Abstract
Mercury, a heavy metal, is widespread and persistent in the environment and has been elucidated as a possible risk factor in cardiovascular diseases. Mercury has been reported to selectively impair the nitric oxide (NO) pathway in the vascular endothelium as a consequence of oxidative stress. Conversely, mercury per se causes endothelium-dependent vasorelaxation at lower concentration via the NO pathway. Little is known about the effects of mercury per se on other endothelial mediators. To elucidate possible mechanisms involved in this action, isometric tension was measured in aortic rings precontracted with phenylephrine (10 µM) from Wistar rats. Responses to increasing concentrations of inorganic mercuric chloride (10−12–10−5 M) were obtained in the presence and absence of endothelium. Inorganic mercury produced a biphasic response in endothelium-intact aortic rings and produced only vasoconstriction in endothelium-denuded aortic rings. To study the possible underlying mechanisms for the biphasic response of mercury, increasing concentrations of mercuric chloride (10−12–10−5 M) were used before and after NG-nitro-
Introduction
Vascular endothelium plays an important role in vascular homeostasis. It maintains a delicate balance between endothelium-derived smooth muscle relaxing factors and contracting factors to regulate the vascular tone and blood flow to tissues/organs of the body. 1 Any pollutant when enters the body reaches various organs through the circulatory system. Therefore, blood vessels are the primary site of exposure to the toxic effects of pollutants.
Mercury is a well-known environmental pollutant, and the role of mercury toxicity as a possible risk factor in cardiovascular disease has been discussed and documented by several workers. 2 –4 One of the most commonly observed cardiovascular effects of mercury exposure is loss of endothelial function. 5 –8 Mercury exposure reportedly causes oxidative stress leading to endothelial dysfunction and vasoconstriction. 7,8 Previous study from our group have reported endothelial dysfunction in blood vessels from chronically inorganic mercury-exposed rats due to oxidative stress resulting in the selective loss of nitric oxide (NO)-mediated relaxation 5 and persistence/upregulation of an endothelium-derived hyperpolarizing factor (EDHF) pathway. In endothelial dysfunction, there is either decrease in endothelial NO synthase (eNOS) activity and NO production or decreased bioavailability of NO due to oxidative stress. In vascular smooth muscle, NO stimulates production of cyclic guanosine monophosphate (cGMP), which promotes vasodilation by lowering intracellular calcium (Ca2 +) ion concentration and reducing the sensitivity of the contractile proteins to Ca2 +. 9 The treatment of endothelial cells with Ca2 + channel blocker, nifedipine increases the release of NO that is not due to an altered expression of eNOS messenger RNA (mRNA) and protein but because of an enhanced availability of NO. 10 Contrary to the above, Golpon et al. 6 have demonstrated that in the isolated aortic tissue, mercury induced an endothelial-dependent vasorelaxation and altered structure and function of vascular endothelial cells. Acute methyl mercury chloride exposure also modulates the vasorelaxant response by inducing NOS and increasing the NO production along with inducing oxidative stress. 11 From the above studies, it is conceivable to hypothesize that mercury exerts its effects on the NO-mediated relaxation, which in turn may contribute to the mercury-induced cardiovascular diseases in the populations exposed to mercury exposure.
Therefore, the aim of this study was to evaluate whether mercury, a redox inactive metal, per se affects the endothelial cells by involving NO, adenosine triphosphate-sensitive potassium channel (K+
ATP channel), reactive oxygen species (ROS), and
Methods
Wistar rats (n = 12), weighing 250–300 g, of either sex were used. The animals were maintained according to recommendations by National Accreditation Board of Testing and Calibration Laboratories (NABL), and the study was approved by the VP Chest Institute’s animal ethical committee. Rats were anesthetized with urethane (1.5 g/kg body weight intraperitoneally). The thoracic aorta was quickly removed, cleaned of adhering fat, and connective tissue. Ring segments (3–5 mm) were mounted between two stainless steel wires in 10 ml organ baths filled with modified Krebs solution, under passive tension of 2 g for 60 min. The Krebs solution had the following composition: sodium chloride (118 mM); potassium chloride (KCl; (4.7 mM); calcium chloride (2.5 mM); magnesium sulfate (1.2 mM); mono potassium phosphate (1.17 mM); sodium bicarbonate (20 mM); ethylenediaminetetraacetic acid (0.01 mM), and glucose. 12
The rings were maintained at 37°C and gassed with 95% oxygen and 5% carbon dioxide mixture (pH = 7.4). The contractile response (isometric tension, in grams) was measured by a force transducer (model FT-03, Grass Technologies, Warwick, Rhode Island, USA) and coupled to a data acquisition system (AD Instruments, Dunedin, New Zealand). Vessels were initially exposed to 75 mM KCl to check their functional integrity, and the presence of endothelium was confirmed by the ability of acetylcholine (ACh; 10 µM) to relax segments precontracted with phenylephrine (PE) at a concentration that produces close to 50% of the contraction induced by 75 mM KCl. Endothelium was removed by rubbing the lumen with forceps. Response to ACh (10 µM) was observed to confirm denudation (no vasorelaxation), which was validated by histology. 12 For each set of experiments described below four rats were used and three tissues from each animal were studied. There was equal distribution of female and male rats in each set.
Protocols
Effects of mercury on aortic rings
To evaluate the effects of mercury on the aortic rings, experiments were performed using rings with intact endothelium as well as denuded endothelium. After equilibration, aortic rings were precontracted with PE (10−7 M) and once the plateau was attained, concentration-dependent response curves of mercuric chloride (10−12–10−5 M) were obtained.
Effects of mercury on endothelial mediators
To evaluate the effects of mercury on the endothelial mediators: NO and EDHF, two different experimental designs were used.
Endothelium-intact aortic rings were preincubated separately with the NO synthase inhibitor NG-nitro-
Role of free radical scavengers on mercury-induced relaxation and contraction
To investigate the possible involvement of ROS in the vasorelaxing and vasoconstricting effects of mercury, endothelium-intact aortic rings were preincubated with the superoxide dismutase (SOD; 10 U/ml) + catalase (100 U/ml), and
Effect of l -type Ca2+ channel inhibitor on mercury-induced contraction
To investigate the possible involvement of
Statistical analysis
The responses produced by mercuric chloride are expressed in grams. Data are presented as mean ± standard error of the mean. Statistical evaluations were performed using Student’s unpaired t test and the value of p < 0.05 was considered significant.
Results
Effect of mercury on aortic rings tension development
Inorganic mercury (mercuric chloride) produced a biphasic response—a significant vasorelaxation with concentrations 10−7–10−6 M and vasoconstriction with concentration 10−5 M in endothelium-intact aortic rings of normal rats precontracted by PE (10−7 M; Figure 1).

Biphasic response of aortic rings precontracted with PE (10−7 M) to cumulative concentrations of mercuric chloride (10−12–10−5 M) in control rats. e+: endothelium intact (n = 12), e−: endothelium denuded (n = 12) n: number of tissues. Data represent mean ± SEM *p < 0.05: compared to baseline value; ¥p < 0.05: compared with the corresponding value before endothelium denudation. PE: phenylephrine.
Endothelial dependence of the vasorelaxant effects of mercury
Concentration–response curves for cumulative concentration of mercuric chloride (10−12–10−5 M) treatment (Figure 1) showed a biphasic response in endothelium-intact aortic rings. However, in endothelium-denuded aortic rings, mercury produced vasoconstriction with concentrations 10−7–10−5 M (Figure 1). These results suggest that the vasorelaxation produced by mercury is endothelium dependent and the vasoconstriction produced is by its effect upon smooth muscle activity. The endothelium-dependent vasorelaxation is a primary event and it masks the vasoconstrictor effect of mercury at lower concentration.
Effects of endothelial mediators in mercury-induced vasorelaxation
To investigate the role of NO or EDHF concentration–response curves for cumulative mercuric chloride (10−12–10−5 M) treatment in endothelium-intact aortic rings precontracted with PE (10−7 M) were performed before and after treatment with
These results were very similar to those obtained with endothelium-denuded preparations, suggesting that vasorelaxant actions of mercury methyl chloride affected by removal of endothelium were mediated by NO and EDHF (Figures 2 and 3).

Inhibition of vasorelaxant responses of aortic rings precontracted with PE (10−7 M) to cumulative concentrations of mercuric chloride (10−12–10−5 M) in control rats after incubation with
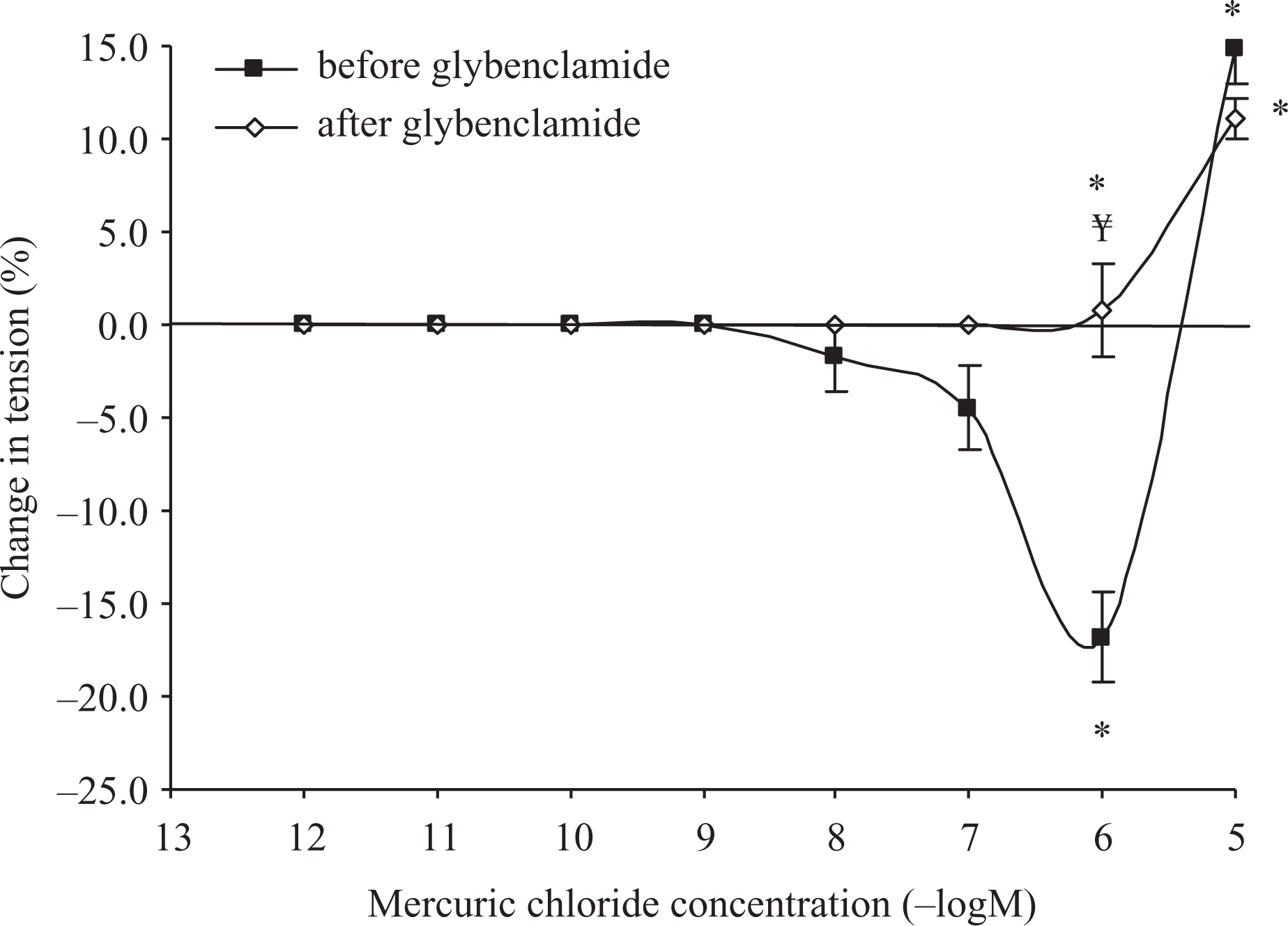
Inhibition of vasorelaxant response of aortic rings precontracted with PE (10−7 M) to cumulative concentrations of mercuric chloride (10−12–10−5 M) in control rats before incubation with glybenclamide (n = 12) and after incubation with glybenclamide (n = 12). n: number of tissues. Data represent mean ± SEM. *p < 0.05: compared to baseline value; ¥p < 0.05: compared to corresponding value before incubation with glybenclamide. PE: phenylephrine.
Effects of free radicals in mercury-induced vasorelaxation
Additional experiments were performed to elucidate the mechanisms behind the dual effects of mercury on the aortic vasculature. Vascular responsiveness to mercury was examined after incubation of endothelium-intact aortic rings with free radical scavengers SOD and catalase. Mercuric chloride, at concentrations 10−11–10−5 M, produced significant (p < 0.05) vasorelaxation (Figure 4) suggesting that mercury induces oxidative stress resulting in vasoconstriction. When aortic rings were incubated with free radical scavengers along with

Responses of aortic rings precontracted with PE (10−7 M) to cumulative concentrations of mercuric chloride (10−12–10−5 M) in control rats before incubation with SOD and catalase (n = 12) and after incubation with SOD and catalase (n = 12) and SOD + catalase along
Effect of Ca2+ channel inhibitor on mercury-induced contraction
Treatment with Ca2+ channel inhibitor, nefidipine abolished the vasoconstrictor response of mercury. There was a significant increase in the vasorelaxant response (p < 0.05) at concentrations (10−8–10−5 M) of mercuric chloride, though at 10 µM, there was a tendency for a reduction in the vasorelaxant response (Figure 5).

Inhibition of vasoconstrictor responses of aortic rings precontracted with PE (10−7 M) to cumulative concentrations of mercuric chloride (10−12–10−5 M) in control rats after incubation with nifedipine (n = 12). n: number of tissues. Data represent mean ± SEM. *p < 0.05: compared with the baseline value; ¥p < 0.05: compared with the corresponding values before incubation with nifedipine. PE: phenylephrine.
Discussion
In the present study, inorganic mercury (mercuric chloride) produced a biphasic response—a vasorelaxation at lower concentrations and vasoconstriction at higher concentration (10 µM) in isolated endothelium-intact aortic rings. However, in aortic rings whose endothelium was removed, mercury produced only vasoconstriction. These results suggest that the vasorelaxation produced by mercury is endothelium-dependent and the vasoconstriction produced is by its effect upon smooth muscle activity. Our results are consistent with previous studies that mercury at concentrations of 0.5–10 µM produces vasoconstriction. 7 Thus, we can say that endothelium-dependent vasorelaxation is a primary event and it camouflages the vasoconstrictor effect of mercury. A mercury level of 15 µg/l of blood is used as the biological exposure index, 13 therefore we used concentrations of mercury between 10−12 and 10−5 M for our experiments. The concentration of 10 µM of mercury approximately corresponds to 15 µg/l of mercury in blood. The fact that the endothelium is affected by mercury below the reference values highlights the importance and the need to better understand the mechanisms behind the dual effects of mercury on the aortic vasculature.
One of the most commonly observed vascular effects of mercury exposure is the loss of endothelial function.
5
–8
Endothelial dysfunction is attributed to reduction in NO bioavailability because of increased ROS production.
5,8,14
Previous studies from our group have demonstrated that chronic mercuric chloride exposure selectively impairs the NO pathway as a consequence of oxidative stress, while EDHF is able to maintain endothelium-dependent relaxation at a reduced level.
5
Although an increase in NO was observed in our study, reductions in NO bioavailability due to the increased ROS production was responsible for the endothelial dysfunction.
7
–9
In contrast, interestingly, previous study by Golpon et al. has reported that mercury in vitro induces an endothelium-dependent vasorelaxation, which is totally blocked by the NOS inhibitor
In the present study, the vasorelaxant response of mercury was abolished after incubation of aortic rings with
In the presence of free radical scavengers SOD and catalase, we observed augmentation of the vasorelaxant response of mercury and the abolishment of the vasoconstrictor response. The vasorelaxation response occurring even with as low a concentration as 10−12 M of mercury suggests that free radicals play an important role in this response. Numerous studies have shown that mercury induces oxidative stress with subsequent damage to several organs or systems
4,8,9,17
–19
and also reduces NO production and increases the inducible NO synthase gene expression.
20,21
Although multiple processes may lead to endothelial damage, the generation of oxygen-derived free radicals and subsequent lipid peroxidation may be key components in the cascade of events.
22
In the presence of mercury, the vascular endothelium releases NO that interacts with free radicals to form peroxynitrite, a potent vasoconstrictor and reduces the bioavailability of NO resulting in increased Ca2+ in the cytosol of the vascular smooth muscle and a decrease of vasorelaxation. SOD, and catalase scavenge the free radicals and make most of NO available to the vascular tissue. Hence, there is potentiation in the endothelium-dependent vasorelaxation response of mercury.
23
In the presence of free radical scavengers along with
When the aortic rings were treated with nifedipine, only the vasorelaxant response of mercury was observed suggesting that the vasoconstrictor response may be dependent on
The results reveal that mercury per se produces an endothelium-dependent vasorelaxation which is mediated through production of NO which phosphorylate K+
ATP channels. This endothelium-mediated vasorelaxation is potentiated in presence of free radical scavengers and
Footnotes
Funding
This work was funded by the Indian Council of Medical Research, New Delhi, India.
