Abstract
Ferric nitrilotriacetate (Fe-NTA) induces tissue necrosis as a result of lipid peroxidation (LPO) and oxidative damage that leads to high incidence of renal carcinomas. The present study was undertaken to evaluate the effect of diallyl sulphide (DAS) against Fe-NTA-induced nephrotoxicity. A total of 30 healthy male rats were randomly divided into 5 groups of 6 rats each: (1) control, (2) DAS (200 mg kg−1), (3) Fe-NTA (9 g Fe kg−1), (4) DAS (100 mg kg−1) + Fe-NTA (9 mg Fe kg−1) and (5) DAS (200 mg kg−1) + Fe-NTA (9 mg Fe kg−1). Fe-NTA + DAS-treated groups were given DAS for a period of 1 week before Fe-NTA administration. The intraperitoneal administration of Fe-NTA enhanced blood urea nitrogen and creatinine levels with reduction in levels of antioxidant enzymes. However, significant restoration of depleted renal glutathione and its dependent enzymes (glutathione reductase and glutathione-S-transferase) was observed in DAS pretreated groups. DAS also attenuated Fe-NTA-induced increase in LPO, hydrogen peroxide generation and protein carbonyl formation (p < 0.05). The results indicate that DAS may be beneficial in ameliorating the Fe-NTA-induced renal oxidative damage in rats.
Introduction
Experimental evidences suggest the role of reactive oxygen species (ROS) in ferric nitrilotriacetate (Fe-NTA)-induced renal toxicity in rats, which are thought to play a role in mutagenesis, carcinogenesis and ageing. Fe-NTA is also known to induce lipid peroxidation (LPO) leading to acute tubular necrosis 1,2 and apoptosis in mouse renal proximal tubules. 3 Nitrilotriacetic acid (NTA) is used as polyphosphate substitute in detergents in various countries and forms water-soluble chelate complexes with metal cations at neutral pH. 4 The major target organ for Fe-NTA damage is the kidney, and it results in acute nephrotoxicity and a high incidence of renal cellular carcinoma in Wistar rats and mice. 5
Fruits, vitamins, spices and other several herbs have been shown to be rich sources of cancer chemopreventive agents with diversified pharmacological properties. 6 –9 Many such agents possess antioxidant and free-radical scavenging properties. 10 –15 Phenolic antioxidants when added to human and animal food lower toxicity either by interception of harmful free radicals or activating the detoxifying enzymes of the body or by inhibiting the formation of ultimately carcinogenic metabolites and their binding to DNA and modifying the immune response of the organism.
However, the action of each compound is influenced by their own chemical structure, selected dose, tissue upon which they are exposed to and time of administration in relation to the time of the carcinogen exposure. Garlic is a popular spice added to several edible preparations and is a remedy for a variety of ailments. Garlic has been thought to metabolize into diallyl disulphide, diallyl sulphide (DAS), and allyl methyl sulphide and show protection through a number of mechanisms such as increasing gluathione levels, increasing the activities of enzymes such as glutathione-S-transferase, catalase, inhibition of cytochrome P4502E1, DNA repair mechanisms, prevention of chromosomal damage and so on. 16
Since DAS is a major component of garlic, which is known to possess antioxidant properties, 17,18 the present study was designed to investigate whether the prophylactic treatment of DAS could modulate renal oxidative stress and toxicity in rats receiving Fe-NTA.
Materials and methods
Preparation of Fe-NTA
The Fe-NTA solution was prepared by the method described by Awai et al. 19 The concentration of ferric (Fe) was 1 mg Fe mL−1, the molar ratio of Fe to NTA 1:3, and the pH was 7.4.
Animals and treatments
Male albino rats of Wistar strain (4–6 weeks old) weighing 125–150 g were used in this study and were housed in an air-conditioned room and had free access to pellet diet and water ad libitum. All animal studies were approved by the Animal Care and Use Committee.
Study design
For various sets of biochemical studies, different groups of animals were used. For studying the effect of prophylactic treatment of DAS on Fe-NTA-mediated generation, 30 rats were taken and were divided into 5 groups of 6 rats each. Group I received saline (0.85% sodium chloride (NaCl)) injected intraperitoneally (ip) at 10 mg kg−1 body weight (b.w.) and served as negative control. Group II received an oral treatment of higher dose of DAS (200 mg kg−1 b.w. mL−1 corn oil) and served as negative control. Groups IV and V received DAS at a dose level of 100 mg and 200 mg kg−1 b.w. mL−1 corn oil, respectively, for a period of 1 week administered through the gavage. Group III received only corn oil, 1.0 mL kg−1 b.w. (vehicle of DAS), for a period of 1 week through the gavage. Twenty-four hours after the last treatment of DAS or corn oil, the animals of groups III, IV and V were injected ip with Fe-NTA (9 mg Fe kg−1 b.w. 10 mL). All the animals were killed 12 h after the treatment of Fe-NTA or saline by cervical dislocation. Just before the killing, blood samples of these animals were collected in test tubes from retro-orbital sinus for serum analysis to estimate blood urea nitrogen (BUN) and creatinine (CRN) levels. Kidneys were collected and processed for the estimation of antioxidants including glutathione (GSH), glutathione reductase, and glutathione-S-transferase.
Estimation of CRN
Protein-free filtrate was prepared. To 1.0mL of plasma/serum, 1.0 mL of sodium tungstate (5% w/v), 1.0 mL of sulphuric acid (0.6 N) and 1.0 mL of distilled water were added, mixed thoroughly and centrifuged at 800g for 5 min. The supernatant was added to a mixture containing 1.0 mL of picric acid (1.05% w/v) and 1.0 mL of sodium hydroxide (NaOH) (0.75 N). The absorbance was recorded exactly after 20 min at 520 nm using a spectrophotometer against a reagent blank. 20
Estimation of BUN
Protein-free filtrate was prepared. To 0.5 mL of protein-free filtrate, 3.5 mL of distilled water, 0.8 mL diacetylmonoxime (2%) and 3.2 mL of sulphuric acid–phosphoric acid reagent (reagent was prepared by mixing 150 mL of 85% w/v phosphoric acid with 140 mL of water and 50 mL of concentrated sulphuric acid) were added. The reaction mixture was placed in a boiling water bath for 30 min and then cooled. The absorbance was recorded at 480 nm using a spectrophotometer against a reagent blank. 21
Preparation of PMS and microsomes
Kidneys were quickly removed, perfused immediately with ice-cold saline (0.85% w/v NaCl) and homogenized in chilled phosphate buffer (0.1 M, pH 7.4) that contained potassium chloride (KCl; 1.17% w/v) using a homogenizer. The homogenate was filtered through a muslin cloth and was centrifuged at 800g for 5 min at 4°C to separate the nuclear debris. The aliquot obtained was centrifuged at 10,500g for 20 min at 4°C to obtain postmitochondrial supernatant (PMS), which was used as a source of enzymes. A portion of the PMS was centrifuged and the pellet was considered to be the microsomal fraction and was suspended in phosphate buffer (0.1 M, pH 7.4) containing KCl (1.17% w/v).
Lipid peroxidation
The reaction mixture contained 0.58 mL phosphate buffer (0.1 M, pH 7.4), 0.2 mL of microsome (10% w/v), 0.2 mL ascorbic acid (100 mM) and 0.02 mL of ferric chloride (100 mM) and was incubated at 37°C in a shaking water bath for 1 h. Reaction was stopped by the addition of 1.0 mL trichloroacetic acid (TCA; 10% w/v). Following the addition of 1.0 mL thiobarbituric acid (0.67% w/v), all tubes were placed in a boiling water bath for a period of 20 min and centrifuged at 2500g for 10 min. The amount of malondialdehyde (MDA) formed in each of the sample was assessed by measuring the optical density of the supernatant at 535 nm against a reagent blank. The results were expressed as nanomolar of MDA formed per hour per gram of tissue at 37°C using a molar extinction coefficient of 1.563105 M cm−1. 22
Estimation of GSH
An aliquot of 1.0 mL of PMS (10% w/v) was precipitated with 1.0 mL of sulphosalicylic acid (4% w/v). The samples were kept at 4°C for at least 1 h and then subjected to centrifugation at 1200g for 15 min at 4°C. The assay mixture contained 0.1 mL filtered aliquot, 2.7 mL phosphate buffer (0.1 M, pH 7.4) and 0.2 mL 5,5′-dithiobis-(2-nitrobenzoic acid) (40 mg/10 mL of phosphate buffer 0.1 M, pH 7.4) in a total volume of 3.0 mL. The yellow colour developed was read immediately at 412 nm. The amount of GSH estimated was expressed in millimoles per gram of tissue. 23
Assay of antioxidant enzymes
Glutathione reductase activity
The assay system consisted of 1.65 mL phosphate buffer (0.1 M, pH 7.6), 0.1 mL ethylenediaminetetraacetic acid (0.5 mM), 0.05 mL oxidized GSH (1 mM), 0.1 mL nicotinamide adenine dinucleotide phosphate (NADPH; 0.1 mM) and 0.1 mL of PMS (10% w/v) in a total volume of 2.0 mL. The enzyme activity was quantitated at 25°C by measuring the disappearance of NADPH at 340 nm and was calculated in nanomoles of NADPH oxidized per minute per milligram of protein using a molar extinction coefficient of 6.223103 M cm−1. 24
Glutathione-S-transferase activity
The reaction mixture consisted of 1.425 mL phosphate buffer (0.1 M, pH 6.5), 0.2 mL reduced GSH (1 mM), 0.025 mL 1-chloro-2,4-dinitrobenzene (CDNB; 1 mM), 0.3 mL and 0.05 mL of PMS (10% w/v), respectively, in a total volume of 2.0 mL. The changes in absorbance were recorded at 340 nm and enzyme activity was calculated as nanomoles of CDNB conjugate formed per minute per milligram of protein using a molar extinction coefficient of 9.63 M cm−1. 25
H2O2 assay
Microsomes of 2.0 mL (10% w/v) each were suspended in 1.0 mL of solution containing phenol red (0.28 nM), horseradish peroxidase (8.5 U), dextrose (5.5 nM) and phosphate buffer (0.05 M, pH 7.0) and were incubated at 37°C for 60 min. The reaction was stopped by the addition of 0.01 mL NaOH (10 N) and then centrifuged at 800g for 5 min. The absorbance of the supernatant was recorded at 610 nm against a reagent blank. The quantity of hydrogen peroxide (H2O2) produced was expressed in nanomoles of H2O2 per hour per gram of tissue based on the standard curve of H2O2-oxidized phenol red. 26
Determination of protein carbonyl content
Protein oxidation was measured by estimating the carbonyl groups with slight modifications using 2,4-dinitrophenylhydrazine (DNPH) reagent. 0.1 mL of serum was treated with 0.5 mL of 10 mM DNPH, dissolved in 2 M hydrochloric acid (HCl) as a sample or with 0.5 mL of 2 M HCl as control blank. The reaction mixtures were allowed to stand for 1 h at room temperature and in dark, with stirring at 15 min intervals. Then, 0.5 mL of ice-cold 20% TCA was added, and the sample was left on ice for 15 min. The tubes were then centrifuged at 11,000 r min−1 for 5 min to obtain the protein pellet. The precipitated proteins were subsequently washed three times with 1 mL of ethanol–ethyl acetate (1:1). Each washing step was followed by centrifugation at 3000 r min−1 for 7 min. Final protein pellet was dissolved in 0.25 mL of 6 M guanidine hydrochloride and incubated at 37°C for 10 min. The carbonyl content was calculated from peak absorption (360 nm) using an absorption coefficient (e) of 22,000 M−1 cm−1 . 27
Statistical analysis
Values were expressed as means ± standard error of mean. The level of significance between different groups is based on Dunnett’s t test followed by analysis of variance (ANOVA). One-way ANOVA was used to calculate the statistical significance between various groups. The value of p < 0.05 was considered to be statistically significant.
Results
Effect of DAS on Fe-NTA-mediated enhancement in the value of serum CRN and BUN
Fe-NTA administration led to about 2.7–3-fold enhancement in the levels of serum CRN and BUN, respectively. Pretreatment of DAS to rats on receiving Fe-NTA led to a significant decrease in both of these values. The effect was dependent on the dose of DAS. However, at a higher dose of 200 mg kg−1 b.w. of DAS, the values of CRN and BUN reached almost 1.6–1.92-fold respectively compared with the Fe-NTA-treated group (group III). The changes in levels of BUN and CRN are shown in Figure 1.
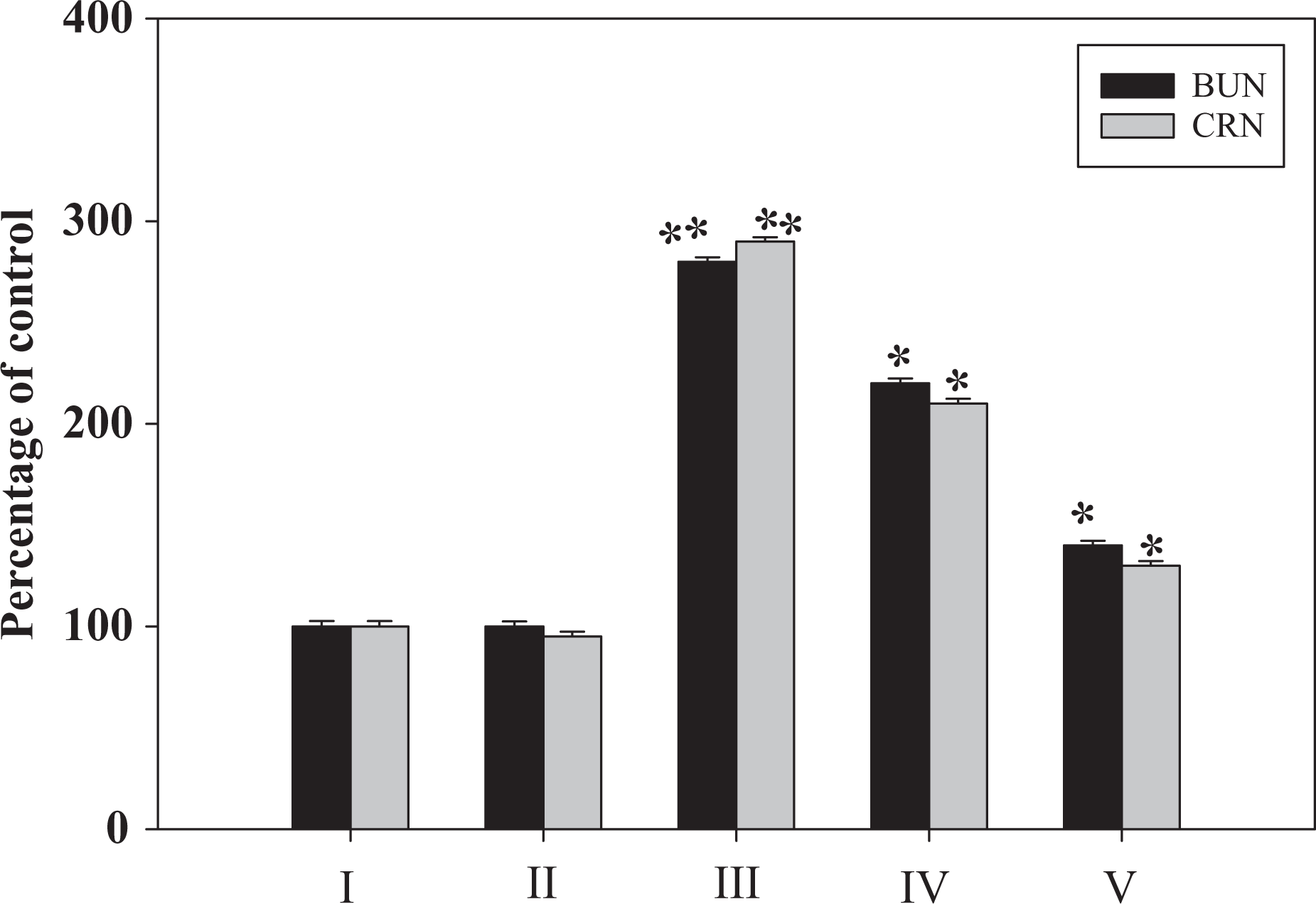
Effect of DAS on Fe-NTA-induced levels of BUN and CRN. I: saline, II: DAS (D2), III: Fe-NTA, IV: DAS (D1) + Fe-NTA, and V: DAS (D2) + Fe-NTA. Data represent mean ± SE of six animals. Dose regimen and treatment protocol are described in the text. Saline-treated group served as control for group III. Fe-NTA-treated group served as control for groups IV and V. *p < 0.05; **p < 0.001. Fe-NTA: ferric nitrilotriacetate; DAS: diallyl sulphide; D1: dose 1; D2: dose 2; BUN: blood urea nitrogen; CRN: creatinine.
Effect of DAS on Fe-NTA-induced changes in antioxidants
The effect of pretreatment of rats with DAS on Fe-NTA-mediated depletion in renal antioxidant molecules is shown in Table 1. Fe-NTA treatment results in the depletion of renal GSH, glutathione reductase, and glutathione-S-transferase to the level of 68, 44 and 37%, respectively, of the control value. Pretreatment with DAS resulted in recovery of these enzymes to approximately 75%, whereas at the higher dose this recovery was approximately 91% as compared to Fe-NTA-treated group.
Effect of DAS on Fe-NTA-mediated renal levels of GSH and the activities of GSH-metabolizing enzymes.a

DAS: diallyl sulphide; Fe-NTA: ferric nitrilotriacetate; GSH: glutathione; NADPH: nicotinamide adenine dinucleotide phosphate; D1: dose 1; D2: dose 2.aEach value represents mean ± SE of six animals. D1 and D2 represent administration of 100 mg and 200 mg DAS per animal per day, respectively, given orally 1 week before the treatment of Fe-NTA. Saline-treated group served as control for group III. Fe-NTA-treated group served as control for groups IV and V.
b p < 0.001.
c p < 0.05.
Effect of DAS on Fe-NTA-induced LPO and H2O2 generation
Fe-NTA treatment enhances microsomal LPO to 157%, whereas there is an enhancement of H2O2 generation to more than twofold of the corresponding control. DAS pretreatment inhibited dose dependently the enhancement of Fe–NTA-mediated LPO as well as H2O2 generation. Although there is a significant reduction at both the doses of DAS studied, at the higher dose of 200 mg kg−1 b.w. the level of LPO reached almost to the value of the control group, while H2O2 generation was also significantly reduced (p < 0.001) in renal tissues of rats as shown in Table 2.
Effect of DAS on Fe–NTA-induced renal microsomal lipid peroxidation and generation of hydrogen peroxide.a

DAS: diallyl sulphide; Fe–NTA: ferric nitrilotriacetate; MDA: malondialdehyde; H2O2: hydrogen peroxide; D1: dose 1; D2: dose 2.aEach value represents mean ± SE of six animals. Dose 1 and dose 2 represent administration of 100 mg and 200 mg DAS/animal/day respectively given orally 1 week before the treatment of Fe–NTA. Saline-treated group served as control for group III. Fe–NTA-treated group served as control for group IV/V.
b p < 0.05.
c p < 0.001.
Effect of pretreatment of rats with DAS on Fe-NTA-mediated induction of protein carbonyl
Fe-NTA (alone) treatment results in a maximum of 2.7-fold increase in the 2,4-DNPH incorporation into cystosolic proteins in rat kidneys. In DAS-pretreated animal, this increase in the incorporation of 2.4-DNPH was less as compared to Fe-NTA (alone)-treated group. The decrease in the incorporation of 2,4-DNPH into cystosolic protein was dependent on the dose of DAS used. At the higher dose of DAS, the decrease in the incorporation of 2,4-DNPH into cystosolic protein was reduced to about 1.2-fold (p < 0.05) as shown in Table 3.
Effect of DAS on Fe-NTA-mediated induction of protein carbonyl.a

DAS: diallyl sulphide; Fe-NTA: ferric nitrilotriacetate; D1: dose 1; D2: dose 2.aEach value represents mean ± SE of six animals. D1 and D2 represent administration of 100 mg and 200 mg DAS per animal per day, respectively, given orally 1 week before the treatment of Fe-NTA. Saline-treated group served as control for group III. Fe–NTA-treated group served as control for groups IV and V.
b p < 0.05.
Discussion
Fe-NTA leads to generation of ROS and plays an important role in toxicity causing DNA–protein cross-link formation, DNA base modifications, apoptosis, 28 DNA damage leading to single-strand and double-strand DNA breaks. 29 –31 Previously it has been shown that Fe-NTA induces oxidative stress in kidney by decreasing antioxidant enzyme activities with the concomitant increase in the production of lipid peroxides and H2O2. 32 The involvement of oxidative stress in Fe-NTA-treated rats is also evident from the present study with an increase in the formation of renal protein carbonyl, an indication of oxidation of cellular protein as a result of increased generation of oxidant.
Intake of natural antioxidants in diet including garlic, therefore, has been suggested to be useful against oxidant-mediated toxicity. 33 –37 It has been reported that the members of allium family are excellent sources of organic sulphides and thus act as potent modifiers of tissues oxidant response. DAS, a naturally occurring thioether with strong antioxidant properties, has been shown as a cancer chemopreventive agent. 38 –41 In the present study, DAS diminished Fe-NTA-mediated renal oxidative stress and toxicity as a result of its counteracting effect on Fe-NTA-mediated generation of oxidants.
A sharp decrease in the level of BUN and serum CRN in DAS-pretreated groups suggests that prophylaxis of DAS is effective in improving renal functions in Fe-NTA-treated animals. Recently, similar studies were carried out where diallyl disulphide had a renoprotective effect in gentamicin-induced acute renal failure in rats may be related, at least in part, to the amelioration in the oxidative stress and the preservation in the activity of the antioxidant enzymes in kidney. 42 Other studies suggest that short-term modulation of carcinogen bioactivation in situ by DAS may contribute to the chemoprevention of nitrosamine tumorigenesis. 43
It has also been known that garlic and its related compounds have inhibitory effects on chemical carcinogenesis and mutagenesis. 44 –46 These compounds may inhibit the activation of other carcinogens at low efficiency and the induction of glutathione -S-transferase and phase II enzymes may also play a role. 47 –50 Since DAS is a major constituent present in garlic oil and the pattern of protection between DAS and garlic oil is not very much different as observed in earlier studies, it could be interpreted that the earlier protection by garlic oil may be due to the effect of DAS. Hence, DAS-dependent attenuation of Fe-NTA-mediated renal damage may be the result of the scavenging effect of DAS to protect cellular membrane damage against the deleterious effect of ROS.
In summary, the results show that DAS is a potent protector of Fe-NTA-mediated kidney damage and may act by modulating the oxidative stress and cellular proliferation response. Also, garlic and its thioether constituents may be of great importance in affording protection against environmental renal carcinogens which inflict their toxicity by oxidative mechanisms.
Conclusion
Present data suggest that DAS has the potential to be used as a potent chemopreventive agent and may offer protection against Fe-NTA-mediated renal damage in rats. However, the levels of DAS used in many studies are much higher than what is normally consumed by humans. A major concern that must be addressed when extrapolating animal findings to humans is the dosage of the agent studied. This will have to be taken into consideration in the design of clinical studies and in evaluating the role of garlic and its related compounds as chemopreventives.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research project was supported by a grant from the “Research Center of the Center for Female Scientific and Medical Colleges”, Deanship of Scientific Research, King Saud University, Riyadh, Saudi Arabia.
