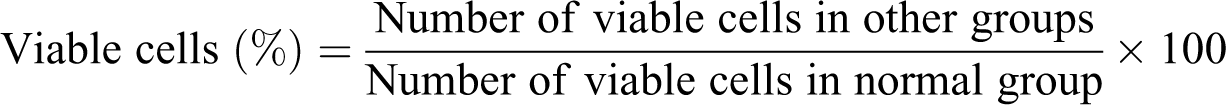Abstract
Biochanin-A (BCA), a potent phytoconstituent, has been previously used as an antitumour, a dopaminergic neuron protective agent, an antioxidant, an anticholinergic and on other pharmacological activities including neuroprotection. The present study was aimed to evaluate the behavioural and neurochemical evidence of BCA in cognitive-deficit mice in scopolamine challenged and natural aged-induced amnesia models in young and aged mice, respectively. BCA has exhibited decrease in the transfer latency and increase in step through latency significantly (p < 0.001) in scopolamine-treated and natural aged mice of exteroceptive behavioural models such as elevated plus maze and passive shock avoidance paradigm, respectively. A decrease in acetylcholinesterase activity of whole brain was seen in scopolamine and aged mice with standard piracetam (Pira; p < 0.001) and BCA in dose-dependent manner. The antioxidant property of BCA was proven by increase in GSH (p < 0.01) and decrease in thiobarbituric acid reactive substances level significantly in a scopolamine-challenged and aged mice. The scopolamine-treated mice exhibited significant (p < 0.01) increase in the content of noradrenalin and dopamine, which is a sign of dementia, and these excess increased neurotransmitters were reversed by BCA 40 mg kg−1 (p < 0.05), BCA 20 mg kg−1 (p > 0.05), BCA 10 mg kg−1 (p < 0.05) and standard Pira (p < 0.05) when compared with scopolamine group. Furthermore, in histopathology of hippocampus, the Pira and BCA-treated mice were protected from the formation of pyknotic neurons, increases in the viable cells count and decreases in the number of degenerative cells compared with the scopolamine group. Hence, BCA could be potential enough for the betterment of Alzheimer’s disease.
Introduction
Dementia is a syndrome of failing memory and other intellectual functions, which shows progressive loss of personality and behavioural disturbance. 1 This syndrome occurs highly in Alzheimer’s disease (AD). Dementia related to AD has gained foremost attention in the last decade due to its increasing number of patients suffering from the disease. 2 It disrupts various cortical functions such as memory, judgement, intelligence, learning ability and diminishes the individual’s ability to correlate with occupational function and day-to-day social activities. 3,4 AD induces mainly loss of memory including loss of neurons in cerebral cortex and certain subcortical regions of brain. 5,6
AD is the third largest highly paid disease, affecting one person in eight over 65 years of age and almost half over 85 years; it is the fifth leading death-causing disease especially in elderly people. It is estimated that there are 7.7 million of new cases reporting dementia each year and the number will be doubled by 2030 and can even be tripled by 2050. 7 The number of people with dementia in India is 3.7 million and the number is set to be double in the next 20 years. 8
Various etiological factors responsible for the development of learning and memory deficits are decrease in cholinergic release, 9 change in biogenic amines and increase in free radicals formation including oxidative stress. 10 Memory enhancers and cholinesterase inhibitors such as piracetam (Pira) and donepezil, respectively, are being used to improve the memory with wide variety of side effects such as diarrhoea, insomnia, bronchitis and muscular cramps, which make them their use limited. 11
Herbal medicines containing phytoconstituents can modify the ageing of brainand it has been proved scientifically. 12 Biochanin-A (BCA) is a pharmacologically active phytochemical present in the seeds of Glycine max (soya bean/soy) belonging to the family Fabaceae. Soy is an edible bean and traditionally being used to improve the cognitive performance in postmenopausal women 13 and also to prevent the degeneration of central nervous system and development of AD. 14,15 BCA is an o-methylated natural isoflavone exhibiting various pharmacological properties such as anti-inflammatory, anti-carcinogenic and hypolipidemic effects. Also, BCA is scientifically proven for its dopaminergic neuronal protection, 16 antioxidant 17 and anticholinergic activities. 18 All these characteristics play an important role in the treatment of AD. Besides, further literature survey reveals that BCA has an anti-tumour activity interlinked in relation with the modulation of antioxidant enzymes, 19 inhibits P-glycoprotein activity in human intestinal Caco-2 cells 20 and protects dopaminergic neurons by inhibiting microglial activation. 16 The chemical name of BCA is 5,7-dihydroxy-3-(4-methoxyphenyl) chromen-4-one 21 (Figure 1). Hence, in the current study, an effort has been made to estimate the effects of BCA on various biochemical parameters like reduced glutathione levels (GSH), brain thiobarbituric acid reactive substances (TBARS) and brain acetylcholinesterase (AchE) activity, including neurotransmitter assays such as dopamine and noradrenaline to correlate them with cognition concern.

Molecular structure of Biochanin-A.
Materials and methods
Chemicals
BCA was purchased from Sigma Aldrich (Bangalore, Karnataka, India). Pira and Scopolamine were obtained from Elite pharmaceuticals (Ahmedabad, Gujarat, India) and Alkaloids Corporation of India (Kolkata, West Bengal, India), respectively, as a gift sample. 5, 5′-Dithiobis (2-nitro benzoic acid) (DTNB), trichloroacetic acid (TCA) and thiobarbituric acid (TBA) were procured from Hi media (Bangalore, Karnataka, India). Other chemicals used were of analytical grade.
Animals
Experiments were carried out in Swiss albino male mice, aged 3 months(weighing 22–28 g) and 14 months (weighing 35–42 g). Experiments were conducted after getting approval from the Institutional Animal Ethical Committee (Approval Number: SETCP/IAEC/2008-2009/242). Animals were kept in an animal house of S.E.T’s College of Pharmacy, Dharwad, Karnataka, India, under controlled conditions of temperature (23 ± 2°C), humidity (50 ± 5%) and 12-h light/dark cycle. Animals were fed with standard diet pellets (obtained from Venkateshwara Enterprises, Bangalore, Karnataka, India) and water ad libitum. All the animals were acclimatized for seven days before the starting the experimental studies.
Preparation of doses
The selected doses of BCA 40, 20, 10 mg kg−1 body weight (b.w.) depend on literature survey 22 were dissolved in dimethyl sulfoxide and administered intraperitoneally (i.p.) to different groups of mice. Pira (400 mg kg−1, b.w.), a standard drug, was dissolved in distilled water (DW) and administered periorally (p.o.). Amnesia was induced by scopolamine (3 mg kg−1, b.w.) 23 –25 i.p. by dissolving it in DW.
Experimental design
Mice were divided into different groups for interoceptive and exteroceptive behavioural models, each group comprising of a minimum of six animals. Normal young mice received DW in the dose of 10 ml kg−1 b.w. orally. BCA (40, 20 and 10 mg kg−1) was administered i.p. for 14 successive days to young and aged mice. After 90 min of the administration of the last dose (on day 14), amnesia was induced in young mice by injecting scopolamine (3 mg kg−1, i.p.) and in a aged mice, natural ageing itself acts as an amnesia-inducing agent. Young and aged mice were exposed to the training session after 30 min of scopolamine injection (only young mice) using elevated plus maze (EPM) and passive shock avoidance (PSA; step through) apparatus. Retention memory was recorded after 24 h (on day 15). Pira (400 mg kg−1, p.o.) was used as an established standard nootropic agent and was injected for 14 days to positive control groups.
Elevated plus maze
EPM served as the exteroceptive behavioural model to evaluate short-term memory in mice. Briefly, EPM for mice consisted of two open arms (16 × 5 cm2) and two covered arms (16 × 5 × 12 cm3) extended from a central platform (5 × 5 cm2), and the maze was elevated to a height of 25 cm from the floor. On the first day (i.e. 14th day of BCA administration), each mouse was placed at the end of an open arm, facing away from the central platform to measure the acquisition transfer latency (TL). TL was defined as the time (in seconds) taken by the animal to move from the open arm into one of the covered arms with all its four legs. The mouse was allowed to explore the maze for another 2 min and then returned to their home cage. Retention of this learned task (memory) was examined 24 h after the acquisition trial. A significant reduction in TL indicates improvement in the memory. 26
PSA(step through) paradigm
The PSA was measured in an apparatus consisting of one light and one dark compartment. After treatment, each animal was placed in the light compartment and the time taken to move from light compartment to dark compartment was measured as acquisition latency. A shock of 0.4 mA for 2 s was applied as soon as the animal stepped into the dark compartment. Immediately after that, the animal are allowed to return to their home cages, and after 24 h, all the animals were again placed in the light compartment in order to measure the time it took to move from the light to the dark compartment as a retention latency. Animals which did not enter dark compartment even after 180 s, which was considered as an upper cut-off time of retention, were removed from the apparatus. 27
Estimation of whole brain AchE
The estimation of whole brain AchE activity was carried out based on Elman’s method with slight modifications. The animals were decapitated and brains were dissected out immediately and placed in ice-cold saline. The tissue was weighed and homogenized in 0.1 M phosphate buffer pH 8 (10% w/v), the homogenized tissue was centrifuged to 15,375g. for 10 min. Then, 0.4 ml aliquot of the supernatant was added to a cuvette-containing 2.6 ml phosphate buffer (0.1 M, pH 8) and 100 μl of DTNB. The contents of the cuvette were mixed thoroughly by bubbling air and absorbance was measured at 412 nm in a Lab India spectrophotometer. When absorbance reached a stable value, it was recorded as the basal reading. To this, 20 μl of substrate, that is, acetylthiocholine was added and the change in absorbance is recorded for a period of 10 min at intervals of 2 min. The change in the absorbance per min was determined. AchE activity was calculated using the following equation
28
Where R is the rate in moles of substrate hydrolyzed per minute per gram of brain tissue; A is the change in absorbance per minute and CO is the original concentration of the tissue in milligram per milliliter.
Biochemical estimation of brain biogenic amines
The animals were killed and the whole brain was dissected out. The weighed quantity of tissue was homogenized in 10 ml hydrochloric acid–butanol (0.85 ml of 37% hydrochloric acid in one litre n-butanol for spectroscopy) for 1 min in a cool environment. The samples were then centrifuged for 10 min at 3075g. Then, 0.08 ml of supernatant phase was removed and added to an eppendorf reagent tube containing 0.2 ml of n-heptane (for spectroscopy) and 0.025 ml of 0.1 M hydrochloric acid. After 10 min of vigorous shaking, the tube was centrifuged under same conditions to separate two phases. Upper organic phase was discarded, and to the aqueous phase of 0.02 ml, 0.05 ml 0.4 M and 0.01 ml ethylenediaminetetraacetic acid/sodium acetate buffer (pH 6.9) were added, followed by 0.01 ml iodine solution (0.1 M in ethanol) for oxidation. The reaction was stored after 2 min by addition of 0.01 ml sodium sulphite in 5 M sodium hydroxide. After 1,5 min, acetic acid was added, and the solution was then heated to 100°C for 6 min. When the sample again reached room temperature, excitation and emission spectra were read at 395–485 nm for noradrenaline and 330–375 nm for dopamine, respectively. The values were expressed as fluorescent excitation spectral height intensity corresponding to its concentration of biogenic amines present in the sample and percentage decrease in the content of neurotransmitter was calculated. 29
Determination of TBARS
The animals were killed and the whole brain was dissected out and homogenized in phosphate buffer (pH 8, 10% w/v). The homogenates were centrifuged at 15,375g at 4°C for 20 min using a refrigerated high speed centrifuge (C-24; Remi Laboratory Instruments, Remi Eletrochnik Ltd., Thane, India). Then, 2.0 ml of the tissue homogenate(Supernatant) was added to 2 ml of freshly prepared 10% w/v TCA, and the mixture was allowed to stand in an ice bath for 15 min. After 15 min, the precipitate was separated by centrifugation and 2.0 ml of clear supernatant solution was mixed with 2 ml of freshly prepared 0.67% TBA. The resulting solution was heated in a boiling water bath for 10 min. It was then immediately cooled in an ice bath for 5 min. The colour developed was measured at 532 nm against reagent blank. The values were expressed in micromoles per litre. 30 The value of extinction coefficient for TBARS was 1.56 × 105 M−1 cm−1. 31
Determination of GSH
The animals were killed and the whole brain was dissected out and homogenized in phosphate buffer (pH 8, 10% w/v). The homogenates were centrifuged at 15,375g at 4°C for 20 min using Remi C-24 high speed cooling centrifuge. Equal volumes of tissue homogenate (supernatant) and 20% TCAwere mixed. The precipitated fraction was centrifuged and to 0.25 ml of supernatant, 2 ml of 0.6 mM DTNB reagent was added. The final volume was made up to 3 ml with phosphate buffer (0.2 M, pH 8.0). The colour developed was read at 412 nm against reagent blank. The values were expressed in micromoles per litre. 32 The value of extinction coefficient for GSH was 13.6 × 103 M−1 cm−1. 31
Histopathology of hippocampus of brain
After completion of behavioural studies, animals were transcardially perfused with cold saline followed by 4% formalin in phosphate-buffered saline (0.1 M; pH 7.4). The brains were removed from the skull and fixed in the same fixative for 24 h. Thereafter, the brains were embedded in paraffin and 5 µm thick sections were coronally cut at the level of the dorsal hippocampus by a rotator microtome. Tissue sections of hippocampal CA1 regions were stained with haematoxylin and eosin, and hippocampal damage was determined by counting the population of viable and neurodegenerative cells in the stratum pyramidal within the CA1 subfield region at a higher magnification using a CH 20i microscope (Olympus, Olympus opto systems, India Private Ltd., Noida, U.P., India). 33 The results were expressed as a total number of viable and neurodegenerated (pyknotic black neurons and karryorhexis) cells for the region 34,35 and also expressed in terms of percentage by pathologist who was unaware of drug treatments in Jawaharlal Nehru Medical College, Belgaum, Karnataka, India.
Calculation for percentage of viable cells
Calculation for percentage of degenerative cells
Statistics
The data were analyzed statistically using analysis of variance followed by Tukey’s post test. Values are expressed as Mean ± SEM. The value of p < 0.05 was considered as significant and p > 0.05 was considered as non-significant. Statistical comparisons were performed by Tukey’s post test using Graph Pad Prism version 5.0 (GraphPad Software, San Diego, California, USA).
Results
Effect of BCA on EPM and PSA
The effects of standard drug Pira and BCA was found to be nootropic as they decreased (p < 0.001) the TL and increased (p < 0.001) the STL in EPM and PSA, respectively, with both the scopolamine-challenged and aged mice (Tables 1 and 3). The administration of scopolamine (3 mg kg−1, i.p.) induced amnesia in scopolamine control animals compared with the normal control group animals. Natural ageing was considered as an amnesic model for aged mice. Furthermore, scopolamine-administered mice and aged mice significantly increased TL and decreased the STL compared with the normal group of animals in EPM and PSA, respectively. These observations suggested that scopolamine and ageing produced an impaired learning and memory. However, Pira and BCA per se produced a significant improvement in memory scores (p < 0.001; Tables 1 and 3). Scopolamine- and ageing-induced memory deficits were successfully reversed by BCA 40 mg kg−1 (p < 0.001), 20 mg kg−1 (p < 0.001), 10 mg kg−1 (p < 0.01) as indicated by decreased in TL (Tables 1 and 2) and increased in STL (Tables 3 and 4). Pretreatment with Pira (400 mg kg−1; p.o.) reversed the scopolamine- and ageing-induced memory deficits (p < 0.001) as expected.
Effect of BCA on TLs of normal young and scopolamine-challenged mice on EPM.a

BCA: biochanin-A; TLs: transfer latencies; EPM: elevated plus maze; DW: distilled water; Pira: piracetam; i.p.: intraperitoneally; p.o.: periorally.
aEach group consists of six animals (n = 6). Values are mean ± SEM.
b p < 0.001 as compared to normal group.
c p < 0.01 as compared to normal group.
d p < 0.001 ascompared to scopolamine group.
e p < 0.01 as compared to scopolamine group.
Effect of BCA on TLs of aged mice on EPM.a

BCA: biochanin-A; TLs: transfer latencies; EPM: elevated plus maze; DW: distilled water; Pira: piracetam; i.p.: intraperitoneally; p.o.: periorally.
aEach group consists of six animals (n = 6). Values are mean ± SEM.
b p < 0.001 as compared to normal group.
c p < 0.001 as compared to aged group.
d p < 0.05 as compared to aged group.
e p < 0.01 as compared to aged group.
Effect of BCA on STLs of normal young and scopolamine challenged mice on passive shock avoidance.a

BCA: biochanin-A; STLs: step-through-latencies ; EPM: elevated plus maze; DW: distilled water; Pira: piracetam; i.p.: intraperitoneally; p.o.: periorally.
aEach group consists of six animals (n = 6). Values are mean ± SEM. p < 0.05 is considered as significant.
b p < 0.001 as compared to normal group.
c p < 0.001 as compared to scopolamine group.
Effect of BCA on STLs of aged mice on PSA.a
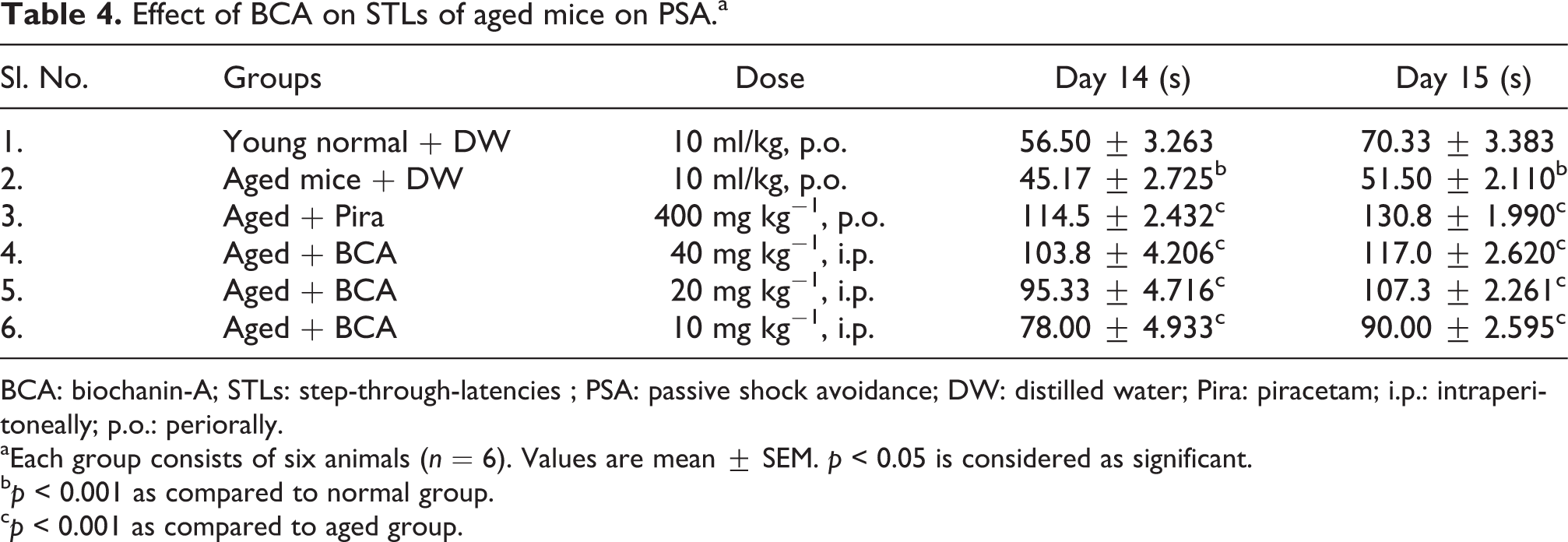
BCA: biochanin-A; STLs: step-through-latencies ; PSA: passive shock avoidance; DW: distilled water; Pira: piracetam; i.p.: intraperitoneally; p.o.: periorally.
aEach group consists of six animals (n = 6). Values are mean ± SEM. p < 0.05 is considered as significant.
b p < 0.001 as compared to normal group.
c p < 0.001 as compared to aged group.
Effect of BCA on brain AchE activity
Statistically significant differences were observed in whole brain AchE activity of Pira-treated mice (p < 0.01) compared with the normal control group, whereas BCA per se-treated mice did not produce a significant (p > 0.05) results compare to normal control in normal young mice (Figure 2(a)). On the other hand, administration of scopolamine (3 mg kg−1, i.p.) and aged animals significantly (p < 0.001) increased the brain AchE activity, and this elevated AchE was reversed by BCA in a dose-dependent manner 40 mg kg−1 (p < 0.001), 20 mg kg−1 (p < 0.01), 10 mg kg−1 (p < 0.0 5) (Figure 2(b) and (c)). Pira (400 mg kg−1; p.o.) being a standard drug, also reversed increased AchE (p < 0.001) in scopolamine and aged animals.
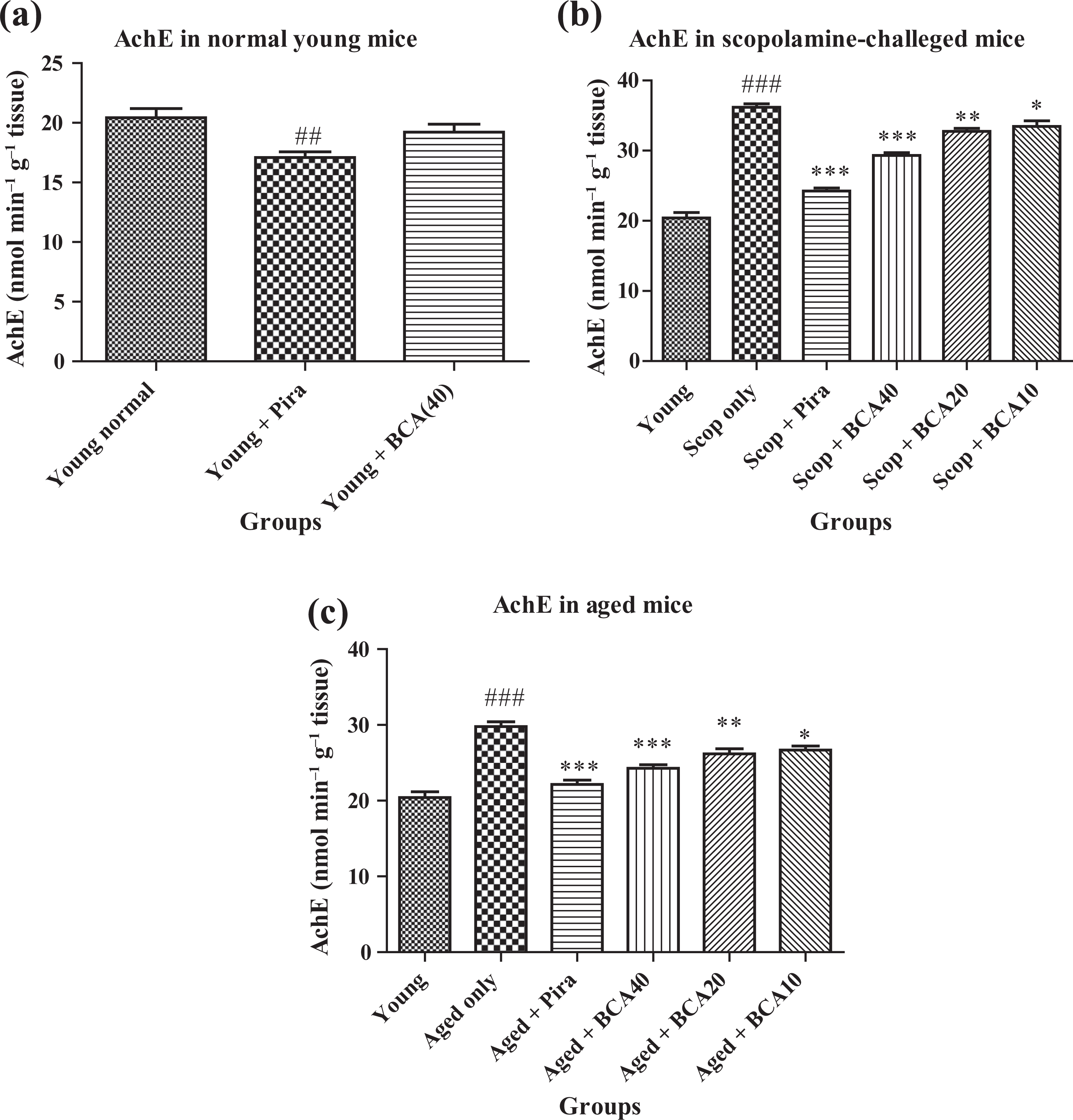
(a) Each group consists of six animals (n = 6). Values are mean ± SEM. p < 0.05 is considered as significant. ## p < 0.001 as compared to normal control. (b). Each group consists of six animals (n = 6). Values are mean ± SEM. p < 0.05 is considered as significant. ***p < 0.001, **p < 0.01, *p < 0.05 compared with scopolamine group and ### p < 0.001 as compared to normal group. (c) Each group consists of six animals (n = 6). Values are mean ± SEM. p < 0.05 is considered as significant. ***p < 0.001, **p < 0.01, *p < 0.05 compared with aged group and ### p < 0.001 as compared to normal group.
Effect of BCA on brain biogenic amines
The administration of Pira and BCA to a normal group of mice indicated decrease in the height intensity of emitted spectra of dopamine and noradrenaline insignificantly (p > 0.05) except standard drug in dopamine, which was significant (p < 0.01) comparative to young normal group of mice treated with plane DW (Table 5).
Effect of BCA on brain biogenic amines in normal young and scopolamine-challenged mice.a

BCA: biochanin-A; DW: distilled water; Pira: piracetam; i.p.: intraperitoneally; p.o.: periorally.
aEach group consists of five animals (n = 5). The values obtained are fluorescent excitation height of biogenic amines. Values are mean ± SEM. p < 0.05 is considered as significant.
b p < 0.001 as compared to normal control.
c p < 0.001 as compared to scopolamine group.
d p < 0.01 as compared to scopolamine group.
Animals treated with scopolamine exhibited significant (p < 0.001) increase in the height intensity of emitted spectra of noradrenaline and insignificant increase of (p > 0.05) dopamine comparative to young normal group (Table 5). The insignificant (p > 0.05) reversal effect of increased height intensity of emitted spectra of dopamine and noradrenaline was seen with different doses of BCA, except BCA 40 mg kg−1, which is significant (p < 0.01) in noradrenalin. Pira exhibited insignificant (p > 0.05) reversal effect of increased height intensity spectra of dopamine and significant (p < 0.01) reversal effect of noradrenalin as compared to scopolamine group of animals. The percentage of dopamine and noradrenaline content has also been indicated in Table 5.
Effect of BCA on brain antioxidant
Young mice pretreated with Pira (p < 0.05) exhibited significant increased antioxidant activity, whereas BCA exhibited insignificant (p > 0.05) increase in antioxidant activity comparative to young normal control group, which were pretreated with plane DW (Figure 3(a)). Administration of scopolamine (3 mg kg−1, i.p.) and natural age-induced amnesia increased the brain TBARS levels (p < 0.001) and decreased brain GSH levels (p < 0.001) which were considered as an increase in oxidation activity in brain when compared to normal control group. The administration of BCA 40 mg kg−1 (p < 0.001), 20 mg kg−1 (p < 0.01) and 10 mg kg−1 (p < 0.05) and Pira significantly (p < 0.001) reversed both scopolamine-and ageing-induced deficits increased TBARS level and decreased GSH level of brain (Figure 3(b) and 3(c)).

(a) Each group consists of six animals (n = 6). Values are mean ± SEM. p < 0.05 is considered as significant. ### p < 0.001, # p < 0.01 as compared to normal control. (b) Each group consists of six animals (n = 6). Values are mean ± SEM. p < 0.05 is considered as significant. ***p < 0.001, **p < 0.01, *p < 0.05 compared with scopolamine group and ### p < 0.001 as compared to normal group. (c) Each group consists of six animals (n = 6). Values are mean ± SEM. p < 0.05 is considered as significant. ***p < 0.001, **p < 0.01, *p < 0.05 compared with aged group and ### p < 0.001, ## p < 0.01 as compared to normal group.
Effect of BCA on histopathology of hippocampus of brain
Photomicrograph of hippocampus of brain of (a) normal control, (b) scopolamine control, (c) piracetam-treated group and (d) BCA-treated group are indicated in Figure 4. In the figure, (a) represents normal hippocampus of mice with normal cells, which are intact and no neuron degeneration was observed, (b) indicates scopolamine induced neuronal cell death consistently throughout the hippocampus, indicated by the appearance of pyknotic black neurons, karryorhexis and karyorlysis with condensed nucleus, (c) indicates the mild damage to the hippocampus pretreated with Pira 400 mg kg−1, indicated by the presence of less number of degenerative cells compared with the scopolamine group and (d) indicates less damage to the hippocampus pretreated with BCA indicated by the presence of mild to moderate number of degenerative cells and increased viable cells count as compared to the scopolamine group. The detailed quantification of viable cells and neurodegenerative cells has been carried out and indicated in Table 6.

(a) Represents normal hippocampus of mice which shows normal cells, which are intact and no neuron degeneration is observed. (b) Scopolamine-induced neuron death was observed consistently in hippocampus, as indicated by the appearance of pyknotic black neurons, karryorhexis and karyorlysis with condensed nucleus. (c) Shows mild damage to the hippocampus area pretreated with Pira 400 mg kg−1, indicated by the presence of less number of degenerative cells as compared to scopolamine group. (d) Shows moderate damage to the hippocampus area pretreated with BCA 40 mg kg−1, indicated by the presence of medium number of degenerative cells compared to Scopolamine group. Solid arrow indicates the presence of viable neuron cells. Broken arrow indicates the presence of degenerated neuron cells. Pira: piracetam.
Quantification of viable cells and neurodegenerative cells in the photomicrographs of hippocampus.

Discussion
Dementia is a clinical syndrome characterized by the development of multiple cognitive defects that are severe enough to interfere with daily social and professional functioning of life. 36 AD is the most common cause of dementia in the elderly, accounting for 60–70% of all demented cases. In spite of availability of various treatment strategies, the severity and prevalence of this disease are not yet under the control. Therefore, alternative and complementary medicines including herbal supplements are being utilized in the management of the disease. 37,38 Hence, BCA was screened for behavioural and biochemical parameter to develop a novel phytochemical for the betterment of AD.
The EPM has a spatial component since the animals should remember the configuration of the open and enclosed arms and they should escape from the unsafe open arm more rapidly on the second trial. Shortened TL on day 2 trial is used as a parameter for retention or consolidation of memory, while treatment with drugs prior to day 1 acquisition trial. 39 Passive avoidance task is a hippocampal and amygdala-dependent test, 40 which evaluates long-term (24 h) emotional memory. The animals learn to avoid an inescapable electrical shock, and longer retention latencies indicate a better learned experience seen with prior treatment of test (BCA) and standard drug, Pira.
According to the cholinergic hypothesis, memory impairment in patients with senile dementia is due to selective and irreversible deficiency in the cholinergic function of the brain. This serves as a rationale for the use of AchE inhibitors for the symptomatic relief of AD in its early development stage. The drugs that reduce cholinergic function, such as muscarinic receptor antagonist, scopolamine cause profound memory impairment in animals and human beings. 41,42 Moreover, the scopolamine-induced amnesia model is being widely used as a primary screening test for anti-Alzheimer drugs. 43 –45 Scopolamine-induced amnesia leads to an increased calcium influx followed by oxidative stress which in turn increased the activity of AchE. As expected, in the present study, the AchE activity of the whole brain was increased in mice treated with scopolamine when compared with the normal, while scopolamine-induced increased AchE activity was attenuated by standard piracetam and BCA in dose-dependent manner. Hence, BCA may be utilized in the symptomatic relief of AD, which may act in the similar manner of anticholinesterases.
Despite extensive experimental and clinical studies, the neurochemical basis of learning and memory remains controversial. 46 Learning and memory processes involve interactions between various neurotransmitters due to the complex brain system. 32 The neurotransmitters play an important role in learning and memory in experimental animals. 47,48 The ‘inverted U-shaped dose-effect curve’ (IUSDEC) with respect to learning and memory shows that as the dose of the test compound increases the effects initially reach the maximum level and then it starts decreasing. 48 When the IUSDEC is compared with administration of monoamines in normal experimental animals, memory is improved at moderate doses and impaired at high doses. 49 –54 Noradrenaline and dopamine are known as excitatory neurotransmitters of brain biogenic systems. A subchronic effect of an environmental carcinogen shows that elevated level of excitatory neurotransmitters is related to loss of memory and amnesia. 55 Hence, excessive release of dopamine and noradrenaline leads to dementia and memory impairment finally leading to AD. 56 –60 Excessive decrease in the level of neurotransmitters may lead to various pathological conditions like Parkinson’s disease. 61 A standard nootropic memory-enhancing drug piracetam 62 decreases the brain content of dopamine and noradrenaline and their metabolites and overall turnover of central monoamines implicates the involvement of these neurotransmitters in learning and memory processes. In this study, administration of BCA and standard drug piracetam decreases the whole brain content of biogenic amines such as dopamine and noradrenaline insignificantly to a certain extent only, thereby improving the learning and memory, and there is a less chance to have a Parkinson’s disease because only excessive decrease of neurotransmitters lead to Parkinson’s disease. Therefore, BCA improves the mice behavioural performance in PSA that may be linked with decrease in the content of dopamine and noradrenaline levels.
The increase in the production of free radicals increases the lipid peroxidation and reduces the antioxidant enzymatic activity leading to decreased scavenging activity of free radicals, which is been observed in AD brain. 63,64 In the present study, scopolamine and ageing significantly increased the oxidative stress as indicated by the increased TBARS and decreased GSH levels. The administration of BCA for 14 successive days to young and aged mice not only decreased oxidative stress but also attenuated the scopolamine-induced rise in oxidative damage as indicated by the reduced TBARS and increased GSH levels.
The hippocampus is the critical part of the brain concerned with spatial memory and behavioural functions. In addition, lesions of the hippocampus impair performance in many types of tasks that are dependent on the use of spatial information. 65 Hippocampus is the one of first region in brain to suffer in Alzheimer’s disease, encephalitis and medial temporal lobe epilepsy. People with extensive hippocampal damage may experience amnesia and learning and memory disabilities. 66 Neurodegeneration to the hippocampus starts with condensation (pyknosis) of nucleus, followed by the formation of small fragments (karyorrhexis). These fragments get dissolved (karyolysis) and disappearance of nucleus will take place followed by cell necrosis. Pretreatment with Pira and BCA prevented the formation/extent of neuron degeneration by reducing the severity of cell damage and decrease in the count of dead cells and also reduction in the formation of pyknotic black neurons comparative to scopolamine hippocampus. In scopolamine per se-treated group, all these parameters were increased and these results are consistent with the previous findings. 35 As a result, BCA further supports its role as a promising neuroprotective agent.
Conclusion
In the present study, the BCA elevated the ACh indirectly by significant decrease in cholinesterase activity; hence, it improves the memory of both young and aged mice. Due to its antioxidant characteristic by reducing the lipid peroxidation (TBARS) and increasing the GSH levels, it may delay the natural ageing process and neurodegeneration progress. Moreover, this potent neuroprotective characteristic of BCA was further supported by brain biogenic amines and histopathogical findings of brain. In light of the above results, it may be worthwhile to explore the potentiality of BCA as a neuroprotective agent in the management of AD.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Acknowledgement
The authors are thankful to Sri Dambal HV, President, and Dr Kulkarni VH, Principal, SET’s College of Pharmacy, Dharwad, Karnataka, India, for providing the necessary facilities required to carry out the experimental study.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.