Abstract
The present study investigated the effect of rutin, a natural flavonoid, on the expression of caspase activation recruitment domain (CARD) and pyrin domain (PYD) of apoptosis-associated speck-like protein (ASC), a mediator of inflammation, in the pancreas of rats administered with ethanol (EtOH) and high-fat diet (HFD). Pancreatitis was induced in male albino Wistar rats by administering EtOH (8–12 g/kg/day) and HFD (22% fat) for 90 days. In addition, rats also received 100 mg rutin/kg body weight orally from 31st day till the experimental period. Serum levels of cytokines, interleukin 18 (IL-18) and IL-6; activity levels of caspase-1 and myeloperoxidase (MPO); messenger RNA (mRNA) expression of tumor necrosis factor α (TNF-α), caspase-1, CARD and PYD of ASC; and histological changes in pancreas were assessed. We observed a significant elevation in serum IL-18, IL-6, caspase-1 and MPO activities, mRNA expression of PYD, TNF-α and caspase-1 in the pancreas of rats administered with EtOH and HFD. Rutin administration along with EtOH and HFD significantly upregulated the mRNA expression of CARD and downregulated PYD, caspase-1, and TNF-α expressions. Rutin supplementation was also found to reduce IL-18 and IL-6 levels; and inflammatory changes in tissue architecture were evidenced by histological observations. The anti-inflammatory activity of rutin might be due to its effect on modulating the expression of ASC complex that mediates inflammation.
Introduction
The assembly of large protein complexes known as inflammasomes during apoptotic and inflammatory signaling involves protein interaction domains belonging to the death domain-fold superfamily of six helix bundles. 1 One of the domains, the adaptor protein, apoptosis associated-speck-like protein (ASC) containing a caspase activation recruitment domain (CARD) and pyrin domain (PYD) connects several pattern-recognition receptors (PRRs) such as the nucleotide-binding oligomerization domain-like receptor PYD containing 3 (NLRP3) with caspase-1. These PRRs recognize danger signals from injured cells (adenosine triphosphate and uric acid) and along with ASC, they effectively carry out the proteolytic maturation of caspase-1 that proteolytically cleaves pro interleukin (IL)-1β and pro IL-18 to make them active cytokines responsible for inflammation. 2
It has been shown that PYD–CARD-containing ASC activates procaspase-1 by ASC–CARD and CARD–caspase-1 interaction and oligomerization that results in proteolytic cleavage of procaspase-1. Furthermore, the isolated CARD of ASC was found to be a dominant regulatory inhibitor of IL-1β generation. 3
Caspases are synthesized as zymogens and activated through interaction with adaptor molecules that contain CARD. 4 Any drug that acts as an anti-inflammatory agent might interact with CARD to modulate caspase-1 activation and thereby to reduce cytokines formation.
Chronic pancreatitis is one of the major gastrointestinal diseases characterized by progressive inflammation in pancreas and severe abdominal pain. 5 Alcoholism is the most common etiologic factor for chronic pancreatitis 6 where the free radicals formed during ethanol (EtOH) metabolism initiate tissue injury. High-fat diet (HFD) along with EtOH causes hypogranulation and apoptosis of acinar cells that results in chronic pancreatitis. 7 The tissue injury and lesions have been characterized by fat necrosis, mononuclear cell infiltration, fibrosis, acinar atrophy, ductal dilatation, and intraductal mucinous or proteinaceous plugs. The incidences of focal acute pancreatitis have been reported to be increased with higher dietary fat content. Gallstone disease is the other cause of pancreatitis that blocks the release of pancreatic enzymes and autodigest the cells of the pancreas causing inflammation and injury. It is known that the components of inflammasomes, ASC and caspase-1 are involved in the development of pancreatitis and their antagonism could provide a new therapeutic strategy for the treatment.
The ever-increasing problem of pancreatitis demands the identification of new drugs. Herbal medicines and isolated compounds have stood best for their safety, efficacy, cultural acceptability, and lesser side effects. The flavonoid rutin is a flavanol glycoside comprised of the flavanol quercetin and the disaccharide rutinose. Rutin is richly present in black tea and apple skin peels. The richest source of rutin is the buckwheat plant Fagopyrum esculentum Moench and Emblica officinalis Gaertn. Chemically rutin is 5,7,3′,4′ tetrahydroxy flavonol-3-rhamnoglucoside and has been reported to exhibit anti-inflammatory and vasoactive properties. 8,9 Rutin is an important antioxidant and has been reported to be a potent scavenger of hydroxyl and superoxide radicals 10,11 and an inhibitor of lipid peroxidation. 12
The aim of the present study is to evaluate whether rutin acts as an anti-inflammatory agent by altering the gene level expression of CARD and PYD domains of ASC in rats subjected to experimental pancreatitis by EtOH and HFD ingestion.
Materials and methods
Chemicals and reagents
Rutin was obtained from Santa Cruz Biotechnology, Inc. (Canada). Enzyme-linked immunosorbent assay (ELISA) kits for IL-18, IL-6, and caspase-1 were purchased from Invitrogen (Camarillo, California, USA), Biosource International, (Camarillo, California, USA), and Genei (Bangalore, Karnataka, India), respectively. All other chemicals and reagents used were of analytical grade and purchased through the authorized dealers.
Rutin preparation
Rutin was dissolved in distilled water and mixed thoroughly. Freshly prepared rutin was administered orally by intragastric intubation.
Animal maintenance and groupings
Male albino rats of Wistar strain weighing about 175–200 g were purchased from King’s Institute (Guindy, Chennai, Tamil Nadu, India). The rats were housed in proper cages maintained with good sanitary conditions and under 12-h light and 12-h dark cycle at 22°C. After 3 days of acclimatization, the rats were segregated into 4 groups of equal number (n = 6). Groups 1 and 2 were given standard rat chow containing 5% fat. Group 3 and 4 rats were fed HFD (25% fat) along with EtOH for 90 days. The dose of EtOH was progressively increased to 8 g/kg/day (b.w.) for the first 4 weeks and to 12 g/kg/day for the remaining experimental period. The energy gained by the normal diet was 3.43 Kcal/g and by the HFD + EtOH was 5.24 Kcal/g. Hence, the carbohydrate content of the feed was isocalorically adjusted. In addition, group 2 rats were treated with 100 mg/kg b.w. of rutin and group 4 rats were administered with 100 mg/kg b.w. of rutin from 31st day till the experimental period.
This study was conducted according to the guidelines approved by the Institutional Animal Ethics Committee. At the end of the experimental period, the rats were subjected to ketamine hydrochloride (30 mg/kg b.w.) administration and killed by cervical decapitation; immediately, the blood samples were collected and the plasma/serum separated was stored at 4°C until analyses. In order to rule out the day-to-day variation in results, one animal from each group was killed per day. The following analyses were carried out to evaluate the pancreatic protective nature of rutin.
Biochemical investigations
Tissue homogenate preparation
Pancreas was removed immediately, carefully washed, and homogenized in 0.1 M Tris hydrochloric acid buffer at pH 7.4 and centrifuged at low speed to remove any cell debris. The supernatant was used for the determination of caspase-1 activity.
Determination of serum MPO activity
Myeloperoxidase (MPO) activity in the serum was estimated according to the modified method of Matheson et al. 13 and expressed in picomolar per liter.
Assay of IL-18
The assay was carried out as per the instruction in kit manual (KRC2341). The serum sample and standards were pipetted into antibody-immobilized wells. After incubation, biotinylated secondary antibody was added. After removal of excess secondary antibody, streptavidin–peroxidase was added. Then, the substrate solution was added to react with the bound enzyme to produce color, and the intensity was measured at 450 nm. The activity of IL-18 was expressed as picogram per milliliter.
Assay of IL-6
Serum immunoreactive IL-6 levels were also determined in duplicate by a rat-specific ELISA method with a detection limit of 30 pg/ml (KRC0061; Biosource International). Assay specificity was confirmed, and the percentage recoveries determined by spiking experimental serum samples with recombinant rat IL-6 was used as the standard. 14
Assay of caspase-1
Caspase-1 (EC: 3.4.22.36) activity was determined colorimetrically in serum or pancreatic extract, prepared according to the method of Thornberry and colleagues, 15 as the enzyme source. Briefly, the pancreas was homogenized in a lysis buffer (25 mM HEPES (pH 7.5), 1 mM ethylenediaminetetraacetic acid, 10 μg of aprotinin per ml, 10 μg of leupeptin per ml, 2 mM dithiothreitol) at 5 ml/100 mg of pancreas tissue. Extracts were centrifuged at 15,000g for 30 min at 4°C, and the supernatant was centrifuged again at 200,000g for 1 h at 4°C. The cytosol was used for caspase-1 activity measurements. The assay in undiluted serum or pancreas extract was performed as per the kit manufacturer instruction (ICT098). Reactions with enzyme preparation alone, with enzyme mixed with caspase-1 substrate (Ac-YVAD-pNA) or inhibitor (Ac-YVAD-CHO), and with substrate alone were also run as controls. The activity was measured by proteolytic cleavage of Ac-YVAD-pNA for 4 h at 37°C. The plates were read at 405 nm. A recombinant caspase-1 enzyme was used as a positive control.
mRNA expression of caspase-1, CARD, PYD, and TNF-α
RNA isolation
For extraction of total RNA from frozen (tissues, RNeasy) RNeasy Mini Kit (Qiagen, Hilden, Germany) was used according to the manufacturer’s instructions. First-strand complementary DNA (cDNA) was synthesized using oligo-dT primers. For real-time polymerase chain reaction (PCR) ABI PRISM Sequence Detection System 7700 (Applied Biosystems, CA, USA) was employed with Sybr Green (Cambrex Bioscience, Rockland, Maine, USA) as fluorescent and 6-carboxy-X-rhodamine (Molecular Probes, Eugene, Oregon, USA) as reference dye. The Sequence Detector 1.7 software (Applied Biosystems, CA, USA; www.appliedbiosystems.com) was applied to determine the threshold cycle (C t) values. To control the specificity of the amplification product, dissociation curves were analyzed for each experiment. The following primer sequences were used for real-time PCR analysis: Caspase-1 (forward 5′-CACAGCTCTGGAGATGGTGA-3′, reverse 5′-TCTTTCAAGCTTGGGCACTT-3′), ASC-CARD (NM-172322.1) (forward 5′-CAACACAGGCAAGCACTCAT-3′, reverse 5′-TGTTCCAGGTCTGTCACCAA-3′), ASC-PYD (NM-172322.1) (forward 5′-TGAAAACTTGACAGCGGATG-3′, reverse 5′-AGCTCCTGTATGCCGATGTC-3′), TNF-α (forward 5′-TGCCTCAGCCTCTTCTCATT-3′, reverse 5′-CCCATTTGGGAACTTCTCCT-3′), β-actin (forward 5′-GAGAAGATTTGGCACCACAC-3′, reverse 5′-CATCACAATGCCAGTGGTAC-3′). PCR conditions using heat-activatable Taq polymerase (Hot Master Taq, Eppendorf, Germany) were as follows: 40 cycles of 95°C for 20 s (denaturation), 55°C for 30 s (annealing), and 60°C for 30 s (extension). The cDNA samples were stored at −20°C and run on 1.5% agarose gel electrophoresis for confirmation.
Histological examination
A portion of pancreatic tissue was fixed overnight in 4% buffered neutral paraformaldehyde solution, embedded in paraffin, and deparaffinized by standard procedures. Ultrathin sections (5 µm) were stained with hematoxylin and eosin and observed under light microscope. All histological analyses were performed in randomly selected fields of sections by two investigators blinded to the identity of the treatment groups.
Statistical analyses
Data were analyzed using a commercially available statistics software package (Statistical Package for Social Sciences for Windows; version 10; SPSS, Chicago, Illinois, USA). The statistical significance of mean values between different groups was determined by applying one-way ANOVA with post hoc Bonferroni’s test; and the value of p < 0.05 was considered significant.
Results
The biochemical analyses revealed the following results:
Effect of rutin on serum MPO
Table 1 shows the levels of MPO activity in the serum of experimental animals. In EtOH + HFD-fed rats (group 3), the level of MPO was found to be increased in serum. Rats coadministered with rutin (group 4) showed decrease in the level of MPO activity.
Levels of serum IL-18, IL-6, and MPO activity in experimental animals.a

IL: interleukin; ANOVA: analysis of variance; HFD: high-fat diet; EtOH: ethanol; MPO: myeloperoxidase.
aData were analyzed by one-way ANOVA followed by post hoc Bonferroni’s test. Values are expressed as mean ± SD for n = 6 in each group.
b p = 0.000 for control versus EtOH + HFD, EtOH + HFD + rutin versus EtOH + HFD; p = 1.000 for control versus rutin control (not significant).
Effect of rutin on serum cytokines
The levels of serum IL-18 and IL-6 on experimental animals are shown in Table 1. Elevated levels of these serum cytokines are observed in EtOH + HFD-fed rats (group 3) and significant reduction was observed in rats coadministered with rutin (group 4).
Effect of rutin on CARD mRNA expression
Figure 1 shows the gene level expression of CARD in the pancreas of experimental animals. The expression level of CARD was found to be decreased in EtOH + HFD-fed rats (group 3) when compared with that of control rats that received normal diet. There was a significant increase in the mRNA expression of CARD in rats coadministered with rutin (group 4).

Alterations in mRNA expression of CARD in EtOH + HFD and rutin-treated rats. Data were analyzed by one-way ANOVA followed by post hoc Bonferroni’s test. Values are expressed as mean ± SD for n = 6 in each group. *p = 0.000 for control versus EtOH + HFD, EtOH + HFD + rutin versus EtOH + HFD and p = 1.000 for control versus rutin control (NS). mRNA: messenger RNA; CARD: caspase activation recruitment domain; EtOH: ethanol; ANOVA: analysis of variance; HFD: high-fat diet; NS: not significant.
Effect of rutin on mRNA PYD expression
The gene expression level of PYD in the pancreas of experimental animals is shown in Figure 2. Rats fed with EtOH + HFD showed elevated mRNA expression of PYD. Rutin-coadministered rats (group 4) showed significant decrease in the level of PYD expression.

mRNA expression of PYD in pancreas of experimental rats. Data were analyzed by one-way ANOVA followed by post hoc Bonferroni’s test. Values are expressed as mean ± SD for n = 6 in each group. *p = 0.000 for control versus EtOH + HFD, EtOH + HFD + rutin versus EtOH + HFD and p = 1.000 for control versus rutin control (NS). mRNA: messenger RNA; PYD: pyrin domain; EtOH: ethanol; ANOVA: analysis of variance; HFD: high-fat diet; NS: not significant.
Effect of rutin on TNF-α and caspase-1 mRNA expression
Figures 3 and 4 show the mRNA expression levels of TNF-α and caspase-1, respectively, in the pancreas of experimental animals. In EtOH + HFD-fed rats (group 3), the mRNA expression was found to be increased, whereas rats coadministered with rutin (group 4) showed a significant decrease in expression level.

mRNA expression of TNF-α in pancreas of experimental rats. Data were analyzed by one-way ANOVA followed by post hoc Bonferroni’s test. Values are expressed as mean ± SD for n = 6 in each group. *p = 0.000 for control versus EtOH + HFD, EtOH + HFD + rutin versus EtOH + HFD and p = 1.000 for control versus rutin control (NS). mRNA: messenger RNA; TNF-α: tumor necrosis factor α; EtOH: ethanol; ANOVA: analysis of variance; HFD: high-fat diet; NS: not significant.

mRNA expression of caspase-1 in pancreas of experimental rats. Data were analyzed by one-way ANOVA followed by post hoc Bonferroni’s test. Values are expressed as mean ± SD for n = 6 in each group. *p = 0.000 for control versus EtOH + HFD, EtOH + HFD + rutin versus EtOH + HFD and p = 1.000 for control versus rutin control (NS). mRNA: messenger RNA; ANOVA: analysis of variance; HFD: high-fat diet; EtOH: ethanol; NS: not significant.
Effect of rutin on caspase-1 activity
The serum and tissue caspase-1 activity level are shown in Table 2. The activity of serum caspase-1 was found to be increased in EtOH + HFD-fed rats (group 3). There was a significant decrease in the caspase-1 activity in rats coadministered with rutin (group 4). Rats received normal diet and rutin (group 2) showed no significant change in the activity of caspase-1 (p = 1.000). The activity of caspase-1 in pancreas was also significantly elevated in group 3 rats, and the levels were maintained in rats that received rutin (group 4).
Activity levels of caspase-1 in serum and pancreas of experimental animals.a

ANOVA: analysis of variance; HFD: high-fat diet; EtOH: ethanol.
aData were analyzed by one-way ANOVA followed by post hoc Bonferroni’s test. Values are expressed as mean ± SD for n = 6 in each group.
b p = 0.000 for control versus EtOH + HFD, EtOH + HFD + rutin versus EtOH + HFD; p = 1.000 for control versus rutin control (not significant).
Effect of rutin on the histology of pancreas
The histological changes in the pancreas of control and experimental rats are shown in Figure 5. The pancreas of control rats fed with normal diet and given no treatment showed normal tissue architecture with acini and islets of Langerhans (Figure 5(a)). Rats that received normal diet and rutin showed tissue architecture similar to that of control rats (Figure 5(b)). Rats that received EtOH + HFD showed steatosis, fatty deposition, and inflammation (Figure 5(c)), hemorrhage (Figure 5(d)) and rats fed with EtOH + HFD + rutin showed reduced inflammatory cell filtration, steatosis, and hemorrhage and preservation of pancreatic tissue architecture (Figure 5(e)).
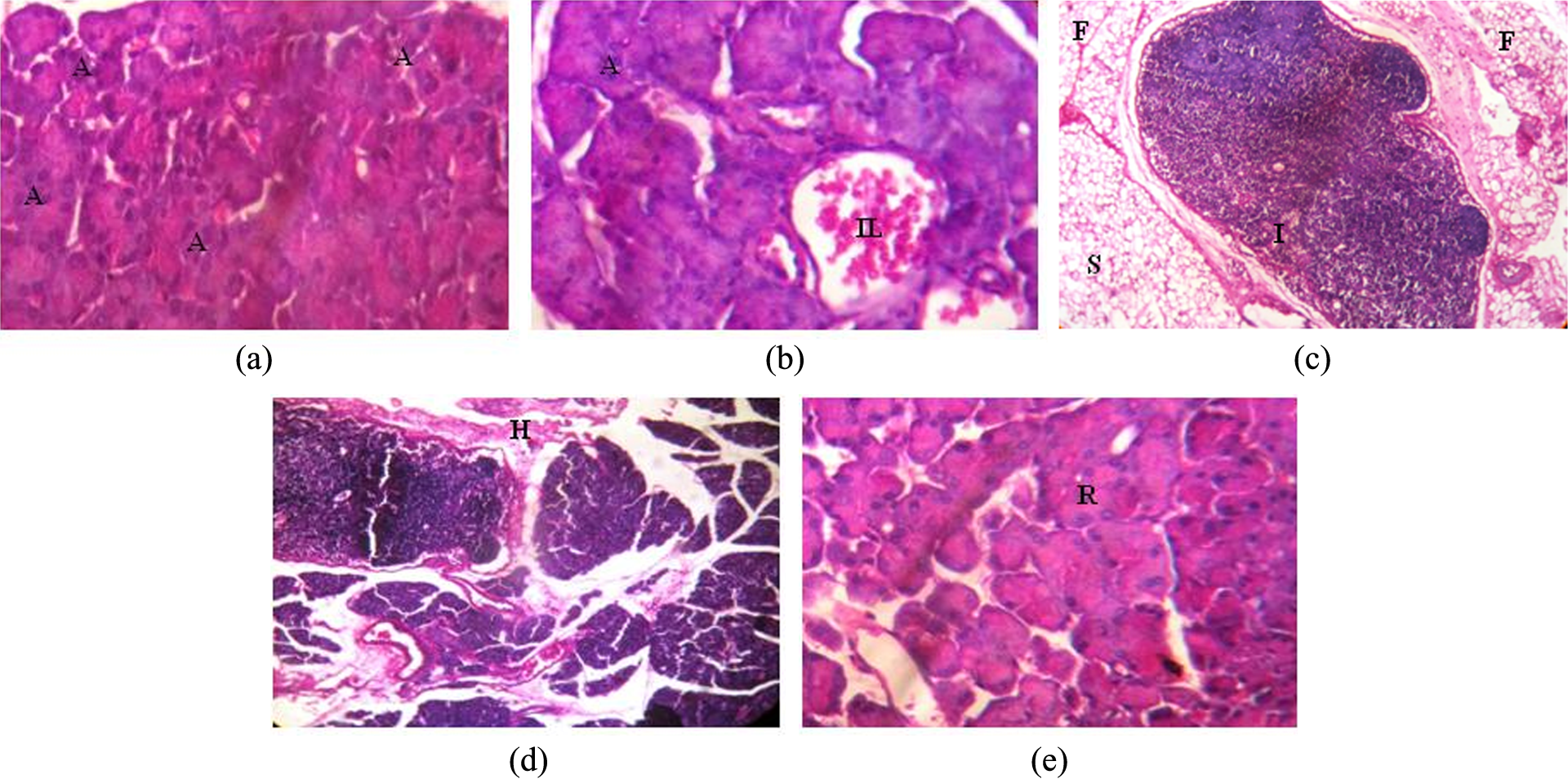
Histology of pancreas. (a) Photomicrograph of pancreas showing normal tissue architecture with acini (A) and islets of Langerhans of control rats fed with normal diet and given no treatment (H&E stain, ×40). (b) Photomicrograph of pancreas in rats fed with normal diet and rutin (100 mg/kg b.w.) showed tissue architecture similar to that of control rats (acini (A) and islets of Langerhans (IL); H&E stain, ×40). (c) Photomicrograph of pancreas in rats fed with EtOH and HFD showing steatosis (S), fatty deposition (F), and inflammation (IL; H&E stain, ×40). (d) Photomicrograph of pancreas in rats fed with EtOH and HFD showing hemorrhage (H; H&E stain, ×40). (e) Photomicrograph of pancreas in rats fed with EtOH and HFD and also supplemented with rutin showed reduced inflammatory cell infiltration, steatosis, hemorrhage, and preservation of pancreatic tissue architecture (tissue regeneration (R); H&E stain, ×40). H&E: hematoxylin and eosin; HFD: high-fat diet; EtOH: ethanol; b.w.: body weight.
Discussion
MPO is an inflammatory marker enzyme secreted at the site of injury to kill pathogens that may invade the injured tissue. The physiological substrate of MPO is hydrogen peroxide (H2O2), which is used to oxidize chloride, resulting in the formation of the proinflammatory oxidant, hypochlorous acid (HOCl). It requires heme as the cofactor and oxidizes tyrosine to tyrosyl radical using H2O2 as an oxidizing agent. 16 HOCl and tyrosyl radicals are cytotoxic that worsen the tissue injury. The activity level of serum MPO is elevated in EtOH + HFD-fed rats. This elevation in MPO activity may be attributed to detoxify the free radicals that cause tissue injury. Rutin-supplemented rats showed reduced activity of MPO in serum. The pancreatic protective activity of rutin is evidenced by the low level of serum MPO in rutin-coadministered rats.
Cytokines are pleotropical regulatory peptides synthesized by nucleated cells that produce both proinflammatory and anti-inflammatory cytokines. 17 One such cytokine is IL-18, which is expressed at the sites of chronic inflammation and in the context of numerous infectious diseases. 18 IL-18 appears to modulate inflammation at multiple checkpoints and has direct effects on target host tissue cells. The level of serum IL-18 was elevated in EtOH + HFD-fed rats that might worsen the tissue damage. The level of serum IL-18 was found to be significantly decreased in rutin-coadministered rats showing the anti-inflammatory nature of rutin.
IL-6 acts as proinflammatory or anti-inflammatory cytokine. IL-6 is secreted by T cells and macrophages to stimulate immune response, during infection and after trauma, especially burns or other tissue damage leading to inflammation. 19 IL-6 can be secreted by macrophages in response to specific microbial molecules referred to as pathogen-associated molecular patterns (PAMPs). These PAMPs bind to highly important group of detection molecules of the innate immune system called PRRs, including Toll-like receptors. These are present on the cell surface and intracellular compartments and induce intracellular signaling cascades that give rise to inflammatory cytokine production. 20 The activity level of serum IL-6 is elevated in EtOH + HFD-received rats and significant decrease in rutin-supplemented rats.
Caspase-1 is a cysteine protease that proteolytically cleaves other proteins such as the precursor forms of proinflammatory cytokines into active matured peptides. Caspase-1 itself needs activation by inflammasomes, a protein platform containing different domains such as CARD and PYD. 21 ASC is a specific procaspase-1 binding partner and its interaction with procaspase-1 induces activation and processing of pro IL-18 and pro IL-1β and secretion of active IL-18 and IL-1β. ASC uses its CARD to interact with the CARD of procaspase-1 and certain caspase-1 activators to regulate the activation of this protease and secretion of cytokines. Caspase-1 also triggers a form of cell death with characteristic necrosis and apoptosis that has been called pyroptosis. 22 In EtOH + HFD-fed rats, caspase-1 activity was found to be elevated significantly both in serum and pancreas showing the role of caspase-1 in inflammation. Coadministration of rutin was found to reduce the activity of the caspase-1 with the concomitant reduction in the levels of IL-18 and IL-1β.
ASC is a protein complex with PYD-CARD domain structure generated at the time of onset of inflammation in organs. The major role of ASC is to activate procaspase-1 to caspase-1 which in turn activates pro IL-1β and pro IL-18 for their maturation. ASC mRNA is encoded by three exons: exons 1 and 3 encode a PYD and CARD, respectively, and exon 2 encodes a proline- and glycine-rich (PGR) domain. The glycine present in PGR domain is known to confer flexibility to protein structures, 23 and α-helix conformational changes can be generated by a proline hinge in transmembrane proteins. 24,25 Thus, the PGR domain imparts a bending property to ASC and can be considered to be a hinge between the PYD and CARD domains that is folded in the inactive form thereby affecting the three-dimensional structure of ASC and hence playing a vital role in the activation of ASC. 26 The mRNA expression of ASC has been reported to be upregulated with the subsequent activation of caspase-1 and release of IL-1β has been reported in human skin inflammation, 27 spinal cord lesion 28 and respiratory syncytial virus infection. 29 The CARD of ASC exerts its dominant negative effect by interacting directly with the CARD of procaspase-1. CARD of caspase-1 has been shown to bind to ASC and its isolated CARD but not to PYD, indicating that the CARD of procaspase-1 interacts with ASC via CARD–CARD interactions, and it is a regulatory domain to limit the activation of procaspase-1 to avoid excess tissue injury. 30 In EtOH + HFD-fed rats, the mRNA expression of CARD was found to be decreased when compared with rutin-supplemented rats. Rats coadministered with rutin showed increased mRNA expression of CARD. The results of this investigation clearly show that CARD of ASC is upregulated by rutin to control the activation of procaspase-1.
Secondary structure analysis revealed that the PYD is a member of the six-helix bundle death domain-fold superfamily that includes death domains, death effector domains, and CARDs. 31,32 The PYD is found at the N terminal of the ASC suggesting that this domain might play a role in caspase-1 activation. 33 The PYD of ASC have been shown to increase IL-18 and IL-6 secretion. 34 This effect might be attributed to PYD–PYD interactions between ASC and procaspase-1, which may enhance oligomerization of ASC and activation of caspase-1. In EtOH + HFD-fed rats, the PYD mRNA expression was found to be significantly high. Rats appended with rutin showed decrease in the mRNA expression of PYD. The anti-inflammatory activity of rutin might be due to its effect on ASC complex by downregulating the PYD expression which is a key domain in activating procaspase-1. Although many scientific reports proved the upregulation of NLRP3-associated ASC, a study on the mRNA expression of various domains of ASC complex is limited, and our work is the first of its kind. Hence, the study on the mRNA expression of CARD and PYD domains of ASC is found essential to evaluate the mode of action of any new anti-inflammatory drug.
TNF-α recruits inflammatory cells and triggers the production of other cytokines, which initiate the healing process leading to fibrogenesis. TNF-α accelerates the synthesis of other cytokines and enhances neutrophil chemotaxis that leads to severe inflammatory response. 35 In EtOH + HFD-fed rats, the mRNA expression of TNF-α was found to be elevated significantly. Rutin-coadministered rats showed decrease in the expression of TNF-α. The anti-inflammatory activity of rutin might be due to the effect on TNF-α expression.
The present study shows that rutin coadministration in EtOH + HFD-fed rats significantly reduces the acinar cell damage, inflammation, and hemorrhage in pancreas. The histopathological evidences further confirm that rutin supplementation provide protection to the pancreas in rats fed with EtOH + HFD.
Conclusion
The results of the study revealed that rutin acts as an anti-inflammatory agent to prevent tissue damage in pancreas probably by influencing the upregulation of CARD and downregulation of PYD domains of ASC protein complex, which might have resulted in the reduced activation of caspase-1, cytokine production, and tissue injury. However, a study on the PGR domain of ASC complex is warranted to throw more light on the mechanism of action of rutin as an anti-inflammatory agent.
Footnotes
Acknowledgment
The authors acknowledge the help rendered by Dr A. Anbu, School of Pharmaceutical Sciences, Vel’s University, Chennai, Tamil Nadu, India, to obtain ethical approval for the study protocol.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
