Abstract
Chronic mercury exposure impairs vascular function, leading to the depression of endothelium-dependent vasodilatation. Loss of the nitric oxide (NO) pathway has been implicated, but little is known about effects on other endothelial mediators. This study investigated the mechanisms of endothelial dysfunction in rats subjected to chronic mercury chloride exposure. The endothelium-dependent relaxation of rat aorta evoked by acetylcholine (ACh) and isoproterenol was impaired in a dose-dependent manner by chronic mercury chloride exposure. Endothelium-independent responses to sodium nitroprusside (SNP) were not affected by chronic mercury chloride exposure. In healthy vessels, ACh-induced relaxation was inhibited by
Introduction
Mercury has been there on this earth from its very beginning, with it being used for medicinal (laxative, dental amalgam etc.) as well as commercial purposes (CFL bulbs, batteries etc.) for centuries and man has been exposed to it. There is vast amount of data indicating deleterious effects of mercury exposure on human health. Numerous studies have reported cardiovascular abnormalities like tachycardia, high blood pressure and heart palpitations after exposure to mercury. 1 Significant increases in systolic blood pressure and diastolic blood pressure have been found in volunteers with dental amalgam containing mercury. 2,3 Elevated body levels of mercury, due to fish consumption by humans, have been hypothesized as a risk factor in coronary heart disease. 4,5 Recently, cardiovascular abnormalities attributed to parasympathetic dysfunction have been reported in a community exposed to mercury living near to an industrial complex in Korea. 6 A growing mass of data now strongly indicates that damage to the blood vessel endothelium occurs following exposure to mercury, leading to a loss of endothelium-dependent vasodilation 7,8 due to induction of oxidative stress. 9,10 The exposure to mercury causes a decrease in the endothelial nitric oxide 11 which in turn leads to altered vasoreactivity. In contrast, a cohort study is suggestive that high blood mercury levels do not influence the vascular reactivity but increases the risk of hypertension. 12 Clarification of the effects of mercury exposure on blood vessels and study of mechanisms underlying may therefore help to identify therapeutic interventions that can improve the treatment of cardiovascular disease associated with mercury exposure.
The aim of this study was to determine the effect of mercury exposure and to study the mechanisms underlying that develops in rat aorta after chronic mercury chloride exposure. We were particularly interested to determine whether or not mercury exposure selectively inhibits the nitric oxide (NO)-mediated component of endothelium-dependent vasodilatation by involving free radicals and its effect on other endothelial mediators.
Materials and methods
The present study was conducted on adult, healthy Wistar rats of either sex weighing between 250 and 300 g. The animals were maintained according to the recommendations by National Accreditation Board of Testing and Calibration Laboratories (NABL) and the study was approved by the institute’s animal ethical committee. To study the effect of chronic inorganic mercury exposure on the aorta, the rats (n = 10) were treated with 1.25 × 10–6 M/L of mercuric chloride in drinking water 13 for 30 days and the experiments were performed the next day. The control rats (n = 10) were given the same drinking water without mercury for 30 days.
Biochemical studies
Total quantity of mercury in plasma of rats was measured through cold vapour atomic absorption spectrometry (CV–AAS) to observe whether 1.25 × 10−6 M/L of mercuric chloride in drinking water for 30 days was sufficient enough to cause a significant increase in blood mercury levels from baseline value.
Malondialdehyde (MDA) a stable product of lipid peroxidation, which is generally used as an index of free radical production, was determined by reaction with thiobarbituric acid (TBA). 14
Estimation of NO was done indirectly by measuring the serum levels of the products of NO metabolism, as recovery of nitrite and nitrate from serum. Nitrites and nitrates (NOx) were measured using the Greiss Reagent and the resultant purple azo derivative was measured spectrophotometrically at an absorbance of 540 nm. 15
Vasoreactivity studies
Experiments were performed on ring preparations (2–3 mm wide) of rat thoracic aorta, prepared with care in order to keep the endothelium intact.
16
Aortic rings were mounted isometrically under a resting tension of 2 g in an organ bath, between a stationary stainless steel hook and an isometric force transducer (Grass FT-03, USA) and changes in isometric tension were recorded by a Power Lab data–acquisition system (8SP 20B, AD Instruments, Australia) with a computerized analysis programme (Chart 5.4.2, AD Instruments). Vessels were maintained at 37°C in an organ bath containing 10 mL of modified Krebs-bicarbonate buffer solution of the following composition (in m
Statistical analysis
The relaxant response was expressed as decrease (in percentage) in steady state contraction.
Statistical software Graph pad prism and Microsoft Excel (version 2000, SR-I Professional) for windows were used for statistical analysis. Data were expressed as mean ± SEM. Student’s t-test was used for un-paired data wherever appropriate. The values were considered significant at p < 0.05.
Results
Mercuric chloride in drinking water (1.25 × 10–6 M/L) for 30 days was sufficient enough to cause a significant increase in blood mercury levels in rats from baseline value (Table 1). The serum malondialdehyde levels increased significantly in mercury-exposed group (Table 1). An increase in serum NO level was observed on chronic inorganic mercury exposure, but it was not statistically significant (Table 1).
Malondialdehyde (MDA), nitric oxide (NO) and mercury levels in control and chronic inorganic mercury-exposed group (n = 10)

Abbreviation: n: number of rats.
a p < 0.05, compared to control group.
The effect of mercury exposure on sensitivity and responsiveness of the vascular wall to ACh and isoproterenol
Acetylcholine evoked concentration-dependent relaxation of rat thoracic aorta precontracted with phenylephrine (10 µ

Comparison of responses of aortic rings with intact–endothelium precontracted with phenylephrine (PE) (10–5 M) to cumulative concentrations of acetylcholine (ACh; 10–12–10–4 M) between control and inorganic mercury exposure chronically, n = 12 (n = number of tissues). Data represent mean ± SEM. *p < 0.05, compared to corresponding value in the control group.
Additional confirmation for the impairment of endothelium-dependent vasorelaxation was obtained by observing the responses of aortic rings to isoproterenol. In the normal (control) rat, the relaxant response to isoproterenol was blunted by mechanical removal of endothelial cells, suggesting that the relaxant response to isoproterenol was mediated in part through endothelium. Figure 2 acompares the effect of isoproternol on thoracic aorta from unexposed and mercury-exposed rats. In healthy tissue, isoproterenol produced a maximum relaxation (R m) of 59.0% ± 7.3% and on aorta from mercury-exposed animals, it induced a smaller maximum relaxation (R m) of 32.6% ±4.7% (p < 0.05 vs control value). The isoproterenol response was similar in both endothelium-intact and endothelium-denuded aortic rings from mercury-exposed animals (Figure 2b).

a, Comparison of responses of aortic rings with intact–endothelium precontracted with phenylephrine (PE) (10–5 M) to cumulative concentrations of isoproterenol (10–12–10–4 M) between control and inorganic mercury (HgCl2) exposure chronically, n = 12 (n = number of tissues). Data represent mean ± SEM. *p < 0.05, compared to corresponding value in the control group. b, Responses of endothelium-intact and endothelium-denuded aortic rings precontracted with PE (10–5 M) to cumulative concentrations of isoproterenol (10–12–10–4 M) in inorganic mercury (HgCl2) exposure chronically, e+ endothelium–intact (n = 12), e– endothelium–denuded (n = 12); n = number of tissues. Data represent mean ± SEM.
The effects of mercury on vascular smooth muscle responsiveness
To study the effect of mercury on smooth muscle activity, precontracted thoracic aorta from both exposed and unexposed groups were treated with sodium nitroprusside (SNP). The SNP response was similar in both endothelium-intact and endothelium-denuded aortic rings from healthy and chronic inorganic mercury-exposed animals (Figure 3a and b).

a, Comparison of responses of aortic rings with intact–endothelium precontracted with phenylephrine (PE) (10–5 M) to cumulative concentrations of sodium nitroprusside (10–12–10–4 M) between control and inorganic mercury (HgCl2) exposure chronically, n = 12; n = number of tissues. Data represent mean ± SEM. b, Responses of endothelium-intact and endothelium-denuded aortic rings precontracted with PE (10–5 M) to cumulative concentrations of sodium nitroprusside (10–12–10–4 M) in inorganic mercury (HgCl2) exposure chronically; e+ endothelium–intact (n = 12), e– endothelium–denuded (n = 12); n = number of tissues. Data represent mean ± SEM.
The effects of mercury exposure on the EDRF/NO but not the EDHF-dependent component of ACh-induced vasorelaxation
To test whether altered release or action of endothelium-derived relaxing factors might contribute to the loss of responsiveness to ACh at 30 days post-mercury exposure, we tested the effects of agents that interfere with endothelium-dependent pathways on responses of vessels from healthy and mercury-exposed animals.
When L-NAME was added to inhibit the synthesis of NO, the responses of healthy vessels to ACh was reduced (13.6 ± 3.5%, p < 0.05) indicating the involvement of NO. In contrast, responses of vessels from chronically exposed mercury animals which were already depressed were unaffected by 100 µM L-NAME (26.3% ± 1.2% before and 16.3% ± 3.8% after adding 100 µM L-NAME; Figure 4).

Histogram comparing relaxations to 10 μM ACh in aortic rings from control animals (n = 12) and animals exposed to mercury (n = 12), in the absence of any drug and in the presence of 100 μM L-NAME. Relaxations are expressed as per cent decrease in the tension evoked by 10−5 M phenylephrine. *P < 0.05, compared with the response to ACh in the absence of drugs from control. **P < 0.05, compared with the response to ACh in the absence of drugs from mercury exposed.
To assess the potential contribution of endothelium-derived hyperpolarising factor (EDHF) to the ACh-induced relaxation and its modulation by mercury exposure, we replaced

Histogram comparing relaxations to 10 μM ACh in aortic rings from control animals (n = 12) and animals exposed to mercury (n = 12), in the absence of any drug and in the presence of 10 μM Glybenclamide. Relaxations are expressed as per cent decrease in the tension evoked by 10−5 M phenylephrine. *P < 0.05, compared with the response to ACh in the absence of drugs from control. **P < 0.05, compared with the response to ACh in the absence of drugs from mercury exposed.
To explore whether the free radicals produced interfere with endothelial function, concentration response curves of ACh were generated after incubating aortic tissues with antioxidant enzymes SOD and catalase. SOD plus catalase did not induce any change in the vasorelaxant response to ACh in normal animals (Figure 6). There was a significant increase in the relaxation response to ACh in chronically exposed animal (26.3% ± 1.2% before and 40.2% ± 1.3% after adding SOD and catalase, p < 0.05). In comparison, response of vessels exposed to mercury were unaffected by incubation with L-NAME along with SOD and catalase (26.3% ± 1.2% before and 28.4% ± 0.8% after adding L-NAME, SOD and catalase, n = 12).
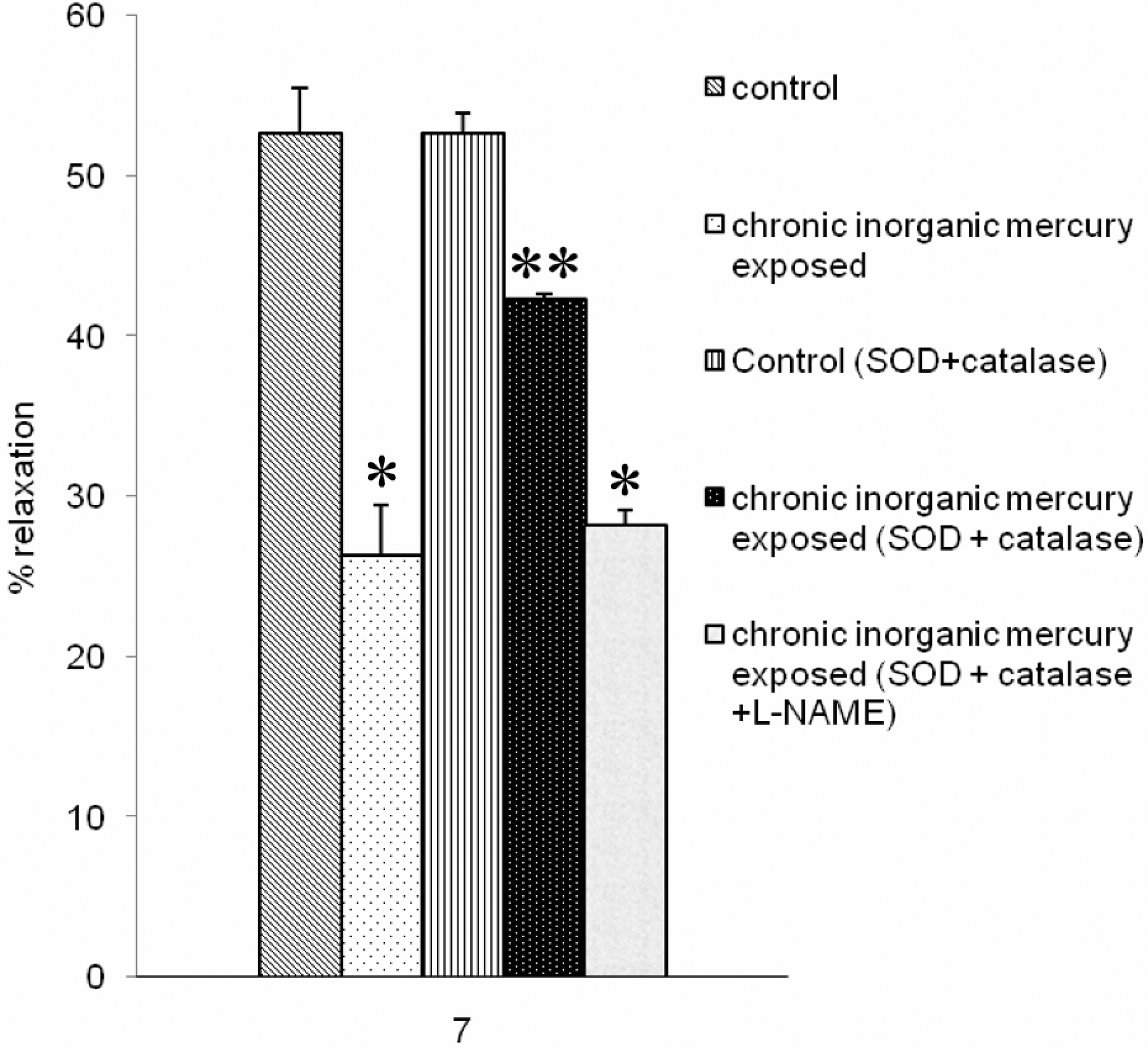
Histogram comparing relaxations to 10 μM ACh in aortic rings from control animals (n = 12) and animals exposed to mercury (n = 12), in the absence of any drug and in the presence of SOD and catalase, and SOD, catalase and L-NAME. Relaxations are expressed as per cent decrease in the tension evoked by 10−5 M phenylephrine. *P < 0.05, compared with the response to ACh in the absence of drugs from control. **P < 0.05, compared with the response to ACh in the absence of drugs from mercury exposed.
Discussion
Healthy Wistar rats exposed to inorganic mercuric chloride in drinking water (1.25 × 10–6 M/L) for 30 days showed an increase in the blood mercury levels with a significantly increased MDA and NO levels, suggesting that mercury exposure induces oxidative stress (Table 1). These findings are in agreement with a previous report in rats. 13 Even though there was an increase in the NO level on chronic inorganic mercury exposure in comparison to control, this increase was not statistically significant (Table 1). The failure to attain significance may be because along with NO, there was a significant increase in free radical production which may have interacted with NO and reduced its level. 17,18 By this process, the free radicals reduced the bioavailability of NO and reduced the endothelium-dependent relaxant response mediated by NO.
The loss of endothelial function is one of the most commonly observed vascular effects of oxidative stress.
19
In our study, chronic mercury exposure caused a loss of endothelium-dependent vasodilatation in response to ACh, at concentrations above 100 n
In healthy vessels, the relaxation evoked by ACh is mediated by a combination of NO and EDHF pathways. This was suggested by the finding that the relaxation induced by ACh is only partially inhibited by
In contrast to its effect in healthy vessels,
In conclusion, the data indicate that endothelial function is impaired in blood vessels from mercury-exposed rats as a consequence of the selective loss of NO-mediated relaxation without causing any structural changes in the vasculature. Following the loss of the NO pathway, endothelium-dependent vasodilatation is maintained at a reduced level due to the persistence/upregulation of an EDHF pathway that is resistant to mercury exposure. Contrary to endothelium-dependent responses, the endothelium-independent responses to NO donor are unaffected. Therefore, NO-mediated relaxation is likely to be more important in the development of the cardiovascular disorders seen in the populations exposed to mercury exposure.
Footnotes
This work was funded by the Indian Council of Medical Research, New Delhi.
