DNA. This effect was smaller, in decreasing order, for Fe and for Mn, suggesting that these metals interact with the biochemical replication or repair system machinery to induce the chromosomal alterations observed (Table 1; Figure 1). This could be the reason why the cells have to be in proliferation for Fe and Mn to be able to exert their effects on the DNA molecule. However, in a preliminary screening, we can conclude that all three metals studied have genotoxic potential.52,74,87

Numerical and structural chromosome aberrations and cytotoxicity of aluminum (Al), iron (Fe) and manganese (Mn)a
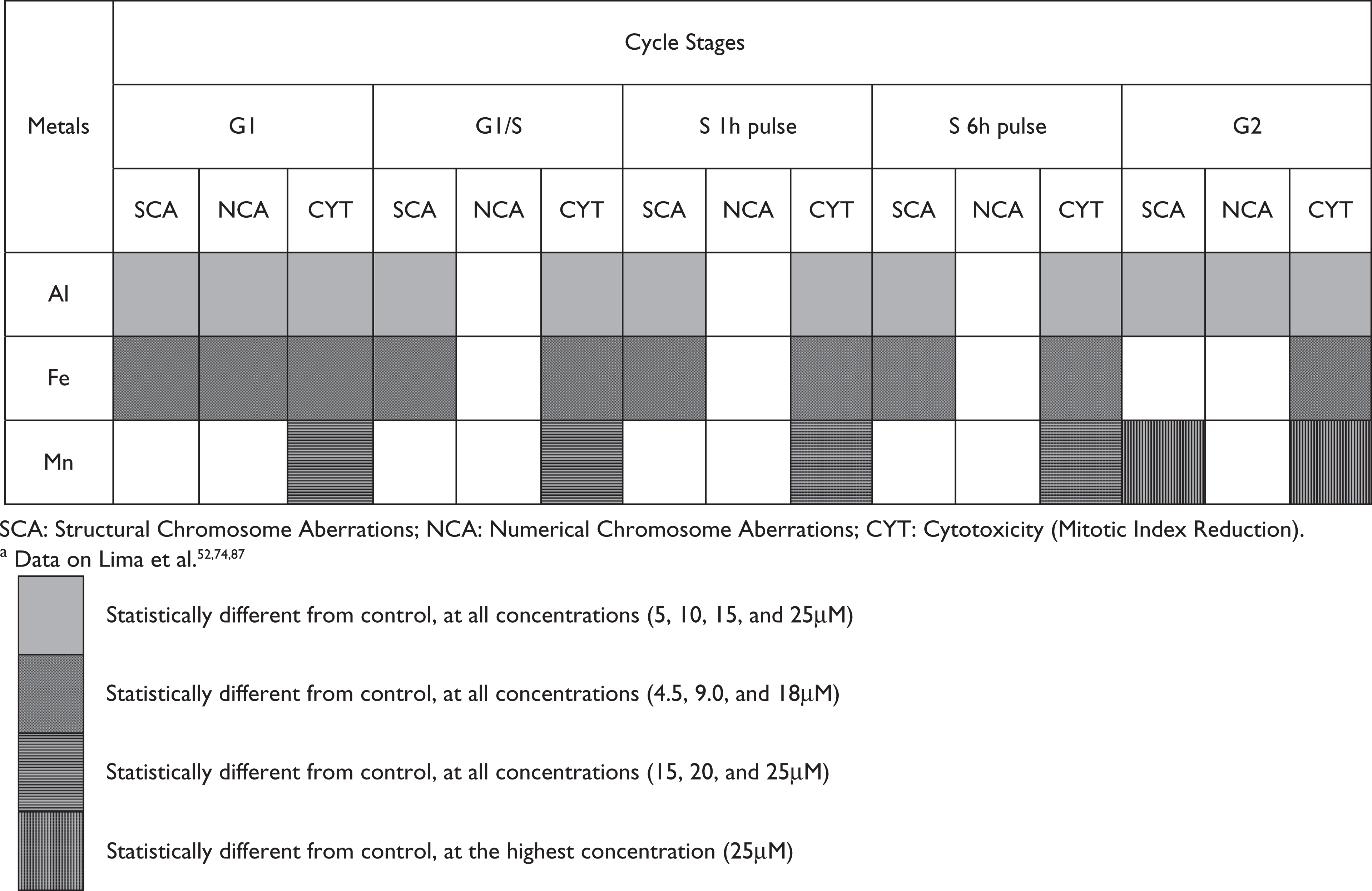
The objective of our studies was to evaluate the genotoxicity of the three metals in similar concentrations of those tested in studies about the toxicity of the metals in human neural cells. Thus, the concentrations of Al, Fe and Mn used in our studies are based on the literature references, adapted to lymphocyte cell cultures. The final concentrations used were 5, 10, 15 and 25 μM for AlCl3; 4.5, 9 and 18 μM for FeSO4 and 15, 20 and 25 μM for MnCl2.52,74,87
An important observation was that Mn was the only metal assumed to have exerted an induction effect on the repair system, probably because it is less toxic than Al and Fe. Al also interacts with the mitotic apparatus preventing its polymerization, probably by direct interaction with tubulin, whereas Fe, based on the results presented, indirectly prevents the building-up of the mitotic apparatus, interfering with the tubulin synthesis. Mn did not present any evidence, with the techniques used, of blocking the formation of microtubuli. This observation is pertinent since the morphology and physiology of the nervous cells depend directly on the formation of microtubular structures, which are responsible for the construction of axons and dendrites. Thus, in our evaluation, Al appears to be the most toxic metal for nervous cells.
Fe seems to have an indirect effect, via tubulin synthesis, and Mn does not seem to have any influence on the microtubular structures. We can however see that all metals analyzed have a cytotoxic action, where Al is the one with the strongest effect, followed by Fe and Mn.
In conclusion, based both on our results and on a review of the data from the literature, Al in the concentration range of 5 to 25 μM in lymphocytes, 1 mM in plant cultured cells and 34 mg/kg mice body weight, is the metal with the greatest toxicity to the DNA molecule and the mitotic apparatus, followed, in decreasing order, by Fe whereas genotoxicity can be observed in the concentration range of 4.5 μM in lymphocytes and 250 to 1000 μM in colon cancer cells and Mn whereas genotoxic activity can be seem around 25 μM in cultured lymphocytes and 0.05 M in bacteria.
Footnotes
The authors declare that there are no conflicts of interest.
The studies conducted by our research group were supported by Financiadora de Estudos e Projetos (FINEP CT-INFRA/FADESP) Grant No. 0927-03; Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
