Abstract
Atherosclerosis is morphologically an inflammatory disease, where endothelial dysfunction plays a key role in all the stages. The nitric oxide (NO) synthase 3 (NOS3) gene is responsible for the synthesis of endothelial NO synthase (eNOS) in humans and some genetic polymorphisms are considered “polymorphisms associated with risk” for the development of coronary artery diseases, such as acute coronary syndrome. Thus, the present study aimed to evaluate the influence of the -786T>C polymorphism of the eNOS gene on inflammatory and oxidative process. A prospective cohort study of 125 consecutive patients with clinical diagnosis of non-ST-elevation acute coronary syndromes was conducted. Patients were assessed using a standardized questionnaire. Blood samples were drawn to measure serum levels of high-sensitivity C-reactive protein, soluble CD40 ligand, interleukin-6 (IL-6), N-terminal prohormone of brain natriuretic peptide, immunoglobulin G antibodies against oxidized low-density lipoprotein. The genotypes for the -786T>C polymorphism in the 5′-flanking region of eNOS gene were determined. The -786C allele was found in 92 of 250 alleles (38.8%). No statistical association was observed between demographic and clinical characteristics and distribution of eNOS-786T>C polymorphism. We found that -786CC was associated with lower levels of IL-6. No significant differences were observed between the distribution of -786T>C polymorphism and other investigated markers.
Introduction
Coronary artery disease (CAD) is acknowledged as cardiac manifestation of generalized systemic disease, the atherosclerosis (disease that affects large and medium-sized arteries at different vascular sites). 1 Acute coronary syndrome (ACS) involves a group of signs and clinical symptoms that reflect different degrees of acute myocardial ischemia. ACS should be understood as a moment of instability of the atherosclerotic plaque, that establishes an underlying thrombotic process, with subsequent risk of total or subtotal occlusion of the coronary arteries. 2 This fact determines propitious conditions of acute myocardial infarction (AMI) that can be fatal or nonfatal, making these conditions unfavorable in clinical results of great importance. 2,3 It has shown that inflammation is an important part in the ACS development. Atherosclerosis is morphologically an inflammatory disease, and histological findings demonstrate that rupture sites of coronary atherosclerotic plaques are characterized by the presence of activated macrophages and smooth muscle cells. 4,5 The endothelium covers all the vessels, and its integrity contributes to the maintenance of adequate blood flow to body cells and tissues, and in any situation of disruption in the continuity of their cells, they reorganize themselves to remake the intercellular connection. 6,7 The endothelial cell is capable, through an extensive data transmission network, to detect minimal changes in arterial pressure, blood flow, oxidative balance, coagulation, sign of inflammation, and immune system activation. 6,7 The need of an integrated network is because the major product released by the endothelium, the nitric oxide (NO), has a short half-life. 7 Thus, loss of endothelial integrity is one of the criteria to consider a vulnerable plaque and, consequently, one of the criteria for classifying a patient as “vulnerable” (with higher probability of developing cardiac events in a short period of time). 5 It has been suggested that the main mechanism by which endothelial cells confront vascular diseases is through the synthesis of endothelial NO synthase (eNOS) that will generate NO that is considered vascular protective. 8 Evidence suggests that derangements in metabolism or in endothelial regulatory functions are associated with the pathophysiology of many cardiovascular diseases. NO synthase 3 (NOS3) gene is responsible for the synthesis of eNOS in human beings. 9 Some polymorphisms described in this gene are considered “polymorphisms associated with risk” for ACS, because of their association with the atherosclerotic process. 10,11 NOS3 gene variations more commonly studied include numerous single nucleotide polymorphisms (SNPs) located either in regulatory or coding regions and also a variable number of tandem repeats (VNTR). 11,12 In a previous study, Piccoli et al. 13 demonstrated that polymorphisms in the NOS3 gene (eNOS) -786T>C, 894G>T, and intron 4 a/b VNTR may be an additional risk factor for the development of ACS. In several studies, the polymorphic variant -786T>C has been associated with CAD, although the results are still conflicting. 14 –18 A SNP, -786T>C was identified in the 5′-flanking region of the eNOS involving a substitution of thymine (T) to cytosine (C) at a locus 786 base pairs upstream of the eNOS. 15 This polymorphism results in a significant reduction in the eNOS gene promoter activity, leading to the reduced NO production and the endothelial dysfunction. 15,16 Currently, the important role of the inflammatory process during initiation and progression of CAD is unquestionable. 19 –21 Such pathogenetic achievement has led to the use of inflammatory cells and proteins as prognostic markers in these syndromes. A number of markers have been proposed, including proinflammatory cytokines such as the interleukin-6 (IL-6), IL-1, and tumor necrosis factor-alpha, adhesion molecules, and markers of cellular activation. In this way, the present study aimed to evaluate whether the gene -786T>C polymorphism of eNOS is associated with the inflammatory and oxidative process, expressed by ultrasensitive C-reactive protein (CRP), IL-6, N-terminal prohormone of brain natriuretic peptide (NT-ProBNP), CD40 ligand (CD40L), immunoglobulin G (IgG) antibodies against oxidized low-density lipoprotein (LDLox) levels and total leukocytes count in a population sample treated in an intensive care unit (coronary-ICU) with ACS.
Materials and methods
Design and subjects
The Ethics Committee of the Pontificia Universidade Católica do Rio Grande do Sul (PUCRS) approved the study protocol. A prospective cohort study of 125 consecutive patients with clinical diagnosis of non-ST-elevation acute coronary syndromes (NSTE-ACS) admitted to a coronary care unit of a teaching hospital was conducted. Eligibility criteria included patients presenting with NSTE-ACS within 24 h of symptom onset. Patients presenting with acute chest pain, electrocardiographic (ECG) abnormalities, elevated serum markers of myocardial injury, in whom hospital admission and management assumed as a principal diagnosis of ACS were included. The diagnosis of myocardial infarction (MI) was based on the redefined AMI classification proposed by the Joint European Society of Cardiology and American College of Cardiology Committee for the Redefinition of Myocardial Infarction. 22 The study was conducted in accordance with the Declaration of Helsinki; the Ethics Committee of our institution approved the study, and all patients signed written informed consent form.
Data collection
Patients were assessed using a standardized questionnaire that included data on clinical history, physical examination, laboratory examinations, and hospital management. The ECG was performed on admission and repeated daily during the stay in the hospital. Blood samples were drawn on an average 12 h after admission to measure serum levels of high-sensitivity CRP (hs-CRP), soluble CD40L, IL-6, NT-proBNP, IgG antibodies against LDLox. Blood samples were drawn for DNA extraction.
Laboratory analysis
Blood samples were collected and serum was stored in aliquots at −70°C. This serum was used to measure NT-proBNP and CD40L. The NT-proBNP concentration was measured with a sandwich immunoassay (Elecsys 2010, Roche Diagnostics, Indianapolis, Indiana, USA), CD40L was measured by enzyme-linked immunosorbent assay (ELISA) (R&D Systems, Minneapolis, Minnesota, USA), and hs-CRP was measured by immunonephelometry using intensifying particles (Dade Behring, Newark, Delaware, USA). The determination of IL-6 was performed using a commercially available ELISA kit from DPC Medlab Laboratory (Brasil). The detection of IgG antibodies against LDLox generated in case and control sera was made by the technique of ELISA. Each assay was performed according to the manufacturers’ specifications. Attending physicians and investigators were blinded to the test results until the study was completed. Blood samples from a peripheral vein were drawn according to the venoclysis system with a disposable vacuum device (Vacutainer) and stored in tubes with 0.1% ethylenediaminetetraacetic acid (final volume at the concentration of 1 mg dL−1). After that, the material collected was maintained at 4°C until DNA extraction for up to 24 h. Genomic DNA was isolated from peripheral blood leukocytes using a GFX Genomic Blood DNA Purification kit (Amersham Biosciences Inc., Co.).
Genotype determination
Genotypes for the -786T>C polymorphism in the 5′-flanking region of eNOS were determined by polymerase chain reaction (PCR) amplification using the primers 5′-TGGAGAGTGCTGGTGTACCCCA-3′ (sense) and 5′-GCCTCCACCCCCAC CCTGTC-3′ (antisense) and in the PCR conditions as described previously by Gonzáles-Ordóñez et al. 23 The amplified products (180 pb) were digested with MspI for at least 3 h, at 37°C, producing fragments of 140 and 40 bp for the wild-type allele (T allele), or 90, 50, and 40 bp in the case of a polymorphic variant (C allele). -786T>C fragments were separated by electrophoresis in 3% agarose gels and visualized by ethidium bromide staining.
Cardiovascular risk factor variables
(1) Diabetes mellitus (DM): individuals with glycemic levels above 126 mg dL−1 and those in use of medicines to lower glucose were considered diabetics. (2) Obesity: those with body mass index ≥30 were considered obese; the body mass index was calculated by dividing weight (kg) by height squared (m2). 24 (3) Tabagism: tobacco use was assessed by related history, and the individuals were classified as smokers or nonsmokers. (4) Dyslipidemia: subjects with total cholesterol ≥190 mg dL−1, LDL cholesterol (LDL-C) ≥110 mg dL−1, or elevated triglycerides (≥150 mg dL−1) were considered dyslipidemic as well as those who used drugs to lower cholesterol (III Diretrizes Brasileiras sobre Dislipidemia e Diretriz de Prevenção da Aterosclerose). 24 (5) Hypertension: we used standard desk mercury sphygmomanometers and stethoscopes to assess the blood pressure (BP) measurement (VI Diretrizes Brasileiras de Hipertensão): 25 subjects with systolic BP levels ≥140 mmHg or diastolic BP levels ≥90 mmHg, or both, were considered as hypertensive.
Statistical analysis
The allelic and genotypic frequencies were tested to equilibrium by the Hardy–Weinberg law. Descriptive analysis was performed using continuous variables (hs-CRP, total cholesterol, HDL-C, LDL-C, triglycerides, Pro-BNP, CD40L, leukocytes, glucose, and age). The normality of continuous variables was tested through the one-sample Kolmogorov-Smirnov Test. The analysis of comparison between the genotypes of -786T>C and the nonparametric continuous variables was performed using the nonparametric Kruskal-Wallis test. Already data that showed normal distribution curve are used for the one-way analysis of variance (ANOVA), followed by post-hoc Tukey test. To test the associations between categorical data and the genotypes of -786T>C, we used the chi-square test. Statistical analyses were performed by SPSS/PC Statistical Package Version 18.0 (SPSS, Inc., Illinois, USA). All p values were two-tailed. A value of p < 0.05 was considered statistically significant.
Results
The study population consisted of 125 consecutive patients (mean age 61.4 ± 12.2 years, 46.4% male) admitted with NSTE-ACS (Table 1). The frequency distribution of -786T>C polymorphism was 16 (12.8%) in -786CC-homozygotes, 60 (48%) in -786TC-heterozygotes, and 49 (39.2%) in -786TT-homozygotes. The -786C allele was found in 92 of 250 alleles (38.8%). Table 2 summarizes the characteristics of the study population according to the distribution of the investigated polymorphism. No statistical association was observed between demographic and clinical characteristics and distribution of eNOS-786T>C polymorphism (Table 2). There was no association between the distribution of -786T>C polymorphism and sex (χ2=0.72; p = 0.70), DM (χ2 = 0.95; p = 0.62), current or former smoking (χ2 = 1.27; p = 0.87), hypercholesterolemia (χ2 = 0.95; p = 0.62), hypertension (χ2 =1.04; p = 0.60), family history (χ2 = 1.00; p = 0.61), and obesity (χ2=1.33; p = 0.51). The continuous variables were tested to normality with one-sample Kolmogorov-Smirnov Test. Total serum cholesterol, IgG antibodies against LDLox, and white blood cell count demonstrated normal distribution. Other continuous variables analyzed did not demonstrate normal distribution (CRP, triglycerides, NT-proBNP, CD40L, glucose and IL-6). No statistically significant differences were found between -786T>C genotypes and total serum cholesterol, triglycerides, and serum blood glucose levels.
Baseline characteristics of patients admitted with non-ST elevation acute coronary syndromes (n = 125).a

CHD: coronary heart disease.
aData expressed as n (%) and mean ± SD for continuous variables.
Demographic and clinical characteristics of the participants, according to the distribution of eNOS-786T>C polymorphism.a
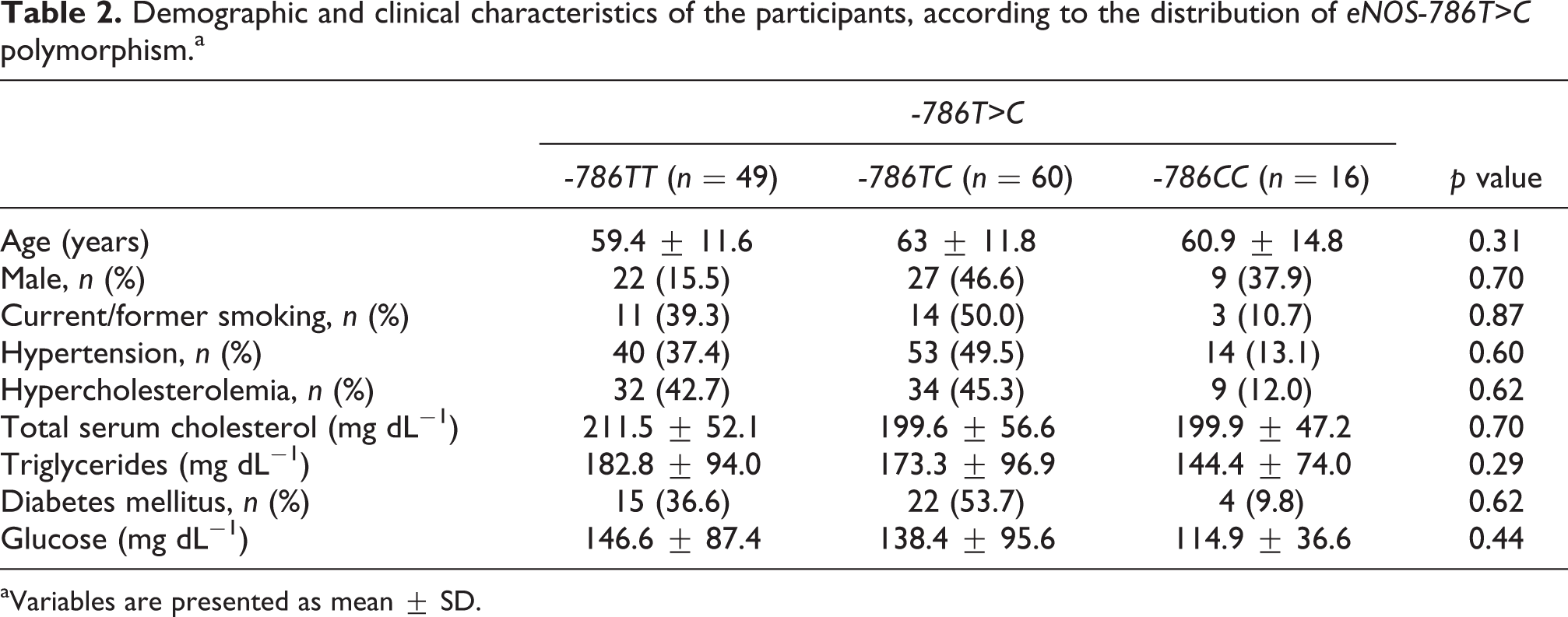
aVariables are presented as mean ± SD.
Mean values of the investigated inflammatory and oxidative markers and eNOS polymorphism are presented in Table 3. Thus, after nonparametric Kruskal-Wallis test, we found that -786CC was associated with lower levels of IL-6 (χ2 = 6.78; p = 0.03). No significant differences were observed between the distribution of -786T>C polymorphism and other investigated markers.
Levels of the investigated inflammatory and oxidative stress markers, according to the distribution of eNOS-786T>C polymorphism.a

CD40L: CD40 ligand; NT-proBNP: N-terminal prohormone of brain natriuretic peptide; LDLox: oxidized low-density lipoprotein.
aVariables are presented as mean ± SD.
b p values derived from the multivariate analysis of variance.
Discussion
In this study, we analyzed that the association between the polymorphism -786T>C of the eNOS gene and the IL-6 levels, an inflammatory marker associated with ACS. The -786CC genotype was associated with lower levels of circulating IL-6. There was no statistically significant association between the polymorphism studied and the serum levels of IgG antibodies against LDLox, hs-PCR, NT-ProBNP, CD40L, and total leukocyte count. The NO role in the pathogenesis of ACS is very well documented. 10,26 –28 It regulates vasodilatation in endothelial level, conferring a vasculoprotective action, mainly attributed by its biological potentialities as inhibition of platelet aggregation and leukocytes adhesion, which is considered an important regulator of the vascular tonus and of the endothelial integrity. 26 The two fundamental mechanisms for impaired bioavailability of NO are reduced synthesis and increased oxidative inactivation by reactive oxygen species. However, the loss of expression of NO leads to an increase in the expression of molecules of adhesion, while a higher production of these mediators promotes inflammation and contributes to the thrombogenic status, which is a mechanism involved during the development of atherosclerosis. 10
An impaired NO production is associated with inflammatory and oxidative processes as related in the development of CAD. A reduction in eNOS expression may also be due to the polymorphisms in the promoter region of NOS3 gene. Studies have suggested that the -786T>C polymorphic variant has an effective role in endothelial function. 15
Interestingly, the -786TT individuals were more hypertensive and presented slightly higher levels of glucose, triglycerides, and total cholesterol when compared with -786CC individuals, even though this increase was not statistically significant.
Considering that NO facilitates the glucose uptake, -786TT individuals can present shorter time of glucose uptake than the -786CC. In this study, individuals with -786CC genotype showed smaller rates of circulating glucose. Although these results were not statistically significant, a hypothesis that can be considered is that lesser the available glucose, lesser is the formation of electron donators (nicotinamide adenine dinucleotide and flavin adenine dinucleotide) during its metabolism. Consequently, less superoxide anion would be formed, so the rate of free radical production would be lower, which could explain the association between the -786CC with lower levels of IL-6. Besides, as hyperglycemia induces the expression of several adhesion molecules (vascular cell adhesion molecule-1 and intercellular adhesion molecule-1) and cytokines, 29 we could expect that individuals with higher indices of circulating glucose present a more inflamed profile.
In addition, despite the importance of the eNOS-derived NO in maintaining homeostasis in the interaction between endothelium and inflammatory cells under basal physiological conditions, we also know that the same eNOS-derived NO in pathologic conditions, as in the case of ACS, may act as a proinflammatory, thus increasing vascular permeability and angiogenesis. 30 This ambiguous mechanism indicates that NO, in different conditions, can act either in physiological or pathological processes, acting in a prooxidant manner. 29,30
Other studies showed that NO can modulate many of the processes leading to ventricular remodeling. 31 Endothelium-derived NO causes systemic vascular relaxation, 32 thereby reducing cardiac preload and afterload. Evidence suggests that NO can increase angiogenesis, decrease cardiac fibrosis, and decrease angiotensin II-induced cardiac myocyte hypertrophy, 33 all of which could limit ventricular remodeling after MI. The study by Çiftçi et al. 34 demonstrated that the high frequency of the mutant (C/C) and heterozygous (C/T) genotypes found in ACS patients may be linked to left ventricule remodeling after MI.
Our study has certain limitations that need to be taken into account. As this study associates polymorphism with reduced NO synthesis, the lack of measurements of nitrite and nitrate levels has not allowed the establishment of a straight correlation between the -786T>C polymorphism of eNOS, the effective levels of circulating NO, and the inflammatory markers studied. Further studies correlating eNOS gene polymorphisms, inflammatory markers, leukocytes adhesion proteins, and ACS could provide a better understanding of the results obtained here.
Our findings suggest that an interaction may exist between the -786T>C variant and the inflammatory process. Although this is a complex interaction, it could explain the association between the eNOS gene and the cardiovascular risk.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
