Abstract
Introduction
Phthalates are plasticizers derived from phthalic acid (1,2-benzenedicarboxylic acid). Based on molecular weight, phthalates are classified into two groups including low molecular weight phthalates (di-n-butyl phthalate (DnBP), diethyl phthalate (DEP), and dimethyl phthalate (DMP)) and high molecular weight phthalates (di-(2- ethylhexyl) phthalate (DEHP), di-isononyl phthalate (DINP), and di-isodecyl phthalate (DIDP)). 1 Phthalates are frequently used in manufacturing of plastic materials such as polyvinyl chloride (PVC) to improve the flexibility and workability of polymers. 2 Phthalates have a short half-life in an outdoor environment from hours to days. The exposure to phthalates occurs through ingestion, inhalation, skin absorption, and intravenous administration. 1 These compounds are subjected to hydrolysis by esterases and lipases during the phase I of biotransformation. These hydrolytic monoesters: (I) enter the phase II of biotransformation to form glucuronide-conjugated monoesters excreted in the urine; (II) undergo phase I biotransformation to generate more polar products before glucuronidation; and (III) are excreted in the urine in the form of unconjugated (free) monoester and/or secondary metabolites. 3 Phthalates are well known endocrine-disrupting compounds (EDCs), which are classified as anti-androgens; they imitate natural hormones features and disrupt their cellular functions resulting in reproductive and behavioral disorders. 4 Among various types of phthalates, DEHP has a high male reproductive toxicity and acts as a suppressor of testosterone biosynthesis leading to the atrophy of seminiferous tubules and degeneration of testes. 5 After exposure to the DEHP, this compound is metabolized to MEHP and 2-ethylhexanol (2-EH) through hydrolytic enzymes of tissues including lipases and/or esterases. 6 The MEHP metabolite induces oxidative stress in testicular tissue, which in turn the mitochondrial dysfunction resulting in the apoptosis of spermatocytes. 7 The MEHP metabolite triggers early embryo loss by inhibiting trophoblast invasion. This effect results from its ability to the activation of proliferator-activated receptor (PPAR)/retinoid X receptor (RXR) pathway; this pathway induces oxidative stress-induced DNA damage leading to the inhibition of methyl-CpG binding proteins (MeCP) and subsequent changes in chromatin organization. 8 These findings suggest that antioxidant agents may be able to inhibit DEHP-induced reproductive toxicity. 9
Gallic acid (3,4,5-trihydroxybenzoic acid, GA) is a naturally occurring polyphenol in many natural sources, including mango, grapes, green tea, oak bark, and different types of berries.10,11 Bioactive phenolic compounds such as GA are a group of small molecules characterized by an aromatic ring with one or more hydroxyl groups. These compounds possess various pharmacological properties, including antioxidant, anti-inflammatory, and anti-carcinogen effects. 12 Gallic acid exerts antioxidants properties through scavenging reactive free radicals and increasing cellular antioxidant; this antioxidant activity of GA could protect various tissues against toxic agents-induced damage. 13 The regulatory effect of GA against inflammatory responses results from its ability to the inhibition of nuclear factor-κB (NF-κB) pathway; this effect leads to the down-regulation of the expression of pro-inflammatory cytokines including interleukin-1β (IL-1β), IL-6, IL-8, and tumor necrosis factor α (TNF-α). 13 Considering the important etiological roles of oxidative stress and inflammation in the DEHP-induced toxicity, GA may be a useful agent to inhibit DEHP-induced male reproductive toxicity. Current work was, therefore, carried out to evaluate the effects of different doses of GA (25, 50, and 100 mg/kg/day) on DEHP-induced oxidative stress and inflammation in the testicular tissue.
Materials and methods
Chemicals
Gallic acid (GA, (HO)3C6H2CO2H; Cas. Number: 149–91-7), di-(2-ethylhexyl) phthalate (DEHP, C24H38O4; Cas. Number: 117–81-7) and all the other reagents were obtained from Sigma-Aldrich Chemical Company (St. Louis, MO, U.S.A).
Animals and study design
After an initial acclimatization for one week, 35 healthy adult male NMRI mice (age 4–5 weeks), purchased from animal house, Ahvaz Jundishapur University of Medical Sciences, Iran, were used to this study. Mice were housed in groups of three to four/polycarbonate cages at 22 ± 2.0°C with a 12 h light:12 h dark cycle and free access to food and water. Animals were randomly divided into five groups including (I) normal saline and corn oil (5 mL/kg/day, p. o) treated group, (II) DEHP (2 g/kg/day, dissolved in corn oil, p. o.) treated group, and (III-V) DEHP (2 g/kg/day, dissolved in corn oil, p. o) and GA (25, 50 and 100 mg/kg/day, p. o.) treated groups; treatment duration was 14 days, as previously described14,15 and all agents and vehicles were administered through oral gavage. All experimental procedures were accepted by Institutional Animal Care and Use Committee of Ahvaz Jundishapur University of Medical Sciences (Ethic code: IR. AJUMS.ABHC.REC.1399.038).
Measurement of body and testes weights and sample collection
The measurement of mice body was done in the beginning and at the end of the experiment. On day 15 of the experiment, mice were anesthetized using ketamine and xylazine (100/10 mg/kg; i. p.) injection and blood samples were collected from the heart. The blood samples were centrifuged (10 min at 1000 × g) for separation of serum. Mice were then sacrificed by decapitation and both testes were removed immediately. The weight of testes was measured. The left testis was used to histological examination and the right testis was used to biochemical analysis.
Measurement of serum hormone levels
Mouse testosterone, luteinizing hormone (LH) and follicle-stimulating hormone (FSH) ELISA kits (MyBioSource Co, cat num: MBS843463, MBS041300 and MBS2507988, respectively) were used to determine the serum levels of testosterone, LH and FSH. The detection limits of the assay were 0.066 ng/mL, 0.15 mIU/ml and 1.406 mIU/ml for testosterone, LH and FSH, respectively. The detection range of the assay was 0.1–25.0 ng/mL, 0.3–60 mlU/ml, and 2.344–150 mIU/ml for testosterone, LH, and FSH, respectively.
The value of testosterone was expressed as ng/mL and the values of FSH and LH were expressed as mlU/ml.
Tissue biochemical parameters
For the assessment of biochemical parameters in the right testes, tissues were homogenized (1:10 w/v) in ice-cold Tris-HCl buffer (0.1 M, pH 7.4) and centrifuged (20 min at 4,000 x g) at 4°C. The obtained supernatants were collected and stored at −20°C. The total protein concentration of testicular tissue samples was determined using Bradford method.
Malondialdehyde, protein carbonylation, and nitric oxide assay
The MDA level in the testicular tissue was measured using MDA assay kit (Teb Pazhouhan Razi (TPR), Tehran, Iran) according to manufacturer’s protocol. Briefly, testicular tissue homogenates were mixed with butylated hydroxytoluene (BHT) to prevent further peroxidation. Samples were transferred to a tube containing chromogenic solution (thiobarbituric acid, alkali, and acetic acid) and detergent to form a pink color. The absorbance of developed color was measured at 532 nm using Synergy HT Microplate Reader (BioTek Instruments, Inc, Winooski, VT, USA). A standard curve of MDA was performed and MDA level was expressed as nmol/mg protein.
The protein oxidation in different groups was determined by measuring the level of protein-bound carbonyl as a biomarker for oxidative stress. 16 Carbonylated proteins are derivatized with 2, 4-dinitrophenylhydrazine (DNPH). The absorbance was measured at 375 nm and expressed as nmol/mg protein.
The level of NO in the samples was measured using the Griess method 16 by conversion of nitrate to nitrite using nitrate reductase (Griess diazotization reaction). A standard curve of sodium nitrite (NaNO2) was performed and the level of NO• was expressed as nmol/mg protein
Antioxidant enzymes activities
The activity of superoxide dismutase (SOD) and catalase (CAT) in the testicular tissue was measured using CAT and SOD assay kits (Teb Pazhouhan Razi (TPR), Tehran, IRAN) according to manufacturer’s protocol. The activity of SOD was assessed by the colorimetric method based on SOD ability to inhibit the reduction of tetrazolium salt by superoxide. The absorbance of developed color was measured at 450 nm and expressed as units of SOD/mg of protein (U/mg protein).
CAT activity was determined by measuring the generation of formaldehyde via the reaction of methanol and hydrogen peroxide (H2O2) in the presence of CAT. A standard curve of formaldehyde was performed and the absorbance was measured at 540 nm. The CAT activity was expressed as units of CAT/mg of protein (U/mg protein).
The activity of glutathione peroxidase (GPx) was assessed via the addition of NADPH, GR, and GSH to samples of testicular tissues. The mixture was incubated at room temperature for 15 min. The absorbance of obtained color was measured at 340 nm and the GPx activity was expressed as units of GPx/mg of protein (U/mg protein).
The level of glutathione and total antioxidant capacity
Testicular GSH level was measured by a method described previously. 11 Briefly, tissue samples were mixed with Ellman’s reagent (DTNB) in tris–EDTA buffer and incubated at room temperature for 20 min. The absorbance of obtained yellow color was measured at 412 nm. The GSH level was calculated using GSH standard curve and expressed as nmol/mg protein.
The total antioxidant capacity in the testicular tissue was measured using TAC assay kit (Teb Pazhouhan Razi (TPR), Tehran, Iran) according to manufacturer’s protocol. The level of TAC was determined by measuring the production of water-soluble chromogen following the oxidation of peroxidase chromogenic substrate by ferryl myoglobin radicals. The absorbance of obtained green color was measured at 734 nm and expressed as μg/mg protein.
The level of inflammatory cytokines
The level of inflammatory cytokines including tumor necrosis factor-α (TNF) and interleukin-1β (IL-1β) was measured in testes using mouse TNF-α ELISA kit and (Catalog Number: IB49688, IBL company) and mouse IL-1β ELISA kit (Catalog Number: IB99538, IBL company). The level of cytokines was expressed as pg of IL-1β or TNF-α/mg of protein.
Histological study
Johnsen scoring system histopathological examination of testicular tissue.

Sperm analysis
The right cauda epididymis was used for the evaluation of sperms. The cauda epididymis was minced and suspended in 1 mL of T6 medium containing 4 mg/mL bovine serum albumin (BSA) and incubated at 37°C and 5% CO2 for 1 h. The sperm suspension (5 μL) was observed with an optical microscope in a magnification 400 X and a total of 100 sperm was analyzed. Each spermatozoon was classified as motile and progressive sperms. The sperm vitality was evaluated using eosin B (0.5% in normal saline) staining by analyzing a total of 100 spermatozoa; spermatozoa were classified as motile living sperm, immotile living sperm, or dead sperm.
Statistical analysis
Statistical analysis was performed using Graph Pad Prism version 6 for Windows (Graph Pad Software, USA). Results were presented as mean ± standard deviations (SD). The normality test of the results was performed using Kolmogorov-Smirnov test and because the data distribution was normal, parametric tests were used. Individual differences between the groups were determined using one-way ANOVA followed by Tukey’s post hoc test and p < 0.05 was considered statistically significant.
Results
The impact of gallic acid on di-(2-ethylhexyl) phthalate-induced alterations in body and testes weights
The impact of GA on DEHP-induced alterations in body and testes weights.

The impact of gallic acid on di-(2-Ethylhexyl) phthalate-induced alterations in serum sex hormones
The serum level of testosterone significantly decreased and the serum LH and FSH level significantly increased in mice exposed with DEHP compared to the control group (p < 0.001, Figure 1). Treatment with GA in the dose of 100 mg/kg significantly restored DEHP-induced alterations in testosterone and FSH levels compared to the DEHP-treated group (p < 0.05 and p < 0.001, respectively). The doses of 50 and 100 mg/kg of GA inhibited DEHP-induced elevation of LH level (p < 0.05 and p < 0.01, respectively). The impact of GA on serum testosterone, LH and FSH levels in mice exposed to DEHP. Values are means ± SD. (n = 7). ***p < 0.001, **p < 0.01 and *p < 0.05 as compared to control group. ###p < 0.001, ##p < 0.01 and #p < 0.05 as compared to DEHP group.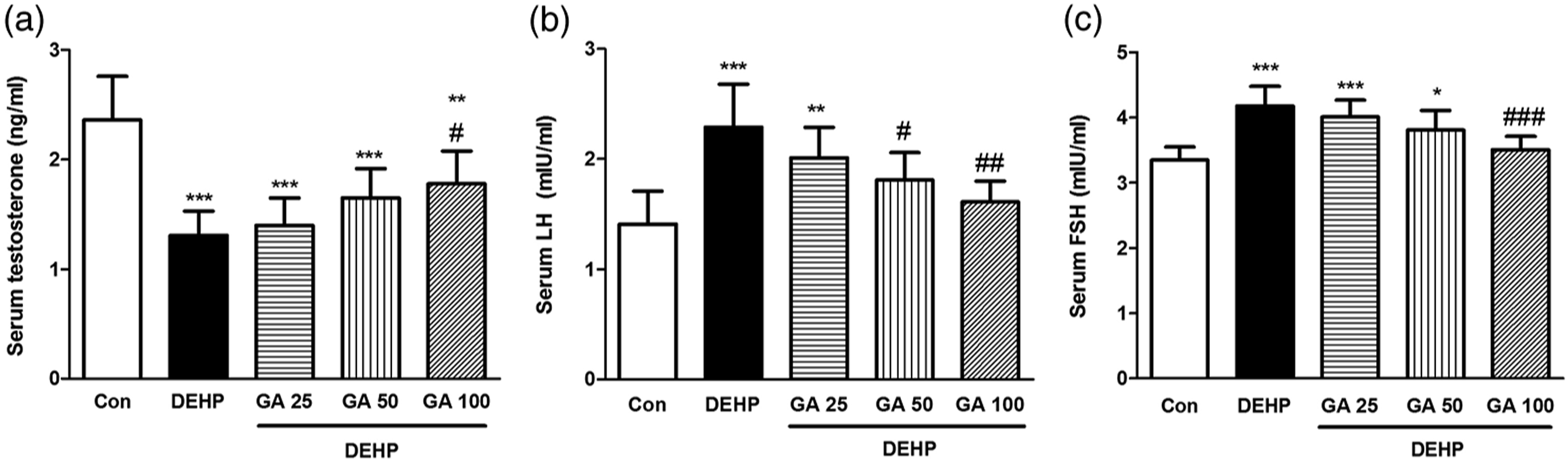
The impact of gallic acid on di-(2-Ethylhexyl) phthalate-induced oxidative stress in testes
The level of MDA (p < 0.001), PC (p < 0.01) and NO· (p < 0.001) significantly increased in DEHP-treated mice in comparison with the control group. Administration of GA inhibited DEHP-induced elevation of MDA in doses of 50 and 100 mg/kg (p < 0.001), number in doses of 25 (p < 0.05), 50 (p < 0.001) and 100 mg/kg (p < 0.001), and PC in the dose of 100 mg/kg (p < 0.05, Figure 2). The impact of GA on MDA, PC, NO and TAC levels in testes of mice exposed to DEHP. Values are means ± SD. (n = 7). ***p < 0.001, **p < 0.01 and *p < 0.05 as compared to control group. ###p < 0.001 and #p < 0.05 as compared to DEHP group.
The activity of antioxidant enzymes (SOD, CAT, and GPx) and the content of GSH and TAC significantly reduced in testicular tissues of mice exposed to DEHP compared to the control animals (p < 0.001). Treatment with GA significantly inhibited DEHP-induced reduction in the activity of SOD (50 and 100 mg/kg; p < 0.001), GPx (50 mg/kg; p < 0.05 and 100 mg/kg; p < 0.001) and CAT (50 and 100 mg/kg; p < 0.001) in the testicular tissue (Figure 3). Furthermore, GA significantly inhibited DEHP-induced reduction in the level of GSH (25 mg/kg; p < 0.01, and 50 and 100 mg/kg; p < 0.001) and TAC in the testicular tissue (50 mg/kg; p < 0.05 and 100 mg/kg; p < 0.001, Figure 2). The impact of GA on GSH level and GPx, SOD and CAT activity in testes of mice exposed to DEHP. Values are means ± SD. (n = 7). ***p < 0.001, **p < 0.01 and *p < 0.05 as compared to control group. ###p < 0.001, ##p < 0.01 and #p < 0.05 as compared to DEHP group.
The impact of gallic acid on di-(2-ethylhexyl) phthalate-induced inflammatory responses
The expression of pro-inflammatory cytokines including TNF-α and IL-1β significantly increased in mice exposed to DEHP compared to the control group (p < 0.001). Treatment with GA could significantly inhibit DEHP-induced elevation level of IL-1β (50 mg/kg; p < 0.05) and TNF-α (50 and 100 mg/kg; p < 0.001) in comparison with the DEHP group (Figure 4). The impact of GA on TNF-α and IL-1β levels in testes of mice exposed to DEHP. Values are means ± SD. (n = 7). ***p < 0.001 as compared to control group. ###p < 0.001 and #p < 0.05 as compared to DEHP group.
The impact of gallic acid on di-(2-ethylhexyl) phthalate-induced sperm and testicular damages
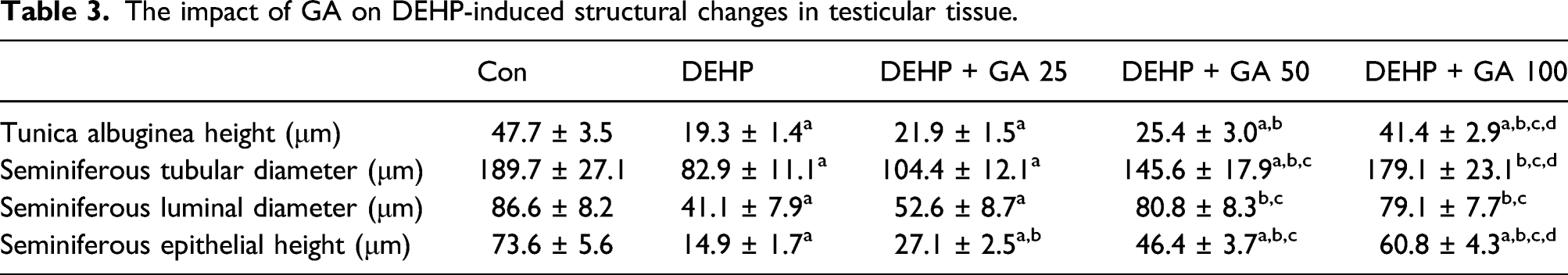
The impact of GA on DEHP-induced structural changes in testicular tissue.

Cross section of testicular tubules showing effects of GA on DEHP-induced testicular toxicity. (A) Control group (B) DEHP group. (C) DEHP+GA (25 mg/kg/day) group. (D) DEHP+GA (50 mg/kg/day) group. (E) DEHP+GA (100 mg/kg/day) group. White arrows show the desquamation of epithelial germ cells of seminiferous tubules, black arrows show hyperemia and yellow arrows show sever congestion of intertubular blood vessels in testicular tissue. Seminiferous epithelial; SE, Intertubular space; In and seminiferous lumen; L.

Johnsen’s score in testicular tissue. Values are means ± SD. (n = 7). ***p < 0.001 as compared to control group. ###p < 0.001 as compared to DEHP group.
The impact of gallic acid on the motility and viability of sperm in vitro
The impact of GA on the motility and viability of sperm in vitro.

Discussion
This investigation was assigned to evaluate the protective effects of GA (25, 50, and 100 mg/kg/day) on DEHP (2 g/kg/day)-induced testicular toxicity in adult male mice. Results indicated that two-weeks exposure of mice to DEHP decreased body and testicular weights, increased oxidative stress and inflammation, altered serum testosterone, FSH and LH concentrations, inhibited spermatogenesis, damaged testicular structure, and reduced the quality of sperms characterized by reducing sperm vitality and sperm progressive motility. The DEHP-induced reduction of testicular weight results from a decreased level of testosterone, possibly as a result of that the degeneration of Leydig cells, testosterone-secreting cells localized in the inter-tubular spaces of the testes. 18 Reduced level of testosterone results in the increased secretion of LH and FSH, as positive feedback.19, 20
Seminiferous tubules contain two distinct populations of cells, including spermatogenic cells and Sertoli cells. Spermatogenic cells develop into spermatozoa, and Sertoli cells have supportive and nutrient function. 21 Therefore, DEHP-induced reduction of spermatogenesis and sperm quality results from damage to germ cells and Sertoli cells. Obtained results from this study indicated that DEHP increased MDA, NO˙, and PC levels and decreased the content of GSH and TAC and the activity of antioxidant enzymes including SOD, CAT, and GPx. The MEHP metabolite of DEHP is responsible for the induction of testicular damage through inducing oxidative stress in these cells; 7 the effect of MEHP on Leydig and Sertoli cells contributes to the reduction of testosterone generation and impairment of spermatogenesis. Oxidative stress-induced by DEHP impairs mitochondrial function in testicular cells; this event leads to the excessive generation of ROS/RNS and activation of mitochondrial apoptotic pathways resulting in the induction of testis atrophy. 22 The impairment of ATP generation, resulting from the inhibition of oxidative phosphorylation (OXPHOS) complex II, III, IV, and V subunits expressions, might be a reason for the DEHP-induced reduction of sperm motility. 23
Results from this study demonstrated that administration of GA could prevent DEHP-induced oxidative stress in testicular tissue, which is characterized by reducing MDA, NO˙ and PC levels and elevating GSH and TAC levels and SOD, CAT, and GPx activities. The GA effect on the reduction of DEHP-induced oxidative stress was associated with the improvement of serum hormones levels, sperm motility, germinal cells numbers, and testicular structure. These suggest that GA may inhibit DEHP-induced degeneration of germinal, Leydig and Sertoli cells through suppressing oxidative stress in testicular tissue. These findings agree with previous studies indicating the protective effect of GA on oxidative stress-induced testes damages, which results from its direct antioxidant activity 24 or its stimulatory effect on antioxidant enzymes activities. 24 In Bisphenol A-induced testicular injury, GA enhances male reproductive capacity through inhibiting bisphenol A-induced elevation of ROS generation and reducing antioxidant enzymes activities; this results in the improvement of gonadosomatic index and serum testosterone and prolactin levels. 24 Gallic acid reverses cyclophosphamide-induced elevation of MDA, nitrite, and hydrogen peroxide levels and reduction of anti-oxidant enzymes activities in testes and epididymis of rats; these effects are associated with the improvement of serum hormones levels and sperm motility and viability. 25 Administration of GA inhibits DnBP-induced alteration in testicular structure and germ cell count, which is associated with the improvement in the testis antioxidant profiles. 26 Gallic acid improves mitochondrial function through inhibiting the generation of mitochondrial ROS, peroxidation of mitochondrial membrane lipids, and improvement of mitochondrial antioxidant defense. Therefore, GA could inhibit activation of mitochondrial apoptosis in normal cells; 27 this suggests that GA may increase ATP production in DEHP-exposed testes, which subsequently leads to the improvement of sperm motility and viability.
Results from our study revealed that DEHP increased the expression of pro-inflammatory cytokines, including IL-1β and TNF-α in the testicular tissue. The stimulatory effect of DEHP on inflammatory responses results from the excessive generation of ROS/RNS, inducing nuclear translocation of nuclear factor (NF)-κB, which up-regulates the expression of pro-inflammatory cytokines, including IL-1β, IL-6, IL-8, and TNF-α.28,29 Inflammation has destructive effects on spermatogenesis and leads to the male infertility. 30 Exposure to the low doses of DEHP is reported to be sufficient to induce inflammation in testicular tissue (orchitis). 31 Treatment with GA could remarkably reduce inflammatory responses in the testicular tissues of mice exposed to DEHP through down-regulating the expression of TNF-α and IL-1β. Gallic acid inhibits inflammatory processes through suppression of NF-κB pathway leading to the down-regulation of the expression of pro-inflammatory cytokines, including cyclooxygenase-2 (COX-2), inducible nitric oxide synthase (iNOS), IL-1β, IL-6, IL-8, and TNF-α.13,32 In streptozocin-induced diabetic rats, GA reduces inflammatory responses which are associated with the reduction of TNF-α and iNOS levels in testicular tissue; this effect is suggested to be mediated by NF-kB inhibition and reactive radical scavenger properties of GA. 33
Conclusions
The results of this study showed that GA ameliorated DEHP-induced testicular tissue damage in adult mice, especially in doses of 50 and 100 mg/kg/day. Administration of GA inhibited DEHP-induced oxidative stress and inflammatory responses; these effects were associated with the improvement of serum hormones levels and sperm motility and viability. Therefore, GA might be benefit in the reproductive health protection against endocrine disrupting chemicals such as DEHP through reducing oxidative stress and inflammatory responses.
Footnotes
Author contributions
Conception and design: MG
Performing the literature search, data collection and analysis: AH, SM, AS, ZB and NB
Drafting the article: all authors
Approving the final version: all authors
MG: is responsible for the integrity of the work as a whole.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran (Grant number: MPRC-9919).
