Abstract
Objectives:
The aim of this study was to evaluate, the cytotoxicity of orthodontic composites in vitro as a function of degree of conversion (DC) and the light curing units (LCU) employed on mouse fibroblast (L929).
Materials and Methods:
Cured samples of the composites Light bond (Reliance Orthodontic Products, Itasca, Illinois, USA), Ortho bracket paste (Bisco, Schaumburg, Illinois, USA), Opal bond MV (OPAL, South Jordan, Utah, USA), and Transbond XT (3M, Monrovia, California, USA) were prepared. Polymerization was performed with two LCUs: VALO Ortho (Ultradent, South Jordan, Utah, USA) is a third-generation LCU and Elipar S10 (3M, USA) is a second-generation LCU. Four samples were immersed in cell culture medium to obtain composite extracts. After incubation of L929 cell cultures with the extracts obtained, cytotoxicity was determined using the methyl tetrazolium test. Fourier transform infrared spectroscopy (FTIR) was used to evaluate DC for five samples. A multivariate analysis of variance (ANOVA), two-way ANOVA, and Tukey’s honestly significant difference test were utilized for statistical analyses.
Results:
Cytotoxicity and DC of all tested composites (p < 0.001) and the interaction between composites and LCUs (p < 0.01) were significantly different. LCUs had no significant influence on the cytotoxicity and DC of composite materials (p > 0.05). The correlations between cell viability and DC were positive for three composites but statistically insignificant.
Conclusion:
Composites and LCUs must be matched with one another to result in satisfactory maximal biocompatibility and DC. Opal Bond plasma light-emitting diode combination was a better choice for cell viability. Three composites showed a positive correlation between cytotoxicity and DC. Therefore high-intensity LCUs can be said to efficiently affect polymerization, and so, higher DC rates may achieve higher cell viability rates.
Introduction
An orthodontic composite should ideally have some essential characteristics for good clinical performance in all aspects of treatment, such as ease of use, sufficient physicomechanical properties, nontoxicity, and nonirritation of local tissue. 1,2 For years, light-curing orthodontic composites have been used as bonding agents of brackets to enamel and they are now a preferred alternative to chemical curing adhesives. Light stimulates the photoinitiator system of light-curing composites, thereby achieving polymerization (cross-linking) of the organic matrix molecules. Therefore, during a polymerization reaction, all carbon double bonds (C=C) of matrix monomer are expected to be converted into carbon single bonds (C–C) to form polymer. 3
The final product, polymer, is directly affected by monomer type, filler size, and polymerization conditions such as curing time, light intensity, emission spectrum, and power density. 3–5 Clinically, polymerization processes are usually incomplete owing to difficulties in illuminating the composite evenly from each side of the bracket as well as difficulties involved with the light source reaching all parts of the oral cavity.
Nowadays, high-intensity light curing units (LCUs), which are plasma arc curing (PAC), and solid state light-emitting diode (LED), have a wavelength range of 360–520 nm; the relation between excitation emission spectrum and photosensitizer system has a decisive effect on degree of conversion (DC) of composites and indirectly, the release of cytotoxic residual monomers. 2,3 For the most part, adequate polymerization is linked to wavelength range, power density, and exposure duration of LCU, and powerful light curing is found to eventually result in higher polymerization to a lower residual monomer. 4,6–8 In this way, photoinitiator systems of current light-cured resin-based composites generally do not claim all details by manufacturers, so there might be unknown coinitiators different from camphorquinone (CQ) such as lucirin trimethylbenzoyl-diphenylphosphine oxide (TPO) which has lower spectral extinction range from CQ. 9,10 This circumstance needs a LCU which must present good match with absorption spectrum of possible unknown photoinitiator agents of adhesive resins.
The third generation or poly-wave LED LCUs have a broad emission spectrum to coincide with the absorption spectrum of the photoinitiators at approximately 390–510 nm, which can have a significant influence on the DC and, in the end, on the residual monomer content. 8 – 10 In vitro studies have reported that DC values of orthodontic composites to range from 42 to 70% using PAC. 7 Furthermore, studies have proved that DC values of LED ranged from 44 to 88% and from 49 to 52%. 11,12 DC values of a light-cured composite under brackets to range from 37 to 55%. 13 So far no study has reported any information about the effects of these poly-wave LED LCUs on DC or cytotoxicity of orthodontic composites.
In particular, inadequate polymerization of orthodontic composites results in inferior physicomechanical properties and clinical performance and increased toxicity rates. 1,2,6,13 A higher share of soluble residual monomers or segregated composite constituents could produce local or systemic toxicity, via dentin, pulp, and mucosa. 2,14 Therefore, if light polymerization is incomplete or if less-cured polymer is produced, extracts may have increased cytotoxic effects. Moreover, an insufficiently polymerized composite layer under brackets is highly permeable and cytotoxic substances from the composite can penetrate the enamel, dentin, and pulp.
Thus far, an increasing number of investigations have focused on harmful effects of cytotoxicity of orthodontic composites and components on cultured cells; 12,15 however, studies have not evaluated and correlated the influence of polymerization quality to cytotoxicity using poly-wave LED LCUs. The relative contributions of polymerization indirectly influence the residual share of uncured toxic monomers. 2,6 The potential cytotoxicity levels of different orthodontic composites pose an interesting research topic. However, to date there is no knowledge about which combinations of LCUs and orthodontic composites cause the least toxic effects.
The aims of this investigation were to assess the possible cytotoxicity levels of various orthodontic composites differing in types of LCU, in order to evaluate and correlate the DC of these resins as a function of light curing type to cytotoxicity. The null hypothesis assumed is that the plasma-emulating LED LCU would show similar effects on levels of cytotoxicity and DC of various composites compared with conventional LED LCU observed in the experimental protocols.
Materials and methods
Specimen preparation
Four different composites currently used were investigated for cytotoxicity and DC (Table 1). In total, 72 disk-shaped test specimens were prepared in sterile Teflon molds containing 5 mm diameter (depth 2 mm) under aseptic conditions in an ultraviolet-sterilized laminar air flow chamber. Samples were hardened between Mylor and glass slabs (2 mm) to minimize the oxygen inhibition and maximize the surface smoothness. The curing time for plasma-emulating LED (plasma LED) was 3 s, while that for LED was 20 s as instructed by manufacturers. The light outputs from LCUs were measured and averaged three times using a dental radiometer (Table 2).
‘Composites’ (Based on MSDS).

LB: light bond; BR: ortho bracket paste; OBM: opal bond MV; TB: transbond XT; GMA: glycidyl methacrylate; UDMA: Urethane Dimethacrylate; TEG-DMA: Triethylene Glycol Dimethacrylate.
Light-curing units.

LED: light-emitting diode.
aBluephase Meter by Ivocar Vivadent.
Eluate preparation and cytotoxicity
Four specimens from each curing were randomly selected and immediately transferred in 7 mL of culture medium containing 8 tubes for 24 h at 37°C to extract residual monomer. The eluates containing material extracts were filtered for sterilization to use on cell cultures.
Mouse fibroblast (L929) cells (ATCC-CCL-1, Şap Enstitüsü, Turkey) were cultivated in 10% fetal calf serum and 1% antibiotic antimycotic solution at 37°C and in a fully humidified air atmosphere containing 5% carbon dioxide (CO2). Cells of 12th to 15th passage were used to minimize differences in cell cycle.
Cells of the L929 culture were seeded in a density of approximately 10,000 cells into per well of 96-well microplates for 24 h at 37°Cand 5% CO2. After 24 h of incubation, 200 µL eluate was covered to cell layer per well, and also, original culture medium served as control. In this manner, 9 parallel test batches were acquired from each composite–LCU combination, plus 12 control batches for 24 h at 37°C and 5% CO2. Afterward, (3-[4,5- dimethyl-thiazol-2-yl]-2,5-diphenyl-tetrazolium bromide) (MTT) test was used to evaluate cell viability of the L929, which is reasonably representative of the activity of mitochondrial dehydrogenases in the cells and reflects both cell number and activity.
The old medium was cleaned, cells were the rinsed in phosphate-buffered saline solution, and 200 µL MTT solutions were added each well, and the plates were incubated overnight at 37°C and 5% CO2. The mitochondrial dehydrogenases converted the yellowish extracellular MTT dye (tetrazolium salt) into the insoluble intracellular formazan. To this, 200 µL dimethyl sulfoxide was added for solubilization and then optical densities (OD) were measured with a spectrophotometer at 570 nm. The cell viability was calculated using the following formula: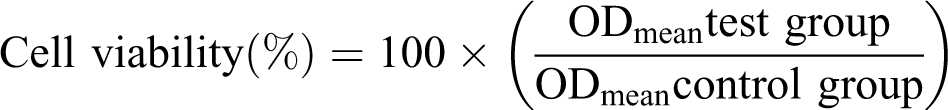
DC assessment
A Fourier transform infrared (FTIR) spectrometer (Shimadzu, 8300-FTIR, Kyoto, Japan) was employed to analyze DC with using a potassium bromide (KBr) pellet transmission method. Remaining five specimens from each group were stored in light-proof boxes to prevent further exposure to light and for 24 h in artificial saliva at 37°C and then mechanically powdered with mortar and pestle. A standardized quantity of resin powder (1%) and IR-grade KBr was manually mixed and pressed (5000 psi, 2 min) into a transparent pellet (12 mm in diameter). FTIR spectrum of unpolymerized composites, which were smeared onto KBr disks, was also recorded. Absorbance spectra were analyzed, using 4000–400 cm−1 wave number range, 30 coadded scans at 4 cm−1 in transmission.
The calculation of DC of methacrylate-based composites firstly begins with estimation of aliphatic C=C bond stretching vibrations of uncured material from absorption (A) peak occurring at 1637 cm−1. After polymerization, diminished concentration of aliphatic C=C is then redetermined from diminished absorption peak. The aromatic (C=C) bond stretching vibrations are convenient to take the reference aromatic peak, which does not enter into reaction during curing at 1605 cm−1.
Residual double bonds (RDB) were proportioned and recorded as percentage (%) using the following equation:
DC is calculated using the following equation:
Statistical evaluation
The mean and standard deviation values were obtained (Table 3). A multivariate analysis of variance (MANOVA) was conducted to test the interaction between cell viability and DC in a way of composites and LCUs. Then, in order to see clearly the source of differences, two-way ANOVA was also carried out separately with Bonferroni’s post test. Tukey’s honestly significant difference test was used for multiple comparisons. Cell viabilities in treated groups were compared with that in the untreated controls. The percentage values of DC and cell viability were correlated by Pearson’s correlation test. The level of significance was set at p < 0.05.
L929 cell viability of preincubated specimens expressed as a percentage of controls (cultures without specimens) and percentages of degree of conversions: means (standard deviations).

Results
Results of MANOVA show that cytotoxicity and DC of all four tested composites (p < 0.0001) and interaction between composites and LCUs (p < 0.0001) were significantly different, while cytotoxicity and DC of LCUs (p > 0.050) were not significantly different.
For cytotoxicity, composites had statistically significant effect (p < 0.05); however, LCUs had no statistically significant influence (p > 0.05). Conversely, interaction between composite and LCU was found to play a statistically significant role on cytotoxicity (p < 0.05). Light bond (LB)-plasma LED showed the highest levels of cytotoxicity (Table 4).
Cell viability percentages of composites with different light sources in order to highest to lowest.

aMeans with the same letter were not significantly different at the α = 0.05 level.
The interaction between composite and LCU reflected that LB and ortho bracket paste (BR)-plasma LED produced higher cytotoxicity than with LED, whereas transbond XT (TB)-LED produced higher cytotoxicity than with plasma LED; moreover, opal bond mw (OB)-plasma LED/LED produced similar cytotoxicity rates (Figure 1).

Cell viability percentages of composites with different light sources.
For DC, composites had statistically significant effect (p < 0.05); however, LCUs had no statistically significant influence (p > 0.05). Conversely, interaction between composite and LCU was found to play statistically significant role on DC (p < 0.05). LB-LED displayed the highest levels of DC, and TB-LED showed the lowest levels (Table 5).
Degree of conversion percentages of composites with different light sources in order from highest to lowest.
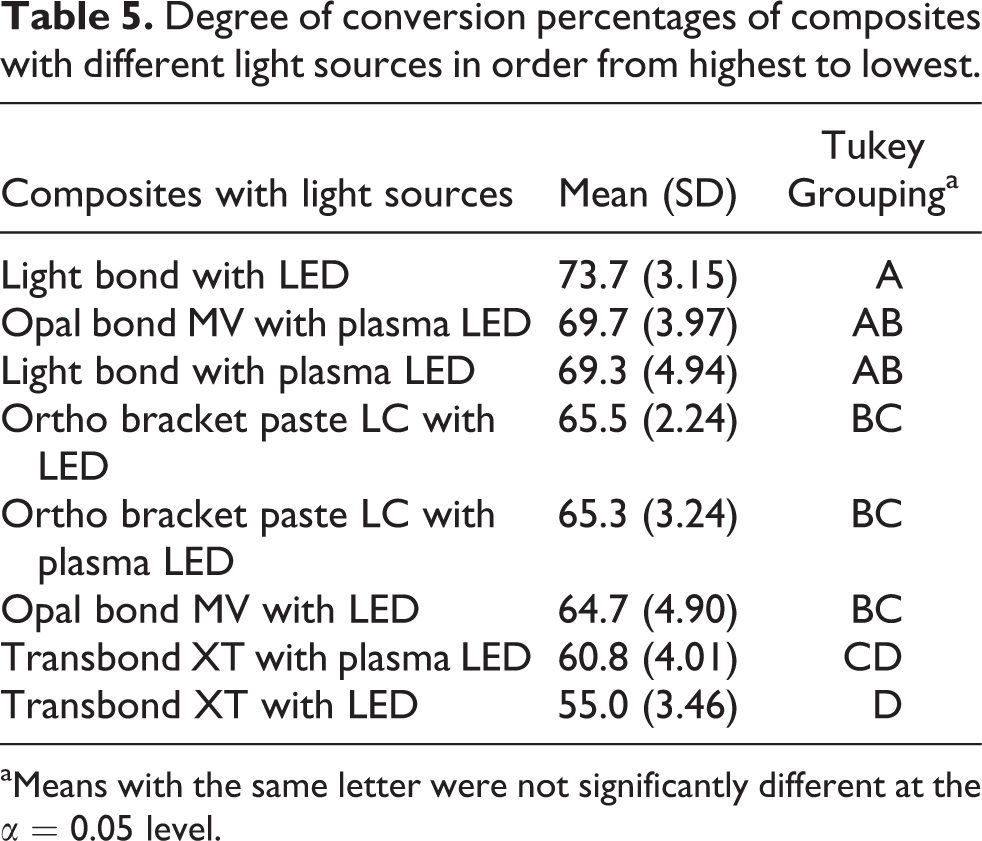
aMeans with the same letter were not significantly different at the α = 0.05 level.
The interaction between composite and LCU reflected that TB-plasma LED and OB-plasma LED produced higher DC than with LED; whereas in LB-LED produced higher DC than with plasma LED. Moreover BR-plasma LED and LED produced similar DC rates (Figure 2).

Degree of conversion percentages of composites with different light sources.
The correlations between cell viability and DC were positive for all the materials tested except for BR. But these positive correlations were not statistically significant (p > 0.05; Table 6).
Pearson correlation between cell viability and degree of conversion expressed by composites.
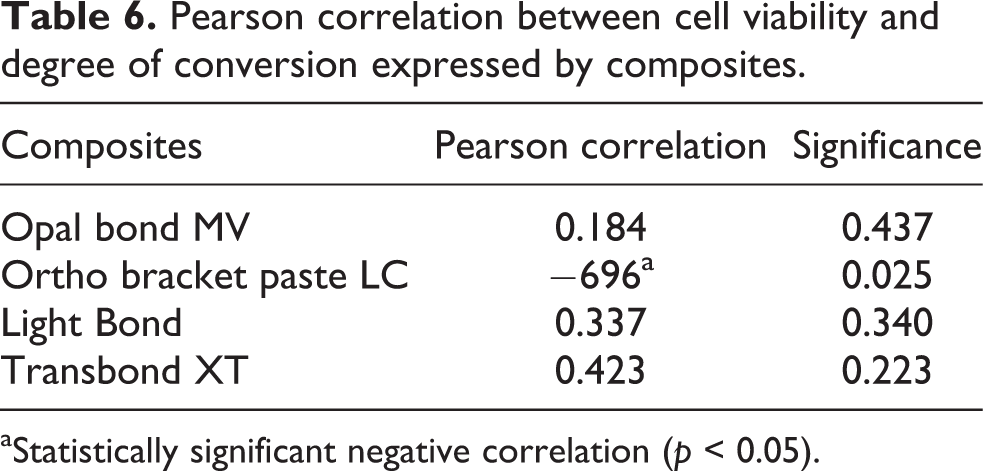
aStatistically significant negative correlation (p < 0.05).
Discussion
As part of this investigation, we tested cytotoxicity and DC of four composite-two LCU combinations. Our results reflected that all tested composite-LCU combinations had statistically significant influences on cytotoxicity (Table 4). DC rates of composite-LCU combinations were also statistically different (Table 5). Therefore, the null hypothesis was rejected. Furthermore, there were insignificant positive correlations between cell viability and DC in all tested composites except BR.
The underlying purpose of the study design is twofold: (1) Thus far, researchers have not investigated and correlated cell viability and DC of composites with different high-intensity LCUs and (2) a suboptimal cure of monomer can lead to a greater amount of toxic residual monomers in composite. 6 Most of the cytotoxic capacity of composites is well known to depend on unbound free resinous monomers. 2,6 Moreover, the power density and exposure duration of LCUs are the most critical factors affecting monomer conversion of composites. 6 –8,16 After covering L929 cells to 24 h eluates, we observed a significant reduction in cell survival rate induced by composite–LCU combinations. The LCU combinations with TB and LB showed the highest cytotoxicity rates, whereas the combination with OB produced favorable consequences depending on LCU. The superior composite–LCU combinations were OB-plasma LED, LB-LED, OB-LED, TB-Plasma LED, and BP-LED, because these resulted in a cell viability of nearly 95%. Additionally, a clear diminishing of cell viability was reported in LB-plasma LED and TB-LED combinations (Table 4 and Figure 1). Cytotoxicity rates for TB-LED, BR-LED, and LB-LED were in agreement with those recorded by a previous study. 15
The strong cytotoxicity results for LB and TB composites can be explained not only by differences in their chemical compositions (Table 1) but also because they released greater amounts of leachable substances (which resulted in degradation over time) into the culture medium (24 h). Therefore, cellular activity and survival of L929 could have been sufficiently disturbed. One study has provided supporting evidence for this owing to the fact that unpolished surfaces on specimen body promote faster elution of unbound surface monomers. 17 It can be concluded that enamel surfaces of a bracket bonding composite or a slight bit excess of adhesive after setting are also considered as unpolished surfaces and this can possibly interact there with the surrounding dental tissue. 14,17
Except for BR composite, cell viability was positively correlated with DC. A possible reason is that polymerization with high-intensity LCUs used (Table 2) might have a crucial impact on the cell viability. This finding was in contrast to the results of another study. 12 In that study, low-intensity LCU and DC values ranged from 39 to 52%, values that are relatively lower than those obtained in the present study. 12 Therefore, it can be said that higher DC rates may achieve higher cell viability rates.
Bisphenol A, bis-glycidyl methacrylate (GMA), and CQ are ingredients in polymers that are easily lixiviated among other components. 18 These substances are known to be toxic and are thought to affect cellular metabolism when in very low concentrations. 2,16,19–21 Three of the four test composites—LB, OB, and TB—contained bis-GMA, and all four composites used CQ and some other unclear coinitiators as photoinitiator. The most toxic among them were bis-GMA and bisphenol A, whose cytotoxic effects become pronounced after hydrolysis through the production of methacrylic acid. Methacrylic acid is a metabolite that can affect cell viability by affecting membrane permeability. 19 Methacrylic acid also exhibits estrogenicity and can be detected in pulp, oral mucosa, and dental cement. 2,19,21
Orthodontic composites exhibit a potential high risk when there is an incomplete cross-linking of polymer because these composites are employed in thin layers that favor inhibition by oxygen and difficulties in illuminating under brackets. 2,3,12 This is also clinically problematic under brackets where the bonding composite is very thin. Through the experimental design of the present study, we also proved that polymerization was not completely accomplished on the surface of the test material due to the presence of oxygen, and as a result, the surface may contain residual monomer available for release. In a previous study, TB composite with halogen-LCU showed a strong restrictive effect of oxygen on polymerization, which decreased on cell viability. 12
Thus far, studies have not evaluated cell and tissue responses or DC, of OB in the literature. Of the four composites assessed, OB had the highest potential cell viability and the most favorable DC values. It was therefore possible to consider the chemical interaction among the components of OB. Nonetheless, composite resin viscosity has a decisive effect on the thickness of oxygen-inhibited layer; 22 therefore, lower viscosity results in higher cytotoxicity. Clinically, viscosity of OB was higher than that of other composites and this may have decreased cytotoxicity of OB.
In the present study, with the exception of TB, the cell viability estimations of released extracts displayed a tendency to be higher when LED was used; however, the toxicity rates of released extracts showing varying tendencies when high-intensity poly-wave plasma LED was used. Moreover, DC measurements of composites showed a similar trend when both LED and plasma LED were used, with exception of TB. As a result, fast activation by powerful plasma LED can achieve a high DC as well as a low degree of cytotoxicity with an intense polymeric network. 23 While some cytotoxicity and DC values of tested composites-plasma LED were found to be in contrast with that proof (Tables 4 and 5). This is probably because inorganic and organic components and viscosities of composites were different. Besides, in current experiment, LED curing time was 20 s which was enough to produce a strong polymeric structure at the end of the irradiation period in some composites.
In this study, there was an obvious interaction between LCU and composite especially in cases of LB and TB, where polymerization capacity possibly diminished the amount of unbound toxic substances. The increase in cytotoxicity of LB and TB extracts were 11 and 10%, respectively. Therefore, it can be said that plasma LED and LED may be necessary for efficient polymerization of TB and LB, respectively. Likewise, in terms of cytotoxicity, OB and TB seemed to benefit significantly from the use of plasma LED; in terms of DC, LB seemed to benefit significantly from the use of LED. Similar cytotoxicity results were also observed in two other studies that used LED for LB 15 and TB 12 (Tables 4 and 5).
Currently, plasma LED or LED units are popular LCUs in orthodontic practice because of their numerous advantages such as shorter curing time, different curing modes, high power density, light output stability, beam profile match with CQ and other coinitiators, and cordless design. However, the current investigation reveals that there remains scope for improvement. Based on the findings of this research, composites and LCUs must be matched with one another in order to obtain maximal biocompatibility and a satisfactory DC rates. Further studies should focus on assessing long-term toxic effects and DCs for orthodontic composites as well as different irradiation times or LCUs.
Conclusion
All tested composite–LCU combinations reduced cell viability of L929 which may be indicative of potential cytotoxicity to dental tissue.
The composites and LCUs must be matched with one another to result in a satisfactory DC and maximal biocompatibility.
Opal bond–plasma LED combination was the preferred choice for cell viability.
Three composites—LB, OB, and TB—showed a positive correlation between cytotoxicity and DC. Therefore high-intensity LCUs may efficiently affect the polymerization; corresponding higher DC rates may achieve higher cell viability rates.
Footnotes
Acknowledgments
We would like to thank Prof Dr Hamit Coşkun and Department of Chemistry, Faculty of Art and Science, Abant İzzet Baysal University for their support.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
