Abstract
The aim of this study was to evaluate the effect of lead (Pb)-contaminated drinking water on magnetic resonance imaging (MRI)-estimated cardiac function, vascular reactivity, and serum lipids in rats. For 3 months, male Wistar rats, aged 4–6 weeks, were given drinking water with the addition of lead acetate at a concentration of 100 ppm Pb (10 rats) or water free from Pb (8 control rats). The cardiac MRI was performed at rest and under β-adrenergic stimulation on a 4.7 T scanner using electrocardiogram-triggered gradient echo (FLASH) cine sequence. After 1–2 weeks of the MRI test, experiments were performed ex vivo. After stabilization of perfusion pressure (PP), norepinephrine at doses from 0.01 to 5.0 μg was dissolved in Krebs solution, injected in a volume of 100 μl, and next infused at a concentration of 0.5 μg/ml into the isolated mesenteric artery. In this manner, preconstricted mesenteric bed was used to determine PP changes induced by acetylcholine, given at doses from 0.05 to 5.0 μg, before and during the infusion of nitric oxide synthase inhibitor (1.0 μg/ml). At the end, dobutamine (5 mg), followed by potassium chloride (10.5 mg), was injected. Lipid levels were determined enzymatically, blood Pb level was measured by the atomic absorption spectrophotometer. This study showed that Pb impairs the left ventricular systolic and diastolic function. Pb-induced changes in response to resistance of vessels to vasoactive agents may be secondary to the reduced left ventricular ejection fraction. The high-density lipoprotein subfraction 2 (HDL2) is involved in the cardiovascular effect of Pb.
Introduction
The environmental exposure to lead (Pb) is associated with increased cardiovascular morbidity and mortality. 1 –3 Prospective cohort and clinical studies suggest that low-level cumulative exposure to Pb is connected with disturbances in cardiac conductivity, left ventricular diastolic dysfunction, decreased heart rate (HR) variability, and more frequent incidence of tachycardia. 4 –8 It has been well documented that environmental exposure to Pb is associated with arterial hypertension. 9 –12 Increased blood pressure is associated with moderately elevated concentrations of Pb in the blood or bone. 3,13,14 However, a very low blood Pb level (i.e., 5 µg/dl) is also associated with hypertension. 15
The hypertension induced by Pb in experimental animals is mediated, inter alia, by an increased vessel wall reactivity to adrenergic stimulation and an impaired vasodilatatory effect of acetylcholine. 16 –18 Also the Pb effect on the cardiac muscle contractility and conductance has been documented. 19 However, results of studies performed on Pb-poisoned animals often differ, depending on multiple endo- and exogenous factors. Nevertheless, Pb effects on the heart and blood vessels’ function have rarely been studied simultaneously in the same animals.
The main mechanism of Pb toxicity is an increased generation of reactive oxygen species. 20,21 In rats, a diet supplemented with antioxidants, such as ascorbic acid, tocopherol, or N-acetylcysteine, mitigate the toxic effects of Pb. 22 –24 Pb can also have an impact on lipid metabolism, causing decrease in serum high-density cholesterol (HDL-C). 25 Consequently, decreased HDL may be responsible, at least partially, for disturbances in cardiovascular system. The blood lipoproteins, especially high-density lipoprotein (HDL), exert a vasodilatory effect on blood vessels. 26 In rats, the HDL capacity to produce a vasodilatory effect decreases with age. 27 In people, HDL-C and apolipoprotein A1 levels had a strong, positive correlation with small artery reactive hyperemia. 28 Aside from the vasodilatory action, HDL influences the cardiac function. 29 In rat models, a short-term infusion of reconstituted HDL improved the left ventricle (LV) function, reduced the development of early-stage myocardial lesions, and attenuated postinfarction left ventricular remodeling. 30,31
The aim of this study was to evaluate the effect of Pb-contaminated drinking water on MRI-assessed cardiac function in experiments performed in vivo, vascular reactivity to vasoactive agents examined ex vivo, and serum lipid pattern in the same rats.
Materials and methods
Animals
The study was performed on 16 male Wistar rats of age 4–6 weeks, weighing 200 ± 15 g. The animals were kept in the same room with natural lighting cycle, at a stable temperature of 20°C and a humidity of 55 ± 5%. Animals had free access to drinking water and to standard laboratory chow (Labofeed H; WPIK, Kcynia, Poland). For 3 months, the animals were fed with drinking water with the addition of lead acetate at a concentration of 100 ppm Pb (9 rats) and water free from Pb (7 control rats). The water contaminated with Pb was prepared daily. This study was approved by the Local Ethics Committee no 61/2010.
Experiments in vivo: Measurement of cardiac function using MR imaging
The cardiac magnetic resonance imaging (MRI) was performed under inhaled anesthesia (2% isoflurane in oxygen (O2) and air mixture (40:60)) on the scanner consisting of a 4.7 T/310 mm magnet (Bruker, Ettlingen, Germany), a MARAN DRX console (Resonance Instruments Ltd., Witney, UK), Inner diameter (ID) 90 mm gradient coils (Resonance Research, Billerica, Massachusetts, USA), and a custom-built 8-rung radiofrequency birdcage coil (ID 60 mm). Rats were placed in a supine position, their temperature was stabilized using warm air home-built system and controlled with endorectal probe (SA Instruments, Stony Brook, New York, USA). Bright blood cine images were collected using flow-compensated FLASH sequence with cardiac gating (SA Instruments) and the following parameters: ecsho time: 2.5 ms, slice thickness: 1 mm (1.5 mm for stress-test protocol), matrix size: 128 × 128 pixels, spatial resolution: 420 × 390 µm2; repetition time: 5.5–6.4 ms, number of excitations: 8, and a flip angle adjusted for obtaining appropriate contrast between myocardium and blood. The average cardiac cycle time was 157 ± 28 ms, allowing 19 to 32 frames/cycle. Images were collected in short axis covering LV with the 15% gap between slices. Basal image for the stress-test protocol was acquired from the single slice positioned at the papillary muscle level. Then, dobutamine was administered intraperitoneally in a bolus through a catheter (1.5 mg/1 kg of body weight (b.w.)) and two cardiac measurements (8 and 12 min after injection) from which the data describing the most profound changes were chosen for the analysis of cardiac reserve.
Images were segmented semiautomatically with Aphelion software (ADCIS, Saint-Contest, France) to evaluate the LV endocardium area. The volume of LV was calculated as the sum of the slice volumes (area multiplied by slice thickness) evaluated in corresponding time points. Interpolation was made in case of differences in HR between slices. Cardiac function parameters were estimated from the time–volume curve or corresponding time–area curve plot (Figure 1) in case of stress test measurements.

Cardiac function parameters assessed at rest and in stress condition from the time-area curve: end-systolic area (ESA) and end diastolic area (EDA) the minimal and maximal value at the plot before normalization to the EDA (in mm2); fractional area change (FAC) expressed as [(EDA-ESA)/EDA]•100%; ejection and filling rates (ER and FR) were calculated as slopes at the beginning of systole (rapid ejection) and diastole. If that points were not clearly visible ER and FR were obtained by fitting straight line to the points from the first 50% of the phase (both normalized to EDA and expressed as EDA/ms).
Experiments ex vivo: Study of the mesenteric superior artery reactivity
After 1–2 weeks of the MRI test, experiments were performed ex vivo. Rats were fasted from the night before the start of the experiment, and the next day they were anesthetized intramuscularly with ketamine at a dose of 300 mg/kg. An isolated superior mesenteric artery preparation was obtained, which was immediately placed in the chamber of the Harvard perfusion system and flushed with modified Krebs solution in the following composition (in mM): sodium chloride 112.0, potassium chloride (KCl) 5.0, monosodium phosphate 1.0, magnesium chloride 0.5, calcium chloride 2.5, sodium bicarbonate 25.0, and
After stabilization of basal PP, norepinephrine (NE) in doses of 0.01, 0.1, 0.5, 1.0, 3.0, and 5.0 μg was dissolved in Krebs solution and injected in a volume of 100 µl. Subsequently, NE was infused at a concentration of 0.5 μg/ml into the mesenteric artery. In this manner, preconstricted mesenteric arterial bed was used to determine PP changes induced by acetylcholine (ACh), given in doses of 0.05, 0.2, 0.5, 2.0, 3.0, and 5.0 µg, before and during the infusion of nitric oxide (NO) synthase inhibitor (N-ω-nitro-
Each drug was administered every 10 min, after the value of PP returns to the value before administration. The concentrations of all the drugs were expressed in terms of free base and prepared ex tempore. The drugs used for the study were lead acetate (Sigma Chemical Co, St. Louis, MO, USA), norepinephrine hydrochloride (Sigma Chemical Co, St. Louis, MO, USA), L-NOARG (Sigma Chemical Co, St. Louis, MO, USA), acetylcholine chloride (Fluka Riedel-de Haën Laboratory Chemicals, Seelze, Germany), dobutamine-Hexal (Sandoz, Warszawa, Poland), KCl (Polfa SA, Warszawa, Poland).
Biochemical measurements
After preparation of the mesenteric artery, blood samples from the heart were collected, and the animals were killed by cervical dislocation. Blood Pb levels were determined by the atomic absorption spectrophotometer (type SOLAAR M6, Thermo Elemental, Cambridge, UK). The serum total cholesterol, triglycerides, HDL cholesterol, HDL subfraction 2 (HDL2), and HDL subfraction 3 (HDL3) cholesterol were determined using enzymatic assay (SPINREACT, S.A. Ctra Santa Coloma, 7 E-17176 Sant Esteve de Bas, Girona, Spain) by the spectrophotometer Beckman DU 650 (Fullerton, CA, USA). The serum non-HDL-C was calculated as a difference between total- and HDL-C concentrations.
Statistical analysis
Values are presented as a mean ± SEM. Statistically significant effect of treatment was analyzed using one-way analysis of variance, followed by post hoc comparison performed by test RIR Tukey’s or Student’s test. The strength of the linear association between variables was determined by the Pearson correlation coefficient (r). Values of p < 0.05 were considered significant. Statistical analysis was performed using STATISTICA 10.0 software.
Results
Measurement of cardiac function using MRI method
At a similar HR, the resting end-systolic volume or area (ESV or ESA) and end-diastolic volume or area (EDV or EDA) were similar in both Pb-exposed and control rats. However, the mean cardiac ejection fraction (EF) in rats exposed to Pb was lower (p < 0.05) than that of the control. Also in dobutamine test, the mean basal value of fractional area change (FAC), which is analogous to basal EF, was significantly lower in this group in comparison with the control. Dobutamine injected intraperitoneally at a dose of 1.5 mg/kg b.w. significantly (p < 0.001) increased FAC in both groups of rats to values that were slightly (p = 0.14) lower in rats treated with Pb in comparison to control. Simultaneously, dobutamine decreased the ESA (p < 0.001) in all animals and decreased EDA (in both groups p < 0.01) as shown in Table 1. Additionally, dobutamine accelerated the ventricular filling rate (FR) in the control group, while in Pb-exposed rats, dobutamine induced a small change in FR. Thus, dobutamine stimulated FR in Pb-exposed rats, which was lower than that in the controls (p < 0.05) as shown in Table 1.
The resting values of ESV, EDV, EF, and dobutamine test: values of ESA, EDA, FAC, ER, and FR measured before (basal) and after (1.5 D) dobutamine injection (1.5 mg/kg b.w., intraperitoneally) in rats poisoned with lead and controls.a
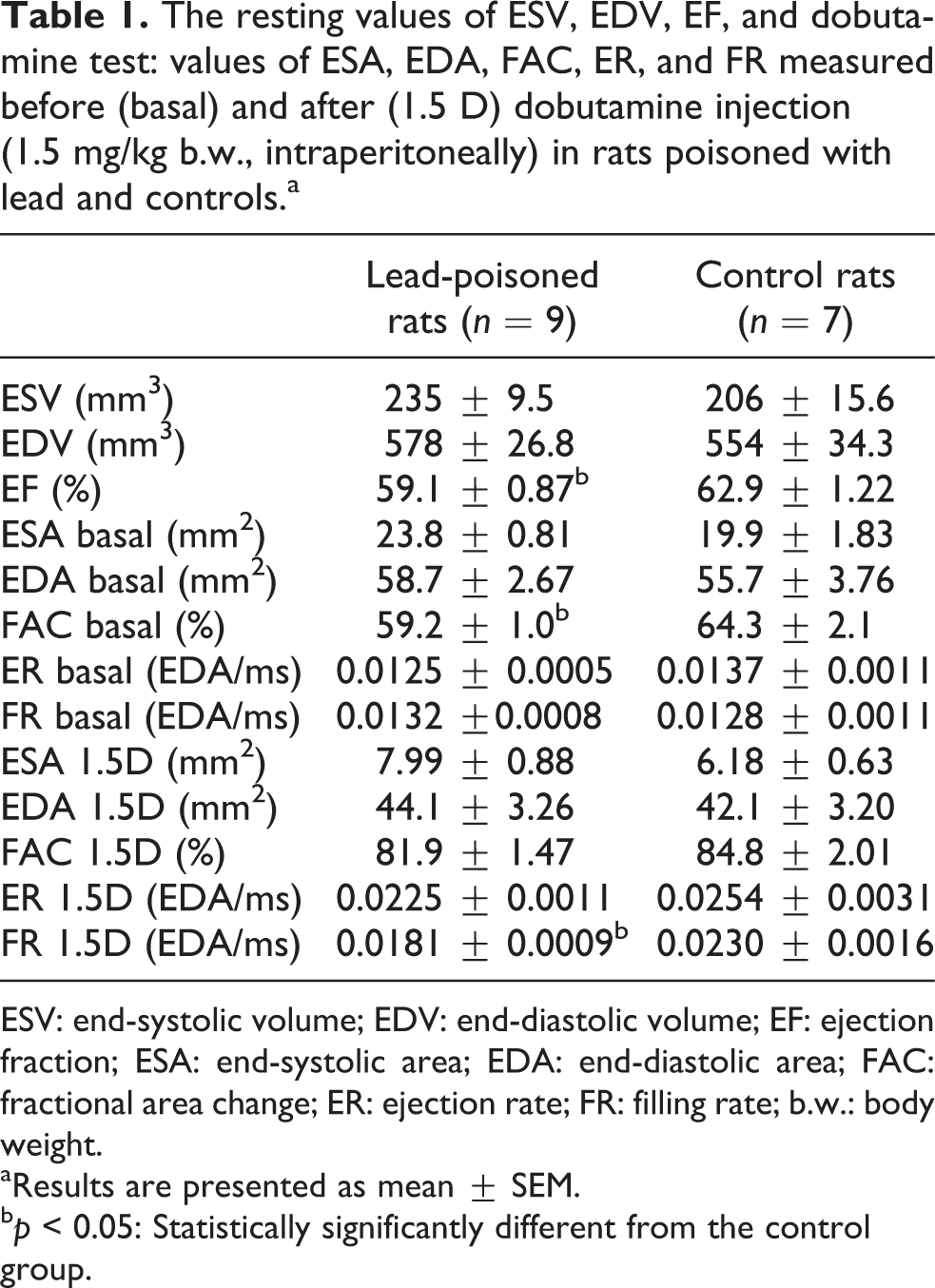
ESV: end-systolic volume; EDV: end-diastolic volume; EF: ejection fraction; ESA: end-systolic area; EDA: end-diastolic area; FAC: fractional area change; ER: ejection rate; FR: filling rate; b.w.: body weight.
aResults are presented as mean ± SEM.
b p < 0.05: Statistically significantly different from the control group.
There was a negative linear correlation between blood Pb level and EF (r = −0.4615; p < 0.05), a positive correlation between blood Pb level and ESA 1.5D (r = 0.5117; p < 0.05), and a negative correlation between blood Pb level and FR 1.5D (r = −0.6110; p < 0.01) in dobutamine test (Figure 2(a) to (c)).

The dependence between blood lead level (Pb-B) and: ejection fraction (EF) at rest (a); end-systolic area (ESA) in dobutamine stress test (b); filling rate (FR) in dobutamine test (c).
Surprisingly, there was also a linear correlation between serum HDL2 cholesterol level and the resting EF (r = 0.6115; p < 0.01), as shown in Figure 3, and between HDL2 and basal FAC (r = 0.5790; p < 0.01).
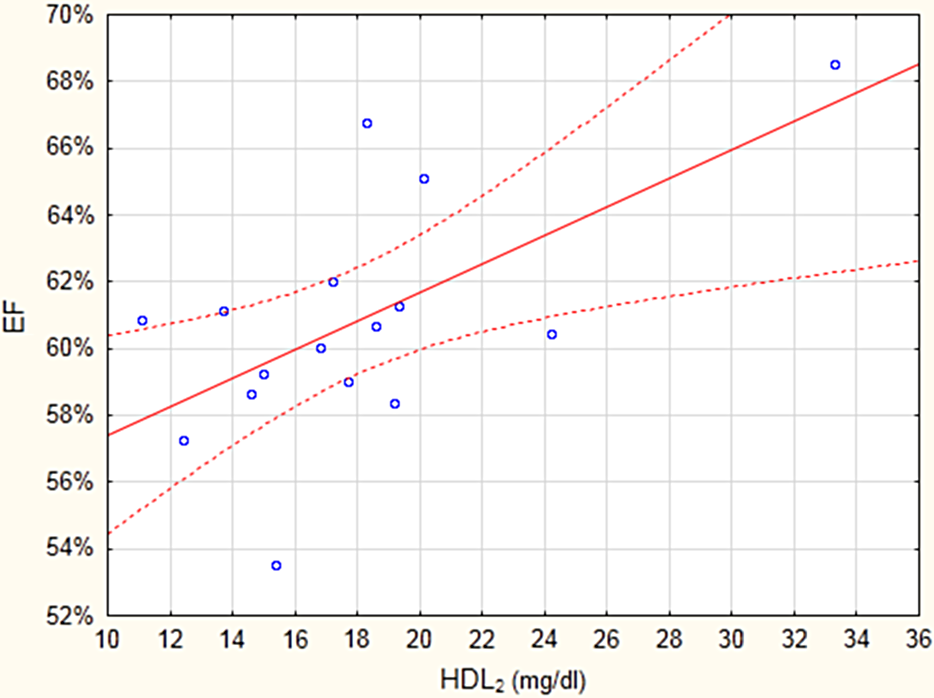
The correlation between serum HDL2 cholesterol and the resting ejection fraction (EF) in rats.
The isolated mesenteric artery reactivity to vasoactive agents
In comparison with the controls, the mean basal PP in mesenteric bed of rats exposed to Pb was about 5 mmHg lower, whereas the reactivity of mesenteric vessels to NE in doses of 0.01–5.0 µg (absolute and relative PP changes) was similar (Tables 2 and 3 and Figure 4). Only the response to NE at a dose of 0.01 µg in rats poisoned with Pb was significantly (p < 0.05) greater than that in controls (Table 3), and there was a linear correlation between blood Pb level and vascular response to NE injected at a dose of 5.0 µg (r = 0.6451; p < 0.001; Figure 5(a)). Simultaneously, there was a linear correlation between FAC measured during dobutamine MRI stress test and PP changes in the mesenteric bed induced by NE at a dose of 5.0 µg (r = −0.5351; p < 0.05; Figure 6). The mean 50% dextrose in water (D50)/NE in Pb-poisoned rat group amounted to 0.65 ± 0.07 µg, whereas in controls it was 0.66 ± 0.08 µg.

The relative changes of the perfusion pressure (expressed in % of the maximum response) induced by norepinephrine (NE) injected at a doses of 0.01 – 5.0 μg into the mesenteric artery of rats treated with lead and controls.

The correlation between blood lead level (Pb-B) and response of the mesenteric artery to: norepinephrine at a dose of 5.0 μg (a); acetylcholine at a dose of 0.05 μg (b), and dobutamine at a dose of 5.0 mg (c).

The correlation between fractional area changes measured in vivo in MRI study during dobutamine stress test (FAC d) and pressor effect of NE injected in vitro into the mesenteric artery at a dose of 5.0 μg.
Values of perfusion pressure in the mesenteric artery measured before vasoactive agents’ injection or infusion into mesenteric bed obtained from rats poisoned with lead and controls.a
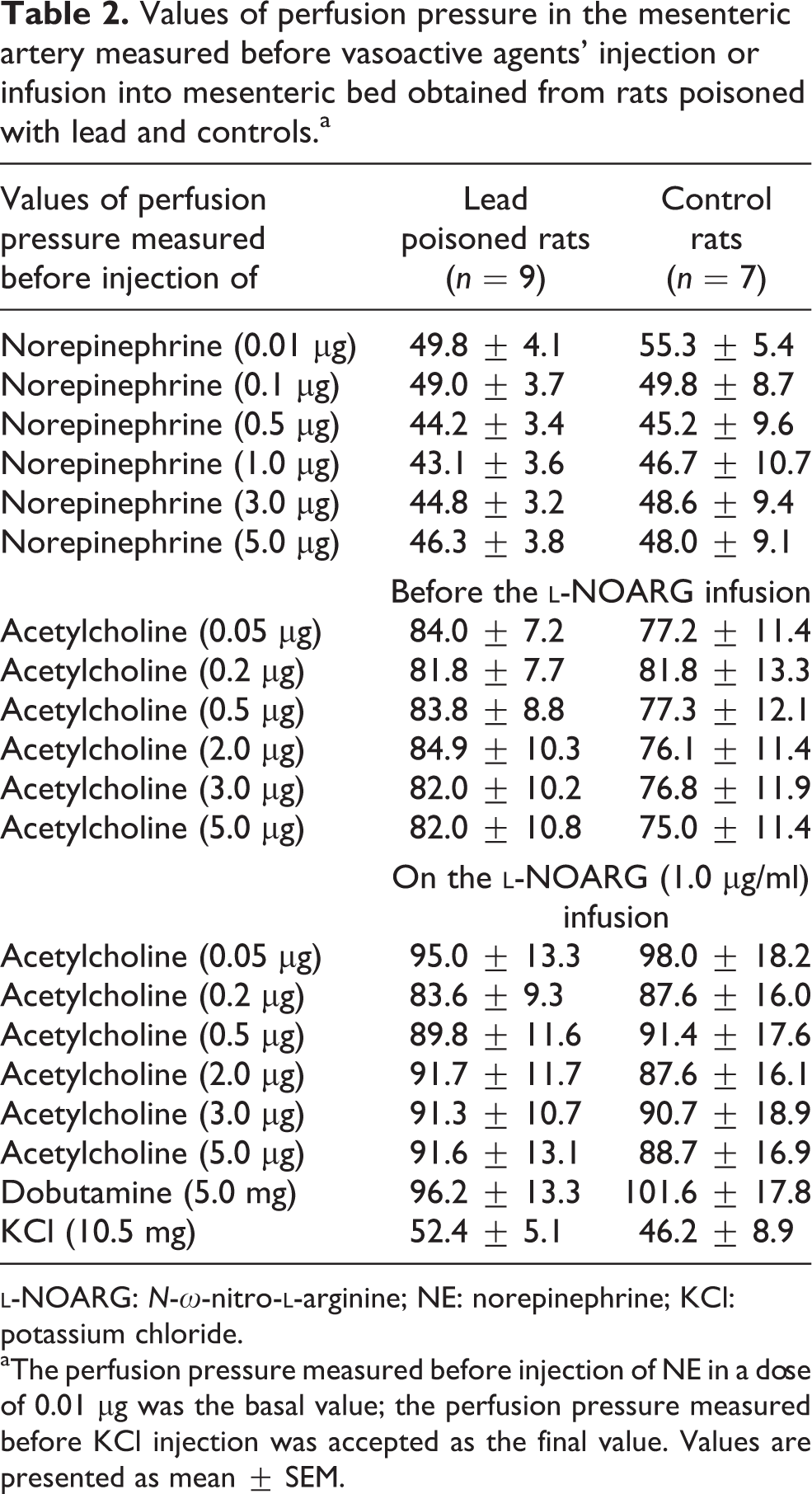
aThe perfusion pressure measured before injection of NE in a dose of 0.01 µg was the basal value; the perfusion pressure measured before KCl injection was accepted as the final value. Values are presented as mean ± SEM.
The perfusion pressure changes (in millimeter of mercury) induced by vasoactive agents in the isolated mesenteric artery bed extracted from rats poisoned with lead and controls.a
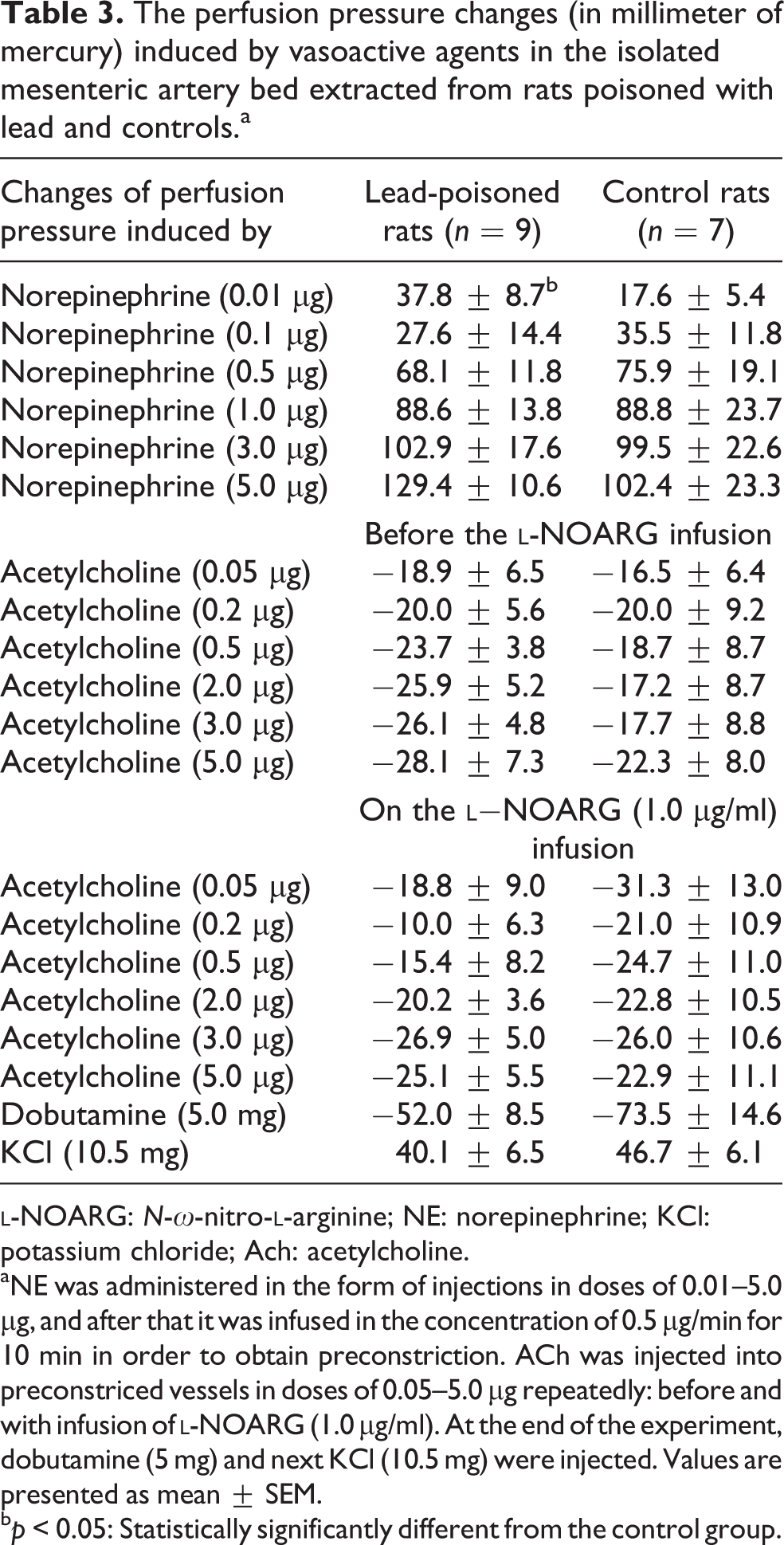
aNE was administered in the form of injections in doses of 0.01–5.0 µg, and after that it was infused in the concentration of 0.5 µg/min for 10 min in order to obtain preconstriction. ACh was injected into preconstriced vessels in doses of 0.05–5.0 µg repeatedly: before and with infusion of
b p < 0.05: Statistically significantly different from the control group.
ACh injected into preconstricted (perfused with NE in concentration of 0.5 µg/min) vessels in doses of 0.01–5.0 µg induced similar PP decreases in all animals (Table 3). However, there was a linear correlation between blood Pb level and vascular effect of ACh injected at a dose of 0.05 μg (r = 0.3993; p < 0.05, Figure 5(b)). The D50 estimated for ACh amounted to 0.99 ± 0.31 µg in Pb-exposed rats and 0.92 ± 0.27 µg in controls. The vasodilatory effect of ACh injected during infusion of the inhibitor of NO synthesis
The vasodilatation induced by dobutamine injected into the mesenteric bed at a dose of 5 mg was lower in Pb-exposed rats by on average of 25 mmHg than that in controls (p = 0.064). Similar to the vascular effect of ACh, there was a linear correlation between blood Pb level and vasodilatory effect of dobutamine (r = 0.4364; p < 0.05; Figure 5(c)). In case of both vasoactive agents, the higher the blood Pb level, the weaker the vasoditatory reaction.
The comparison of PP values measured before ACh injection at a dose of 5.0 µg into preconstricted vessels and in the 10th minute of infusion of
Also, after 40 min of
Blood Pb level and serum lipid concentration
The mean blood Pb level in rats receiving this metal in drinking water was higher (p < 0.001) in comparison with the controls (Table 4). The significant effect of Pb on the cholesterol pattern appeared on the HDL2 cholesterol level. The HDL2 cholesterol was significantly (p < 0.05) lower in animals treated with Pb than that in the controls. There were no significant differences in serum non-HDL-C level between the two groups of rats. Triglycerides’ concentration in serum of rats supplemented with Pb did not differ significantly in comparison with the controls; however, it was about 20 mg/dl higher (Table 4). Body mass of rats measured before the experiment was 212 ± 4.6 g for rats treated with Pb and 228 ± 8.0 g for controls; and before the end of the experiment, it was 452 ± 9.8 g and 438 ± 6.7 g, respectively.
Blood lead level and serum lipid concentration in rats after 3 months of treatment with lead and in control animals.a

Pb: lead; HDL-C: high-density lipoprotein cholesterol; HDL3: HDL subfraction 3; HDL2: HDL subfraction 2.
aValues are presented as a mean ± SEM.
b p < 0.001: Statistically significantly different from the control group.
c p < 0.05: Statistically significantly different from the control group.
Discussion
MRI has been used to identify the function of normal and chronically failing animals’ hearts in vivo, whereas dobutamine-stress MRI allowed noninvasive assessment of systolic and diastolic components of heart failure. 32 In this study, MRI was used for the cardiac function assessment in vivo as it is more accurate and versatile tool than the typically used ultrasound. Heart function is calculated based on changes in left ventricular volume estimated from delineated endocardium. The process of the proper delineation needs sufficient level of the contrast between myocardium and blood. Contrast level may potentially be altered by changes in relaxation processes being consequence of the interaction between protons and paramagnetic ions. However, no significant changes of this kind were observed, so in our opinion such effects even if present does not influence cardiac function assessment.
In our study using MRI, rats exposed to Pb displayed the impairment of systolic cardiac function: a lower EF (p < 0.05) and a lower basal FAC (p < 0.05) in comparison with the control. The existence of the negative linear correlation between blood Pb level and EF (Figure 2(a)) and the positive linear correlation between blood Pb level and ESA in dobutamine stress test (Figure 2(b)) can confirm the weakening of myocardial contractility caused by Pb.
In the MRI stress test, the heart of control rats produced changes typical for adrenergic stimulation, which showed a significant increase (p < 0.001) in FAC, corresponding to the increase in LV EF and a significant decrease in the LV ESA (p < 0.001) and the EDA (p < 0.01) in comparison with the basal values. In comparison with the controls, the dobutamine stimulated systolic response of the hearts of Pb-exposed rats was similar, whereas the stimulated diastolic function was impaired, as proven by the lower FR (p < 0.05) and a slightly greater (n.s.) LV EDV/EDA. The existence of the negative linear correlation between blood Pb level and FR (Figure 2(c)) supports the hypothesis about the diastolic dysfunction induced by Pb. In this aspect, the MRI study showed the weakness of the heart’s contraction at rest and impairment of diastolic reserve in rats exposed to Pb. These results are consistent with some results obtained by others; however, the results of the earlier studies of cardiac stimulation in Pb-poisoned rats are controversial. The Pb-induced rats showed increased cardiac contractility and the level of cAMP, or contrarily, the Pb-induced rats showed decreased β-adrenoceptor and cAMP in the hearts of rats. 17,19 Our data are coherent with the results of Kasperczyk et al. They found that people who are chronically exposed to Pb showed a decreased, in comparison with control, EF assessed by echocardiography. 33 Furthemore, in our study, LV FR failed to increase after dobutamine stress. It is consistent with left ventricular diastolic dysfunction observed in echocardiography in workers exposed to Pb. 6
Experimental poisoning with Pb can lead to various changes in vascular function. Aortic rings taken from rats poisoned with Pb showed an increase or reduction in contractile response to phenylephrine. 18,34 The studies on the impact of Pb on the resistance arteries reactivity showed the shift in the dose–response curve to phenylephrine or metoxamine to the left or unchanged vascular reactivity to α-1 receptor stimulation. 35 –37 The reactivity of mesenteric artery measured in our study using norepinephine in increasing doses was similar in all animals (Figure 4). However, the higher the blood Pb level, the greater was the pressor response to NE at a high dose (Figure 2(a)). Simultaneously, the higher the blood Pb concentration, the lower was the heart EF (Figure 2(a)). Therefore, an increased vascular response to NE, together with an increased blood Pb level, can be a sign of resistance of artery hypersensitivity to catecholamines in case of a non-specific decrease in heart’s contractility. The existence of reverse dependence between FAC and changes in PP induced by NE at a dose of 5.0 µg (Figure 6) points to the physiological and compensatory nature of these changes. The base of this hypersensitivity may be a homeostatic mechanism to maintain a constant flow through the resistance vessels (which include the mesenteric artery bed). In this study, the only difference in the pressor action of NE concerned a very low dose of NE (0.01 µg), comparable to physiological adrenergic stimulation. The response of vascular wall in rats poisoned with Pb was higher (p < 0.05) than that in controls, which may confirm the thesis about the hypersensitivity.
According to this reasoning, the reduced, as compared with control, resting EF in Pb-exposed rats, could be associated with a clearly weaker (p = 0.06) vasodilatory action of dobutamine in the mesenteric bed (Table 3). The negative correlation between blood Pb level and EF (Figure 2(a)), coexisting with reverse dependence between blood Pb level and vasodilatation induced by dobutamine (Figure 5(c)), suggests the existence of a common mechanism of cardiac and vascular Pb toxicity, for example, the impaired function of β-adrenergic receptors. In the heart, dobutamine stimulates β-1-adrenoreceptors; while in the mesenteric bed, dobutamine induces vasodilatation stimulating β-2-adrenoreceptors. 38 Thus, the obtained results could be consistent with the hypothesis of Pb-induced impairment of β-adrenergic response in the cardiovascular system. 19,39 Simultaneously, it is possible that weaker vasodilatatory effect of dobutamine in Pb-treated rats is related to the left ventricular systolic dysfunction induced by Pb.
In rats poisoned with Pb, similar to other researchers, we have observed unaffected relaxation of vascular wall induced by ACh.
40
However, the vasodilatory effect of ACh at a small dose (0.05 µg), similar to dobutamine at a high dose, was smaller, and the concentration of Pb in blood was higher (Figure 5(b) and (c)). The lowering response to vasodilating agents associated with an increase in Pb concentration, leading to increased peripheral resistance, may be involved in the mechanism of Pb-induced hypertension. Another argument pointing to the increased peripheral resistance is the observation of the PP during infusion of
The results obtained thus far might be interpreted, considering some limitations. The main limitation is relatively high blood Pb level in rats drinking Pb-contaminated water was much higher than that of the values measured in populations environmentally exposed to Pb. The degree of exposure to Pb was comparable to one observed in the occupational exposure to this metal. In the future, we will use smaller concentrations.
Our study showed that decreased HDL2 subfraction is involved in the cardiovascular effect of Pb. The association of the low HDL-C with LV diastolic dysfunction was observed in clinical studies. 29 In this study, the impaired cardiac function in rats poisoned with Pb was associated with decreased, in comparison with the controls, serum HDL2 cholesterol (p < 0.05). The linear correlation between serum HDL2 cholesterol level and the resting EF (r = 0.6115; p < 0.01, Figure 3), or basal FAC (r = 0.5790; p < 0.01) indicates the association between HDL2 and heart’s contractility.
As blood lipoproteins, especially HDL, exert a vasodilatory effect on blood vessels, a deficit of HDL may play a role in an increased peripheral resistance. 26,27 A deficit of HDL may be connected with an attenuation of NO-mediated vasodilatation, because HDL regulates the vascular tone via the lysophospholipid receptor S1P3-mediated NO release. 26 It is possible that vascular effect of Pb can involve impaired NO availability resulting from a decrease in HDL concentration. The role of a decreased HDL2 subfraction in such a hypothetic action of Pb needs further studies.
According to the different types of studies (population, clinical, or experimental), the kind of experimental animals (pigeon, rats, rabbits, and calfs), the way of achieving intoxication, and degree of the poisoning, the impact of Pb on lipid metabolism is ambiguous. Most often, hypercholesterolemia is observed. 44 –47 In our study, the lipid pattern in Pb-exposed rats was unchanged, except for a decreased HDL2 cholesterol subfraction, compared with the control. These results are similar to those obtained by Allouche et al. who studied the effects of long-term exposure to low or moderate doses of Pb (0.025, 0.05, 0.1, and 0.3% lead acetate in drinking water for 11 months) on serum lipid profile. The concentrations of total cholesterol, HDL-C, low-density lipoprotein cholesterol, and triglycerides remain unchanged in all Pb-exposed groups, compared with the control. 48 However, the serum HDL-C subfractions were not measured. The selective impact of Pb on HDL2 subfraction is probably observed for the first time in our study.
To conclude, this study showed that exposure to lead is associated with metabolic disturbances, that is, changes in lipid pattern and functional changes in the cardiovascular system. Pb impairs the left ventricular systolic function. It also causes diastolic dysfunction, which was highlighted in the test with dobutamine. Pb-induced changes in response to resistance of vessels to vasoactive agents may be secondary to the reduced left ventricular EF. The HDL2 subfraction is involved in the cardiovascular effect of Pb.
Footnotes
Conflict of Interest
The authors declared no conflicts of interest.
Funding
This study was cofunded by The European Fund of Regional Development, Operational Programme Innovative Economy 2001–2013 Priority 1 Measure 1.3 IE OP, Measure 1.3.1. Project No.: POIG.01.03.01-00-133/08.
