Abstract
Introduction:
Anticholinesterase pesticides are widely used in agriculture and domestic settings throughout the world, and they are responsible for great morbidity and mortality. In Egypt and other developing countries, there is a pressing need for new affordable antidotes to treat anticholinesterase pesticide poisoning. Hence, this study was conducted to evaluate the safety and effectiveness of moderate doses of clonidine in the management of adult patients with acute anticholinesterase pesticide poisoning.
Methodology:
This study was an open-label, phase II pilot clinical trial. Sixty patients with acute anticholinesterase pesticide poisoning gave consent to participate in the study. They were divided into 2 equal groups, with 30 patients in each group. Group I received clonidine plus the routine treatment, while group II received only the routine treatment. Patients were subjected to full history taking, and their vital and clinical data were recorded. Serum cholinesterase levels and routine laboratory investigations were measured. The different outcomes of the patients were assessed.
Results:
The baseline characteristics of both groups were similar. Thirteen (43.3%) patients developed significant hypotension during clonidine treatment. The clinical outcomes (including mortality, need for assisted ventilation, length of hospital stay, and total doses of atropine) showed no significant differences between the two groups.
Conclusion:
The use of clonidine in acute anticholinesterase pesticide poisoning may be associated with a high incidence of hypotension requiring intervention. The clinical outcomes may not significantly improve in clonidine-treated patients.
Introduction
Anticholinesterase pesticides, including organophosphorus (OP) compounds and carbamates, are a diverse group of highly toxic chemicals. 1 The World Health Organization reported about 1 million accidental and 2 million suicidal poisonings with OP insecticides per year, with more than 300,000 fatalities. 2 In Egypt, OP intoxication is a common cause of morbidity and mortality and represents more than 50% of insecticide-poisoned patients. 3
All anticholinesterase pesticides exert toxicity via binding to and phosphorylation of the enzyme acetylcholinesterase causing its inhibition. 4 Exposure to these pesticides can induce acute toxicity (muscarinic, nicotinic, and central nervous system (CNS) manifestations), chronic toxicity, and delayed syndromes (intermediate syndrome, encephalopathy, peripheral neuropathy, and behavioral toxicity). 5
Diagnosis of acute anticholinesterase pesticide poisoning is based on clinical suspicion, characteristic clinical signs, smell of pesticides or solvents, and reduced butyrylcholinesterase or acetylcholinesterase activity in the blood. 6
The traditional antidotes (atropine and oximes, such as pralidoxime and obidoxime) are effective in most cases, but fatalities in some severe cases with such therapies are still occurring. Therefore, new drugs for treatment of this kind of poisoning are urgently needed. 7
Clonidine is a centrally acting α-2 adrenergic agonist, which is traditionally used as an antihypertensive drug. It is known to decrease the presynaptic synthesis and release of acetylcholine. 8 Hence, the aim of this work was to evaluate the safety and effectiveness of moderate doses of clonidine as an adjuvant in the management of adult patients presenting with symptoms and/or signs of acute anticholinesterase pesticide poisoning.
Methods
Study design
An open-label, phase II pilot trial was conducted on 60 patients suffering from acute anticholinesterase pesticide poisoning admitted to the Poison Control Center, Tanta University Emergency Hospital, from April 2013 till October 2013. This study obtained approval from the Research Ethics Committee of Faculty of Medicine, Tanta University.
Patients
Sixty adult patients with history of exposure to, symptoms and/or signs of acute anticholinesterase pesticides poisoning were included after obtaining an informed written consent from each patient or patient’s close relatives for participation. Pregnant females, patients younger than 18 or older than 70 years old, patients who ingested other substances in addition to the pesticide, patients with other serious medical conditions (e.g. cardiovascular disease, renal or hepatic impairment), and patients presenting more than 12 h of exposure to the OP compound were excluded from the study.
Intervention
Patients were allocated to two equal groups. Group I received Catapress® (clonidine hydrochloride) 0.1 mg tablets (the marketing authorizations for Catapress are held by Boehringer Ingelheim Limited, Berkshire, UK, and the tablets are manufactured at Delpharm Reims SAS, France) orally in a loading dose of 0.3 mg, followed by 0.5 mg (divided over 24 h). Blood pressure was measured using sphygmomanometer SK-MINIATUR 300 B, Germany. Hypotension due to clonidine intake was treated with intravenous fluid infusion and temporary delay of clonidine administration. In addition, the standard treatment measures including supportive care, decontamination (by gastric lavage and oral single dose-activated charcoal in case of oral ingestion within 1–2 h), and administration of atropine and obidoxime (Toxogonin®) were provided. Atropine was administered in a bolus dose of 2–5 mg intravenously (IV) and repeated every 10–15 min. Atropine dosing was monitored to resolve bronchorrhea and bronchospasm and to maintain adequate blood pressure and heart rate for tissue oxygenation (systolic blood pressure greater than 80 mmHg and heart rate greater than 80 beats/min). Regular assessment for signs of under-atropinization or over-atropinization was considered to guide the use of further dosing. 9,10 Toxogonin (1 ampule contains 0.25 g of obidoxime chloride in 1 ml, produced by Merck, Darmstadt, Germany), with a loading dose of 250 mg, was administered as a slow IV, followed by continuous IV infusion at a rate of 0.5 mg/kg/h to a maximum of 750 mg/day until at least 12 h after atropine was no longer required. 11 Group II received the same treatment as group I except clonidine.
All patients were subjected to full history taking with emphasis on age, gender, occupation, level of education, circumstances of poisoning, route of exposure, and time passed before hospital admission. In addition, complete physical examination included assessment of consciousness level, general clinical examination, regular monitoring of vital signs (pulse, blood pressure, respiratory rate, and temperature), and laboratory investigations. Although plasma cholinesterase level was used to confirm the diagnosis, it could not be used to correlate with the severity of poisoning.
12,13
Patients in both groups were divided according to the severity of presenting symptoms as follows:
14,15
Mild acute poisoning: fatigue, headache, blurred vision, dizziness, nausea, vomiting, excessive sweating, salivation, abdominal pain, and chest tightness. Moderate acute poisoning: symptoms of mild poisoning in addition to muscle fasciculations, weakness, inability to walk, chest crepitations, and miosis. Severe acute poisoning: symptoms of moderate poisoning plus unconsciousness, flaccid paralysis, respiratory distress, cyanosis, and marked miosis with loss of pupil reflexes.
Outcome measures included reduction of mortality, need for assisted ventilation, length of hospital stay, and total dose of atropine.
Statistical analysis
The values are presented as median values. Multiple-group comparisons were made with Kruskal–Wallis one-way analysis of variance and two-group statistical comparisons were made using Mann–Whitney U test. GraphPad Prism version 6 for Windows (GraphPad Software, San Diego, California, USA, http://www.graphpad.com) was used. The values of p < 0.05 were taken as statistically significant.
Results
The study found no significant difference between the studied groups regarding the patient’s demographics (Table 1) and clinical characteristics (Figure 1). Most cases (65%) presented with accidental ingestion, but some suicidal cases were documented in this study as well. Before treatment the two groups showed no difference regarding their vital data except for oxygen saturation. Routine laboratory investigations and serum cholinesterase levels were almost the same for the two studied groups.
Demographic characteristics (age, gender, marital status, residence, educational level, and work) of the studied groups.

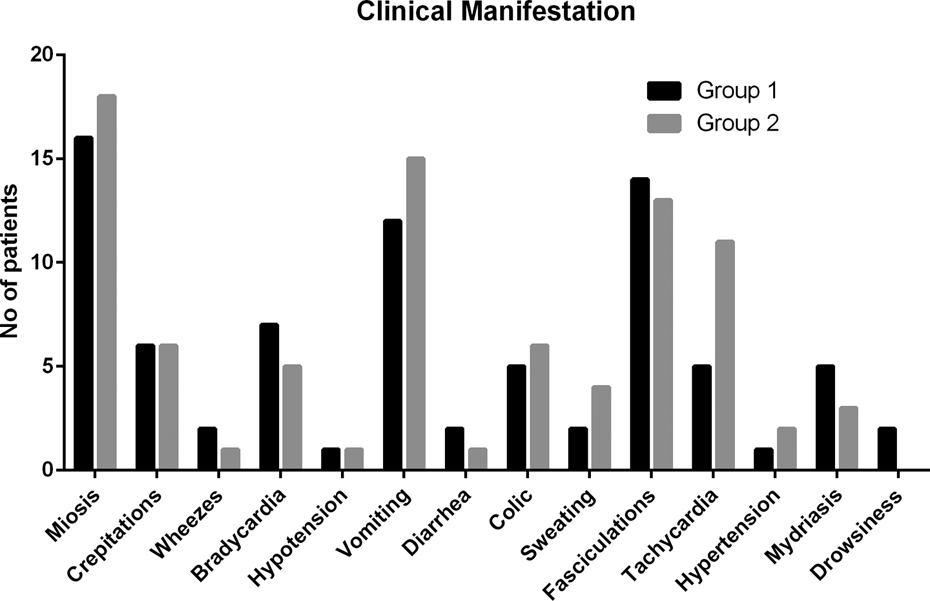
Incidence of different clinical manifestations in the studied groups before treatment.
Table 2 shows the number of hypotensive patients 8, 16, and 24 h after the initiation of clonidine therapy. Of the 30 patients, 13(43.3%) developed significant hypotension (blood pressure less than 80/40 mm Hg) during the early hours of clonidine treatment. Clonidine administration in hypotensive patients was temporarily delayed and IV fluids were given.
The number of patients who developed significant hypotension during clonidine intake in group I.

The study found a significant decrease in the pulse rate and oxygen saturation, but a significant increase in the time passed till normalization of vital signs in clonidine-treated group compared with the other group (Table 3). All patients involved in this study were discharged after treatment without complications except for two patients in group I who suffered from chest infection.
The pulse (beats/min), oxygen saturation (%), and time passed till normalization of vital signs (h) after treatment in the studied groups.

aSignificant.
No significant differences between both groups regarding the length of hospital stay or the total doses of atropine administered were reported. In addition, no mortality was recorded in both groups, and there was no need for assisted ventilation (patients in both groups had either mild or moderate but not severe presentation).
Discussion
This study found no significant difference between the studied groups regarding patient’s demographics, exposure, and clinical characteristics except for oxygen saturation, which showed a slight decrease in group I below normal values. This may be explained in light of the development of respiratory complications in some patients in this group. The observed hypoxemia in these cases may result from respiratory depression, bronchospasm, increased bronchial secretions, and muscle weakness. 16
Cases included in the present study were classified according to the severity of clinical manifestations into mild and moderate cases. No severe cases were included in this study. Both groups included equal number of mild (19 patients) and moderate (11 patients) cases.
The most frequent adverse effects associated with clonidine use are low blood pressure and sedation, but these effects are generally not serious or life threatening. 17 In the present study, hypotension but not sedation was observed during clonidine administration. Clonidine in the setting of acute anticholinesterase poisoning may possibly be less sedating due to the effect of acetylcholine as a CNS stimulant. 18 In group I, blood pressure was measured 8, 16, and 24 h after the start of clonidine intake, and the number of hypotensive patients was recorded to assess for clonidine safety. Hypotension was encountered in 43.3% of patients after 8 h and decreased by time to 30% and 26.6% of patients at 16 and 24 h, respectively, following the initiation of clonidine treatment. Hypotension was treated with IV fluid infusion and temporary delay of clonidine intake, and the blood pressure thereafter returned to normal. None of these patients required vasopressor therapy. The episodes of hypotension may simply be explained by synergism between clonidine and the muscarinic effects of the pesticide. 18
In the current study, vital signs and oxygen saturation were assessed at the end of treatment as well as time elapsed till their normalization. All vital signs normalized after treatment but there was a significant difference regarding pulse between the two groups. This slight reduction in pulse observed in group I compared with group II might be due to clonidine, which acts primarily as a presynaptic CNS α-2 agonist, stimulating receptors in the nucleus tractus solitarii of the medulla oblongata. This inhibits sympathetic outflow, which results primarily in a reduction of sympathetically mediated vasoconstriction, cardiac inotropy, and chronotropy resulting in bradycardia and hypotension. 19 The current study found that the mean time passed till normalization of vital signs was significantly longer in group I compared with group II. This might be due to the time spent to treat hypotension and bradycardia resulting from clonidine intake in group I.
All patients involved in this study recovered after treatment without complications except for two patients in group I who suffered from chest infection. The encountered chest infection in such cases may be due to accumulated secretions, inadequate oxygenation, inadequate atropinization, or muscle weakness. Other studies reported a similar complication in patients with acute OP poisoning. 20
Perera et al. 18 explored the safety of a range of doses of clonidine in acute OP poisoning. The authors found clonidine to be well tolerated at the lowest dosing levels, with hypotension seen only with higher doses. On the other hand, the current study was designed to evaluate the safety and effectiveness of moderate doses of clonidine as an adjuvant in the management of acute anticholinesterase pesticide poisoning. We reported hypotension as the major adverse event for clonidine use, and it was found with the moderate dosing levels used in our study. Both studies found that the use of clonidine therapy didn’t improve the patients’ outcomes significantly. However, both studies were inadequately powered because of the small sample size, and further clinical trials using larger number of patients are required.
Conclusions
We conclude that although clonidine administration as an adjuvant therapy for anticholinesterase pesticide poisoning has been recommended by some authors, the present study showed that clinical outcomes did not improve significantly in clonidine-treated patients compared to those treated with atropine and obidoxime only. In addition, the use of clonidine in these cases was associated with a high incidence of hypotension requiring intervention. However, its use might sound reasonable and acceptable particularly for those cases presenting with hypertension and tachycardia (as nicotinic manifestations) since this drug has been used to treat hypertension for many years without substantial adverse effects.
This study was a small pilot clinical trial designed to guide future studies rather than powered to reach a definitive conclusion about efficacy. Lack of randomization was another important limitation, as awareness of treatment allocation by the treating physician might have influenced the patient selection, monitoring, or the treatment decisions.
Footnotes
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
