Abstract
Uranium (U) accumulates and produces its toxic effects preferentially in the kidneys, especially in the proximal tubular structure. U disturbs the balance of pro-/antioxidants in the renal cortex after acute exposure. Other nephrotoxic agents, such as medications, also cause oxidative stress, but the effects of coexposure are not known. The aim of this study was to analyze the effect of chronic exposure to U and acute gentamicin treatment on the pro- and antioxidant status of the renal cortex of rats. Animals were chronically exposed (9 months) to a nonnephrotoxic level of U (40 mg/L) and then treated with daily injections of gentamicin at a range of doses (0, 5, 25, 100, and 150 mg/kg) during the last week of contamination. We studied changes in the gene expression, protein expression, and enzyme activity of key factors involved in the pro-/antioxidant balance in the renal cortex. At and above a dose of 100 mg/kg, gentamicin decreased the messenger RNA (mRNA) levels of catalase (CAT), copper/zinc superoxide dismutase (SOD) and increased the mRNA levels of heme oxygenase-1 in contaminated rats. This treatment decreased CAT activity, but did not significantly change the SOD protein level. Chronic exposure to U did not worsen these effects in our experimental conditions. In conclusion, gentamicin treatment disturbed the oxidative balance in our model’s renal cortex, but the chronic exposure to U at this nonnephrotoxic level did not appear to reinforce these effects.
Introduction
Uranium (U), because it is both naturally present in the earth’s crust and used by humans for various purposes, is found in the environment, where exposure to it presents risks to local populations. Because of their properties as heavy metals, both natural and depleted U (DU), a by-product of enriched U, are toxic chemicals. In view of its low specific radioactivity, this toxicity dominates its adverse effects on humans. The kidneys are the major target of acute U toxicity. 1 Chronic exposure produces functional kidney changes in humans, 2 and morphological and functional modifications of the proximal tubular cells of rats. 3,4
Induced oxidative stress is one of the principal explanations for these disturbances. It is a common cause of kidney failure induced by numerous metals, including mercury, chromium, cadmium (Cd), and iron 5 as well as U. 6 Oxidative stress is caused by an imbalance between the production of reactive oxygen species (ROS) and a biological system’s ability to detoxify the reactive intermediates or repair the resulting damage easily. Under stress or toxic conditions, ROS product ions can lead to lipid peroxidation, DNA alteration, or sugar oxidation. Cell and tissue functions can be impaired, resulting in various chronic diseases such as kidney failure, diabetes, atherosclerosis, and Alzheimer’s disease.
The body’s antioxidant defense system includes various actors, including enzymes (both catalase (CAT) and superoxide dismutase (SOD)), glutathione (GSH), 7 and transcription factors such as NF-E2-related factor (Nrf2). 8 Lestaevel et al. studied the antioxidant system involved in the brain effects of chronic exposure to U and showed that it induced gene expression of the principal antioxidant enzymes, such as CAT or SOD. 9 However, the effects of chronic exposure on the kidneys have not yet been shown.
Indeed, few published studies have described the in vivo oxidative stress status of the kidneys after chronic exposure to U. 10 No study has examined the effects on oxidative stress of U exposure combined with other nephrotoxic xenobiotics such as gentamicin. In a previous study, we have shown that those renal tubular necrosis were aggravated in DU-exposed rats treated with the highest gentamicin concentration. 11 These alterations were not confirmed by specific nephrotoxic biomarkers analysis (such as kallikrein or Kidney Injury Molecular 1(KIM-1)). But, oxidative stress being a common marker of exposure to DU and gentamicin, it is important to pursue this work by studying the effects of DU and gentamicin coexposure on the pro-/antioxidative system in the renal cortex. Indeed, different mechanisms are known to cause gentamicin nephrotoxicity, but the induction of oxidative stress has been studied in most detail. Gentamicin is an aminoglycoside antibiotic commonly prescribed to treat gram-negative infections, and its nephrotoxicity is well established. 12,13 In the nephron, its target is the proximal tubular cells. 14 Cuzzocrea et al. reports that SOD, an antioxidant enzyme, plays a key role in gentamicin-mediated nephropathy. 15 Similarly, Silan et al. showed that ROS also has a role in gentamicin-induced kidney injury. 12
The objective of this study was to determine whether rats coexposed to DU and gentamicin developed signs of oxidative stress in the kidneys. Adult male rats were exposed for 9 months to a nonnephrotoxic dose (40 mg/L) of DU and were then treated with increasing doses of gentamicin (0, 5, 25, 100, and 150 mg/kg) during the last week of DU exposure.
To assess nephrotoxicity and oxidative status, we studied the following variables in the renal cortex of rats: standard indicators of kidney dysfunction (urea, creatinine, and creatinine clearance) and lipid peroxidation (by measuring thiobarbituric acid reactive substances (TBARS)), as well as the gene, protein, and activity levels of various antioxidant enzymes (CAT, copper-/zinc-SOD (Cu/Zn-SOD) and manganese SOD (Mn-SOD), glutathione peroxidase (GPx), glutathione reductase (GR), and heme oxygenase-1(HO-1)).
Materials and methods
Animals
The study was performed on Sprague Dawley rats provided by Charles River (L’Arbresle, France). Animals were housed in pairs with a 12-h light/12-h dark cycle and under constant room temperature condition (21 ± 1°C). The study was approved by the IRSN Animal Care Committee and conducted in accordance with French regulations for animal experimentation (Ministry of Agriculture Act No. 2011-110, June 2011).
Experimental design
Animal DU exposure
The rats in the contaminated group were exposed via their drinking water to depleted uranyl nitrate (238U: 99.74%, 235U: 0.26%, and 234U: 0. 001%; AREVA-NC Pierrelatte, France) for 9 months at a dose of 40 mg/L (about 1 mg/kg/day). This dose is equivalent to twice the highest concentration found naturally in well water in Finland. 16 Control animals drank no contaminated water. Each group contained 40 animals (Figure 1).
Animal gentamicin treatment
During the last week of U exposure, all the rats in both groups (DU-exposed or not exposed (NE)) received gentamicin or vehicle (sodium chloride (NaCl) 0.9%) treatment (Figure 1). The gentamicin sulfate (Sigma Aldrich, Lyon, France) was administered by subcutaneous injection for four consecutive days (either 0, 5, 25, 100, or 150 mg gentamicin per kilogram of body weight) in 0.9% NaCl, injected at a volume of 1 mL/kg (n = 8 for each condition). The rats were weighed daily during the week of gentamicin treatment.

Groups of rats exposed (DU) or not (NE) to uranium and treated by increasing concentration of gentamicin (5–150 mg/kg) or 0.9% NaCl. DU: depleted uranium; NE: not exposed; NaCl: sodium chloride.
Kidney collection, preparation, and conservation
Animals were anesthetized by inhalation of isoflurane and then euthanized by intracardiac puncture. Both kidneys were collected and weighed for each rat. The cortex and the medulla were meticulously separated, flash frozen in liquid nitrogen, and stored at −80°C. All substances were measured in the renal cortex, which is the principal kidney site for the accumulation of both U 17 and gentamicin. 15
Plasma and urine analysis
Blood was centrifuged at 4000g for 10 min (4°C) to obtain plasma in heparin tube and serum that were stored at −80°C. Urine was collected for 24 h after the last gentamicin injection and then centrifuged at 3000g for 10 min (4°C). The supernatants were collected and stored at −80°C.
The levels of gentamicin, creatinine, and urea in plasma and of gentamicin and creatinine in urine were measured with an automated Konelab 20 analyzer (Thermo Scientific, Cergy-Pontoise, France) (all biological chemistry reagents were obtained from ThermoElectron (Thermo Fischer Scientific, Illkirch, France)). Creatinine clearance was calculated according to this equation
Gene expression analysis
Total RNA from the cortex renal was prepared with the RNeasy Total RNA isolation kit (Qiagen, Courtaboeuf, France), according to the manufacturer’s instructions. High-capacity complementary DNA (cDNA) reverse transcription kits (Life Technologies, Villebon-sur-Yvette, France) were used for the reverse transcription.
Real-time polymerase chain reactions (PCRs) were used to analyze the messenger RNA (mRNA) levels of enzymes involved in the oxidative balance: CAT, GPx, GR, HO-1, Mn-SOD, and Cu-/Zn-SOD. Table 1 reports the sequences for the forward and reverse primers used in this study. In each well, the total volume (10 µL) was adjusted to reach a final concentration of 1 ng/µL of cDNA, with 83% v/v SYBR (Life Technologies), 14.5% v/v sterile water, and 2.5% v/v primers (Life Technologies). Optimized PCR used the AbiPrism 7900 Sequence Detection System (Life Technologies). Samples were normalized to the housekeeping gene, hypoxanthine–guanine phosphoribosyltransferase (HPRT).
Primer sequences for gene encoding antioxidatives enzymes.

HPRT: hypoxanthine–guanine phosphoribosyltransferase; HO-1: heme oxygenase-1; Cu-/Zn-SOD: copper/zinc sodium dismutase; Mn-SOD: manganese sodium dismutase; GPx: glutathione peroxidase; CAT: catalase.
Western blot
Mn-SOD, Cu/Zn-SOD, and HO-1 were semiquantified by Western blotting. Proteins from renal cortex homogenate underwent 10% sodium dodecyl sulfate polyacrylamide gel (SDS-PAGE) electrophoresis and were blotted onto a nitrocellulose membrane. After blocking with 5% nonfat dry milk in Tris Buffered Saline (TBS) for 1 h, the blots were incubated overnight with a primary antibody diluted in 2% nonfat dry milk in TBS at 4°C. Anti-Cu-/Zn-SOD and anti-Mn-SOD polyclonal rabbit antibodies (Merck Millipore, Molsheim, France) and anti-HO-1 polyclonal goat antibody (Santa Cruz Biotechnology, Heidelberg, Germany) were diluted at 1:500, 1:2000, and 1:200, respectively. Immune complexes were revealed by goat anti-rabbit or rabbit anti-goat immunoglobulin G (Santa Cruz Biotechnology) coupled to horseradish peroxidase and luminol-based derivative of Immobilon Western (Merck Millipore). Band densities were quantified with a camera and computer-assisted densitometry (Fuji Las3000, Raytest, Paris, France) and normalized to glyceraldehyde-3-phosphate dehydrogenase or beta-actin (β-actin) detected, respectively, with rabbit anti-goat or goat anti-mouse antibody (Santa Cruz Biotechnology).
Analysis of enzymatic activities
For each assay, 25 mg of renal cortex was homogenized in 10 volumes of appropriate buffer (for CAT and GPx, sample buffer was provided by the manufacturer and for GR, the buffer used was composed of 50 mM potassium phosphate, pH 6–7, and ethylenediaminetetraacetic acid, 1 mM). Tissue samples were then centrifuged at 12,000g for 10 min at 4°C, and the supernatants collected for analyses. Protein concentrations were determined by the Bradford method with serum albumin as the standard.
CAT, GPx, and GR activities were determined with commercial kits supplied by Cayman Chemical (Bertin Pharma, Montigny-le-Bretonneux, France). CAT activity, expressed in nanomoles per minute per milligram of proteins, was assessed via its peroxidatic function, with formaldehyde formation measured spectrophotometrically with a chromogen. GPx activity was assessed indirectly by coupled reaction with GR; the oxidized GSH was recycled to its reduced state by GR and NADPH. NADPH oxidation is accompanied by decreased absorbance at 340 nm directly proportional to GPx activity in the sample. The activity was expressed in nanomoles per minute per microgram of proteins. GR was measured by the NADPH oxidation rate. Its oxidation is also accompanied by decreased absorbance at 340 nm, directly proportional to GR activity in the sample. Results were expressed in nanomoles per minute per microgram of proteins.
Lipid peroxidation
Lipid peroxidation was evaluated by measuring TBARS with a commercial kit supplied by Cayman Chemical (Bertin Pharma). This kit used the reaction between malondialdehyde (MDA) and thiobarbituric acid (TBA) at high temperatures (90–100°C) and acidic conditions to form MDA-TBA and water, measured spectrophotometrically at 530–540 nm.
Statistical analysis
A two-way analysis of variance test was used. The two groups differed according to their exposure to DU (DU and NE). Half the rats in each of these groups were exposed to gentamicin. All results were expressed as mean ± SEM. Statistical software Sigma Plot 11.0 (SPSS, France) was used for all statistical analysis. The level of significance was set at p < 0.05.
Results
Kidney biochemical parameters
Kidney function for all animals was evaluated by standard markers: plasma creatinine and urea and creatinine clearance. No significant effect was observed at doses below 100 mg/kg of gentamicin for DU and NE groups. Plasma urea and creatinine levels increased, as evidenced by the nephrotoxicity (p < 0.001), at doses higher than 100 mg/kg of gentamicin. This increase began at that level in both the DU and NE groups. In NE rats, the increase was significant for both urea and creatinine (by factors of 1.8 and 1.5, respectively, p < 0.01 for both; Figure 2). In DU rats, on the other hand, at the highest dose of gentamicin, only plasma creatinine increased, to a level 1.5 times higher than in the control group (DU without gentamicin). Accordingly, exposure to U had no effect on plasma urea or creatinine.

Kidney physiological and plasmatic parameters in nonexposed (NE) and in DU-exposed (DU) rats treated or not by gentamicin (5–150 mg/kg) for 4 days. Asterisk represents a significant difference between gentamicin treated and untreated (0) rats (n = 8 for each group; two-way analysis of variance, ***p < 0.001). DU: depleted uranium; NE: not exposed.
A dose-dependent reduction in creatinine clearance was observed for NE rats exposed to gentamicin from 25 mg/kg. Creatinine clearance also decreased in DU rats, and gentamicin treatment had no significant effect on this parameter for DU rats. These kidney changes showed that gentamicin treatment disrupted kidney integrity.
Antioxidant enzymes
Gene expression and enzymatic activity were studied for the major enzymes involved in the pro-/antioxidant balance in the renal cortex. In normal conditions, antioxidant enzymes are mainly found in this part of the nephron. 15
The figures present mRNA quantity (in arbitrary units), activity (nanomoles per minute per milligram or nanomoles per minute per microgram), and protein levels (arbitrary units), according to gentamicin treatment (in milligrams per kilogram). The dotted black line represents the NE group and the continuous gray line the DU group.
Catalase
Figure 3(a) shows a significant decrease in CAT gene expression, at a dose of 100 mg/kg gentamicin in both NE and DU-exposed rats (64%, p < 0.01). CAT enzymatic activity (Figure 3(b)) decreased significantly from 5 mg/kg gentamicin for the NE rats (by a factor of 1.8, p < 0.001) and from 100 mg/kg gentamicin for the DU rats (again, by a factor 1.8, p < 0.01). Gentamicin treatment induced a dose-dependent decrease in CAT activity, while the effect of DU exposure was not cumulative with that of gentamicin.

Catalase gene expression (a) and activity (b) in renal cortex of kidney in nonexposed (NE) and in DU-exposed (DU) rats treated or not by gentamicin (5–150 mg/kg) for 4 days (n = 8 for each group). The ratio is expressed as a ratio of the mRNA levels of housekeeping gene hypoxanthine–guanine phosphoribosyl transferase, in AU. The activity is expressed in nanomoles per minute per milligram of proteins. Asterisk represents a significant difference between gentamicin treated and untreated (0) rats (two-way analysis of variance, **p < 0.01, ***p < 0.001). DU: depleted uranium; NE: not exposed; AU: arbitrary units; mRNA: messenger RNA.
Glutathione peroxidase
As shown in Figure 4(a), gene expression of GPx did not change until the gentamicin dose reached 100 mg/kg in NE rats (i.e. at 100 mg/kg of gentamicin for NE rats (−57%, p < 0.01)). Regardless of the gentamicin dose, the GPx level did not differ between DU-exposed and NE rats. Nor did GPx activity (Figure 4(b)) differ with gentamicin treatment or U exposure.

Glutathione peroxidase gene expression (a) and activity (b) in renal cortex of kidney in nonexposed (NE) and in DU-exposed (DU) rats treated or not by gentamicin (5–150 mg/kg) for 4 days (n = 8 for each group). The ratio is expressed as a ratio of the mRNA levels of housekeeping gene hypoxanthine–guanine phosphoribosyl transferase, in AU. The activity is expressed in nanomoles per minute per microgram of proteins. Asterisk represents a significant difference between gentamicin treated and untreated (0) rats (two-way analysis of variance, **p < 0.01). DU: depleted uranium; NE: not exposed; AU: arbitrary units; mRNA: messenger RNA.
Glutathione reductase
GR gene expression did not differ significantly after either gentamicin treatment or DU exposure, as shown in Figure 5. Similarly, neither exposure affected GR enzymatic activity.
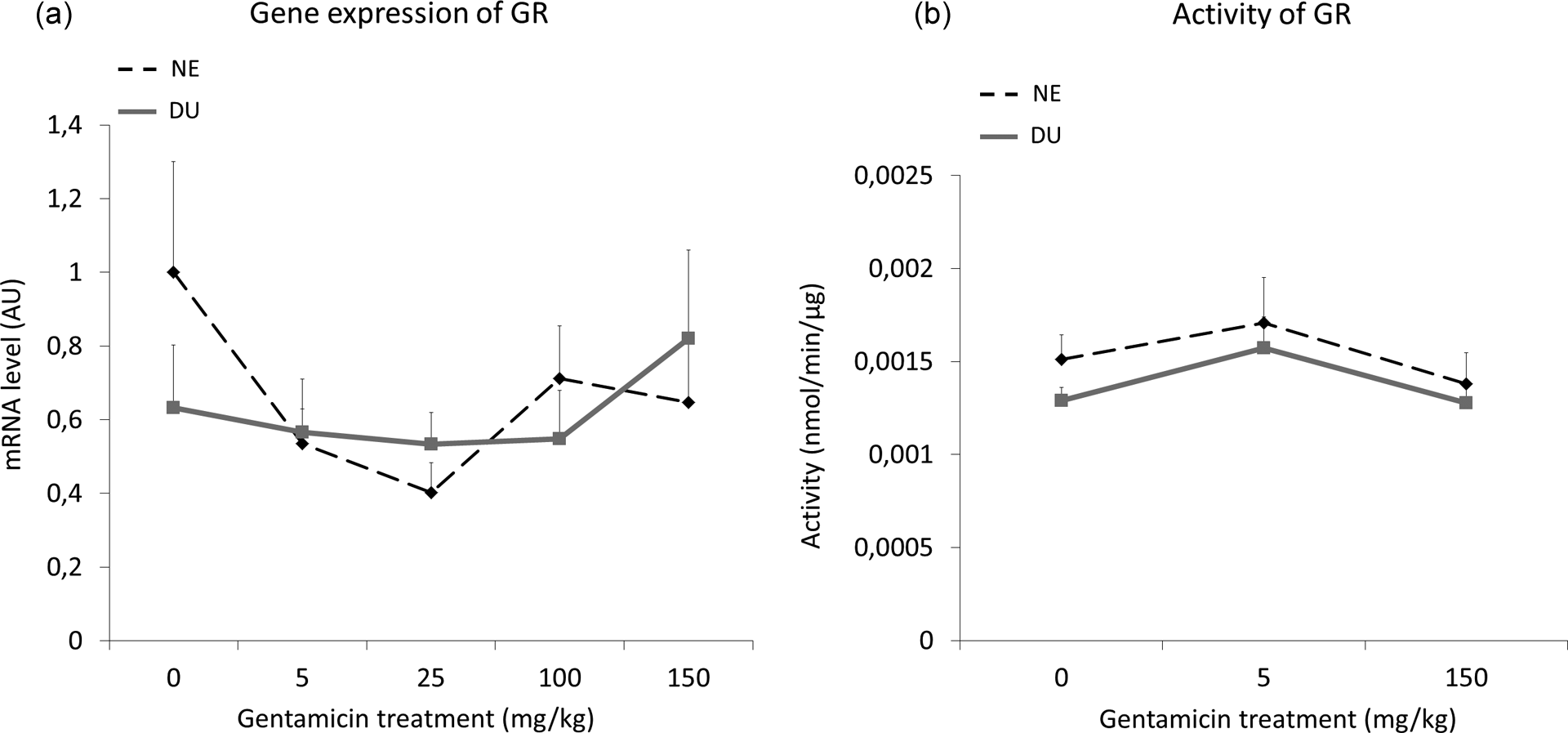
Glutathione reductase gene expression (a) and activity (b) in renal cortex of kidney in nonexposed (NE) and in DU-exposed (DU) rats treated or not by gentamicin (5–150 mg/kg) for 4 days (n = 8 for each group). The ratio is expressed as a ratio of the mRNA levels of housekeeping gene hypoxanthine–guanine phosphoribosyl transferase, in AU. The activity is expressed in nanomoles per minute per microgram of proteins. DU: depleted uranium; NE: not exposed; AU: arbitrary units; mRNA: messenger RNA.
Superoxide dismutases
These metalloenzymes catalyze the dismutation of superoxide anion into hydrogen peroxide and molecular oxygen. There are two principal types of SOD: Cu/Zn-SOD is found in the cytosol and Mn-SOD mainly in the mitochondria.
Mn-SOD
Neither gentamicin treatment nor exposure to U appeared to have any significant effect on either the gene expression or protein levels of Mn-SOD (Figure 6).
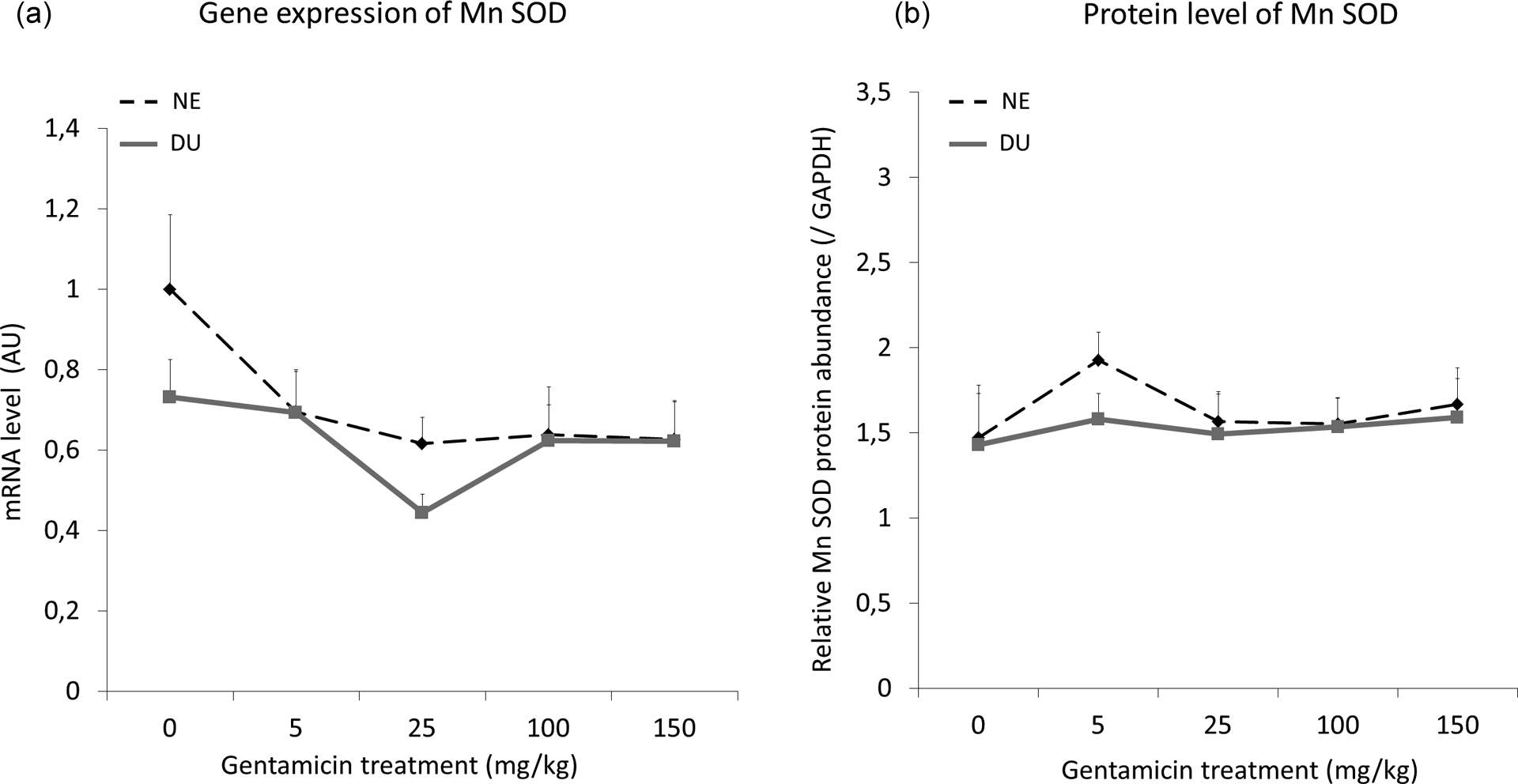
Mn-SOD gene expression (a) and protein levels (b) in renal cortex of nonexposed and in DU-exposed rats treated or not by gentamicin (5–150 mg/kg) for 4 days (n = 8 for each group). In (a), the ratio is expressed as a ratio of the mRNA levels of housekeeping gene hypoxanthine–guanine phosphoribosyl transferase, in AU. For Western blot (b), GAPDH was used as a loading control. Data are expressed as mean ± SEM of the target protein band intensity as compared to GAPDH band intensity. DU: depleted uranium; NE: not exposed; AU: arbitrary units; Mn-SOD: manganese sodium dismutase; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; mRNA: messenger RNA.
Cu/Zn-SOD
Gene expression of Cu/Zn-SOD (Figure 7) decreased significantly NE animals at the highest gentamicin dose (by factors of 2.7, p < 0.001). On the other hand, a significant effect was observed from U exposure in rats treated with 100 mg/kg gentamicin (p < 0.05). Nevertheless, neither gentamicin treatment nor exposure to DU significantly modified protein levels of this enzyme.
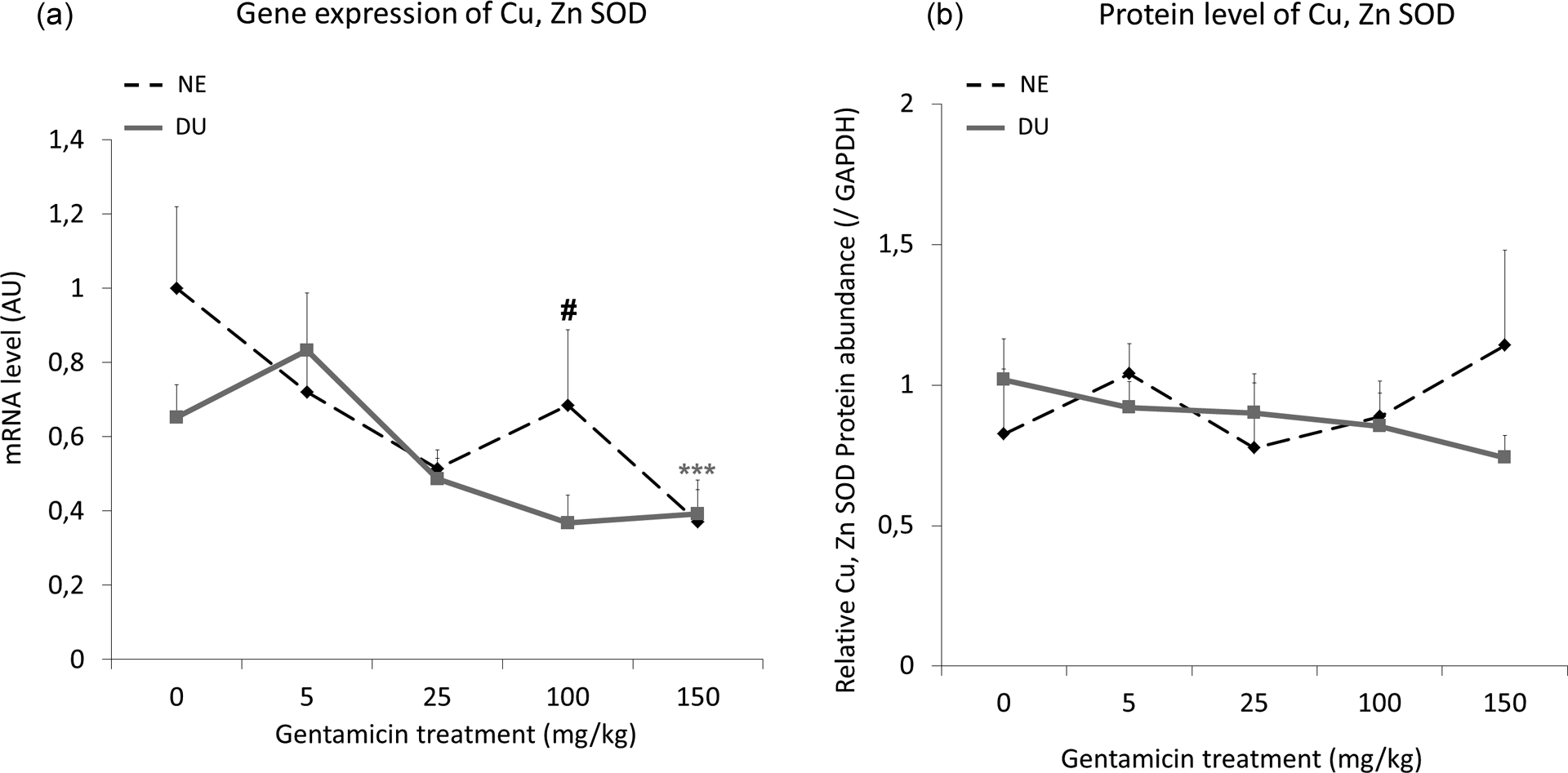
Cu-/Zn-SOD gene expression (a) and protein levels (b) in renal cortex of nonexposed and in DU-exposed rats treated or not by gentamicin (5–150 mg/kg) for 4 days. In (a), the ratio is expressed as a ratio of the mRNA levels of housekeeping gene hypoxanthine–guanine phosphoribosyl transferase, in AU. For Western blot (b), GAPDH was used as a loading control. Data are expressed as mean ± SEM of the target protein band intensity as compared to GAPDH band intensity, n = 8 for each group. Asterisk represents a significant difference between gentamicin treated and untreated (0) rats (two-way ANOVA, ***p < 0.001). Sharp sign represents a significant difference between DU exposed (DU) and nonexposed (NE) rats (n = 8 for each group; two-way ANOVA, #p < 0.05). DU: depleted uranium; NE: not exposed; AU: arbitrary units; Cu/Zn-SOD: copper/zinc sodium dismutase; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; ANOVA: analysis of variance.
Heme oxygenase-1
As shown in Figure 8(a), HO-1 gene expression increased significantly in rats treated with 100 mg/kg of gentamicin in both the DU and NE groups. The protein level did not differ in any of the experimental groups (Figure 8(b)).

HO-1 gene expression in renal cortex of nonexposed and in DU-exposed rats treated or not by gentamicin (5–150 mg/kg) for 4 days (n = 8 for each group). In (a), the ratio is expressed as a ratio of the mRNA levels of housekeeping gene hypoxanthine–guanine phosphoribosyl transferase, in AU. For Western blot (b), beta-actin was used as a loading control. Data are expressed as mean ± SEM of the target protein band intensity as compared to beta-actin band intensity. Asterisk represents a significant difference between gentamicin treated and untreated (0) rats (two-way analysis of variance, **p < 0.01, ***p < 0.001). DU: depleted uranium; NE: not exposed; AU: arbitrary units; HO-1: heme oxygenase-1; mRNA: messenger RNA.
Lipid peroxidation
We measured TBARS to assess lipid peroxidation. They did not vary significantly in any of the groups considered: DU versus NE rats or gentamicin versus NaCl treatment (Figure 9).

Evaluation of lipid peroxidation by TBARS measurement in renal cortex of nonexposed (NE) and in DU-exposed (DU) rats treated or not (0) by gentamicin (5–150 mg/kg) for 4 days (n = 8 for each group). Concentration of MDA is in micromoles per liter. DU: depleted uranium; NE: not exposed; TBARS: thiobarbituric acid reactive substances; MDA: malondialdehyde.
Discussion
It has been clearly established that U is a nephrotoxic agent 1,22 and that the first symptom of this toxicity is tubular nephritis. 1,23,24 The physiological mechanism underlying this renal damage has nonetheless not been completely elucidated. One of the mechanisms for this nephrotoxic action of U mentioned most often is an imbalance in the oxidative status of the kidneys, 6 more precisely in the renal cortex of the nephron, and specifically in the renal proximal tubular cell. 24,25 Increased lipid peroxidation in the renal cortex was observed in rats after intraperitoneal (i.p.) injection of 0.5 mg/kg uranyl nitrate for 5 days. 24 After contamination of mice by i.p. injection (0.5 mg/kg) of uranyl nitrate for 2 days, Taulan et al. reported some changes in the levels of the antioxidant enzymes SOD and GPx, which are involved in oxidative equilibrium. 26
Increased oxidative stress has also been observed in other organs after acute exposure to uranyl nitrate, including the central nervous system (CNS), bones, and lungs. 27 –29 Similar results are reported in other species: uranyl nitrate induces oxidative stress in the proximal tubular cells of rabbit kidneys 30 but also in the liver of Danio Rerio fish. 31 Such stress, however, has not been studied previously in the kidney of rodent after chronic exposure to a nonnephrotoxic U dose. In our work, U concentration in the kidneys was less than 0.15 µg/g of kidney, a nonnephrotoxic concentration.
Here, we used a rat model of chronic exposure developed in the laboratory 11 that enabled us to study simultaneously the effects of chronic exposure to U, acute gentamicin treatment and coexposure to these two potentially prooxidant agents. It is important to note that DU exposure induces any significant modification in the renal plasma biochemical parameters. These results are in line with previous studies conducted on rats exposed to 40 mg/L of DU during 9 months. 32,33
To study if antioxidant status could be modified, we first analyzed lipid peroxidation (measured by the quantity of TBARS formed) in this model and found no significant change after chronic exposure. In comparison, after 3 months of contamination by increasing U concentrations (10–40 mg/kg), 10 no increase in TBARS activity was observed at the lowest dose, which was also the dose closest to that used in this study. But there was a significant increase at the highest concentration, almost 20 times higher than in our study. This increase was correlated with the U concentration in the kidneys. The discrepancy between these studies can be explained by the U dose. Nonetheless, in the study by Linares et al., the renal activity of the antioxidant enzymes evaluated (GPx and GR) did not change as it did in this study.
The effect of chronic exposure to U on pro-/antioxidant status has been studied in other tissues, including the testes and the CNS. Linares et al. 10 found in another study that GR activity and GSH level decreased in the testis, while Lestaevel et al. showed increased activity of some antioxidant enzymes in the CNS. 9
These enzymes are not altered by chronic exposure to U in the kidney at this dose, as mentioned previously in this text. So, adaptation mechanisms could occur beforehand.
These adaptation mechanisms have been reported in a review for various toxic substances. 34 Cd is another toxic heavy metal, which is also a nephrotoxic agent. 35 It can induce oxidative stress. 36 –38 Both increased 36 and decreased lipid peroxidation 39 have been described after chronic Cd exposure. Other authors have shown a biphasic defense to low Cd concentrations, by activation of antioxidant enzymes in the kidneys of chronically exposed mice. 40 Liu et al. reviewed many studies of chronic Cd exposure and hypothesized that compensatory mechanism following the oxidative stress could be developed by the organism in response to Cd chronic exposure. 41 The similarities between Cd and U are that both are heavy metals, nephrotoxic agents, and inducers of oxidative stress; mechanisms of adaptation could appear after chronic exposure to U.
Like heavy metals, some drugs also induce oxidative stress. Gentamicin is a well-known nephrotoxic antibiotic 42 and one of the predominant mechanisms of this nephrotoxicity includes the induction of oxidative stress. 43 In our study, the use of increasing concentrations of gentamicin led to a dose-dependent pro-/antioxidant imbalance, demonstrated by decreased CAT activity and gene expression of CAT and Cu/Zn-SOD. In similar experimental conditions, decreased activities of the antioxidant enzymes CAT, SOD, and GPx were described. 44
The two main isoforms of SOD were studied: Mn and Cu/Zn. Gene expression of Cu/Zn-SOD was altered by gentamicin treatment. These variations were not observed at the protein level. It suggested that the transcription of this gene encoding for these enzymes was disturbed by gentamicin treatment, whereas it had no effect on the protein translation.
Gene expression of Cu/Zn-SOD is significantly different between NE and DU groups only at a dose of 100 mg/kg of gentamicin, which is described as nephrotoxic.
In this work, it can be supposed that the difference in gene expression between both SOD isoforms is probably due to their cellular localization and the effect of gentamicin and U in the different cellular compartments. Indeed, Mn-SOD is mainly localized in the mitochondria, whereas Cu/Zn-SOD is found in the cytosol. Recently, we have shown in an in vitro study that the U distribution is heterogeneous between the nuclear and the cytoplasm, 45 but the U and gentamicin localization in different organelles is not yet known. Nevertheless, this result leads to suggest that U localization being heterogeneous, U effects on organelles are heterogeneous too, that could explain the different effects between Cu/Zn- and Mn-SOD.
As reported above, lipid peroxidation in our model did not vary with gentamicin treatment or exposure to U or both together. Other studies report that gentamicin induces TBARS production during longer treatment, 46 but coexposure to another xenobiotic was not studied. HO-1, which is another important antioxidant enzyme, was significantly induced starting at a dose of 100 mg/kg gentamicin in our model, as reported previously. 47 However, chronic exposure to U at a dose of 40 mg/L produced no synergistic effects.
Overall, according to our model, no potentiations of gentamicin’s prooxidant effect were observed. Only some sporadic changes in the levels of antioxidant enzymes were observed due to U exposure, consistent with what has been reported previously about nephrotoxicity parameters. 11 Nevertheless, the variations were mainly observed at the gene expression level. In order to complete this study, in vitro kinetics studies are necessary to clarify cellular pathways involved in coexposure to U and gentamicin.
Footnotes
Acknowledgments
The authors thank C. Baudelin, T. Loiseau, and F. Voyer for their assistance during the animal experiments.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was part of the ENVIRHOM research program supported by Institute for Radioprotection and Nuclear Safety (IRSN).

