Abstract
Titanium dioxide (TiO2) nanoparticles (NPs) are commonly used materials present in many consumables for which most people are exposed to. The biological hazards of the NPs on human health have been demonstrated previously. In this study, we aimed to assess the cytotoxicity potency of TiO2 NPs on the primary human amniotic fluid cells. The cells derived from amniotic fluid were treated with different dosages of TiO2 NPs for some periods. Cell adhesion status was assessed using a light microscopic observation. Cell proliferation and cell death rates were determined using trypan blue staining and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay. Also, mitotic index was determined using fluorescence in situ hybridization with chromosome 8 centromer-specific DNA probe. Disrupted cell adhesion, decreased proliferation, and increased mortality rates were detected in the cells that were treated with TiO2 NPs depending on the dosage (p < 0.001). Also, reduced mitotic index was determined in the cells depending on the time and TiO2 dosage when compared with the controls (p < 0.0001). These results showed that TiO2 NPs have high cytotoxicity for amniotic fluid-derived cells. Therefore, different products containing TiO2 NPs should be used with care, especially for pregnant women.
Introduction
Nanotechnology and usage of nanoparticles (NPs) have been dramaticaly increasing worldwide. NPs ranging in size from 1 nm to 100 nm have become widely used in our daily life such as industrial applications, environmental studies, food technology, and human health impacts because of their unique physical and chemical properties. 1 Generally, well-known nanosized particles are more toxic than larger particles at identical mass concentration. 2 NPs have higher ability of entering the organism via diffusion to target cells and tissues and then induce cytotoxicity in many ways. 3,4 Among NPs, titanium dioxide (TiO2) is frequently used in the production of paints, paper, plastics as well as in processes of food additives, colorants, cosmetics, and pharmaceuticals. 5,6 Thereby, the development of nanotechnology is considered as a kind of novel industrial revolution due to increasing number of applications and economic impacts. Many studies showed cytotoxicity of TiO2 NPs in different cell models and chiefly resulted from the induction of apoptosis in mammalian cells. 7,8 In addition, TiO2 NPs induce oxidative stress-mediated toxicity in cells by catalyzing the formation of superoxide, hydrogen peroxide (H2O2), and hydroxyl radicals. 9,10 L’Azou et al. (2008) indicated that TiO2 NPs when incorporated into cellular membranes and cytoplasm of mammalian cells induce generation of reactive oxygen species. 11 TiO2 NPs trigger inflammatory processes, pulmonary damages, fibrosis, and even DNA damages that lead to the development of cancerous cells such as lung tumors. 12 –14 Moreover, these NPs cause lipid peroxidation, micronuclei formation, increasing H2O2, and nitric oxide production in human bronchial epithelial cells. 9,15 Numerous in vitro studies on different cell types have shown oxidative stress-based cytotoxicity and genotoxicity to cells treated with TiO2 NPs. 16,17
TiO2 NPs could be absorbed through inhalation, ingestion, and dermal penetration into the body and then delivered in the important organs such as lung, 18,19 lymph nodes, 12 brain, 20 liver, and kidney. 19 Very recent study showed that TiO2 NPs can also change gene expression in human neutrophils, resulting in the change of enzyme activity. 21 Furthermore, the possible interference of TiO2 NPs in human health during prenatal or early childhood has been investigated considering that TiO2 NPs have the ability to cross the placenta. 22 Given the importance of maternal and fetus health during pregnancy, investigation on possible effects of TiO2 NPs on amniotic fluid cells will give important information about TiO2 biohazards. In this study, we aimed to assess the cytotoxicity of TiO2 NPs on primary fetal amniotic fluid cells by determining survival and proliferation rates and mitotic index of the cells. We firstly investigated the possible cytotoxicity of TiO2 NPs and then determined mitotic index of the human amniotic fluid cells in the presence of TiO2 NPs in different dosages and treatment periods.
Materials and methods
Chemicals
Anatase form of TiO2 NPs (Milenium brand) was obtained as a gift from Dr M Ersoz, Faculty of Science at Selcuk University, Turkey. H2O2 was used as a postive control agent for cytotoxity and mitotic index reducer. TiO2 NPs in powder form were sterilized by heating to 120°C for 2 h and were suspended in sterilized phosphate-buffered saline (PBS) as 1 mg/ml concentration for stock solution. This was subsequently diluted in complete culture medium serially to yield concentrations ranging from 10 μg/ml to 200 μg/ml. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) agent for assaying was purchased from Sigma-Aldrich Co. (St Louis, Missouri, USA). For fluorescence in situ hybridization (FISH) study, chromosome 8-specific α satellite DNA probe labeled with rhodamine and counterstain dye including 4′,6-diamidino-2-phenylindole (DAPI) was supplied by Diagen AS (Ankara, Turkey).
Cell culture and chemical treatment
This study has been approved as ethical according to the local ethics committee of Selçuk University. Six different human amniotic fluid samples derived from mothers at 14 weeks gestational age were received from Dr G. Bagcı, Faculty of Medicine, Pamukkale University, Denizli, Turkey. Before beginning the amniotic fluid collection procedure, local anesthesia was given to the mothers, then a needle was inserted through the mother’s uterus wall, and approximately 20 ml amniotic fluid were drawn. Amniotic fluids were cultured in Bio-AMF medium (Gibco®, UK) contained 2%
Cells adhesion
The adherence of amniotic fluid cells on the plate and cellular morphology were screened for every 24 h after seeding the cells using an inverted microscope (400×; Nikon Ts100, Tokyo, Japan).
Viability assay
Viability of cell was evaluated using trypan blue assay. The cells were collected from culture and then washed with PBS. The pellet was suspended in 1 ml of complete medium and then from each sample. Cell suspension (20 μl) was stained with 20 μl of 0.4% trypan blue solution. The mixture was allowed to stand for 2 min at room temperature. Afterward, the cells were loaded onto hematocytometer chamber and then analyzed under a microscope by 100× magnification. Viable and dead cells were scored, considering lucid cells as viable and blue as dead cells.
MTT proliferation assay
After removing the culture medium from the plates, the cells on 96 wells were washed with PBS triple and then 20 μl of MTT solution (10 mg/ml) was added to the cells and were incubated for 3 h at 37°C or until the cells looked blue and crystalline. Afterward, the solution was aspirated by paper towel gently and then the cells were treated with 150 μl of dimethylsulfoxide. The plates were placed on rotator for 20 min or until all the crystals were dissolved. The absorbance was quantified using the Absorbance Micro-plate Reader (Bio-Tek EL×800, Winooski, Vermont, USA) at 540 nm wavelength. There was a direct relationship with absorbance quantity and the rate of cell viability and proliferation.
Mitotic index assay
Mitotic index assay was carried out using FISH technique with chromosome 8-specific centromeric probe labeled with rhodamine. After treatment periods, the cells were washed with PBS and harvested. Then, the cells were pelleted with centrifugation at 200g for 5 min. The pellet was suspended in 5 ml of hypotonic solution (0.015 M potassium chloride) and incubated for 10 min at 37°C. The cell suspension was centrifuged at 200g for 5 min and the pellet was suspended in 1 ml cold fixative solution (methanol: acetic acid in 3:1 ratio) and allowed to air-dry. Hybridization mixture containing 20 ng of centromer 8-specific FISH probe labeled with rhodamine (Diagen AS) was added on the fixed cells on the microscopy slides and a cover slip was then placed on it. Denaturation was carried out at 70°C for 6 min, and hybridization was performed at 37°C overnight. At the end of time, the slides were washed for 5 min with 2× stock solution (SSC) consisting of 300 mM sodium chloride + 30 mM sodium citrate (salt saline citrate) once and with 4× SSC with 0.5% Tween-20 twice. The cells mounted were counterstained with DAPI dye. In order to determine the mitotic index, 200 cells of each treatment groups were analyzed under the fluorescent microscope (Nikon Eclipse 80i, Tokyo, Japan) in terms of chromosome 8 copy number.
Statistical analysis
To determine differences and assosiations between the study groups, p values, odds ratio (OR), and 95% confidence intervals were calculated applying χ 2 or Student’s t test. All statistical analyses were carried out using Graphpad Prism v 5.03 software (La Jolla, San Diego, California, USA). The value of p < 0.05 was considered statistically significant.
Results
Cells adhesion
In order to determine the effects of TiO2 NPs on the adhesion property of amniotic fluid cells, 5 × 104 cells were seeded and cultured in 24-well plates. Chemical treatment was started at the beginning of culture and continued for three different periods as 24, 48, and 96 h. In the absence of TiO2 NPs, adhesion potency of cells was maintained, and the cells were possessing morphological characteristics of adherent cells (Figure 1(a)). In contrast, in the presence of 50 and 100 μg/ml of TiO2 NPs, the adhesion of cells was disrupted and the cells were round shaped and floating in the medium when compared with the cells treated with H2O2 as positive control (Figure 1(b) to (d)).

Microscopic image (×400) of amniotic fluid cells that were cultured for 48 h. Cells that were cultured in the absence of TiO2 NPs were succulent as attached to the plate (a); while the cells in presence of 50 (b) and 100 (c) μg/ml of TiO2 NPs have abnormal morphology and adhesion. The accumulation of TiO2 NPs is shown with arrows. Cells treated with 10 mM of H2O2 were used as positive control (d). TiO2: titanium dioxide; NPs: nanoparticles; H2O2: hydrogen peroxide.
Cytotoxicity of TiO2 NPs
The cytotoxicity of TiO2 NPs on treated and control cells was evaluated using trypan blue and MTT assays. The percentages of death cells are shown in Figure 2. The cellular cytotoxicity was enhanced following increasing treated concentrations of TiO2 NPs for all three 24, 48, and 96 h. In all treatment groups, the most toxic dose of TiO2 NPs was 200 μg/ml when compared with lower doses and the control (Figure 2(a) to (c)). The percentage of the dead cells treated with 200 μg/ml was 76% (p < 0.0001). Even lower doses in particular 50 and 100 μg/ml were also extremely mortal for the cells (p < 0.0001).
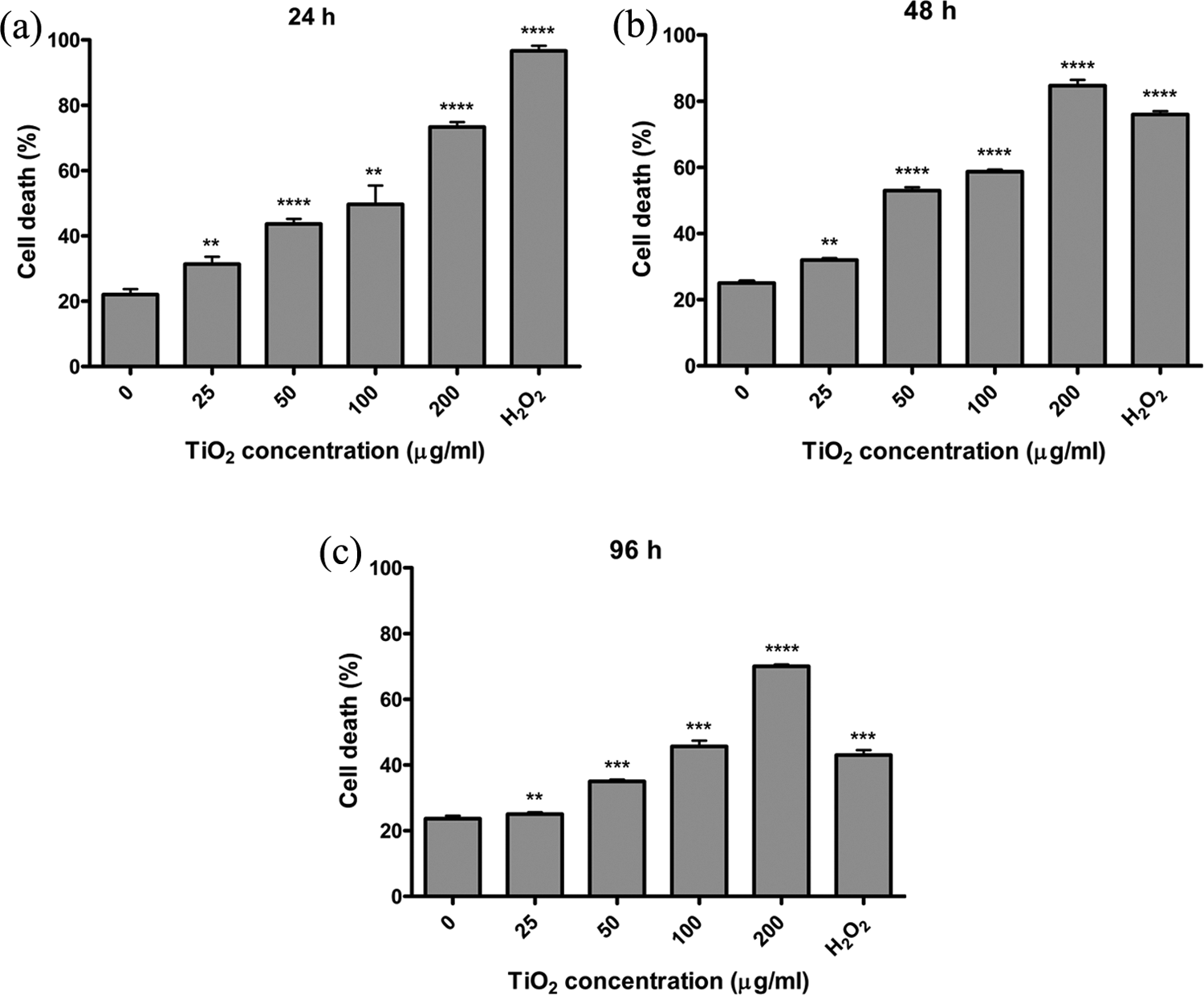
Percentage of dead amniotic fluid cells. Amniotic fluid cells were treated with 0, 12.5, 25, 50, 100, and 200 μg/ml of TiO2 NPs and were then stained with trypan blue after 24 h (a), 48 h (b), and 96 h (c) incubations. Increased dead cells percentage is evident in mid increscent concentration of TiO2 NPs (p < 0.0001). 10 mM of H2O2 was used as a positive control for cytotoxicity (p < 0.0001). **p < 0.01; ***p < 0 .001; ****p < 0.0001. TiO2: titanium dioxide; NPs: nanoparticles.
The results of MTT assay showed a dramatic dose-dependent decline in the proliferation rate (Figure 3(a) to (c)). The lowest proliferation rate was observed in the cells treated with 100 ng/μl TiO2 NPs for 24 h (p < 0.001). Notwithstanding, the same pattern was also applied for 48 h (p < 0.0005). The mean absorbance of 12.5 mg/μl TiO2 NPs concentration was lower, but standard deviation was high due to some inconsistent results (SD = 0.67). Interestingly, despite the reduction of H2O2 cytotoxicity, the cell death capability of TiO2 NPs was maintained over time (Figure 3(c)). Also, cytotoxicity of 200 μg/ml TiO2 NPs was much higher than 10 mM of H2O2 for 96 h treatment (p < 0.0001). As a result, TiO2 NPs are highly cytotoxic on cultured amniotic fluid cells in vitro.

MTT assay and cell proliferation rates of amniotic fluid cells. Amniotic fluid cells were treated with 0, 12.5, 25, 50, 100, and 200 μg/ml of TiO2 NPs for 24 h (a), 48 h (b), and 96 h (c). After these periods, cell proliferation was measured using MTT assay. The proliferation rate correlates with the absorbance value. Increased proliferation rate is evident in mid increscent concentration of TiO2 NPs (p < 0.001). 10 mM of H2O2 was used as a positive control for proliferation inhibitor (p < 0.0001). *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001. TiO2: titanium dioxide; NPs: nanoparticles; H2O2: hydrogen peroxide; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide.
Mitotic index assay
To determine the effects of TiO2 NPs on the mitotic activity of amniotic fluid cells, 200 cells for each treatment group were investigated, based on the analyses of copy number of chromosome 8. According to the results presented in Table 1, the cells treated with 0, 25, 50, 100, and 200 ng/μl of TiO2 NPs exhibited dual fluorescence signals, respectively, in 120 (60%), 166 (83%), 176 (89%), 188 (94%), and 200 (100%) cells, indicating normal disomic cell status. These cells were considered as in S or G1 phase of cell cycle. The remaining cells, respectively, 80 (40%), 34 (17%), 24 (12%), 12 (6%), and 0 (0%) cells showed four fluorescence signals indicating tetrasomic status of cell (Figures 4 and 5 and Table 1). These cells were considered as in the end of the S or G2 phase of cell cycle. These results suggest that TiO2 NPs reduce mitotic index in amniotic fluid cells as a dose-dependent base (p < 0.0001). It can be said that TiO2 NPs are strong mitotic inhibitors for these types of cells.
Frequency of chromosome 8 centromer-specific signals and relative statistical values in amniotic fluid cells with TiO2 NPs for 48 h.

df: degrees of freedom; CI: confidence interval.
aPositive control.

Fluorescence microscopy image (×1000) of amniotic fluid cells. Cells were cultured in the absence (a) and presence of 50 μg/ml (b) of TiO2 NPs for 48 h and then FISH assay was exerted with chromosome 8 centromer-specific α satellite DNA probe. Most of the untreated cells had dual signal and treated cells had quadrat fluoresent signals (alluded by arrows). TiO2: titanium dioxide; NPs: nanoparticles; FISH: fluorescence in situ hybridization.
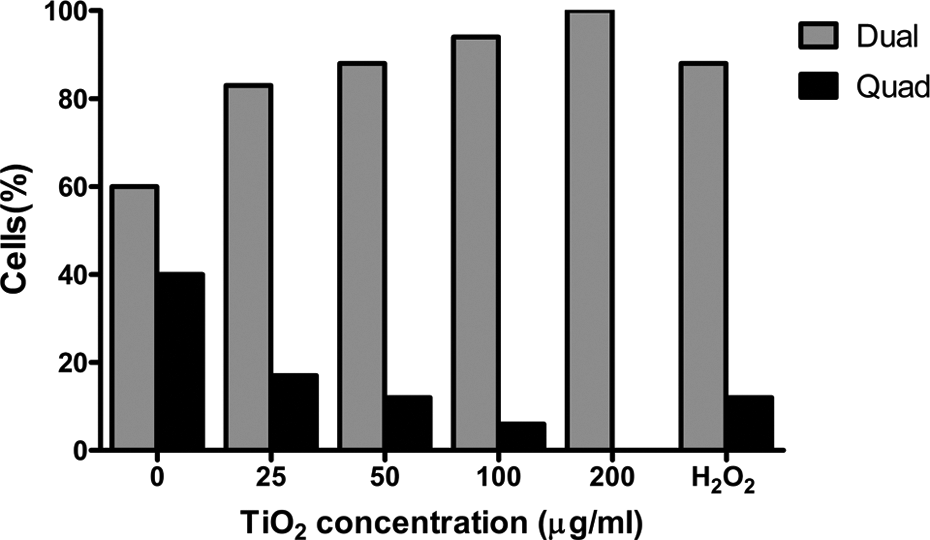
Percentage of amniotic fluid cells with dual and quadrat signals of chromosome 8 specific centromer α satallite DNA probe for FISH assay. Cells were cultured in the absence and presence of different concentrations of TiO2 NPs for 48 h. Rising of dual and declining of quadrat signals in the treated cells are held in considering untreated cells (p < 0.0001). 10 mM of H2O2 was used for positive control (p < 0.0001). TiO2: titanium dioxide; NPs: nanoparticles; FISH: fluorescence in situ hybridization; H2O2: hydrogen peroxide.
Discussion
The tendency of TiO2 NPs to diffuse through the protective cellular barriers may also have serious risks to human health. The NPs could easily pass into brain during the early stages of fetal development and also reach the placental tissue. 23,24 Wick et al. showed the uptake of nanosized materials across the placental barrier in an ex vivo human placental perfusion model. 22 Despite the biological effects of TiO2 NPs were substantiated previously, the influences of this composition on prenatal stages remains unclear yet. Previously, the cytotoxicity of TiO2 NPs on a human amnion cell line was showed in a study. 25 In the present study, we have used primary culture of human amniotic fluid cells to determine cytotoxicity of TiO2 NPs and mitiotic index.
As the first finding, we showed that TiO2 NPs perturbated adhesion ability of human amniotic fluid cells. Given the nature of these types of cells as adherent cells, prevention of adhesion leads to growth cease and eventually cell death. Presumably, this function was responsible for wide range of cell death as it eventuated.
Cell viability assay showed that TiO2 NPs were highly lethal for the cells and killed them with high intensity (p < 0.0001). On the other hand, the proliferation rate in the presence of TiO2 NPs was very low in comparison with absence of it (p < 0.001). Moreover, the proliferation inhibitory effect of TiO2 NPs was confirmed by mitotic index assay. The results showed that mitotic activity ceased in amniotic fluid cells under the presence of TiO2 NPs. Considering the ORs, the mitotic index has reduced progressively depending on TiO2 NPs concentration. These results, likewise, showed the cytotoxicity of TiO2 NPs on amniotic fluid cells in accordance with MTT assay results (p < 0.0001). Rationally, induction of proliferation and mitotic index may be in results of ruined cellular adhesion due to the presence of TiO2 NPs.
With a look up to the cell death, proliferation and mitotic index, it can be seen that the effect of TiO2 NPs was maintained more time than H2O2. This issue can be commended as follows that the cells could tolerate H2O2, while were unable to tolerate TiO2. Metabolization capability, which is an overriding factor in investigating toxicology effects of TiO2 NPs in vitro and in vivo in long-term treatments, can improve the understanding of destructivity power of these NPs.
The results presented here using amniotic fluid cells also supported a number of previous studies using a variety of cell culture systems. 8,17,26 However, there are contradicting results in literature on exposure time and dose and also the type of cells. 28–31 Hussain et al. reported that there was no significant reduction in viability at doses between 10 and 50 μg/ml in rat liver cells, but they did observe a significant effect at higher concentrations (100–250 μg/ml). 3 Another study showed significant cytotoxicity even used of the lower concentrations. 27 In our study, we observed a significant reduction in viability at doses between 10 and 200 μg/ml in amniotic fluid cells. These differences may be because of differences in vulnerability to NPs from one cell type to another. It is speculated that metabolic rate, antioxidant enzyme machinery, and DNA repair capabilities of each cell type may exhibit variability in TiO2 NPs-induced toxic effects on human health. Another reason for different levels of cytotoxicity shown in different types of cells is different crystalline forms of TiO2.
Anatase form of TiO2 NPs have been reported to be more cytotoxic. 14 A mixture of anatase and rutile forms of TiO2 NPs treatment according to the individual treatment has been found to lead to a higher level of oxidative DNA damage. 9 TiO2 NPs in the anatase form have the largest surface area than TiO2 NPs in the rutile form. In the present study, we used 100% anatase form of TiO2 NPs of the Milenium brand. This difference may be depended on the used TiO2 NP forms. Similar result has been reported by Saquib et al. in the cytotoxic effect of TiO2 NPs on amniotic cell line. 25 They reported a deceleration on cell division and a significant decrease in cell number in G2 and S phases of the cell, corresponding to reduction in mitotic index.
In our study, while there is a significant increase in cell death was observed in the transition from 24 h to 48 h, contrary to the expectations, cell death did not increase more on 96 h treatment. Bernardeschi et al. showed a similar finding in leukocytes of dolphin. 6 They interpreted that DNA repair activity would be promoted as a result of DNA damage in cells exposed to TiO2 NPs effectively. Similarly, we speculated this effect was caused due to the cell protection systems that manage them to stay alive. This response may be changed depending on exposure time and its dosage. We only investigated the effect of short-term (24 to 96 h) exposure to TiO2 NPs with high doses. TiO2 NPs have been exposed in a way during the period of embryonic life in the womb until their death.
A wide consumption of TiO2 NPs can pass to mother’s body via feeding, aspiration, and skin, and to fetus through the placenta. Most pregnant women are exposed to these materials, and the fetus is susceptible to poisoning with TiO2 NPs. In conclusion, the present and previous findings suggest that pregnant woman should refrain from the use of TiO2-containing materials to prevent any toxicicity induced by TiO2 NPs.
Footnotes
Acknowledgments
The authors gratefully thank Professor G. Bagcı, Faculty of Medicine, Pamukkale University, Denizli, Turkey, for supplying amniotic fluid samples. We also thank Professor M. Ersoz, Faculty of Science, Selcuk University, Turkey, for TiO2 NPs as a gift. We are grateful to Mehrad Mahmoudian at the University of Turku, Turku, Finland, for critical reading of manuscript. The authors thank Diagen A.S. (Ankara, Turkey) for supplying fluorescence in situ hybridization probe and counterstain.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
