Abstract
Objectives:
The aim of this study was to evaluate the cytotoxicity of four low-shrink composites with new monomer technology on the bovine dental pulp-derived cells (bDPCs).
Materials and methods:
Ten samples were prepared for each group composites, and the samples were immersed in 7 mL of culture medium for 72 h at 37°C to extract residual monomer or cytotoxic substances. The culture medium containing the material extracts was sterile filtered for use on the cell cultures. Materials were incubated in medium with serum for 72 h. bDPCs were maintained in a medium with serum. A real-time cell analyzer was used to evaluate cell survival. After seeding 200 mL of the cell suspensions into the wells (10,000 cells/well) of the E-plate 96, bDPCs were treated with bioactive components released by the composite materials (1:1 and 1:2 dilutions) and monitored every 15 min for 50 h.
Results:
According to analysis of variance, there were significant differences between the cell indexes of the control and GC kalore (p < 0.05) and Bisco Reflexions (p < 0.001) groups for the 1:1 dilutions at 25 h. When evaluated at 50 h, 1:1 dilutions of GC Kalore (p < 0.01) and Bisco Reflexions (p < 0.001) reduced cell survival significantly.
Conclusions:
Although composites resins are being advanced, their cytotoxic effects have been proceeding till this time. However, two of the four materials tested significantly reduced cell viability when compared with control.
Clinical relevance:
Research should focus on the cytotoxicity of composites in addition to their mechanical properties.
Introduction
In recent dental practice, dental composites have been used owing to their esthetic features. For anterior teeth in particular, composites are very important, and currently, they are the best and the most commonly used dental filling material. However, significant concerns remain regarding their biocompatibility 1,2
In addition to their usage as restorative materials, resin composites can be used as dentin adhesives; as luting agents for inlays, crowns, and veneers; or for orthodontic brackets or other orthodontic apparatuses. 2 However, polymerization shrinkage and residual monomers are the negative effects of composites according to the literature. 3 Improvements in chemistry and composition have reduced polymerization shrinkage. 4 To reduce the adverse effects of polymerization shrinkage, new resin matrices with low-shrinkage have been developed. 5 For example, the so-called low-shrinkage composite resins claims shrinkage near 1%, by volume lower than the 2–5% exhibited by some bisphenol A-glycidyl methacrylate (Bis-GMA) composites. 6 Another problem is the release of monomers from dental resin materials, which results in adverse biological effects in mammalian cells. 7 There are many indications that release of monomers and of some additives are potentially hazardous and might have local or even systemic effects. 8 Several ingredients have been shown allergenic, 9 cytotoxic, 10 genotoxic, 11 and toxic to reproductive system. 6 Resin-based composite material releases several components into the oral mucosa, periodontium, and dentinopulpal complex. These substances may be systemically distributed, which could result in adverse systemic effects. 12 It is reported that the amount of components released and the extent of the polymerization reaction are related. 13 In a previous study, the newly developed “low-shrinkage” composites exhibited significantly lower shrinkage rates and shrinkage volumes than the conventional methacrylate composites However, it is unclear whether reducing polymerization shrinkage will reduce the adverse side effects such as biological toxicity. 14
The types of composite resins include Bis-GMA, urethane dimethacrylate (UDMA), triethylene glycol dimethacrylate (TEGDMA), and bisphenol A ethoxylate dimethacrylate. Many researchers demonstrated that these components of composite resins are cytotoxic and mutagenic 15 locally and systemically and may cause pulp reactions and allergic and estrogenic effects, 12 using different methods and different cell culture techniques. Cytotoxic effects of dental composites are connected residual monomers because of incomplete polymerization and degradation process 16 ; moreover, a lot of different major monomers, comonomers, various additives, and reaction products have been shown to be released from polymerized dental composites. 17,18 On the other hand, biologically active resin monomers released from composite restorations may influence the dentine–pulp complex, for instance, its regenerative and reparative capacities. 19
Histomorphometric cell counting of intact odontoblasts and pulp fibroblasts and tetrazolium salt 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assays have been applied for cytotoxicity evaluation. 20 In addition, DNA synthesis (bromodeoxyuridine (BrdU) incorporation), modified Boyden chamber, and immunocytochemical and spectrophotometric assays were used by Theilig et al. 21 All of the methods for assessing cytotoxicity, cell proliferation, and viability are single end-point qualitative measures of cell fitness. 22 However, their disadvantages make the results difficult to understand and reproduce. As a better alternative, “real-time” in vitro cell culture has become an increasingly used method. 22,23 It saves expensive reagents used in conventional cell analysis.
In this study, we investigated the cytotoxicity of the newly developed low-shrinkage composites in bovine dental pulp-derived cells (bDPCs) 24 using real-time cell analyzer (RTCA, xCELLigence; Roche Applied Science, Mannheim, Germany; and ACEA Biosciences, San Diego, California, USA). These materials are widely used in dentistry. Therefore, these materials should be tested whether it is toxic.
Methods
Material preparation
In this study, Bisco Reflexions (Bisco Inc., Schaumburg, IL, USA), Kerr Premise (Kerr Corporation, Orange, CA, USA), GC Kalore (GC Co., Tokyo, Japan), and Filtek Silorane (3M Espe, St. Paul, MN, USA) composites were used. These composites are universal light-cured nanocomposites with new monomer technology. Their components and manufacturers are listed in Table 1. All the specimens were prepared and handled according to the manufacturers’ instructions under aseptic conditions (Laminar flow) with light curing units in standard polytetrafluoroethylene disks measuring 5 mm in diameter and 2 mm in thickness. All the specimens were prepared and handled under aseptic conditions to limit any biologic contamination of the cell culture tests. The specimens were prepared between polyester film and glass slabs to minimize the oxygen inhibition and maximize the surface smoothness. Ten samples were prepared for each group of composites. The substances were stored in 7 mL of culture medium. The samples were immersed in culture medium (Minimum Essential Medium Alpha, Gibco Invitrogen, Germany) supplemented with 5% fetal bovine serum (Biological Industries, Beit Haemek, Israel) for 72 h at 37°C according to ISO 10993-5 standards (surface area-to-volume ratio of the specimen to cell culture medium: 3 cm2/mL) to extract residual monomers or cytotoxic substances. The experiments started after 5 h. The xCELLigence system (Roche Diagnostics GmbH, Mannheim, Germany and ACEA Biosciences, Inc., San Diego, California, USA) was used to evaluate cell survival according to the instructions of the supplier.
Ingredients of tested materials.
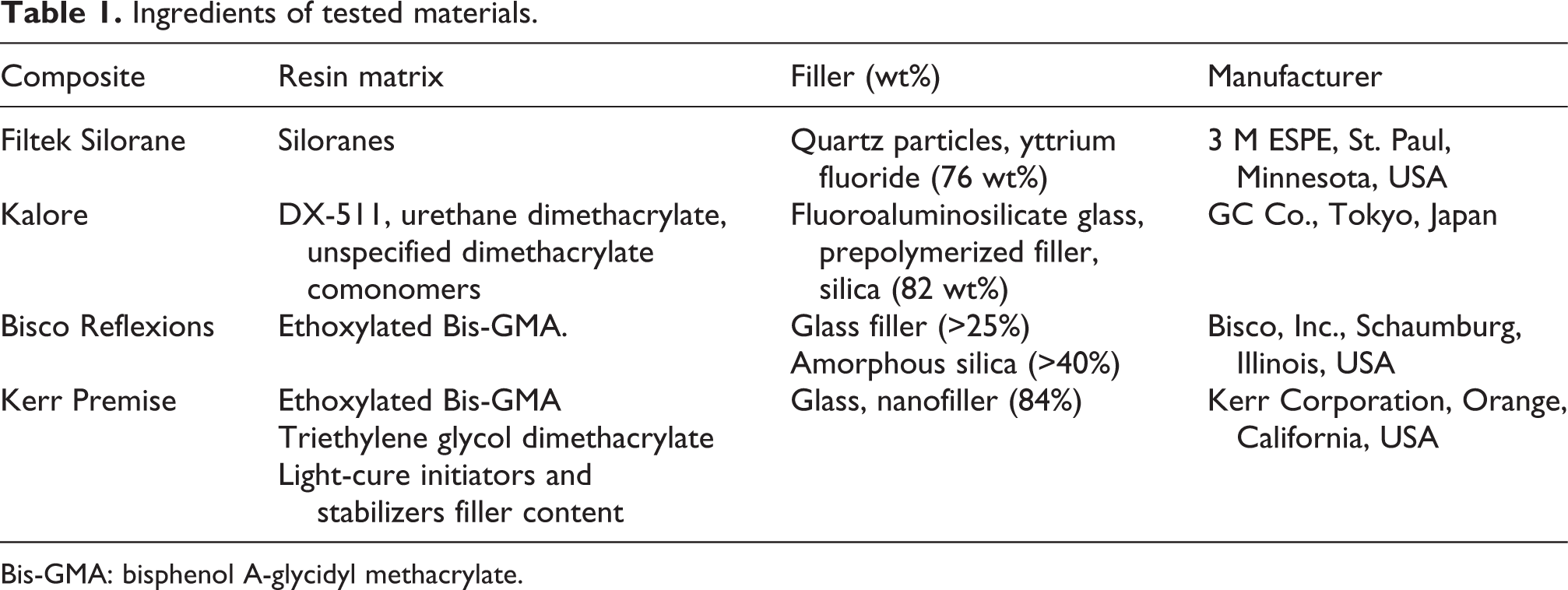
Bis-GMA: bisphenol A-glycidyl methacrylate.
bDPCs were kindly provided by Prof. G. Schmalz from the University of Regensburg. 24 Cells at passage 20 were used for the experiments. After seeding 200 mL (10,000 cells/well) of the cell suspensions into the wells of the E-plate 96, cells were treated with bioactive components released by the composite materials (1:1 and 1:2 dilutions) and monitored every 15 min for 50 h. The morphology of the bDPCs 24 was evaluated after the treatment with the bioactive components of the composites using an inverted microscope.
The xCELLigence system consists of four main components: the impedance-based real-time cell analyzer (RTCA), the RTCA single-plate station, the RTCA computer with integrated software, and a disposable E-plate 96. The RTCA single-plate station fits inside a standard tissue culture incubator. The electronic impedance of the sensor electrodes was measured to allow monitoring and detection of physiologic changes in the cells on the electrodes. The voltage applied to the electrodes during the RTCA measurement was approximately 20 mV (root mean square). The impedance measured between the electrodes in each well depends on the electrode geometry, ion concentration in the well, and attachment of the cells to the electrodes. In the absence of cells, electrode impedance is mainly determined by the ion environment at the electrode-solution interface and bulk solution. In the presence of cells, cells attached to the electrode sensor surfaces act as insulators and thereby alter the local ion environment at the electrode-solution interface, leading to increased impedance. Thus, the obtained values of electrode impedance increase as the number of cells growing on the plate increases. The data expressed in cell index units can be exported to the Excel software (Microsoft, Seattle, Washington, USA) for any type of mathematical analysis.
Statistical analysis
All the calculations were performed using the RTCA integrated software of the xCELLigence system. The RTCA software performs a curve fitting of selected sigmoidal dose–response equations to the experimental data points. The data are presented as means (mmol/L) ± SD (n = 6). For the proliferation experiments, statistical analysis was performed using one-way analysis of variance (ANOVA) and Tukey–Kramer multiple comparison tests. The data are presented as means and SDs. A p < 0.05 was considered statistically significant.
Results
According to ANOVA, when evaluated at 25 h, there were significant differences between the cell indexes of the control group and the GC Kalore (p < 0.05) and Bisco Reflexions groups (p < 0.001) for the 1:1 dilutions (Table 2 and Figure 1). When evaluated at 50 h, the 1:1 dilutions of GC Kalore (p < 0.01) and Bisco Reflexions (p < 0.001) reduced significantly cell viability (Table 2 and Figure 1).
Cell index values by real-time cell analysis and comparisons of 25 and 50 h with analysis of variance and Tukey–Kramer multiple comparison tests.


Dynamic monitoring of cell adhesion and cell proliferation.
Discussion
The RTCA system, which permits label-free dynamic monitoring of cell proliferation and viability in real time, was developed by Roche Applied Science and ACEA Biosciences as a predecessor of the new xCELLigence System. One of the advantages of the newly developed system is its cell impedance measurements. In addition to allowing real-time, rather than end point, measurements of cell proliferation, viability, and cytotoxicity, the xCELLigence System is more economical than its predecessor. 25,26 The new xCELLigence System has recently been used for assaying the cytotoxicity of dental materials. 22,27 However, the limitations of this system is that it can give false positive value at suspensions containing metallic ions.
Previously, the tissue compatibility of dental materials was studied in animal experiments. 27 Moreover, individual test methods are usually adequate only for describing or documenting a single aspect of a certain type of unwanted reaction. Cell culture tests detect only the influence of a material on isolated cells. Isolated cells derived from animal or human tissues are grown in culture plates and then used for these tests. Currently, permanently growing cells are primarily used for this purpose because these cells can be easily amplified and their behavior is well known, relatively consistent, and constant. Frequently, permanent mouse fibroblasts (L929) are used. These cell cultures are “incubated” with the materials or their extracts. Subsequently, a series of various parameters will be measured, such as the number of surviving cells, protein synthesis, enzyme activity, or inflammatory mediator synthesis. 28
Resin composites have been commonly used for years. In clinical practice, dentists prefer composites with low shrinkage, high bond strength, low dye penetration, and low abrasion. However, the clinical success of a material used in humans depends on the physical and chemical properties of the material and its biological safety. In fact, investigations of the cytotoxicity and biocompatibility of composite resins started with their use in dentistry because the organic matrix of dental resin materials, such as TEGDMA, 2-hydroxyethyl methacrylate, 7 Bis-GMA, and UDMA, has been recognized as a source of compounds that cause adverse biological reactions. Furthermore, owing to its high cytotoxic potency, TEGDMA may significantly contribute to adverse local and systemic effects, and the liberation of TEGDMA from resin restorations should therefore be minimized or prevented. 29
Geurtsen et al. found that TEGDMA may be released by polymerized composite resins into water, and Bis-GMA, UDMA, Ethylene glycol dimethylacrylate (EGDMA), diethylene glycol dimethacylate (DEGDMA), 1,6-hexanediol di-methacrylate, methyl methacrylate, camphorquinone, 4-N, N-dimethylamino-benzoic acid ethyl ester, and various other substances were detected in aqueous extracts. 30 In addition, a 100% monomer-to-polymer conversion rate is desired, but as much as 25–50% of the methacrylate monomer double bonds actually remain unreacted in the polymer. 30
Recently, some composite resin materials developed as low-shrinking composites have been introduced, but their complete efficacy in preventing the harmful consequences of the shrinkage stresses generated at the dentin-composite interface has not been demonstrated. 31 A previous study revealed that low-shrinkage composite resin restorative materials have similar mechanical and physical properties as clinically successful composite resins. 32 According to the manufacturer’s information, the filler content and polymerization shrinkage rate of low-shrinkage composite resin restorative materials are different from those of the conventionally used composites. However, there is insufficient information about their cytotoxicity. Although the development and improvement of the dental composite materials are very satisfying and impressive, their biocompatibility remains a problem for clinicians and patients.
According to our results, GC Kalore and Bisco Reflexions at 25 and 50 h, the cell proliferation for the 1:1 and 1:2 dilutions was lower than that of the control group. Thus, the components of GC Kalore and Bisco Reflexions affected the cells more than control group.
The cytotoxicity of resin-based dental composites depends on their chemical composition, their leaching medium, and the amount and type of components that can be extracted from the materials. 33 Our results are consistent with previous findings. 22 The cytotoxicity of utilized materials may be dependent on the filler content or the type of resin matrix. In addition, composite resins may contain unknown monomers. As a result, the concentrations of biologically active ingredients of some of the composite resins may affect pulp cell metabolism.
In this study, we determined the in vitro cytotoxicity of four newly developed low-shrinkage composites in bDPCs, 24 because these cells are most commonly exposed to dental materials. 34
Urcan et al. investigated that influence of cytotoxicity of dental composite components with real-time xCELLigence system. Cell index curves showed that the highest cell index value was found for Hydroxyethyl methacrylate (HEMA) (11.20 ± 0.3). This was significantly different from TEGDMA (3.61 ± 0.2), UDMA (0.20 ± 0.1), and Bis-GMA (0.08 ± 0.1). Bis-GMA was significantly (p < 0.05) highly effective compared with UDMA, TEGDMA, and HEMA. In our study, Cell index value of low-shrink composites were adjacent value to control group. 22
In summary, although composite resins are being improved, their cytotoxic effects are disadvantageous. Therefore, research should focus on the cytotoxicity of composites in addition to their mechanical properties. These materials were no toxic in our study. These materials only reduced cell viability. Therefore, these materials may be a matter of preference than other dental composite materials.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
