Abstract
Objectives:
The study was aimed at evaluating, in vitro, cytotoxicity of four resin-based orthodontic cements (RBOC) as a function of degree of conversion (DC) and the light curing unit (LCU) employed on mouse fibroblast (L929).
Materials and Methods:
Nine samples were manufactured for each group of cements using plasma-emulating light-emitting diode (LED) and conventional LED. Toxicity was assessed by immersing four specimens to culture medium (24 h/37°C) for extracting residual monomer or cytotoxic substance. Cell mitochondrial activity of L929 cell was evaluated using methyl tetrazolium (MTT) test. DC was evaluated by Fourier transform infrared spectroscopy for five samples.
Results:
Cements, LCUs, and interaction between cements and LCUs were found to play a statistically significant role in cytotoxicity (p < 0.0001). Opal band cement (OPAL) plasma LED was found noncytotoxic (90–100% cell viability). The other RBOC–LCU combinations were slightly cytotoxic (60–90% cell viability). Cements (p < 0.01) and LCUs (p < 0.05) had a statistically significant effect on DC. Conversely, interaction between cement and LCU had no statistically significant role on DC (p > 0.05). OPAL plasma LED displayed the highest levels of DC. The correlations between cell viability and DC were positive for three RBOCs.
Conclusion:
Therefore, high-intensity LCUs can be said to efficiently affect polymerization, so higher DC rates may achieve higher cell viability rates.
Clinical Relevance:
Cements and LCUs must be matched to each another to result in higher DC and maximal biocompatibility. Dual cure systems presented relatively high cell survival and higher DC, thus expressing superior to single-cure systems with plasma LED.
Introduction
The foremost advantages of using resin-based orthodontic cements (RBOCs) are ease of use, light activation, high early bond strength, 1 longer working time for optimal bonding of bracket or band, and reduced micro-leakage in enamel–appliance interference. 2 RBOC is used for bonding brackets, bands, and acrylic orthopedic appliances because of its superior strength, chemical adhesion, ability to seal and fill gaps, and also a greater tolerance to moisture. 1 –3 As RBOCs are placed in close relation to and prolonged contact with adjacent soft or hard dental tissue, their toxic effect on local or systemic tissue is very important, especially when the oral hygiene of the patient is poor or gingival inflammation is noted during orthodontic treatment.
When the band is placed subgingivally or excess adhesive is present along the gingival margin or palatal mucosa after cementing the acrylic appliance, RBOC can release certain substances (bisphenol A diglycidylmethacrylate (Bis-GMA), triethylene glycol dimethacrylate (TEG–DMA), and hydroxyethyl methacrylate (HEMA)). These can be swallowed along with saliva or transported by blood penetrating into surrounding soft or hard tissue and therefore influence cells/organisms. 4 –6 Moreover, a deficiently polymerized RBOC layer under the bands or the acrylic appliances is highly permeable, and toxic substitutes from the adhesive material can penetrate dentin, cementum, and pulp as well as oral mucosa. 6
The polymerization reaction of RBOCs is triggered by excitation of photo initiator system with a light curing unit (LCU), which forms the polymeric matrix. 7 However, as metal bands or acrylic appliances interrupt the light path through the crown or dental arch, the polymerization capacity of RBOCs may be especially inadequate because less light can penetrate. 5,8 The degree of polymerization is a measure of the percentage of carbon double bonds (C=C) of matrix monomer that convert into carbon single bonds (C–C) during setting. 7 Inadequate degree of conversion (DC) affects physicomechanical properties, clinical performance, and safety of RBOCs. 4,6,9,10 The quality of polymer is also influenced by monomer type, filler size, photo initiator system, and polymerization conditions such as light source type, material depth, curing time, wavelength range, light intensity, emission spectrum, and power density. 3,7,11,12
Current photo initiator system of resin-based materials is commonly camphorquinone (CQ) that matches the spectral absorption being maximal at 460 nm. However, there are some photo co-initiators that are generally activated in the range of shorter wavelengths. Moreover, most manufacturers do not claim details about their cements containing photo initiators. It is important to choose suitable LCU for polymerization of resin on account of the small bandwidth of photo initiator. 13 This problem was solved by introducing poly-wave light-emitting diode (LED) LCUs emit in the violet (405 nm) and blue (470 nm) range of the light spectrum. The latest poly-wave LED LCU or a third-generation commercial LED LCU version called the VALO ortho has a maximum claimed irradiance of a remarkable 3200 mW/cm−2 and also has four emitters of different wavelengths in a square configuration. So far to our knowledge, no study has compared the effect of this LCU on cytotoxicity and DC of RBOCs.
In several studies, the correlation of cytotoxicity and DC was tested. Less cured materials were more cytotoxic than highly polymerized materials. 14,15 Moreover, clinically, the power density and exposure duration of the LCU have a crucial influence on DC and a lower residual monomer can be achieved using powerful light curing. 4,11
Hitherto, a few in vivo and ex vivo studies have focused on the cytotoxic influences of RBOC and components. 14 –16 However, investigations have not evaluated and correlated the effect of polymerization capacity with cytotoxicity using poly-wave LED and LED LCUs. The relative contributions of LCUs on polymerization indirectly influence uncured residual toxic monomers. 6 There is no suggestion to which combinations of LCUs and RBOCs result in the least toxic effects.
The present study investigates the possible cytotoxicity levels of different combination RBOCs and LED LCUs to evaluate and correlate the DC of these resins as a function of light curing to cytotoxicity. The null hypothesis assumed that the type of LCU does not affect the levels of cytotoxicity and DC of various RBOCs observed in the experimental protocols.
Materials and methods
Specimen preparation
A total of 72 disk-shaped samples of 4 different RBOCs (Table 1) were prepared in sterile Teflon molds of 5 mm diameter (depth 2 mm) under aseptic conditions in an ultraviolet sterilized laminar air flow chamber. Specimens were illuminated between mylor and glass slabs (2 mm) to minimize oxygen inhibition and maximize surface smoothness. The specimens were randomly divided into two groups: the specimens of first group were polymerized with plasma-emulating LED, and the specimens of the second group were polymerized with conventional LED. The technical information and the irradiation conditions are presented in Table 2.
Resin-based orthodontic cements (based on MSDS).

FOLC: Fuji Ortho LC; OPAL: Opal Band Cement; OBPLC: Ortho Band Paste LC; TBP: Transbond Plus; UDMA: urethane dimethacrylate; DUDMA: diurethane dimethacrylate; Bis-GMA: bisphenol A diglycidylmethacrylate; TEG–DMA: triethylene glycol dimethacrylate; MSDS: material safety data sheet.
Light curing units.

LED: light-emitting diode.
Cytotoxicity testing
L929 cells (ATCC-CCL-1, Şap Enstitüsü, Turkey) were cultivated in 10% fetal calf serum and 1% antibiotic antimycotic solution at 37°C and 95% air, 5% carbon dioxide (CO2). Cells of 15th–18th passage were used to decrease differences in cell cycle.
Four randomly selected specimens from each curing were immediately immersed in 7 mL of culture medium containing eight tubes for 24 h at 37°C to extract residual monomer. The sterile medium containing adhesive extracts was filtered to use on cell cultures.
A 96-well microplate was used to seed cells of the L929 culture at a density of approximately 10,000 per well for 24 h at 37°C and 5% CO2. After 24 h of incubation, the culture medium containing RBOC extracts of 200 µL replaced the cell layer in the well and the original culture medium served as a control. In this manner, eight parallel test batches were obtained from each RBOC–LCU combination, plus 12 control batches for 24 h at 37°C under 5% CO2.
Afterward, cell viability of L929 was evaluated using 3-(4,5- dimethyl-thiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) test, which is a reasonable representative of the activity of mitochondrial dehydrogenases in the cells and reflects both cell number and activity. Phosphate-buffered saline solution was used to rinse after cleaning the old medium, then 200 µL MTT solutions were added to each well, and finally the plate was incubated overnight at 37°C and 5% CO2. The yellowish extracellular MTT dye (tetrazolium salt) converted into the insoluble intracellular formazan by mitochondrial dehydrogenases. For analyses, 200 µL dimethyl sulfoxide was added and optical densities (OD) were measured with a spectrophotometer at 570 nm. The cell viability was calculated using the following formula:
L929 cell cytotoxicity was determined by cell viability relative to controls as: 17
no cytotoxic >90% cell viability;
slightly cytotoxic = 60–90% cell viability;
moderately cytotoxic = 30–59% cell viability;
severely cytotoxic ≤30% cell viability.
DC testing
Fourier transform infra-red spectroscopy (FTIR) spectrometer (Shimadzu, 8300-FTIR, Kyoto, Japan) was employed to analyze DC using potassium bromide (KBr) pellet transmission method. The remaining five specimens from each group were stored in light proof boxes to prevent further exposure to light for 24 h in artificial saliva at 37°C and then mechanically powdered with mortar and pestle. A standardized quantity of resin powder (5%) and infrared-grade KBr was manually mixed and pressed (5000 psi, 2 min) into a transparent pellet (12 mm in diameter). The FTIR spectrum of uncured cements, which were smeared onto KBr discs, was also recorded. Absorbance spectra were analyzed, using 4000 to 400 cm−1 wave number range, 30 co-added scans at 4 cm−1 in transmission.
Calculation of DC of RBOC begins with estimation of aliphatic C=C bond stretching vibrations of uncured material from absorption (A) peak occurring at 1638 cm−1. After polymerization, diminished concentration of aliphatic C=C is then redetermined from the diminished absorption peak. The carbon–oxygen (C=O) bond stretching vibrations are a convenient way taking the reference aromatic peak, which does not enter into reaction during curing at 1720 cm−1.
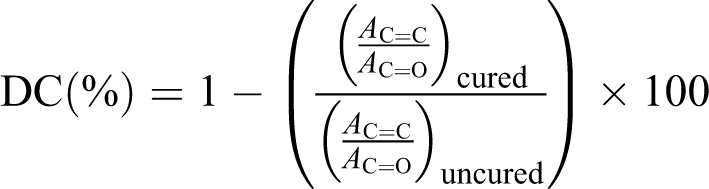
Statistical evaluation
The mean and standard deviation values were obtained (Table 3). A multivariate analysis of variance (MANOVA) was conducted to test the interaction between cell viability and DC in cements and LCUs. To clearly see the differences of source, two-way ANOVA was also carried out separately with Bonferroni post hoc test. Tukey’s honestly significant test was used for multiple comparisons. Cell viabilities in treated groups were compared with those in the untreated controls. The percentage values of DC and cell viability were correlated using Pearson correlation test. The level of significance was set at p < .05.
L929 cell viability of preincubated samples expressed as percentage of controls (cultures without specimens) and percentages of degree of conversions: Means (SDs).

FOLC: Fuji Ortho LC; OPAL: Opal Band Cement; OBPLC: Ortho Band Paste LC; TBP: Transbond Plus.
Results
Results of MANOVA show that cytotoxicity and DC of all four tested cements (p < 0.0001), LCUs (p < 0.05), and interaction between cements and LCUs (p < 0.01) were significantly different (Table 4).
Multivariate analysis of variance.

Cements, LCUs, and interaction between cements and LCUs were found to play a statistically significant role in cytotoxicity (p < 0.0001). OPAL-plasma LED was found noncytotoxic (90–100% cell viability; Table 5, Figure 1). The other RBOC–LCU combinations were slightly cytotoxic (60–90% cell viability).

Cell viability percentages of cements with different light sources.
Cell viability percentages of cements with different light sources in order from highest to lowest.

FOLC: Fuji Ortho LC; OPAL: Opal Band Cement; OBPLC: Ortho Band Paste LC; TBP: Transbond Plus; LED: light-emitting diode.
aMeans with the same letter were not significantly different at α = 0.05 level.
Cements (p < 0.01) and LCUs (p < 0.05) had a statistically significant effect on DC. Conversely, interaction between cement and LCU had no statistically significant role on DC (p > 0.05). OPAL-plasma LED displayed the highest levels of DC, and TBP-LED showed the lowest levels (Table 6, Figure 2).

Degree of conversion percentages of cements with different light sources.
Degree of conversion percentages of cements with different light sources in order to highest to lowest.
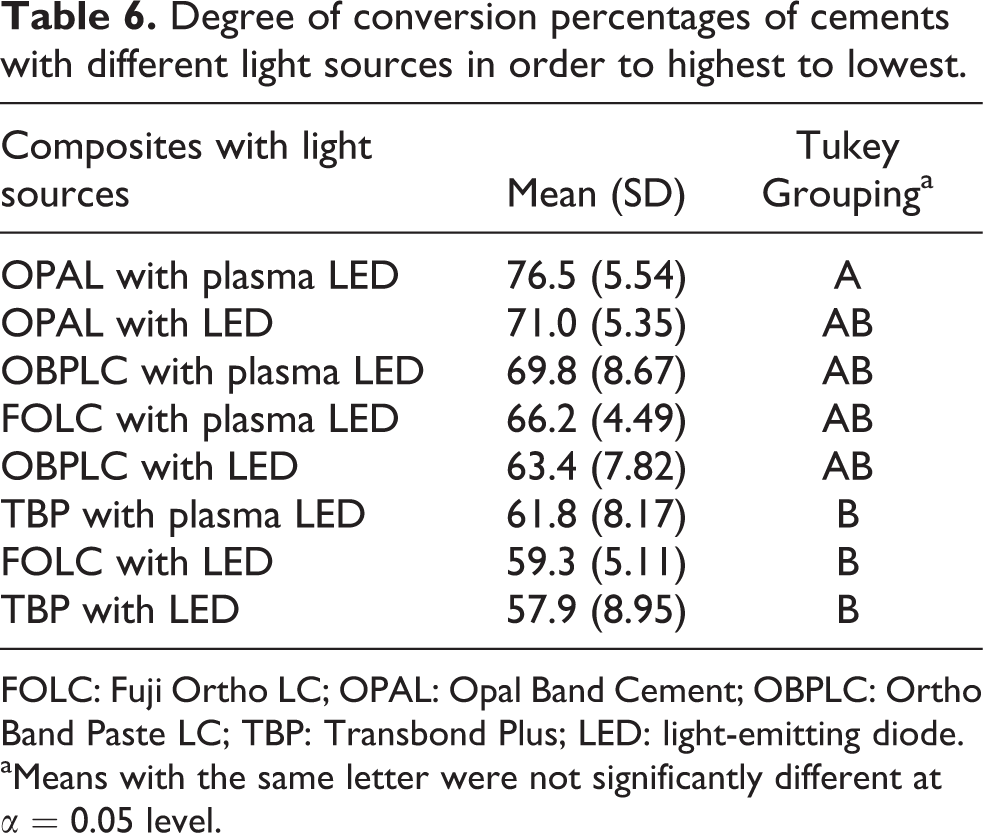
FOLC: Fuji Ortho LC; OPAL: Opal Band Cement; OBPLC: Ortho Band Paste LC; TBP: Transbond Plus; LED: light-emitting diode.
aMeans with the same letter were not significantly different at α = 0.05 level.
The correlations between cell viability and DC were positive for all the materials tested except OBPLC. The correlations of FOLC and OBPLC were statistically significant (p < 0.05; Table 7).
Pearson correlation between cell viability and degree of conversion expressed by cements.

FOLC: Fuji Ortho LC; OPAL: Opal Band Cement; OBPLC: Ortho Band Paste LC; TBP: Transbond Plus.
aStatistically significant positive correlation (p < .05).
Discussion
In the present investigation, we tested cytotoxicity and DC of four cements in two LCU combinations. Our results reflected that all tested cement–LCU combinations had statistically significant influences on cytotoxicity (Table 5). DC rates of cement–LCU combinations were also statistically different (Table 6). Therefore, the null hypothesis was rejected. Furthermore, there were insignificant positive correlations between cell viability and DC in all tested cements except OBPLC.
A review the orthodontic literature showed that researchers have not investigated and compared the effects of different high-intensity LCUs on cell viability and DC of RBOCs. Moreover, incomplete polymerization of RBOCs can produce toxic residual monomers that could cause cellular and metabolic alterations. 4,18 Cytotoxic level of RBOCs differs from little or no toxicity to severe toxicity according to the analytical protocol. In general model, samples are immersed in culture medium and extracts of photocured adhesive material leached in medium in a definite time period. Therefore, subpolymerized or polymerized adhesive material came into indirect contact with cells as single metabolic components. Eluates of photocured RBOCs have been tested under varying experimental protocols. Malkoc et al. 16 and Jagdish et al. 15 evaluated RBOCs with MTT on L929 and showed that eluates were cytotoxic. dos Santos et al. 14 tested extracts from photopolymerized RBOCs with the dye uptake technique on L929 cells and found that they were cytotoxic. The material, the cultivation time, and DC of samples had major influences on toxic effects. 14 –16 In the current study, cytotoxicity evaluation of RBOC–LCU combinations was performed using MTT assay with L929 cell (mouse fibroblasts), a frequently used cytotoxicity test for orthodontic materials. After using 24 h eluates on L929 cells, we found a significant increase in cytotoxicity rates produced by RBOC–LCU combinations (Table 5 and Figure 1). The highest or favorable cell survival rates were obtained by OPAL-plasma LED and FOLC-plasma LED. The other RBOC–LCU combinations showed higher cytotoxic rates.
A suboptimal curing level of monomers into the polymerized adhesive leads to a greater share of toxic substances that can segregate out of the material. 4,6 In the present study, we observed in the case of OBPLC and TBP a marked increase of cytotoxicity in two LCU combinations (Table 5 and Figures 1 and 2). This distinct reduction in cell survival suggests that the organic matrix of these two materials was different and also that these two cements, containing a greater amount of unreacted toxic monomers, leached out some substances at the surface in the culture medium (24 h). Furthermore, it is known that unbound surface components of adhesive samples can easily be released when the surface is left unpolished. 19 In this circumstance, RBOCs used under bonding brackets, bands or acrylic orthopedic appliances are also unpolished and thus can possibly interact there with the dental or oral tissue. 20,21
Tested RBOCs were different in some ways in this study in terms of shade, curing modes, preparation techniques, composition of the organic matrix, and the particle sizes, types, and volume shares of the inorganic matrix (Table 1). The setting mechanism of dual cured or tri cured RBOCs is achieved by acid–base reaction, photoactivated cross-linking of methacrylate to form matrix, and also by self-cure of adhesive monomer. It could be assumed that while dual cured or tri cured RBOC has the advantage of continuation of polymerization after light activation, OPAL and FOLC showed relatively high survival rates with plasma LED. That evidence is also supported by DC levels of these two RBOCs. In a previous study, FOLC showed higher cell survival and DC rates that were similar to the present study. 14 The consequences of cytotoxicity and DC evaluation implied that dual cure systems were superior to single cured systems, which were in contrast with the results of Jagdish et al. 15 Besides, plasma LED showed some favorable results in cell viability and DC levels when compared with LED.
Currently, plasma LED ‘VALO ortho’ is a third-generation LED LCU which has three emitters of different wavelengths incorporating a square configuration. The emitters mounted on curing tip to eliminate the light loss of a light guide. The claimed maximum irradiance of this LCU is 3200 mW/cm−2. Because four custom LED chipset of plasma LED can produce a large emission spectrum from violet (405 nm) to blue (470 nm), the large LED light spectra emission was ideal for photopolymerization of CQ and other photo initiators containing materials in the present study. These properties also give some advantages such as short exposure time (3 s), high power density, light output stability, and beam profile match with CQ and other photo initiators. High power density (3200 mW/cm−2) of this LCU should be assessed with caution, so rapid temperature changes can cause pain in sensitive teeth resulting patient discomfort. Based on the findings of this research, composites and LCUs must be matched to one another in order to obtain maximal biocompatibility and a satisfactory DC rates. Further studies should focus on assessing long-term toxic effects and DCs for orthodontic composites as well as different irradiation times or LCUs.
When using RBOCs with orthodontic appliances, there is a potential high risk for cytotoxicity and DC rates, because not only can thin oxygen inhibition layers can be easily produced but also photoactivation is very difficult under bulky or metal orthodontic appliances. The presence of oxygen can prevent complete conversion of monomer to polymer at the surface of the material, 6,7,15 thus residual toxic substances of RBOCs may cause cytotoxic effects in cells. Moreover, Bis-GMA, urethane dimethacrylate (UDMA), TEG–DMA, and HEMA are cytotoxic methacrylates employed in currently used RBOCs. 6,18,20,22 This toxicity could be provoked by hydrolysis of monomers to methacrylic acid, which has low molecular weight in oral cavity. 5,18,22 Methacrylic acid can easily affect local or systemic tissue via cytotoxicity.
Thus far, there is no assessment of cell and tissue responses or DC, of OPAL in the literature (Table 5). OPAL had no cytotoxicity and also the highest DC levels compared to the other RBOCs. It was probably assumed that the chemical interaction among the components, curing mode, and inorganic filler content of material were distinctly different from those tested cements. Anyhow, the thickness of oxygen inhibited layer of the surface is directly affected by viscosity of resin. 23 It means that lower viscosity results lower cell survival. Clinically, viscosity of OPAL was very high and this may have decreased cytotoxicity of OPAL.
Except for OBPLC cement, cell viability was positively correlated with DC. The correlation between FOLC and OBPLC was statistically significant (Table 6). This is probably because curing with plasma LCUs may have a decisive impact on DC which can in turn indirectly affect cell viability. Jagdish et al. 15 found a significant negative correlation in the results of FOLC. They used low-intensity LCU for polymerization that might have resulted in the lower levels of DC. Therefore, it can be suggested that, higher DC rates may accomplish higher cell viability rates. This study revealed that RBOCs and LCUs must be matched with one another in order to acquire maximal biocompatibility and higher DC rates. In future, in vivo and ex vivo research, into the long-term cytotoxicity and DC of orthodontic cements should be evaluated employing different irradiation times or LCUs.
Conclusions
All tested RBOC–LCU combinations showed a marked relationship between polymerization capacity and cytotoxicity.
All tested RBOC–LCU combinations decreased cell viability of L929 that may be indicative of potential cytotoxicity to dental tissue.
The cements and LCUs must be matched to one another to yield a higher DC and maximal biocompatibility.
Dual cure systems presented relatively high cell survival and higher DC, thus proving themselves superior to single-cure systems with plasma LED.
OPAL-plasma LED combination was the preferred choice for cell viability and DC.
Three cements—OPAL, FOLC and TBP—showed a positive correlation between cytotoxicity and DC. Therefore, high-intensity LCUs may efficiently affect polymerization; correspondingly higher DC rates may achieve higher cell viability rates.
Footnotes
Acknowledgments
We would like to thank Prof. Dr. Hamit Coşkun and Department of Chemistry, Faculty of Art and Science, Abant İzzet Baysal University for their support.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
