Abstract
Selenium (Se) has anti-inflammatory and antioxidant properties and is necessary for the development and normal function of the central nervous system. This study was aimed to compare the in vitro effects of 3-methyl-1-phenyl-2-(phenylseleno)oct-2-en-1-one (C21H2HOSe; organoselenium) and sodium selenate (inorganic Se) on adenosine deaminase (ADA) activity, cell viability, lipid peroxidation, scavenger of nitric oxide (NO) and nonprotein thiols (NP-SH) content in the cerebral cortex slices of the young rats. A decrease in ADA activity was observed when the slices were exposed to organoselenium at the concentrations of 1, 10 and 30 µM. The same compound showed higher scavenger capacity of NO than the inorganic compound. Inorganic Se was able to protect against sodium nitroprusside-induced oxidative damage and increased the NP-SH content. Both the compounds displayed distinctive antioxidant capacities and were not cytotoxic for the cerebral cortex slices in the conditions tested. These findings are likely to be related to immunomodulatory and antioxidant properties of this compound.
Introduction
In the last decade, there has been increasing interest in several nutritional selenium (Se) compounds because of their environmental, biological and toxicological properties, and particularly for their cancer- and disease-preventing activities. Regulation of Se levels is important in balancing the detrimental effects of both Se toxicity and deficiency. 1,2 Se is an essential trace element with a number of biological roles, for humans, animals and some bacteria. 3,4 As a constituent of selenoproteins, it plays a role in the protection of body tissues against oxidative stress, immune function, reproduction, growth and development modulation. 4 The brain is resistant to fluctuations in Se levels, including during conditions of dietary depletion of Se. Therefore, brain Se levels are maintained, whereas levels elsewhere in the body are reduced, suggesting that this trace mineral is essential for normal brain function. 5,6 However, the central nervous system (CNS) is sensitive to Se poisoning, and differences in neurotoxicity between inorganic and organic Se have been demonstrated. 7
Se-containing organic molecules are generally more potent antioxidants than ‘classical’ antioxidants, which is an impetus for the increased interest in the rational design of synthetic 3-methyl-1-phenyl-2-(phenylseleno)oct-2-en-1-one (C21H2HOSe; organoselenium) compounds. 8,9 Previous reports have been published on the glutathione peroxidase mimetic activity of chalcogen compounds, which, such as the native enzyme, relies on the redox cycling of the Se moiety of the compounds. 10,11 Organochalcogens are important intermediates and useful reagents in organic synthesis, which can increase human exposure risk to these chemicals in the workplace. 12,13
The organoselenium (Figure 1) is an α,β-unsaturated ketone-functionalized vinyl chalcogenide that has been found as a potential tool in organic synthesis. Vinylic compounds are important synthetic intermediates because of their easy transformation into other organic compounds with retention of configuration. 14,15

Chemical structure of 3-methyl-1-phenyl-2-(phenylseleno)oct-2-en-1-one (organoselenium).
Several lines of investigations on the cellular metabolism of purines in peripheral tissues have generated considerable interest in adenosine deaminase (ADA; E.C. 3.5.4.4), an enzyme involved in purine metabolism that catalyzes the hydrolytic deamination of adenosine or 2′-deoxyadenosine to inosine or 2′-deoxyinosine. 16 ADA is one ubiquitous, soluble and globular enzyme that can be found at variable amounts in all cell types present in the CNS. 17 Therefore, ADA has been a subject of interest due to its role in the maintenance of intra- and extracellular levels of adenosine. Congenital deficiency of ADA causes severe combined immunodeficiency, which is characterized by the absence of functional T and B lymphocytes. 18 Conversely, increased ADA activities have been observed in diseases such as brucellosis, tuberculosis, rickettsiosis, human immunodeficiency virus and infectious mononucleosis as well as in a variety of malignant conditions. 19
Reactive oxygen species are currently produced during cellular metabolism. Among these, nitric oxide (NO) is regarded as a universal neuronal messenger in the pathophysiology of Alzheimer’s and Parkinson’s diseases and disorders such as stroke, trauma and seizure. 20,21 NO has been used in vitro to promote NO-induced lipid peroxidation. 22 It can mediate biological actions ranging from vasodilatation, neurotransmission, inhibition of platelet adherence and aggregation and killing of pathogens mediated by macrophages and neutrophils. 23 However, high concentrations of NO are toxic and interact with superoxide anion (O2−) to form peroxynitrite (ONOO−), a strong oxidant that forms another potent oxidant with the reactivity of a hydroxyl-like radical, which could initiate lipid peroxidation. 24,25
The rationale for the study of organic and inorganic Se compounds resides in the fact that this element is essential for normal brain function and displays specific roles for developing brain. As organoselenium and inorganic Se compounds might have different neurotoxic behavior, the purpose of the present study was to investigate the in vitro effects of an organic and an inorganic form of Se on ADA activity and cell viability in cerebral cortex slices of young rats, which preserve the interaction between neurons and glia. Considering that oxidative stress is involved in neurochemical changes in the developing cerebral cortex, the total antioxidant capacity and lipid peroxidation of both the compounds were also examined.
Materials and methods
Chemicals
Adenosine was obtained from Merck (Darmstadt, Germany). Sodium selenate (Na2SeO4; inorganic Se), malonaldehyde bis (dimethyl acetal), 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide (MTT), sodium nitroprusside (SNP), sulfanilamide, N-(1-naphthyl)ethylenediamine dihydrochloride, 5,5′-dithiobis-2-nitrobenzoic acid (DTNB) and thiobarbituric acid were obtained from Sigma Chemical Co. (St Louis, Missouri, USA). Organoselenium was synthesized according to Silveira et al. 26 All other chemicals were of analytical grade and were obtained from standard commercial suppliers.
Preparation of cerebral cortex slices
The study was in accordance with the guidelines of the Ethics Committee for Animal Research of this institution, which approved the experimental protocol (number 23081.007418/2007-75). Newly born male Wistar rats were maintained on a 12:12 h light/dark cycle at 24°C. The rats (n = 8) were euthanized at day 7 (P7) and their brains were promptly removed and the cerebral cortices were carefully separated. The slices were cut to have a weight of approximately 5 mg and washed twice with phosphate buffer (50 mmol/L, pH 7.0) before using in the experimental analyses, according to Oyamaa et al. 27 with modifications. The organoselenium (1, 10 and 30 µM) or inorganic Se (1, 10 and 30 µM) was added in the phosphate buffer and the reactions were carried out for 1 h at 37°C, according to Bellé et al. 28
Experimental assays
Enzymatic assays
ADA activity was estimated spectrophotometrically by the method of Giusti, 29 which is based on the direct measurement of the formation of ammonia produced when ADA acts in excess of adenosine. Ammonium sulfate solution (75 μM ammonia) was used as the standard. The values were expressed as unit per liter per milligram of tissue.
The activity of lactate dehydrogenase (LDH) was immediately measured using a commercial kit (Bioclin/Quibasa, Minas Gerais, Brazil). The results were expressed as unit per liter per milligram of tissue.
Parameters of oxidative stress
Measurement of lipid peroxidation
Lipid peroxidation in the cerebral cortex slices was estimated by the thiobarbituric acid reactive substances (TBARS) assay, as described by Niehaus and Samuelsson. 30 First, the slices were incubated with organoselenium (1, 10 and 30 µM) or inorganic Se (1, 10 and 30 µM) in phosphate buffer (50 mmol/L, pH 7.0) for 1 h at 37°C. Thereafter, the slices were homogenized in Tris–hydrochloric acid buffer, centrifuged at 4000 g and 0.1 mL of the obtained supernatant (S1) was used in the assay. 31 In a second step, SNP (50 μM) was added to the reaction mixture for 1 h at room temperature. 32 The above concentrations of the Se compounds were added to the mixture that was incubated for 1 h more before the TBARS levels were measured. At the end of the incubation time, the lipid peroxidation assay was carried out as mentioned above. Calculations were made based on standard curves and presented as nanomoles of malondialdehyde per milligram of protein.
Determination of NP-SH level
Nonprotein thiol (NP-SH) levels in the rat cerebral cortex slices were determined according to the method proposed by Ellman and Boyne, 33 with some modifications by Jacques-Silva et al. 34 A sample of S1 (300 μL) was mixed (1:1) with 10% trichloroacetic acid and subsequently centrifuged at 4000 g for 10 min. After centrifugation, the supernatant fraction was added to a reaction medium containing potassium phosphate (0.5 M and pH 7.4) and DTNB (0.5 mM). NP-SH levels were measured spectrophotometrically at 412 nm. The results were calculated in relation to a standard curve constructed with reduced glutathione and also corrected by the protein content.
Assay of NO-scavenging activity
The procedure is based on the principle that SNP in aqueous solution at physiological pH spontaneously generates NO. For the experiment, SNP (10 mM) was mixed with the different concentrations of the Se compounds and incubated at room temperature for 150 min. The same reaction mixture, without the compounds but with an equivalent amount of water, served as the control. After the incubation period, 0.5 mL of Griess reagent (1% sulfanilamide, 2% phosphoric acid and 0.1% N-(1-naphthyl) ethylenediamine dihydrochloride) was added. The NO generated interacts with oxygen to produce nitrite ions that can be estimated using this reagent. 35 NO scavengers compete with oxygen, leading to reduced production of nitrite ions. The absorbance of the chromophore formed was read at 546 nm. 36 The NO-scavenging activity was given by the following equation
Cell viability
Tetrazolium salt method (MTT assay)
The viability assay was performed by the colorimetric MTT method. This assay for cell survival is an indirect method, where MTT is reduced by active mitochondria in living cells. The tetrazolium salt is metabolically reduced to a colored formazan type of final product. 37 The cerebral cortex slices were incubated in the presence or absence of the above concentrations of organoselenium or inorganic Se for 1 h at 37°C. Immediately after preincubation, 1.2 mM of MTT was added to the medium containing the slices followed by 1 h incubation at 25°C. The formazan product generated during incubation was solubilized in dimethyl sulfoxide and quantified spectrophotometrically at 560 nm. 38 Only viable slices are able to reduce MTT; thus, each value obtained is proportional to the percentage of viable cells. The value of the control was considered as 100%.
Protein quantification
Protein concentration was measured by the method of Lowry et al., 39 using bovine serum albumin as the standard.
Statistical analysis
The results were analyzed by one-way analysis of variance for multiple group comparison. Post hoc analysis was carried out using Tukey’s multiple range test. Data are shown as mean ± SEM. Values of p < 0.05 were regarded as statistically significant.
Results
The present study demonstrated that ADA activity (Figure 2) was reduced in the cerebral cortex slices exposed to the organoselenium compound. However, this effect was not observed when the slices were exposed to the inorganic Se in the same experimental conditions. The MTT assay showed that the organoselenium compound at the concentration of 30 µM caused a significant increase in cell viability in the cerebral cortex slices (p < 0.05; Figure 3). Notwithstanding, this parameter was not altered in the presence of the inorganic Se. Moreover, no difference in LDH activity was observed after the exposure of the cerebral structure to both Se compounds (Table 1).

ADA activity after exposure of cerebral cortex slices of young rats (n = 8) to organoselenium and inorganic Se compounds. Data are expressed as unit per liter per milligram of tissue (mean ± SEM). Asterisk indicates significant differences from control (p < 0.001, one-way ANOVA followed by Tukey’s test). ADA: adenosine deaminase; ANOVA: analysis of variance; Se: selenium.

MTT assay in cerebral cortex slices of young rats (n = 8) after exposure to organoselenium and inorganic Se compounds. Results are expressed as percentage of viable cells (mean ± SEM). Asterisk indicates significant differences from control (p < 0.01, one-way ANOVA followed by Tukey’s test). ANOVA: analysis of variance; Se: selenium; MTT: 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide.
Effects of organic and inorganic Se compounds on LDH activity (n = 8).a

Se: selenium; LDH: lactate dehydrogenase; Na2SeO4: sodium selenate; C21H2HOSe: 3-methyl-1-phenyl-2-(phenylseleno)oct-2-en-1-one.
aData are expressed as mean ± SEM.
When the rat cerebral cortex slices were exposed to both compounds alone, lipid peroxidation was maintained at a background level similar to that observed in the control samples, which demonstrates that the compounds did not promote cell damage (data not shown).
SNP (50 μM) significantly increased the lipid peroxidation in the cerebral cortex slices (p < 0.05). However, distinctive effects were observed when the slices were co-incubated with the different Se forms. Decreased TBARS levels were observed for the inorganic compound at all concentrations tested (p < 0.001), whereas the organoselenium compound did not show this protective effect (Figure 4). Furthermore, as depicted in Figure 5, NO-scavenging capacity of the organoselenium was higher than that of the inorganic Se compound.

Effect of organoselenium and inorganic Se compounds on SNP-induced lipid peroxidation (50 µM) in cerebral cortex slices of young rats (n = 8). Results are expressed as percentage of control (mean ± SEM). Asterisk indicates significant differences from control (p < 0.05, one-way ANOVA followed by Tukey’s test). ANOVA: analysis of variance; Se: selenium; SNP: sodium nitroprusside.
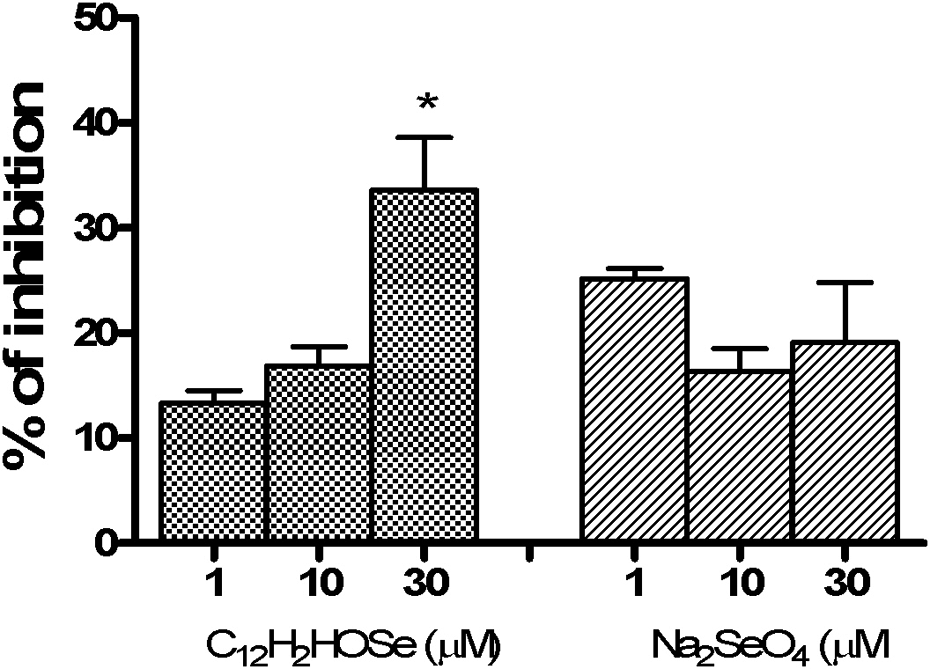
Nitric oxide radical-scavenging activity of Se compounds. Values are presented as mean ± SEM (n = 3). * indicates significant differences from control (p < 0.01, one-way ANOVA followed by Tukey's test). Se: selenium.
We also investigated the status of nonenzymatic antioxidant defenses in the cerebral cortex slices after incubation with the tested compounds, through the measurement of NP-SH levels (Table 2). Our experimental data showed that the exposure of the samples to the organoselenium did not change NP-SH levels. Conversely, inorganic Se was able to increase the NP-SH levels in the cerebral slices at the concentration of 30 μM (p < 0.05).
Effects of organic and inorganic Se compounds on NP-SH (n = 8).a

Se: selenium; NP-SH: nonprotein thiols; Na2SeO4: sodium selenate; C21H2HOSe: 3-methyl-1-phenyl-2-(phenylseleno)oct-2-en-1-one.
aData are expressed as mean ± SEM. * indicates significant differences from control (p < 0.05, one-way ANOVA followed by Tukey's test).
Discussion and conclusion
As far as we know, this is the first study to show distinctive effects of Se compounds on the ADA activity in the cerebral cortex slices of young rats. Since ADA is a sulfhydryl-containing enzyme, one could hypothesize that the decreased enzymatic activity observed is due to the chemical reaction of the organochalcogen with the enzyme thiol groups. 40 – 42 However, the compound tested in this investigation did not present this property since NP-SH levels were not altered in the cerebral cortex slices. Reinforcing this idea, the activity of LDH, another thiol-containing enzyme, was also not altered by the organoselenium. Another view is that the presence of an adjacent heteroatom in close proximity to the central atom of organic compounds may possibly generate small nonbonding interactions, enhancing the reactions of thiols. 43,44 The absence of such condition in the tested organoselenium also explains its nonreactivity with thiol groups. However, the Se atom in the Na2SeO4 exists in its highest oxidation state (+VI) and is hardly reactive with thiols, 45 which is likely to explain, at least to some extent, the reason for the lack of change in the ADA activity.
The purine adenosine is an important neuromodulator, with both excitatory and inhibitory actions within the CNS. 46 This nucleoside is involved in diverse processes including locomotion, sleep and respiration and provides neuroprotection during hypoxia/ischaemia. 47 The control of adenosinergic signaling can be exerted by adenosine uptake via bidirectional transporters followed by intracellular phosphorylation of adenosine to adenosine monophosphate kinase or deamination of inosine by ADA. 48 In brain, ADA occurs in the cytosol and on the outer surface of neurons and synaptosomes, although its activity is more prominent in glia. 49,50 Our findings are strengthened by the use of slices, thereby preserving the interaction between neurons and glia in developing rats, allowing for a more accurate determination of the ADA activity. 38
Furthermore, it is possible to suggest that the inhibition of ADA activity caused by the organoselenium might act in the maintenance of appropriate cellular adenine nucleoside and nucleotide concentrations in the slices assayed. These results support the previous findings from our laboratory. 28 In fact, these organoselenium may have immunomodulatory action since the decrease in ADA activity could favor the anti-inflammatory action of adenosine.
The results of the present study showed that only the organic compound tested improved cell viability, whereas the inorganic compound did not change the MTT assay. MTT is reduced by enzymes of the endoplasmic reticulum in reactions involving nicotinamide adenine dinucleotide and nicotinamide adenine dinucleotide phosphate. 51,52 Thus, it is likely that the organic compound could modulate the antioxidant status of the slices exposed to the compound.
In this study, we showed that the organoselenium did not cause lipid peroxidation per se and, despite the NO-scavenging capacity, the antioxidant property of the organoselenium was not enough to protect the tissue against the damage caused by SNP, which suggests that this compound is ineffective against free radicals involved in the lipid peroxidation process. However, it is important to state that the SNP concentration utilized to induce lipid peroxidation was higher (50 μM) than the organoselenium concentrations (1–30 μM). Thus, the relative lower concentration of organoselenium was likely not enough to overcome the SNP-induced oxidative damage in the brain slices.
The paradoxical effect of these compounds depends on a variety of chemical structure, dose, route and regimen of administration and animal species involved in the studies. 53 It has been demonstrated that organoselenium induces oxidative stress in organ homogenates from immature rats and lacks 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical-scavenging activity. 54 Moreover, it was able to enhance lipid peroxidation in the cerebral cortex after chronic administration in rats. 55,56 In line with this, different inorganic Se concentrations present different effects on the TBARS levels and sulfhydryl groups at distinct brain areas of young rats. 57,58
In summary, we demonstrated the effects of the exposure of cerebral cortex slices of young rats to Se compounds. Inorganic Se was able to protect against the SNP-induced oxidative damage, increased the NP-SH levels and was not cytotoxic for the cerebral cortex slices in the conditions tested, confirming its antioxidant capacity. The organoselenium compound improved the cellular integrity and exhibited higher NO scavenger activity than the inorganic Se. The reduction in the ADA activity observed after the exposure to the organoselenium compound could be related to immunomodulatory properties and maintenance of adenosine levels in the CNS, evidencing a particular feature of this compound. The present findings highlight a promising way to be exploited by both the compounds in CNS.
Footnotes
Acknowledgments
The authors thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and the Federal University of Santa Maria for supporting this study.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.

