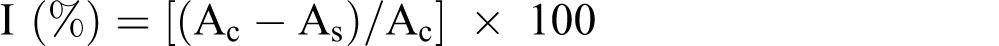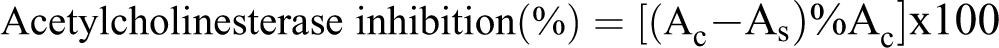Abstract
In the present study, Origanum majorana L. essential oil (EO) was analyzed by gas chromatography-mass spectrometry (GC-MS) and evaluated for free radical scavenging and anticholinesterase activities. GC-MS analysis revealed the presence of 4-terpineol (29.97%), γ-terpinene (15.40%), trans-sabinene hydrate (10.93), α-terpinene (6.86%), 3-cycolohexene-1-1 methanal,a,a4-trimethyl-,(S)-(CAS) (6.54%), and sabinene (3.91%) as main constituents. Origanum majorana L. EO exhibited concentration-dependent inhibitory effects on 2,2′-diphenylpicrylhydrazyl (DPPH•), hydroxyl radical, hydrogen peroxide, reducing power, and lipid peroxidation with IC50 values of 58.67, 67.11, 91.25, 78.67, and 68.75 µg/mL, respectively; while the IC50 values for the standard trolox were noted to be 23.95, 44.97, 51.30, 42.22, and 52.72 µg/mL, respectively. Interestingly, cholinesterase inhibitory activity was also found with IC50 values of 36.40 µg/mL. We can conclude that the marjoram EO has a significant potential to be used as a natural antioxidant and anti-AChE.
Introduction
Free radicals are continuously produced in the body and cause the oxidation of biomolecules (e.g., protein, amino acids, lipids, and DNA) which leads to cell injury and death. The role of free radicals has been implicated in several diseases, including cancer, diabetes and cardiovascular diseases, ageing, etc. 1 Their deteriorative effects can be diminished by natural antioxidants available in foods. Also, the volatile aromatic compounds are employed in the food industry as a flavoring agent. The natural antioxidants present in many plants reduce oxidative damage and help in preventing mutagenesis, carcinogenesis, and aging due to their radical scavenging activities. 2,3 It has been reported that the use of synthetic antioxidants to prevent free radical damage can involve toxic side effects and consumers are becoming more conscious of the nutritional value and safety of their food and ingredients. 4 In this respect, the essential oils and their components of plants have been of interest as sources of natural antioxidants.
Alzheimer’s disease (AD) is frequent in elderly people, as a result of malfunctioning of different biochemical pathways. There are several routes to tackle this problem although the one that has been most successful so far is the “cholinergic hypothesis”. 5,6 The drugs approved for the AD therapy act by counteracting the acetylcholine deficit, that is, they try to enhance the acetylcholine level in the brain. 7 However, according to the cholinergic hypothesis of the pathogenesis of AD, memory impairments in AD patients result from a deficit of cholinergic functions in the brain. Also, an important therapeutic strategy for activating central cholinergic functions has been the use of inhibitors of acetylcholinesterase (AChE), the enzyme responsible for the metabolic hydrolysis of the neurotransmitter acetylcholine. 8 Therefore, the search for new natural AChE inhibitors is of great interest for AD treatment.
Marjoram (Origanum majorana L.) is a tender perennial herb native to North Africa and South West Asia and naturalized in Southern Europe and cultivated in France, Greece, Hungary, the United States, Egypt, and several other Mediterranean countries. It is used as a medicinal plant; marjoram has traditionally been used as stimulant, condiment, and tonic. Furthermore, natural antioxidant from plants (e.g. marjoram) is generally recognized as safe as an extract. 9 Therefore, the present study was undertaken to evaluate the chemical composition, free radical scavenging, and AChE activities of the marjoram (Origanum majorana L.) essential oil through various in vitro models.
Materials and methods
Chemicals
Ascorbic acid was purchased from Sigma–Aldrich Chemie (Steinheim, Germany); 6-hydroxy-2,5,7,8-tetramethychroman-2-carboxylic acid (Trolox) was obtained from Fluka, Buchs, Switzerland; 2-thiobarbituric acid (2, 6-dihydroxypyrimidine-2-thiol; TBA) and 2-deoxyribose from Merck (Germany); 2,2′-diphenylpicrylhydrazyl (DPPH•) from Sigma (St. Louis, Missouri, USA); 5,5'-dithiobis (2-nitro-benzoic acid) (DTNB); acetylthiocholine iodide (ATChI) from Sigma (UK), and sodium phosphate dibasic and monobasic from Alliance Bio (Irvine, California, USA). Kit of haemoglobin (Hb) was obtained from Biodiagnostic, BD, Egypt. All other chemicals were of reagent grades and obtained from the local scientific distributors in Egypt.
Plant material and extraction of essential oil
Origanum majorana L. fresh leaves were collected from local market of Cairo, Egypt, during June 2009. These plants were identified according to the taxonomic characters of Tackholm. 10 The leaves were dried in an oven at 40°C until complete dehydration was achieved, and the essential oil was obtained by hydro-distillation using the Clevenger apparatus. The distillation continued until no more condensing oil could be seen (5 h). The distilled oil was dried over anhydrous sodium sulfate and stored in a freezer at −20°C until analyzed and 1 µL essential oil was diluted in 1 mL diethyl ether for gas chromatography–mass spectrometry analysis (GC-MS) measurement.
Gas chromatography–mass spectrometry analysis
Origanum majorana L. essential oil constituents were analyzed by a gas chromatograph (THERMO TRACE 2000) equipped with a mass spectrometer (FINNIGAN SSQ 7000) GC-MS system (Central Laboratory of the National Research Centre, Cairo, Egypt). The GC was equipped with a fused silica capillary column (DB-5MS; 5% phenyl 95% methylpolysiloxane), 30 m × 0.25 mm x 0.25 µm internal diameter (ID). Helium was used as the carrier gas at the rate of 1 mL/min. The GC-operating conditions were as follows: injector volume, 1 µL; injector temperature, 220°C; column temperature, isothermal at 40°C for 2 min, then programmed to rise up to 250 °C at 5 °C /min; held at this temperature for 2 min; and ion source temperature, 250°C. The effluent of the GC column was introduced directly into the source of the MS. The MS operating conditions were as follows: ionization voltage, 70 eV; scanning interval, 0.5 s, and m/z range, 40–400. Identification of different constituents of Origanum majorana oil was determined by comparing the spectrum fragmentation pattern with those stored in Wiley Mass Spectral Library data.
Animals and care
Eight male albino Wistar rats (Rattus norvegicus) weighing 200−210 g were obtained from Animal Breeding House of the National Research Centre (NRC), Dokki, Cairo, Egypt. Rats were housed in clean plastic cages in the laboratory animal room (22 ± 2°C) on the standard pellet diet and tap water ad libitum, a minimum relative humidity of 40%, and a 12 h dark/light cycle. Rats were allowed to acclimate to laboratory conditions for 1 week. The experimental work on rats was carried out in strict accordance to the guidance for Care and Use of Laboratory Animals as adopted by the Committee of Laboratory Animals of the National Research Centre, Cairo, Egypt.
Blood collection and erythrocytes preparation
A total of 8 mL of blood was collected from all animals (in heparinized tube) under ether anaesthesia by puncturing the retero-orbital venous plexus of the animals with a fine sterilized glass capillary. Within 30 min of blood collection, erythrocytes were isolated by centrifugation at 3500 rpm (600 g) for 10 min at 4°C, using Hereaeus Labofuge 400R, Kendro Laboratory Products GmbH, Germany. The plasma and buffy coat were removed. The erythrocytes were washed three times with ice-cold phosphate buffered saline (PBS: 145 mM NaCl, 1.9 mM NaH2PO4, 8.1 mM Na2HPO4) and centrifuged. The packed erythrocytes after the final wash were used for LPO and AChE assays. Hemoglobin (Hb) concentration was determined spectrophotometrically, based on the colorimetric cyanomethemoglobin method according to Drabkin. 11
Antioxidant activity
Free radical scavenging activity by DPPH• (1-1-diphenyl 2-picryl hydrazyl)
The hydrogen atom-or-electron donation ability of the essential oil was measured from the bleaching of the purple-colored methanol solution of DPPH•. This spectrophotometric assay uses the stable radical, 2,2′-diphenylpicrylhydrazyl (DPPH•), as a reagent.
12
Different concentrations of essential oil (10−160 µg/mL) were mixed with 1.86 mL dimethyl sulphoxide (DMSO) and then added to 1.5 mL of 0.1 mM DPPH• in methanol, and final volume adjusted up to 3.5 mL with DMSO. The mixture was shaken vigorously and allowed to stand at room temperature in the dark for 30 min, and then the absorbance was measured at 517 nm using spectrophotometer (Shimadzu UV-VIS Recording 2401 PC, Japan.). DMSO was used as blank and Trolox (10-160 µg/mL) was used as the reference compound. The absorbance of DMSO and DPPH• radical without oil was measured as control. The radical-scavenging activities of samples, expressed as percentage inhibition of DPPH•, were calculated according to the formula:
Hydroxyl radical scavenging activity
Hydroxyl radical scavenging was carried out by measuring the competition between deoxyribose (2-deoxyribose) and the oil for hydroxyl radicals, which attack deoxyribose leading to the formation of thiobarbituric acid reaction system (TBARS), generated from the Fe3+–ascorbate–EDTA–H2O2 system.
13
Origanum majorana L. essential oil was applied in different concentrations (10−160 µg/mL). Each reaction mixture contained the following reagents, in the final concentrations stated: FeCl3 (100 µM), EDTA (100 µM), H2O2 (2.2 mM), 2-deoxyribose (2.5 µM),
Hydrogen peroxide scavenging activity
The hydrogen peroxide scavenging assay was carried out according to the method of Ruch et al. 15 The principle of this method is that there is a decrease in absorbance of H2O2 upon oxidation of H2O2. A solution of 40 mM H2O2 was prepared in 0.1 M phosphate buffer (pH 7.4). Different concentration of 10−160 µg/mL of essential oil in DMSO was added to 0.6 mL of H2O2 solution (40 mM) and phosphate buffer (pH 7.4) was added up to a final volume of 4 mL. Absorbance of H2O2 at 230 nm was determined after 10 min against a blank solution containing the phosphate buffer without H2O2, using spectrophotometer and Trolox (10-160 µg/mL) was used as the reference compound. The percentage of scavenged [H2O2] = [(Ac-At)/Ac] × 100.
Where Ac was the absorbance of the control and At was the absorbance in the presence of the standard sample or compound oil.
Reducing power
Total reducing capacity of EO was determined according to the method of Oyaizu. 16 One mL of EO at different concentrations (10−160 µg/mL) were mixed with 2.5 mL phosphate buffer (0.2 M, pH 6.6) and 2.5 mL potassium ferricyanide [K3 Fe (CN)6] (1%). The mixture was incubated at 50°C for 20 min, and then a portion (2.5 mL) of TCA (10%) was added to the mixture, which was centrifuged for 10 min at 1000 × g. The upper layer of solution (2.5 mL) was mixed with distilled water (2.5 mL) and 0.5 mL FeCl3 (0.1%). Then, the absorbance was measured at 700 nm. Trolox (10−160 µg/mL) was used as the reference compound. Distilled water was used as blank. A higher absorbance of the reaction mixture indicated a greater reduction power. The reduction powers of samples, expressed as percentage were calculated according to the formula: Reduction powers (%) = [(AC−AS)/AS] × 100 Where: AC and AS are the absorbance of the control and sample, respectively.
Lipid peroxidation assay
The lipid peroxidation (LPO) level is measured as the thiobarbituric acid reactive substance (TBARS), based on the method of Yagi and Rastogi, 17 with some modifications. Erythrocytes were hemolyzed with an equal volume of ice-cold distilled water to yield 50% hemolysate, which was diluted to 1:20 with phosphate buffer (0.1 M, pH 7.4). The final volume of the reaction mixture was 2 mL, which consisted of 1 mL of the hemolysate, 0.6 mL of the buffer containing the essential oil (10−160 µg/mL; the EO was redissolved in the minimum quantity of DMSO), and 0.2 mL of 10 mM H2O2 to start the peroxidation. Samples were incubated for 1 h at 37°C. LPO was measured using the double-heating method of Draper and Hadley. 18 The principle of the method is based on spectrophotometric measurement of the color produced during the reaction of thiobarbituric acid (TBA) with MDA. For this purpose, 2.5 mL of trichloroacetic acid (10%) solution was added into 0.5 mL reaction mixture in test tube and placed in a boiling water bath for 15 min. The mixture was centrifuged at 600 × g for 10 min, and 2 mL of the supernatant was transferred into a new test tube containing 1 mL of TBA (0.67%) solutions and boiling for 15 min. The solution was then cooled and a pink color chromogen read at 532 nm using spectrophotometer. The concentration of malondialdehyde (MDA) was calculated by the absorbance coefficient of MDA-TBA complex (1.56 × 105 cm-1 M-1) and expressed in nmol/gm of Hb. Trolox (10−160 µg/mL) was used as the reference compound.
Erythrocyte AChE activity
Assessment erythrocyte AChE inhibition was carried out using the method of Ellman et al.
19
as modified by Nostrandt et al.
20
Each reaction mixture contained 25 μl in the final concentrations stated of AChE (0.03 U/mL) and DTNB (0.3 mM in phosphate buffer 0.1 M, pH 7 with sodium bicarbonate 0.12 M); 100 μl of EO with different concentrations (10-60 μg/mL) and 1 mL phosphate buffer (0.1 M, pH 8). The mixture was incubated for 15 min at 37°C. Subsequently, the reaction was initiated by adding 25 μL of acetylthiocholine iodide (ATChI) at a final concentration of 0.5 mM and mixture was incubated at 37°C for 10 min. EO was dissolved in DMSO and the final DMSO concentrations in all assays were maintained at 5% (v/v), including controls. The hydrolysis of ATChI was determined by monitoring the formation of yellow 5-thio-2-nitrobenzoate anion as a result of the reaction of 5,5′-dithiobis-2-nitrobenzoic acid with thiocholine released by the enzymatic hydrolysis of acetylthiocholine at 412 nm using spectrophotometer. A control mixture was performed without addition of the EO. The percentage of AChE inhibition was calculated using the following equation:
All determinations were repeated three times (three replicate each) under the same test conditions.
Estimation of IC50 values and statistical analysis
The concentration of the EO induced 50% inhibition (IC50) were performed by probit analysis software. Results were analyzed by SPSS (version 14.0) for Windows and expressed as mean.
Results
Chemical composition of Origanum majorana L. essential oil
The hydrodistillation of leaves of Origanum majorana L. gave colorless oil with a yield of 1.2% (v/w). The chemical composition of EO was analyzed by employing GC–MS, leading to comparison of the relative retention times and the mass spectra of oil components with those stored in Wiley Mass Spectral Library data. The compounds identified and their relative proportions are listed in Table 1 according to their order of elution on DB-5 MS column. The most prominent components were 4-terpineol (29.97%), γ-terpinene (15.40%), trans-sabinene hydrate (10.93), α-terpinene (6.86%), and 3-cycolohexene-1-1 methanal,a,a4-trimethyl-,(S)-(CAS) (6.54%). They constituted about 69.70% of total essential oil.
Chemical composition of Egyptian marjoram (Origanum majorana L.) essential oils

a Compounds listed in order of elution from a DB-5 MS column.
b Rt = Retention time (as minutes).
Free radical scavenging activity
Inhibition of DPPH radical
In DPPH assay, the hydrogen-donating antioxidants were able to reduce the stable radical DPPH to non-radical form DPPH-H. Figure 1 illustrates a decrease in the concentration of DPPH radical due to the scavenging ability of EO and the reference from compound. On DPPH radical, EO and Trolox had scavenging effects with increasing concentration in the range of 10−160 µg/mL, and the scavenging effect of EO was lower. The IC50 were found to be 58.67 µg/mL and 23.95 µg/mL for EO and Trolox, respectively.

DPPH radical scavenging activity of Origanum majorana L. essential oils (EO) and Trolox. Bars represent the means ± SD, n = 9.
Hydroxyl radical (HO•) scavenging activity
In the present study, scavenging activity of HO• was measured on the basis of Fenton reaction. As shown in Figure 2 , Origanum majorana EO (160 µg/mL) exhibited the highest inhibition of about 71.88%, but this is lower than the standard Trolox (160 µg/mL) whose percentage of inhibition is 83.27. The IC50 value of EO was found to be 67.11 µg/mL and 44.97 µg/mL of Trolox, respectively.

Hydroxyl radical scavenging activity of Origanum majorana L. essential oils (EO) and Trolox. Bars represent the means ± SD, n = 9.
Hydrogen peroxide (H2O2) scavenging activity
The hydrogen peroxide scavenging ability of Origanum majorana EO and Trolox are shown in Figure 3 . Hydrogen peroxide scavenging activity of EO at 160 µg/mL was found to be 67.31% and for Trolox at the same concentration was 79.44 µg/mL. The IC50 value of EO was found to be 91.25 µg/mL and for Trolox was 51.30 µg/mL, respectively. The EO was capable of scavenging H2O2 in a concentration-dependant manner.

Hydrogen peroxide scavenging activity of Origanum majorana L. essential oils (EO) and Trolox. Bars represent the means ± SD, n = 9.
Reducing power
As illustrated in Figure 4 , Fe3+ was transformed to Fe2+ in the presence of EO and the reference compound Trolox to measure the reductive capability. As shown in Figure 4 Origanum majorana EO and Trolox had significant inhibition of reducing power with increasing concentration in the range of 10−160 µg/mL. The IC50 were found to be 78.67 and 42.22 µg/mL for EO and Trolox, respectively.

Reducing power of Origanum majorana L. essential oils (EO) and Trolox. Bars represent the means ± SD, n = 9.
Lipid peroxidation
Levels of malondialdehyde (MDA), a major oxidation product of peroxidized polyunsaturated fatty acids, have been considered as an important indicator of LPO. Hence, the ability of Origanum majorana EO to prevent LPO was assessed in RBC by inducing LPO with H2O2. The inhibition of MDA formation in RBC by Origanum majorana EO is shown in Figure 5 . With the increasing concentration of the EO or Trolox from 10 to 160 µg/mL, the inhibition of MDA formation was presented in a concentration-dependent manner. The IC50 were found to be 68.75 µg/mL and 52.72 µg/mL for EO and Trolox, respectively. As shown in Figure 6 , EO and Trolox had free radical scavenging activity, and the scavenging effect of EO was lower than that of Trolox.

Effect of Origanum majorana L. essential oils (EO) and Trolox on lipid peroxidation (LPO) in rat erythrocytes. Bars represent the means ± SD, n = 9.

IC50 values of Origanum majorana L. essential oil (EO) and Trolox of free radical scavenging assays.
Acetylcholinesterase activity
In the present study, Origanum majorana EO showed a concentration-dependent inhibition of AChE activity with IC50 values of 36.40 µg/mL (Figure 7 ). The percentage of AChE inhibition accounted to 5.2, 14.56, 25.89, 58.82, 69.87, and 87.95% at 10, 20, 30, 40, 50, and 60 µg/mL EO, respectively.

Erythrocyte acetylcholinesterase activity of Origanum majorana L. essential oils (EO). Bars represent the means ± SD, n = 9.
Discussion
Reactive oxygen species (ROS) are continuously produced by the body’s normal oxygen usage such as respiration and some cell-mediated immune functions. ROS include free radicals such as superoxide anion radicals (O2 •−), hydroxyl radicals (OH•), and non-free-radical species such as hydrogen peroxide (H2O2) and singlet oxygen (1O2). 21,22 It is increasingly being realized that many of today's diseases are due to the ‘oxidative stress' that results from an imbalance between formation and neutralization of prooxidants. In this respect, ROS play an important role related to the degenerative or pathological processes of various serious diseases, such as aging, 23 cancer, coronary heart disease, Alzheimer’s disease, 24,25 neurodegenerative disorders, atherosclerosis, cataracts, and inflammation. 26 Unfortunately, many synthetic drugs protect against oxidative damage, but they have adverse side effects. An alternative solution to the problem is to consume natural antioxidants from food supplements and traditional medicine. 27,28
In the present study, the most prominent components of Origanum majorana L. EO were 4-terpineol (29.97%), γ-terpinene (15.40%), trans-sabinene hydrate (10.93), α-terpinene (6.86%), and 3-cycolohexene-1-1 methanal,a,a4-trimethyl-,(S)-(CAS) (6.54%). To the best of our knowledge, there are many researches on the chemical composition of the oils isolated from Origanum species. 29,30 These researches indicate that the chemical composition of Origanum oil exist in two forms, one contains terpinen-4-ol and sabinene hydrate as major components while the other one contains thymol and/or carvacrol as predominant compounds. The marjoram essential oil has shown that the volatile aroma composition varies according to the origin of the plant 31,32 and only little artefact formation is found by steam distillation. 33 Also, Nahida et al. 32 reported that the most prominent compounds of the essential oil of Origanum majorana leaf were terpinen-4-ol (47.4%), γ-terpinene (16.4%), α-terpinene (7.9%), and α-terpineol (6.4%), respectively. Our results show a strong similarity with these researches.
The model of scavenging the stable DPPH radical is a widely used method to evaluate antioxidant activities in a relatively short time compared with other methods. The effect of antioxidant on DPPH radical scavenging was thought to be due to their hydrogen-donating ability. Substances capable of donating electrons/hydrogen atoms are able to convert DPPH radical into their non-radical form 1,1-diphenyl-2- picrylhydrazine. Positive DPPH test suggests that the samples were free radical scavengers. In the present study, the scavenging effect of EO and Trolox on DPPH radical was compared. DPPH• was reduced with the increasing of EO concentration in a concentration-dependent manner and a higher DPPH radical-scavenging activity is associated with a lower IC50 value. This result indicates that Origanum majorana EO is considering a good source of natural antioxidants.
The hydroxyl radical (HO•) is the single most toxic radical known and can oxidize all classes of biological macromolecules including lipids, proteins, and nucleic acids at virtually diffusion-limited rates. 34 HO• is known to be capable of abstracting hydrogen atom from membranes and they bring about peroxidic reactions of lipids. 35 In our study, when EO was added to the reaction mixture, it removed the hydroxyl radicals and prevented the reaction. The IC50 value indicates that EO is a better hydroxyl radical scavenger. In this regard, it is anticipated that Origanum majorana EO would show antioxidant effects against LPO on biomembranes and would scavenge HO radicals at the stages of initiation and termination.
Hydrogen peroxide is a weak oxidizing agent that inactivates a few enzymes directly, usually by oxidation of essential thiol (-SH) groups. It can cross cell membranes rapidly; once inside the cell, it can probably react with Fe2+ and possibly Cu2+ ions to form hydroxyl radicals and this may be the origin of many of its toxic effects. 36 From the results, the EO was capable of scavenging H2O2 in a concentration-dependant manner. These results suggest that EO can be a better antioxidant for removing H2O2 and thus protecting living or food systems.
The reducing power reflects the electron-donating capacity of bioactive compounds is associated with antioxidant activity. Antioxidants can be reductants and inactive of oxidants. The reducing capacity of a compound can be measured by the direct reduction of Fe[(CN)6]3 to Fe[(CN)6]2. Addition of free Fe3+ to the reduced product leads to the formation of intense Perl's Prussian blue complex, Fe4[Fe(CN-)6]3, which has a strong absorbance at 700 nm. The reducing ability of a compound greatly depends on the presence of reductones, which have exhibited antioxidative potential by breaking the free radical chain by donating a hydrogen atom. 37 In this regard, increase in Fe3+ to Fe2+ transformation in presence of test sample implies that sample is electron donor and thus can cause reduction of the oxidized intermediates of LPO process. In this assay, the yellow color of the test solution changes to various shades of green and blue depending on the reducing power of antioxidant samples. The reducing capacity of a compound may serve as a significant indicator of its potential antioxidant activity. However, the antioxidant activity of an antioxidant compound have been attributed to various mechanisms, among which are prevention of chain initiation, binding of transition metal ion catalysts, decomposition of peroxides, prevention of continued hydrogen abstraction, reductive capacity, and radical scavenging.
LPO in biological systems has long been thought to be a toxicological phenomenon that can lead to various pathological consequences. 38 The resulting lipid hydroperoxides can affect membrane fluidity and the function of membrane proteins. In addition, lipid hydroperoxides can undergo iron-mediated, one electron reduction and oxygenation to form epoxyallylic peroxyl radicals that trigger a chain reaction of free radical-mediated LPO. The end products of LPO are reactive aldehydes, such as 4-hydroxyl nonenal and malondialdehyde, many of which are highly toxic to cells. 39 However, it has been reported that the damage to lipids (LPO) occurs in three stages: initiation, propagation, and termination reactions. LPO may be induced by radical species, which are sufficiently reactive to abstract a hydrogen atom from the unsaturated fatty acids. This is the starting point for the lipid radical chain propagation reaction. The propagation cycle is broken by termination reactions (two radical species combine to form non-radical final products), which result in the destruction of free radicals.
From the above results in the present study, we can conclude that the EO of Origanum majorana shows potent antioxidant activity and many antioxidant components are found in EO for example, 4-terpineol, γ-terpinene, trans-sabinene hydrate, α-terpinene, sabinene, α-terpinolene, cis-para-2-menthen-1-ol, thymol, and α-thujene. In fact, many EO exhibit antioxidant activity and therefore several studies have been carried out in order to elucidate the activity of their components. 40,41 For example, γ-terpinene retarded the peroxidation of linoleic acid, 40,42 sabinene showed strong radical-scavenging capacity 40,43 as well as α-pinene, 44 while terpinene and terpinolene showed high hydrogen-donating capacity against the DPPH radical, 44,45 terpenes 45 and terpinen-4-ol is a weak antioxidant. 44–46 At higher concentrations, the antioxidant activities of thymol was close to that of α-tocopherol and was in fact responsible for the antioxidant activity of many EOs which contain thymol. 40,41 However, the high potential of phenolic components to scavenger radicals might be explained by their ability to donate a hydrogen atom from their phenolic hydroxyl groups. 47 It has been reported that the antioxidant activity of EOs not only attributed to the major compounds, but also the minor compounds might play a significant role in the antioxidant activity, and synergistic effects were reported. 44
The cholinergic theory of Alzheimer’s disease (AD), originally derived from postmortem studies of the brain, 48 led to the development of licensed drugs based on the inhibition of the enzyme AChE. 49 Treatment with such drugs results in a significant improvement in cognitive function 50 and may also retard the progression of the disease. 51 AChE inhibitors may, however, work more effectively in combination with other agents capable of slowing the neurodegenerative process in AD. 52 In the present study, Origanum majorana EO showed a concentration-dependent inhibition of AChE activity with IC50 values of 36.40 µg/mL. The anti-AChE activity of terpenoids and oils has been previously described. 53,54 It has been reported that the inhibition of AChE by tea tree oil may be due to the presence of more than one terpenoids e.g., terpinen-4-ol, γ-terpinene, α-terpinene, p-cymene and 1,8-cineole, 54 also 1,8-cineole and terpinen-4-ol has been reported to inhibit acetylcholinesterase activity. 55 The cholinesterase inhibition shown by the S. lavandulaefolia oil was shown to be partly due to the cyclic monoterpenes 1,8-cineole and α-pinene, which were shown to inhibit AChE in vitro, with some contribution from other constituents, perhaps by acting synergistically. 56
Recent study have pointed out that AD is associated with inflammatory processes. 57 β-Amiloid peptides contained in the senile plaques found in AD brain can induce these inflammatory processes in which radical oxygen species (ROS) are liberated, among other components. 57,58 ROS are able to damage cellular constituents and act as secondary messenger in inflammation. Antioxidants can scavenge ROS and can also attenuate inflammation pathways. The use of antioxidants may be useful in the treatment of AD. 59
Conclusion
Origanum majorana EO was found to be an effective antioxidant in several in vitro assays (e.g. DPPH•; HO• radical scavenging, hydrogen peroxide scavenging, reducing power, and LPO) and effective as anti-AChE. In this regard, EO could be used in the food industries and other fields that are processing natural products and as anti-AChE.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not for-profit sectors.