Abstract
Objectives
Testicular dysfunction has been associated with chronic hyperglycemia in diabetes mellitus patients. We investigated taurine’s possible mechanisms and protective effects against testicular damage using a rat model of streptozotocin-induced diabetes.
Methods
Wistar rats (N = 56) were divided into seven equal groups. Untreated control rats received saline, and treated control rats received taurine 50 mg/kg orally. To induce diabetes, rats received a single dose of streptozotocin. Metformin-treated diabetic rats received metformin at a dose of 300 mg/kg. Taurine-treated groups received 10, 25, or 50 mg/kg. All treatments were provided orally once a day for 9 weeks following the streptozotocin injection. Levels of blood glucose, serum insulin, cholesterol, testicular tumour necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), interleukin-1beta (IL-1β), malondialdehyde (MDA), superoxide dismutase (SOD), glutathione peroxidase (GPx), glutathione (GSH), and catalase (CAT) were examined. Sperm count, progressive sperm motility, and sperm abnormalities were examined. Body and relative reproductive gland weights were assessed. Histopathological examinations of the testes and epididymis were performed.
Results
Metformin as well as taurine (in a dose-dependent manner) resulted in significant improvements in body and relative reproductive gland weights, blood glucose, serum cholesterol, and insulin levels, as well as cytokine and oxidative parameters. These findings were associated with significant improvement in sperm count, progressive sperm motility, sperm abnormalities, and histopathological lesions in the testes and epididymis.
Conclusion
Taurine can potentially improve hyperglycemia, hypercholesterolemia, and testicular damage associated with diabetes mellitus, possibly by controlling inflammation and oxidative stress.
Keywords
Introduction
The metabolic disease diabetes mellitus (DM) is characterized by hyperglycemia, polyuria, polydipsia, and polyphagia. The disease may be complicated by hypotension, delayed wound healing, foot ulcers, weight loss, retarded development, hypercholesteremia, and arteriosclerosis. It can also cause serious tissue damage, retinopathy, nephropathy, cardiovascular problems, and sexual dysfunction. 1
Type 1 DM (juvenile-onset) and type 2 DM (adult-onset) are the two types of diabetes. Insulin levels, ketone body buildup, and autoimmune antibodies are the main differences between type 1 and 2 DM. 2 Intracellular hypoglycemia results in lipolysis for gluconeogenesis processes that may cause ketoacidosis and arteriosclerosis and decreased protein synthesis that may result in slowed wound healing, stunted growth, and cachexia. Extracellular hyperglycemia results in diabetic coma and osmotic diuresis. 3
DM may affect both male and female fertility. 4 Reduced libido, a high incidence of genitourinary infections, menstrual cycle irregularities, and anovulation may occur in diabetic patients. 5 Diabetes patients have decreased testicular function, spermatogenesis, sperm count, sperm motility, seminal fluid volume, and testosterone levels compared to healthy individuals. 6
Although free radicals are required for normal signaling pathways, oxidation of cellular components may play a role in the development of multiple illnesses. Disturbances in the generation of pro-oxidants and levels of endogenous and exogenous antioxidants cause oxidative stress, which is linked to various pathological disorders, including diabetes and its complications. 7 Oxidative stress, formed during the diabetic pathway due to energy depletion, leads to reduced flagellar protein phosphorylation and increased lipid peroxidation, resulting in spermatozoa deformity and loss of viability and motility. 8
Taurine (TAU), a beta amino acid containing sulfur that is found in almost all mammalian tissues, is derived from the food and the hepatic metabolism of methionine and cysteine. 9 TAU is necessary for organ function due to its regulatory involvement in osmotic pressure, cell membrane stability, bile salts production, and calcium homeostasis. TAU has been found as the primary free amino acid of sperm cells and seminal fluid and has been detected in human testes. 10
TAU has antioxidant qualities, has a membrane-stabilizing impact, modulates intracellular Ca2+ concentration, inhibits apoptosis, lowers pro-inflammatory cytokine levels in numerous organs, and regulates blood pressure. 11 TAU has been reported to have positive benefits in DM and to play a protective function in other pathophysiological disorders. 12
This work aimed to investigate taurine’s putative protective effects against STZ-induced diabetes and its consequences in rat testicular tissue through its possible anti-hyperglycemic, antioxidant, and anti-inflammatory effects.
Materials and methods
Chemicals
STZ (Sigma, St. Louis, MO, United States) was dissolved in a citrate buffer at pH 4.5. A saline solution was used to dissolve TAU pure powder (Foodchem International Corporation, China). Commercially available metformin pills (500 mg) were used (Minapharm Pharmaceuticals SAE, Egypt). All chemicals were dissolved immediately before use. Phosphate buffered saline (NaCl 8 g/L, KCl 0.2 g/L, Na2HPO4 1.44 g/L, and KH2PO4 0.24 g/L). Colorimetric kits (cat no: MAK187D and MAK085, Sigma Aldrich, USA) were used for testicular immunological investigations. The MDA and TAS (Total antioxidant status) kits were purchased from Egypt’s Biodiagnostic Company (Catalog Number: MD 25 29).
Animals
56 adult Wistar rats (150–250 g) were housed four to a cage and maintained in sanitary conditions at 22°C and 55% humidity with a 12-h light/12-h dark cycle. Rats were given conventional rat pellets and had unrestricted access to water. The Institutional Animal Care and Use Committee of the Faculty of Veterinary Medicine at Cairo University accepted the research protocol, and the animal studies were done according to the ethical principles of animal welfare (Vet CU 8/03/2022/415).
Experimental design
After 14 days of acclimatization, the experimental procedures were conducted. Animals were divided into seven groups, with eight rats per group.
A single intraperitoneal injection of 55 mg/kg STZ was provided to DM model rats (Groups III-VI) to induce sickness. 13 Five days later, a shallow tail puncture was used to get a blood sample. Only animals with blood glucose levels >300 mg/dl were used. After being diagnosed with DM, rats were orally administered either TAU (10, 25, or 50 mg/kg), metformin (300 mg/kg), or saline (1.5 mL) once a day for 9 weeks.
Animal groups
Group I (Negative control, CN rats): received 1.5 mL of saline orally once a day for the duration of the trial.
Group II (TAU-treated control group, TAU rats): received 50 mg/kg TAU solution orally, once daily, throughout the experiment.
Group III (Positive control group, STZ rats): following STZ (55 mg/kg b.wt., I.P.), 13 rats received 1.5 mL saline orally, once daily for 9 weeks.
Group IV (STZ-Metformin-treated DM rats): following STZ, rats received metformin 300 mg/kg orally once daily for 9 week. 14
Group V (STZ-TAU-10-treated DM rats): following STZ, rats received TAU 10 mg/kg orally once daily for 9 weeks. 15
Group VI (STZ-TAU-25-treated DM rats): following STZ, rats received TAU 25 mg/kg orally once daily for 9 weeks. 15
Group VII (STZ-TAU-50-treated DM rats): following STZ, rats received TAU 50 mg/kg orally once daily for 9 weeks. 15
Measurements
The subject’s body mass was measured on day one and then once every week throughout the duration of the trial. At the completion of the trial, all rats were weighed to assess the rise in their body weight. The retro-orbital venous plexus was sampled while the patient was under ketamine anesthesia. Real-time blood glucose levels were measured using a glucometer (Accu-Check Active monitoring system, Roche Diagnostics, Rotkreuz, Switzerland). Cholesterol is measured using sera. 16 After 15 min of centrifuging blood at 3000 r/min, insulin, follicle-stimulating hormone (FSH), and testosterone levels were evaluated using radioimmunoassay techniques. The samples were kept at 20°C until analysis. 17 Animals were euthanized using ketamine (90 mg/kg) and xylazine (5 mg/kg) 0.1 mL/100 gm in a 1:1 ratio. Testes and accessory glands (prostate and seminal vesicles) were estimated in proportion to the final body weight/100 g as relative body weight. Testes were extracted and stored at −80°C for antioxidant and immunological evaluation. During the direct examination of the epididymal tissue for sperm index analysis, progressive forward motility and abnormalities were evaluated. 18 Tissue samples were analyzed for tumor necrosis factor (TNF), interleukin-6 (IL-6), and interleukin-one beta (IL-1β) using colorimetric kits (catalog numbers: MAK187D and MAK085, Sigma Aldrich, USA) according to the manufacturer’s instructions. 19 After a careful abdominal incision and testicular excision, the testicles were removed. Then, three sections of testes were homogenized in 5 mL of phosphate buffered saline (NaCl 8 g/L, KCl 0.2 g/L, Na2HPO4 1.44 g/L, and KH2PO4 0.24 g/L) containing NaCl 8 g/L, KCl 0.2 g/L, Na2HPO4 1.44 g/L, and KH2PO After 20 min of centrifugation at 11200 ×g, the supernatants were tested for malondialdehyde (MDA), glutathione (GSH), glutathione peroxidase (GPx), superoxide dismutase (SOD), and catalase (CAT). The remaining portion was identified by fluid histological examination using Bouin’s solution.
Testicular MDA and other antioxidant levels were assessed in organ homogenates using rat-specific colorimetric test kits per the manufacturer’s instructions. Using the QuantiChromTM Antioxidant Assay Kit, other antioxidant capacity was determined (Catalog number: DTAC-100, Hayward, CA 94,545, USA). Malondialdehyde (MDA): kit acquired from Biodiagnostic Company, Egypt (Catalog Number: MD 25 29). Egypt’s Biodiagnostic Company supplied the MDA test kit (Catalog Number: MD 25 29). The test is based on a 30-min reaction at 95°C between thiobarbituric acid (TBA) and MDA in an acidic media. This reaction resulted to the creation of the reactive product of thiobarbituric acid. The absorbance of the resulting colored substance was determined to be 534 nm. Other antioxidants were measured using the QuantiChromTM Antioxidant Assay Kit (DTAC-100, Hayward, CA 94,545, USA) that reduces Cu2+ to Cu+ using an antioxidant. Cu + reacts with a dye reagent to produce a colored compound. The TAC of the sample is proportional to the color intensity at 570 nm.
Semen analysis
Immediately after dissection, the left epididymis was excised and homogenized in sterile phosphate buffer saline solution for semen analysis. The resulting mixture was examined using an oil emersion medium (100×). A high-powered light microscope (40×) was used to assess individual motility. Alkaline methylene violet staining was used to look for sperm cell abnormalities. Morphological characteristics of at least 200 spermatozoa from various areas of the slide were studied, and the number of abnormalities reported as a percentage of the total number of abnormalities.
Sperm was retrieved from the cauda epididymis using a red blood pipette up to the 0.05 L threshold for sperm count testing. Sperm was diluted with normal saline to a concentration of 101. One drop of diluted sperm fluid was introduced to a Neubauer chamber and counted using a microscope (Krüss MBL2000, A. Krüss Optronic GmbH, Germany) at a magnification of 40; the findings were expressed as 106 cells/ml. 20
A Pasteur pipette was used to combine one drop of sperm with one drop of eosin solution and one drop of nigrosine solution in an Eppendorf tube. Using a high-powered light microscope at 40 (Hund Wetzlar H600/12, Germany) and a digital camera, at least 200 spermatozoa were counted (Canon EOS 550D). Unstained spermatozoa were viewed as alive, whereas stained spermatozoa were seen as dead. 21
Weights of reproductive glands
The reproductive glands were dissected and weighed (testis, seminal vesicle, and prostate). Estimated relative organ weights were (organ weight/body weight) ×100.
Histopathological examination
Testis and epididymis samples were fixed with Bouin’s solution for 48 h. Hematoxylin and Eosin staining followed standard histological protocols (dehydration, washing, paraffin embedding, microtomy of 5-m-thick slices). 22 The testes and epididymis were dissected and fixed in Bouain’s fixative for 24 h. The tissues were washed repeatedly with 70% ethanol until the yellow hue of Bouin’s fixative was no longer discernible. The tissues were gradually dehydrated by putting them through more concentrated ethanol solutions (70, 90, and 100%). Paraffin wax was then used to enclose the tissues. Embedded blocks were cut using a 5–6 mm microtome and stained with hematoxylin and eosin as normal (H&E). After the staining operations were completed, slices were viewed under a microscope. Normal and pathological testicular and epididymal structures were evaluated, including lumen diameter, epithelial structure, spermatocytes, and secondary spermatids, as well as sperm presence or absence in the epididymal lumen.
Statistical analysis
The mean and standard deviation of the mean were provided for the data. Statistical significance was established using SPSS version 20.0 software (IBM SPSS Statistic 20.0, Armonk, NY, USA) and a one-way analysis of variance, followed by Tukey’s post hoc test for multiple comparisons. p < 0.05 was considered to be statistically significant. 23
Results
Body weight and relative weight of testes, prostate, and seminal vesicles
After four and 9 weeks, the body weight of normal rats in the negative control group (Group I) and TAU-treated rats (Group II) considerably increased compared to their respective day one weights (p < 0.05). STZ rats saw the least amount of weight gain (Group III). This weight reduction was considerably enhanced in the metformin (Group IV) and placebo (Group TAU) groups (Groups V-VII) (p < 0.05) (Figure 1(a)). Body weight measured on day zero, 4 weeks, and 9 weeks of the experiment. While Relative weight of testes, prostate, and seminal vesicles in relation to whole body weight at the 9th week of the experiment. (Mean ± SE) (each group = eight adult Wistar rats) (Mean values with different superscript letters revealed significant differences from each other at p < 0.05), in between different groups and at the different times from Day of zero till 9 weeks. CN: Group I (negative control group), TAU: Group II (TAU-treated control group), STZ: Group III (positive control group), STZ-MET: Group IV (Metformin-treated diabetic rats), STZ-TAU-10: Group V (TAU-10-treated diabetic group), STZ-TAU-25: Group VI (TAU-25-treated diabetic group), and STZ-TAU-50: Group VII (TAU-50-treated diabetic group).
There was an insignificant difference in the improvement of body weight in Group V and Group VI, treated with TAU 10 and 25, respectively, compared with Group IV, treated with metformin at the end of week four. However, treatment with TAU 50 (Group VII) led to a significant increase in body weight compared with rats treated with either metformin (Group IV), TAU 10 (Group V), or TAU 25 (Group VI) at the end of weeks four and nine (p < 0.05) (Figure 1(a)).
Regarding the relative weight of the reproductive glands, TAU (Group II) produced significant growth of the prostate and seminal vesicles compared with untreated control rats (Group I) (p < .05). STZ in Group III produced a highly significant reduction in the relative weight of the testes, prostate, and seminal vesicles compared with untreated control rats (Group I) (p < .001). This reduction in relative weight showed marked significant improvement by treatment with metformin, TAU 10, TAU 25, and TAU 50 in Groups IV, V, VI, and VII, respectively (p < 0.05) (Figure 1(b)).
There was a dose-dependent improvement in the relative weights of the glands by TAU in Groups V, VI, and VII, with the greatest improvement in Group VII, which succeeded in regaining weight to that of untreated control rats (Group I) (Figure 1(b)).
Testicular oxidant/antioxidant status
The levels of testicular pro-inflammatory cytokines (IL-6, IL-1β, and TNF), MDA, and testicular antioxidants (SOD, GPx, GSH, and CAT) did not change significantly between untreated control rats (Group I) and TAU-treated rats (Group II) (p > 0.05). STZ-treated rats (Group III) demonstrated a very significant increase in testicular concentrations of TNF, IL-6, IL-1β, and MDA (p < 0.05), followed by a highly significant drop in testicular concentrations of SOD, GPx, GSH, and CAT (p < 0.001). (Group I) (Figures 2 and 3). Testicular levels of superoxide dismutase (SOD), malondialdehyde (MDA), catalase (CAT), glutathione (GSH) and glutathione peroxidase (GPx) (Mean ± SE) (each group = eight adult Wistar rats) (Mean values with different superscript letters revealed significant differences from each other at p < 0.05), in between different groups and at the different times from Day of zero till 9 weeks. CN: Group I (negative control group), TAU: Group II (TAU-treated control group), STZ: Group III (positive control group), STZ-MET: Group IV (Metformin-treated diabetic rats), STZ-TAU-10: Group V (TAU -10-treated diabetic group), STZ-TAU-25: Group VI (TAU-25-treated diabetic group), and STZ-TAU-50: Group VII (TAU-50-treated diabetic group). Testicular levels of tumor necrosis factor (TNF), interleukin-6 (IL-6), interleukin-1 and beta (IL-1β). (Mean ± SE) (each group = eight adult Wistar rats) (Mean values with different superscript letters revealed significant differences from each other at p < 0.05), in between different groups and at the different times from Day of zero till 9 weeks. CN: Group I (negative control group), TAU: Group II (TAU-treated control group), STZ: Group III (positive control group), STZ-MET: Group IV (Metformin-treated diabetic rats), STZ-TAU-10: Group V (TAU-10-treated diabetic group), STZ-TAU-25: Group VI (TAU-25-treated diabetic group), and STZ-TAU-50: Group VII (TAU-50-treated diabetic group).
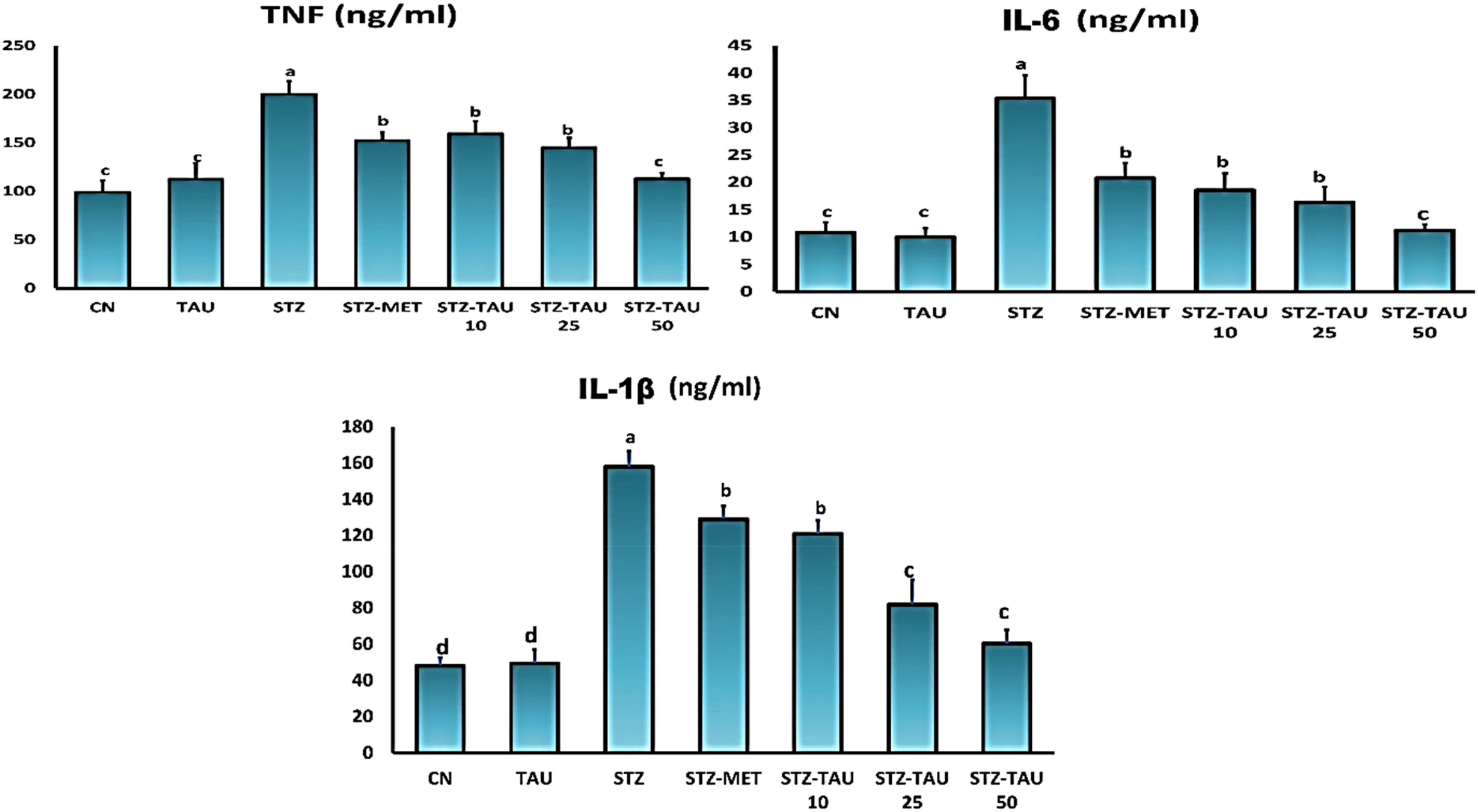
Significantly improved (p < 0.05) were the oxidant/antioxidant indices of diabetic rats treated with metformin, TAU 10, TAU 25, and TAU 50 in Groups IV, V, VI, and VII, respectively. Group VII (diabetic rats treated with TAU 50) exhibited the greatest decrease of STZ-induced oxidative stress when compared to diabetic rats not treated (Group III) (p < 0.001) (Figure 2 and 3).
Serum levels of cholesterol
Untreated diabetic rats (Group III) showed a highly significant rise in serum cholesterol at the end of week nine compared with untreated control rats (Group I) and TAU-treated control rats (Group II) (p < .001). The previous increase in cholesterol level decreased significantly in Groups IV, V, VI, and VII treated with metformin, TAU 10, TAU 25, and TAU 50, respectively (p < 0.001). In Group VII, TAU successfully normalized the altered cholesterol level compared with untreated control rats (Group I) (Figure 4(a)). A: Serum levels of follicular stimulating hormone (FSH) at the 9th week, B: Serum levels of testosterone hormone at the 9th week, C: Serum levels of cholesterol on day zero and at the 9th week (Mean ± SE) (each group = eight adult Wistar rats). (Mean values with different superscript letters revealed significant differences from each other at p < 0.05), in between different groups and at the different times from Day of zero till 9 weeks. CN: Group I (negative control group), TAU: Group II (TAU-treated control group), STZ: Group III (positive control group), STZ-MET: Group IV (Metformin-treated diabetic rats), STZ-TAU-10: Group V (TAU-10-treated diabetic group), STZ-TAU-25: Group VI (TAU-25-treated diabetic group) and STZ-TAU-50: Group VII (TAU-50-treated diabetic group).
Serum level of FSH and testosterone
The serum FSH and testosterone levels of STZ-treated rats (Group III) were considerably lower than those of untreated control rats (Group I) (p < 0.001). After therapy with metformin and TAU at varying dosages (Groups IV-VII, p < 0.001), the levels were significantly recovered. The greatest increase in serum FSH and testosterone concentrations was seen in rats treated with TAU 50. (Group VII) (Figures 4(b) and (c)).
Sperm count, motility, and abnormalities
STZ (Group III) significantly reduced sperm vitality (progressive forward motility) and significantly increased total sperm abnormalities in comparison to untreated control rats (Group I) and TAU-treated rats (Group II) (Group II) (p < 0.001). These reproductive parameters improved after co-administration with either metformin or different doses of TAU (Group IV-VII) (p < 0.05). Regarding the sperm count, the greatest improvement was detected in groups treated with metformin (Group IV) and TAU 50 (Group VII) compared with untreated diabetic rats (Group III) (p < 0.001). The percentage of sperm abnormalities showed significant improvement in TAU-treated groups in a dose-dependent way (Figure 5). A: Percentage of progressive sperm motility, B: sperm count, and C: percentage of sperm abnormalities (Mean ± SE) (Each group = eight adult Wistar rats), (Mean values with different superscript letters revealed significant difference from each other at p < 0.05), in between different groups and at the different times from Day of zero till 9 weeks. CN: Group I (negative control group), TAU: Group II (TAU-treated control group), STZ: Group III (positive control group), STZ-MET: Group IV (Metformin-treated diabetic rats), STZ-TAU-10: Group V (TAU-10-treated diabetic group), STZ-TAU-25: Group VI (TAU-25-treated diabetic group) and STZ-TAU-50: Group VII (TAU-50-treated diabetic group).
Serum insulin and blood glucose levels
Approximately 85% of rats injected with STZ displayed clinical type 1 DM (Group III), which was accompanied by a considerable rise in blood glucose and a drop in serum insulin levels compared to untreated control rats (Group I; p < 0.001). Treatment with metformin and TAU at varied doses (Groups IV-VII) for 9 weeks restored blood glucose levels after four and 9 weeks, with the highest reduction seen with metformin and TAU 50 mg/kg (p < 0.001) (Figure 6). A: Serum insulin levels on day zero and at the 9th week, B: Blood glucose levels on day zero, 4 weeks, and 9 weeks. (Mean ± SE) (each group = eight adult Wistar rats) (Mean values with different superscript letters revealed significant differences from each other at p < 0.05), in between different groups and at the different times from Day of zero till 9 weeks. CN: Group I (negative control group), TAU: Group II (TAU-treated control group), STZ: Group III (positive control group), STZ-MET: Group IV (Metformin-treated diabetic rats), STZ-TAU-10: Group V (TAU-10-treated diabetic group), STZ-TAU-25: Group VI (TAU-25-treated diabetic group) and STZ-TAU-50: Group VII (TAU-50-treated diabetic group).
Histopathology
Examining sections of testes and epididymis from untreated control rats (Group I) and TAU-treated rats (Group II) for histological changes revealed that both tissues had normal histological structure. The former had seminiferous tubules with an oblong or spherical shape and a thin basal lamina. Primary and secondary spermatocytes, spermatids, and spermatozoa were distributed normally. Normal histology of blood arteries and Leydig cells could be found in the interstitial tissues. The ductus epididymis and interstitial tissue had a normal histological architecture. In the epididymal lumens, there were additional clusters of spermatozoa at the Histopathological examination of testes, (H&E-stain, 100×). (Figure 7). Histopathological examination of epididymis, (H&E-stain, 100×). (A), (Negative Control group), (B), STZ, (Positive Control group), (C), STZ-MET, (Metformin-treated diabetic rats), (D), TAU: (TAU-treated control group), (E), STZ-TAU-10(TAU-10-treated diabetic group), (F), STZ-TAU-25: (TAU-25-treated diabetic group) and (G), STZ-TAU-50: (TAU-50-treated diabetic group).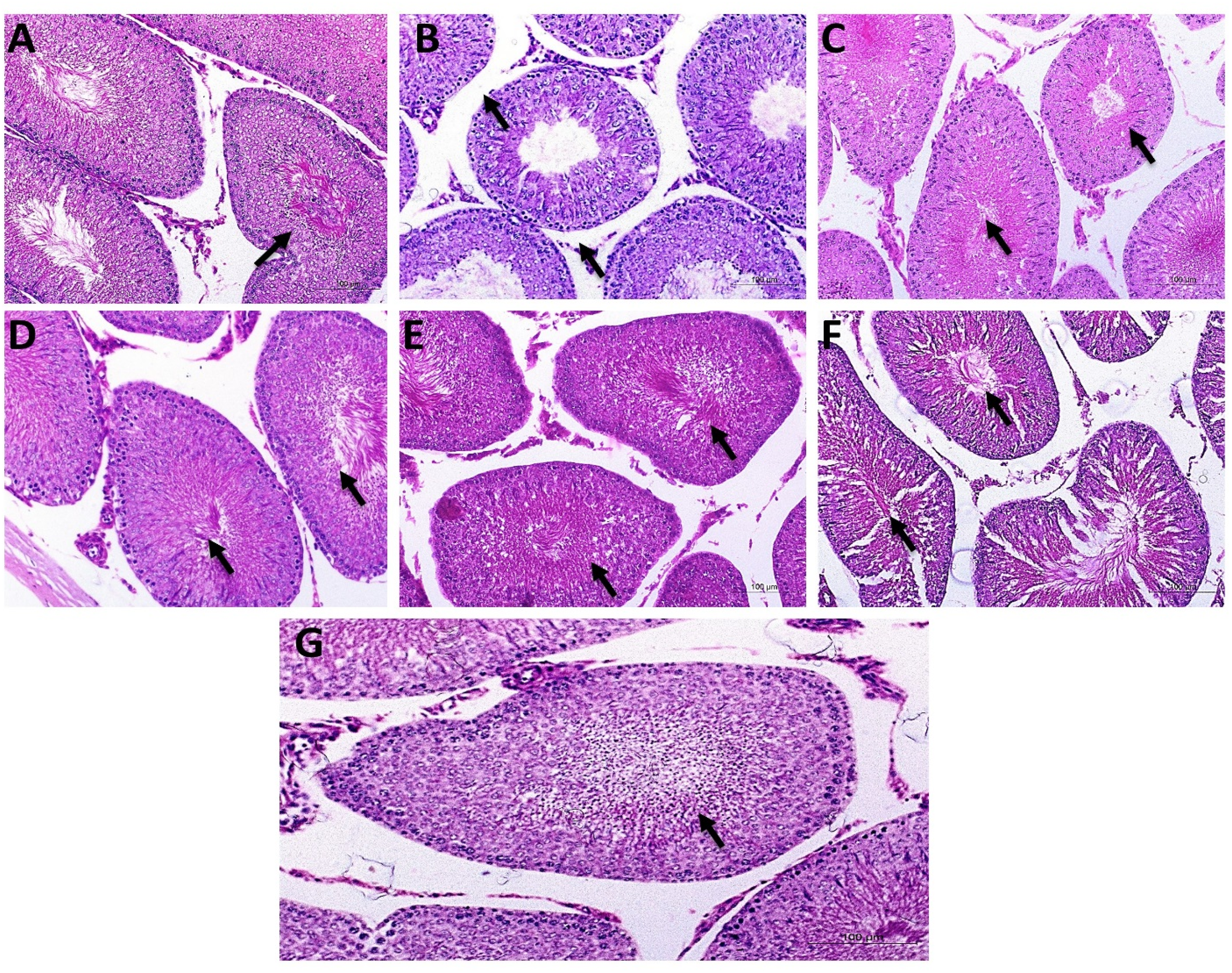
The positive control (STZ) rats had severe pathological lesions defined by disordered seminiferous tubules filled with necrotized germinal cells that were degraded and irregularly distributed. Furthermore, just one or two germinal layers were seen in certain tubules. With fewer spermatozoa, significant vacuolization might also be seen. The ductus epididymis demonstrated a significant decrease in diameter, small irregular lumens, and deteriorated spermatozoa that were less densely packed. The absence of spermatozoa in certain lumens was documented in more severe areas. It was revealed that intracytoplasmic vacuolation was present. Diabetic rats treated with varying doses of metformin and TAU had well-defined seminiferous tubules with relatively normal germinal cell layers, densely packed spermatozoa, and no vacuoles. In these groups, the ductus epididymis exhibited a dose-dependent increase in normal histological structure (Figure 8). The control group (A) had a normal testicular and epididymis structure with normal spermatozoa contents. The contents of spermatozoa were lowered in the epididymis of STZ-treated rats (B) with degenerative testicular alterations appeared in the form of depletion of the secondary spermatocytes, spermatids, and spermatozoa with widening in the lumen and lowered appearance of the internal spermatozoa in the epididymis (Black arrow). TAU-treated rats at different dosages exhibited normal spermatozoa contents in their epididymis with normal spermatogenesis and testicular parenchyma in the testes similar to that of a control animal. Normal testicular and epididymal structure with massive appearance at different degrees in a dose-dependent manner for TAU administrations for spermatids, and spermatozoa in the epididymis (black arrows), TAU with (E–G) or without STZ (D).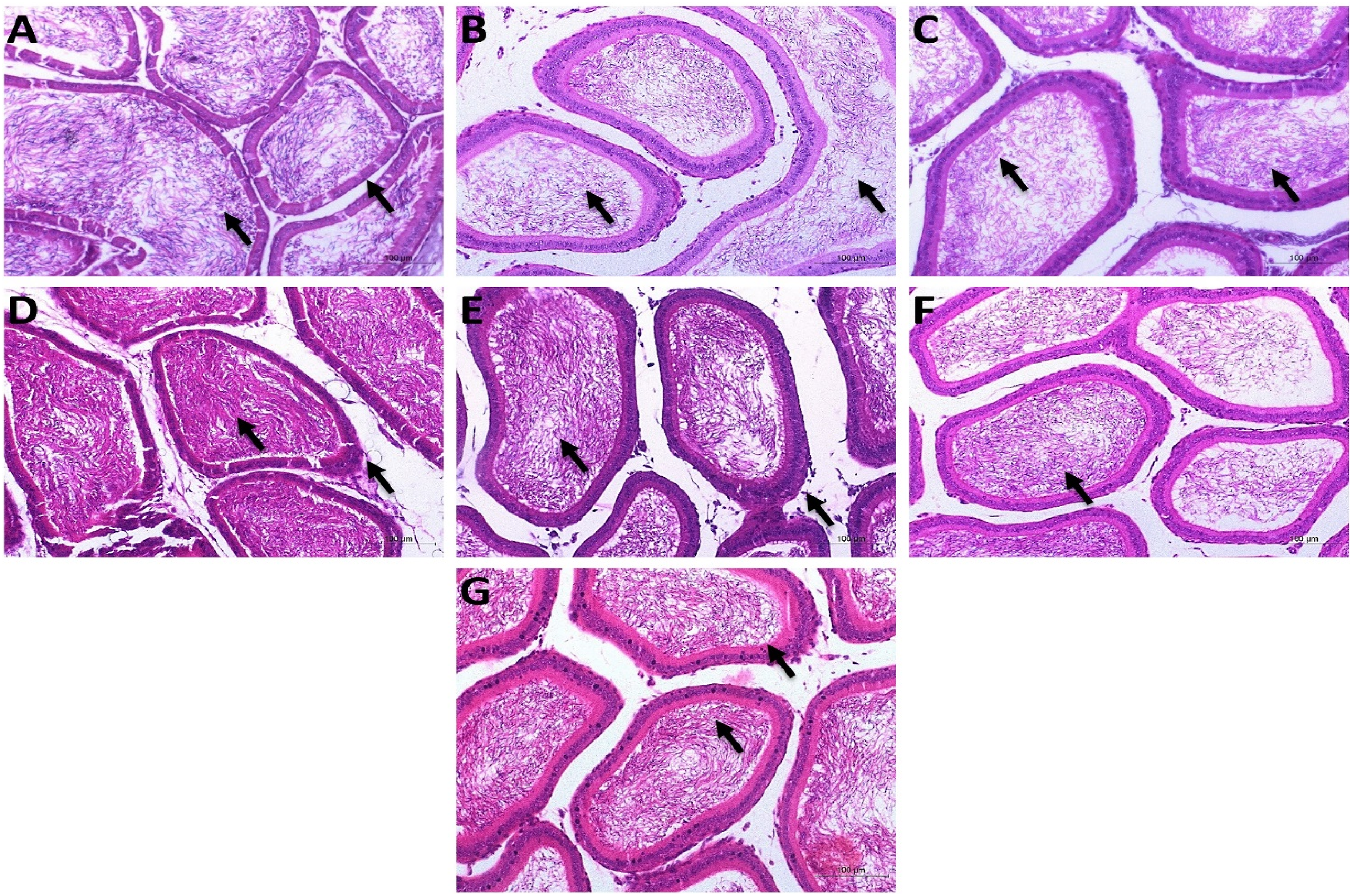
Discussion
Diabetes mellitus is the most widespread metabolic illness and a global health concern. Diabetes-induced oxidative stress results in microvascular problems. Therefore, inadequate blood supply is the cause of testicular dysfunction in diabetic patients. 24 In this study, the protective mechanism of TAU against type 1 diabetes-induced testicular damage in rats was investigated. Oral administration of TAU enhanced absorption, making it more effective than when added to meals. 25
A single injection of STZ may cause diabetes type 1 in animals. STZ detects Glucose moiety-2 (GLUT-2) and guides it to pancreatic cells. Due to the fact that STZ partly kills the pancreas and lowers cell mass, it is an excellent model for researching cell diabetic glucotoxicity, which leads in a large rise in blood glucose levels. 26
In the current investigation, TAU caused dose-dependent lowering in blood glucose level in diabetic rats. TAU may decrease hyperglycemia by boosting insulin production, pancreatic cell protection, enhancing insulin sensitivity, and reducing glucose absorption. 27 TAU therapy can increase basal and insulin-induced tyrosine phosphorylation of insulin receptors in the skeletal muscle and liver of diabetic rats. 28 Also, TAU increases glycogen formation in the liver and glucose absorption in peripheral tissues, which are both plausible explanations for the TAU-mediated improvement in insulin sensitivity and demonstrate an indirect link between TAU and insulin sensitivity. 29
The results of the present study are in agreement with the results detected by Harada et al. (2004) who demonstrated the favorable impact of TAU in decreasing STZ-induced hyperglycemia in a rat model, 30 El-Mesallamy et al. (2010) who demonstrated TAU-induced anti-diabetic effect in a high-fructose diet diabetes model 31 and Wu et al. 32 (2010) who detected that TAU can decrease insulin resistance in hepatic patients.
On opposite side, Li et al. 33 (2005) reported that TAU had no effect on blood glucose levels in diabetic rats. This disparity may be attributable to the length of therapy and mode of administration. In this respect, Long et al. (2018) reported that TAU supplementation with food has lower effect that oral TAU administration. In addition, TAU therapy during the outset of diabetes was more effective in decreasing hyperglycemia in STZ-induced type 1 DM rats. 34 TAU treatment initiated after the induction of diabetes requires an extended period to correct hyperglycemia. 35 Experiments indicated that TAU protects pancreatic cells from damage and preserves normal secretory granule function, hence delaying the development of diabetes. 36
After 9 weeks of therapy with a high dosage of TAU in the present study, insulin secretion was enhanced. Long-term TAU administration may restore the functionality of injured pancreatic islet cells by enhancing islet Ca2+ 37 uptake in response to glucose and increases extracellular glucose concentrations through the glucose transporter GLUT-2. Lin et al. (2017) found that TAU enhanced insulin production through activation of transcription factors. 38 TAU can upregulate insulin receptor substrate one phosphorylation and improve diabetic-related pancreatic dysfunction, possibly through its anti-inflammatory characteristics. 39 Batista et al. (2012) found that pancreatic islets extracted from rats consuming a high-glucose, low-protein diet resumed insulin production when supplemented with TAU 10, Together with the results of Vettorazzi et al. 40 (2014), in insulin who reported that an increase production in the islets of mice fed a high-fat diet while hungry, our study demonstrates a 40 similar effect.
Evidenced by TAU’s regulatory function in redox balance and protein phosphatase activity, Cappelli et al. 41 (2014) demonstrated that TAU maintains glucose tolerance and improves hepatic insulin signal transduction in obese malnourished mice.
Diabetes is characterized by a fast loss of weight. The accelerated breakdown of muscle tissue, lipids, and protein metabolism in diabetic rats causes a reduction in body mass. 42 TAU medication at assorted recommended dosages resulted in a significant difference in average body weights in both normal and diabetic rats compared to untreated rats, which can be explained by Caletti et al. 43 (2015), who stated that the antidepressant effect of TAU may improve the weight loss of diabetic rats.
In the present study, STZ treatment led to increased plasma cholesterol levels as it has been previously reported by Suryawanshi, et al. (2006) who reported that lipid profile changes are prevalent in diabetic patients. They discovered that cells are unable to use blood glucose for energy production and instead draw fatty acids from adipose tissue. The liver accumulates excess fatty acids, which are then turned into triglycerides. Chronic insulin shortage is linked to a decrease in low-density lipoprotein (LDL) receptors, causing a rise in LDL cholesterol levels. 44
The present study showed that, TAU significantly decreased cholesterol levels compared with untreated diabetic rats which may be due to 7-hydroxylase activation, as it is involved in biliary acid production, boosted insulin levels and increased the number of LDL receptors, lowering LDL cholesterol levels. 44
TAU supplementation increases the levels of expression of energy expenditure-related genes in fatty tissue, including peroxisome proliferator-activated receptor, peroxisome proliferator-activated receptor-gamma coactivator 1 (PGC 1), and their target genes lipoprotein lipase, acyl-CoA oxidase, and acyl-CoA synthase, which are associated with cholesterol-lowering impacts. This trait may be the result of direct stimulation of cAMP-dependent protein kinase A (PKA) catalytic activity or indirect PKA stimulation through interference with H2O2 accumulation. 45
TAU has been shown to inhibit the production of lipoprotein products and the incidence of atherosclerosis in animals with hereditary hyperlipidemia 46 and rabbits with Watanabe heritable hyperlipidemia (WHHL), which mirror human familial hypercholesterolemia. 47 Kim et al. 27 (2006) showed that TAU succeeded to reduce triacylglycerol and cholesterol blood levels in diabetic rats for 12 weeks of treatment. Borck et al. 48 (2018) evaluated the effects of TAU on leptin-deficient obese mice for 11 months and found a substantial decrease in plasma cholesterol levels.
The link between oxidative stress and inflammation is robust. At the site of inflammation, immune-activated cells emit reactive species that activates and amplifies intracellular signaling pathways which in turn stimulate the production of pro-inflammatory genes. Apart from the changes in metabolic status, diabetes is regarded as an inflammatory illness. Hyperglycemia induces generation of ROS, cytokines, growth factors and adhesion molecules that activates transcription factor nuclear factor-beta (NF-κβ), which controls expression of gene during inflammation. This causes multiple organs to malfunction. 49 As shown by elevated TNF-α, IL-1β, and IL-6 levels in the current investigation were associated by functional impairment in the testes and accessory glands.
In the present study, metformin and TAU in a dose-dependent manner significantly improved oxidant/antioxidant indices in diabetic rats. By scavenging and producing TAU-chloramine, a stable molecule with anti-inflammatory and cytoprotective properties, TAU may effectively counteract the deleterious effects of oxidative stress and decreases the level of pro-inflammatory cytokines; TNF-α, IL-6, and IL-1β. 50 Sobrido-Cameán et al. 51 (2020) showed that taurine reduced inflammation, neutrophil accumulation, pro-inflammatory cytokine levels and degenerative effects in gray matter, mitigated motor abnormalities and promoted axonal regeneration. Taurine supplementation by reducing oxidative stress prevents cisplatin-induced cardio- and nephrotoxicity. 52
Wang et al. 53 (2016) detected that TAU protects against endothelium dysfunctional by lowering the levels of oxidant compounds as oxidized LDL, evident by the decreased power of aortic contraction in response to noradrenaline. TAU seems to influence connections between leukocytes and endothelial cells as well as hyperglycemia-induced endothelial death. 54
TAU increases GSH, glutathione reductase, and glutathione synthetase and helps to heal pancreatic cell damage in male Sprague Dawley rats when given before STZ administration. Also, TAU supports normal electron transport chain function, shielding mitochondria from excessive O2 production. 55 In addition, You et al. 56 (2013) discovered that obese rats induced by high-fat diet displayed anti-inflammatory effects of TAU on adiponectin and cholesterol levels following 8 weeks of TAU supplementation in their drinking water.
Compared to untreated control rats, STZ diabetic rats had a considerable drop in total sperm count and progressive forward motility, as well as a major rise in the total number of deformed sperm. Total sperm count and motility reversed considerably in diabetic rats treated with varying dosages of TAU, and abnormalities visibly and significantly diminished.
Diabetes mellitus is a significant risk factor for erectile dysfunction in men. In diabetic rats, the neurogenic corpus cavernosum relaxation mediated by nitric oxide (NO) was drastically diminished. An imbalance in parameters controlling cavernosal smooth muscle tone may be the source of altered penile hemodynamic pathways. 57 The intact vascular endothelium produces NO, a vascular protective molecule, by inhibiting platelet and leukocyte adherence to the endothelium. The inhibition of endothelial NO synthase by hyperglycemia leads to the excessive production of ROS in diabetics. Hyperglycemia stimulates the overproduction of vasoconstrictor molecules, such as endothelin 1, and the activation of the renin-angiotensin system. 58
Mohamed and Gawad (2017) confirmed that, TAU supplementation improved sperm counts and motility, as well as degenerative alterations in the spermatogenesis and seminiferous tubules. 59 In addition, Schaalan et al. (2018) reported that in azathioprine-treated rats, TAU improved testes' weights and level of luteinizing hormone and testosterone, which was associated with decreased oxidative stress in testicular tissues. 60
In the current work, anomalies in the pituitary-testicular tract axis of STZ-induced diabetic rats appeared as a decrease in the release of testosterone from Leydig cells. At the histological level, lipid droplets accumulated, the smooth endoplasmic reticulum was diminished, and myelin-like structures appeared. STZ-induced diabetic rats exhibited a decrease in testicular and epididymal weight as well as an increase in apoptotic cell death in their testes.
The testicular and epididymal weights of diabetic rats treated with TAU were partially restored, indicating that the antioxidant therapy has a protective impact inside the testis and on the epididymis. In agreement with the results of another study, diabetic rats had a decrease in testicular weight and atrophy of the seminiferous tubules, 61 suggesting that apoptotic cell death is a key role in the loss of testicular function in diabetic mice used in experiments.
This study’s results are congruent with those of Kamel et al. (2019) who showed that exposure to potassium bromate alters the testicular histology and inhibits spermatogenesis in rats that was treated by TAU. 62 Aydos et al. 63 (2014) found that TAU has antioxidant capabilities and protects against testicular torsion-induced apoptosis and germ cell degeneration. Azab et al. 64 (2020) detected that TAU can protect against gonadotoxicity and testicular apoptosis effects induced by cisplatin in rats.
The results of the present study are matched with results obtained by Ghosh et al. (2019) who showed that TAU prevented inflammation and endoplasmic reticulum stress mediated testicular damage in STZ-induced diabetic rats by influence NF-κB signaling cascade 65 and Liu et al. 66 (2017) who detected that TAU restored testicular steroidogenesis and spermatogenesis in STZ-induced type I diabetic rats, presumably through boosting hypothalamic-pituitary-testicular endocrine activity. Abd El-Twab et al. (2016) described the protective effect of TAU in decrease testicular damage by reduce oxidative stress and up-regulation of the pituitary-gonadal axis. 67
Limitation
The main limitation of this study was we did not conduct any power analysis to calculate the sample size selected for this study.
Conclusion
TAU, in doses of 10, 25, and 50 mg/kg once daily, provided orally for 9 weeks in STZ diabetic adult male rats, exerted dose-dependent anti-hyperglycemic and anti-hypercholesterolemic effects. In diabetic animals, it also stabilized serum insulin levels and body weights. Regarding reproductive functioning, dose-dependent preservation of testis, prostate, and seminal vesicle relative weights in proportion to body weight, as well as blood testosterone and FSH levels, was achieved by TAU in diabetic rats. In addition, sperm count, motility, and abnormalities were recovered by TAU therapy, which was related with testicular and epididymal histological improvements. TAU may generate these effects by decreasing oxidative damage in the testis caused by DM. The current data imply that TAU supplementation has a favorable impact in preventing and treating diabetes as well as mitigating the associated reproductive damage. TAU, an endogenous chemical in the body, is not associated with toxicity or drug dependence; hence, it is widely tolerated when provided properly.
Footnotes
Acknowledgments
The authors acknowledge the support of members of the animal house research team in Faculty of Veterinary Medicine, Cairo University for their help throughout this work.
Authors’ contributions
H.A.E. and F.I.A.E.-E; conceived the research idea and designed the work and wrote the first draft of the manuscript. AHE, ASO, and AHE carried out the experiments and interpretation of results and F.I.A.E.-E performed the statistical analysis. All authors contributed to the editing of the revised manuscript, and approved the final manuscript.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
This study was approved by the Institutional Animal Care and Use Committee of the Faculty of Veterinary Medicine at Cairo University accepted the research protocol, and the animal studies were done according to the ethical principles of animal welfare (Vet CU 8/03/2022 /415).
Data availability
All data and materials are available and can be submitted when needed.
Animal welfare
Animals were handled according to the International Guidelines for Care and Handling of Experimental Animals.
