Abstract
The present study aimed to compare the effect of gender difference on hemodynamic consequences in the development of monocrotaline (MCT)-induced pulmonary hypertension in rat. The effect of antioxidant enzyme systems on the development of pulmonary hypertension mediated by the phytotoxin MCT and the effect of gender on these antioxidant systems were also investigated. For this purpose, the right ventricular pressures (RVPs) and right ventricular/heart weight (HW) ratios were compared between groups and the glutathione (GSH) level and superoxide dismutase (SOD), catalase (CAT) and glutathione-S-transferase (GST) activities were determined in lung and liver tissue samples of rats. RVP and right ventricular/HW ratios significantly increased in the MCT group compared to the control group. In the MCT group, RVP was significantly higher in males than females. MCT-induced pulmonary hypertension resulted in decreased GSH level, decreased GST and SOD activities and increased CAT activity in lung and liver tissues of both male and female rats. In addition, the lung and liver GSH level and GST and SOD levels were higher in female control rats compared to male control rats. The results of the present study, that antioxidant enzyme activities were different between the groups, highlight the possible role of oxidative stress in the pathogenesis of MCT-induced pulmonary hypertension in rats. Moreover, the lower antioxidant defense capacity of male rats than female rats may be considered as a cause of more aggressive course of MCT-induced pulmonary hypertension in males compared to females.
Introduction
Gender differences in the severity and prevalence of cardiovascular and cardiopulmonary diseases have been demonstrated in both human and animal studies. 1 –4 It is well known that men from age 30 to 50 carry a greater risk for cardiovascular disease than women at the same age range. Therefore, it was suggested that premenopausal women carry less risk for these diseases than men. 2,4,5 –7 However, the mortality rate of cardiovascular disease is reported to increase due to reduced estrogen levels in postmenopausal women. 5,7 –9 There are a number of studies showing the protective role of female sex hormones against the development of cardiovascular disease. 5,6,10 Animal studies also demonstrate gender differences in the development of cardiovascular diseases and endogenous estrogen is proposed to be protective against them. 1,3 –5
Although the effect of gender factor on the pulmonary vasculature is not clearly established, it has been proposed that idiopathic pulmonary hypertension occurs more frequently in women than men. 11 –13 In contrast, chronic hypoxia-associated pulmonary hypertension was reported to be associated with less severe symptoms in women than men. 5 –7 There are also some studies demonstrating the protective role of estrogen in pulmonary vasculature. In these studies, pulmonary hypertension models were associated with less severe vascular remodeling and right ventricular hypertrophy in women than men. 13,14 Also, in rat models of pulmonary hypertension (both hypoxia and monocrotaline (MCT)), studies suggest the suppressive and protective effects of estrogens on pulmonary hypertension. 1,15,16
It was reported that oxidative stress is likely to play a role in gender difference found in the incidence of cerebrovascular and cardiovascular disease. 17 Oxidative stress plays a crucial role in the regulation of vascular functions through affecting vascular cell contraction/dilation, cell migration, cell growth/apoptosis, cell membrane lipid peroxidation/oxidation and extracellular matrix protein turnover. Moreover, oxidative stress is known to contribute to the development of vascular remodeling. 18,19 Several studies have shown that oxidative stress is higher in men than women. 20,21 There are also many studies demonstrating gender-related differences in oxidative stress-mediated liver damage induced by hepatotoxins. In these studies, the antioxidant enzyme levels have been found to be higher in females than in males. 19,22
MCT is a toxic pyrrolizidine alkaloid, which induces pulmonary hypertension in rats by increasing the pulmonary artery pressure and leading to right ventricular hypertrophy. 23 This model is frequently used in experimental studies because of the similarities between pulmonary hypertension progression in humans and in animals. MCT rat model was also studied in order to provide information about the genesis of chronic pulmonary vascular disease in people. Although it may not be the exact equivalence, the MCT-treated rat model is considered to be a valuable animal model for investigating primary pulmonary hypertension and the late or proliferative phase of the adult respiratory distress syndrome. 24 Furthermore, human exposure occurs due to consumption of contaminated grains, herbal teas and medicines. 25 To demonstrate its toxic effects, MCT had to be metabolized by cytochrome P450 3A enzyme in the liver to the reactive alkylating pyrrole dehydromonocrotaline. 26 Dehydromonocrotaline is known to cause oxidative stress in both hepatic and pulmonary tissues. 27 –29 Synthetic dehydromonocrotaline reproduces the toxicity of MCT by the formation of reactive oxygen species. 29 Several studies have shown that this metabolite may be detoxified by conjugation with the free radical scavenger–reduced glutathione (GSH). The amount of dehydromonocrotaline available for these presumably intoxicating pathways is affected markedly by the GSH content of the liver. GSH conjugates with dehydromonocrotaline to form GSH dehydropyrrolizidine, a compound of much lower toxicity that is released in high concentration into the bile. 29,30
In the present study, it was aimed to investigate the differences between the sexes in a rat model of MCT-induced pulmonary hypertension. The severity of pulmonary hypertension was evaluated by the measurement of pulmonary artery pressure and assessment of right ventricular hypertrophy in rats. Moreover, catalase (CAT), superoxide dismutase (SOD) and glutathione-S-transferase (GST) enzyme activities and GSH levels were measured in the lung and liver tissue samples of rats in order to clarify the mechanism of the differences between the sexes.
Materials and methods
Animals
Female and male Sprague-Dawley rats weighing 250–300 g were obtained from our own animal facility. Rats were housed under controlled temperature (22°C) and lighting (12/12-h light dark cycle) with free access to food and water. Animal care and research protocols were based on the principles and guidelines adopted by the Guide for the Care and Use of Laboratory Animals (NIH publication no. 85-23, revised in 1985) and approved by the Local Ethics Committee of Anadolu University, Eskisehir, Turkey.
The following four groups were studied: (1) control-female (C-F; n = 10): female rats were given a subcutaneous injection of vehicle. After 3 weeks, hemodynamic studies were performed. (2) MCT-female (MCT-F; n = 10): female rats were given a subcutaneous injection of MCT. After 3 weeks, hemodynamic studies were performed. (3) Control-male (C-M; n = 10): male rats were given a subcutaneous injection of vehicle. After 3 weeks, hemodynamic studies were performed. (4) MCT-male (MCT-M; n = 10): male rats were given a subcutaneous injection of MCT. After 3 weeks, hemodynamic studies were performed.
MCT-induced Pulmonary Hypertension
MCT was dissolved in 1 N hydrochloric acid, and pH was adjusted to 7.4 with 1 N sodium hydroxide. MCT (60 mg/kg) or its vehicle was administered to rats as a single subcutaneous injection. Pulmonary Hypertension developed 21 days after the administration. 31,32
Measurement of RVP
Rats were anesthetized with 60 mg/kg ketamine and 5 mg/kg xylazine. Right ventricular pressure (RVP) was accessed via a blunt dissection in the right third and fourth intercostal space, and a 23-gauge needle placed on the tip of a polyethylene 50 catheter was inserted into the right ventricle (RV), after which direct RVP recordings were obtained by Biopac MP150 Data Acquisition (Biopac Systems,Inc., California, USA).
Measurement of right ventricular hypertrophy
The heart was dissected and weighed after excess blood was removed. The right ventricular wall was separated from the left ventricle and septum to determine the wet weight. The ratio of RV to total heart weight (RV/HW) was calculated to determine the index of right ventricular hypertrophy.
Biochemical measurements
Liver and lung tissues were excised from animals. The liver and lung samples were stored in a refrigerator at −80°C until they were used for biochemical analysis. The tissues were used for the determination of GSH, SOD, CAT and GST levels in groups.
GSH assay
The tissues were washed with phosphate buffered saline (PBS) solution, pH 7.4. They were diluted at the ratio of 1:20 (w:v) with cold buffer (50 mM 2-(N-morpholino)ethanesulfonic acid, pH 6–7, containing 1 mM ethylenediaminetetraacetic acid (EDTA)) and were homogenized. The homogenates were centrifuged at 10,000g for 15 min at +4°C. The supernatants were removed and were deproteinated. Then, the samples were used for total GSH assay. Limit of detection (LOD) of the assay was 0–16 µM.
GST assay
The tissues were washed with PBS solution, pH 7.4. They were diluted at the ratio of 1:20 (w:v) with cold buffer (100 mM potassium phosphate, pH 7, containing 2 mM EDTA) and were homogenized. The homogenates were centrifuged at 10,000g for 15 min at +4°C. The supernatants were removed and were used for assay. LOD of the assay was 24–128 nmol/min/ml.
CAT assay
The tissues were washed with PBS solution, pH 7.4. They were diluted at the ratio of 1:20 (w:v) with cold buffer (50 mM potassium phosphate, pH 7, containing 1 mM EDTA) and were homogenized. The homogenates were centrifuged at 10,000g for 15 min at +4°C. The supernatants were removed and were used for assay. LOD of the assay was 2–34 nmol/min/ml.
SOD assay
The tissues were washed with PBS solution, pH 7.4. They were diluted at the ratio of 1:20 (w:v) with HEPES buffer (pH 7.2, containing 1 mM ethylene glycol diacetate, 210 mM mannitol, and 70 mM sucrose) and were homogenized. The homogenates were centrifuged at 10,000g for 15 min at +4°C. The supernatants were removed and were used for assay. LOD of the assay was 0.025–0.25 U/ml.
Materials
The chemicals and drugs used were obtained from the following sources: ketamine (Ketalar®; Pfizer, lstanbul, Turkey), MCT (Sigma, Missouri, USA), NAC (Asist®; Hüsnü Arsan, Istanbul, Turkey) and xylazine (Sigma, Missouri, USA). For the measurements of GSH, GST, SOD and CAT levels in tissue homogenates, enzyme-linked immunosorbent assay kits from Cayman Chemical Company (Michigan, USA) were used according to the manufacturer’s instructions.
Statistical analysis
RVP, RV/HW, GSH, GST, SOD and CAT ratios were expressed as mean ± standard deviation. Statistical analyses were performed with one-way analysis of variance followed by Tukey’s honestly significant difference test with the GraphPad Prism version 5.0 software (Graphpad Software, California, USA). A p value of <0.05 was considered statistically significant.
Results
RVP measurements
The RVP was significantly higher in male and female rats treated with MCT compared to those in the control group. There was no significant difference in RVPs between the C-F and C-M groups. On the other hand, the MCT-M group had significantly higher RVP than the MCT-F group (Figure 1).

Assessment of right ventricular pressure in anesthetized rats. C-F: control (female) group; MCT-F: monocrotaline (female) group; C-M: control (male) group; MCT-M: monocrotaline (male) group. *Different from C-F (p < 0.05). **Different from C-M (p < 0.05). +Different from MCT-F (p < 0.05).
Assessment of right ventricular hypertrophy
When the male and female rats from the control group and those from the MCT group were compared in terms of RV/HW ratio, the right ventricular mass significantly increased in MCT groups. The RV/HW ratio was significantly higher in the C-M group than in the C-F group. Similarly, the right ventricular mass group is found to be significantly higher in the MCT-M group than in the MCT-F group (Figure 2).

Assessment of right ventricular hypertrophy in rats. C-F: control (female) group; MCT-F: monocrotaline (female) group; C-M: control (male) group; MCT-M: monocrotaline (male) group. *Different from C-F (p < 0.05). **Different from C-M (p < 0.05). +Different from MCT-F (p < 0.05).
Liver GSH level/lung GSH Level
In the liver tissue, decreased level of GSH was observed in the rats treated with MCT compared to control animals (p = 0.07 for female, p < 0.05 for male). In terms of the effect of gender factor on liver GSH levels in control animals, GSH levels in the liver of female rats were higher than those of male rats (p < 0.05). Similarly, the MCT-F group had significantly higher GSH levels than the MCT-M group.
In the lung tissue, decreased GSH level was found in MCT groups compared to male and female rats in the control group (p < 0.05). The lung GSH levels found in female rats were higher than in male rats (p < 0.05). Similarly, the MCT-F group had significantly higher GSH levels than the MCT-M group.
Liver GST activity/lung GST activity
The decreased GST activity was observed in the liver tissue of MCT groups compared to that of control groups (p < 0.05 for female, p = 0.19 for male). In the control group, female rats had higher liver GST activity than male rats (p < 0.05). Similarly, GST activity was significantly higher in the MCT-F group than in the MCT-M group.
The GST activity significantly decreased in the lung tissue of rats treated with MCT than in that of control groups. In the control group, female rats had higher lung GST activity than males (p < 0.05). The GST activity was significantly higher in the MCT-F group compared to the MCT-M group (p < 0.05).
Liver SOD activity/lung SOD activity
In the liver tissue, SOD activity significantly decreased in animals treated with MCT compared to those in the control group. There were no significant difference between the sexes in both MCT and control groups.
In the lung tissue, SOD activity decreased in the MCT group compared to the female rats in the control group (p < 0.05). Conversely, significantly higher SOD activity was found in male rats in the MCT group compared to control animals. In terms of the effect of gender factor, the lung tissue of female control animals had higher SOD activity than that of male control animals. In contrast to other results, SOD activity was significantly higher in male MCT rats compared to female MCT rats (p < 0.05).
Liver CAT activity/lung CAT activity
The CAT activity increased in the liver tissue of male and female rats in MCT groups compared to that of control animals. No significant difference was found between the sexes in both MCT and control groups. The GSH level and the GST, SOD and CAT enzyme activities in the liver tissue of animals from both groups are shown in Table 1.
The mean GSH, GST, SOD and CAT activities of liver tissues in groups.
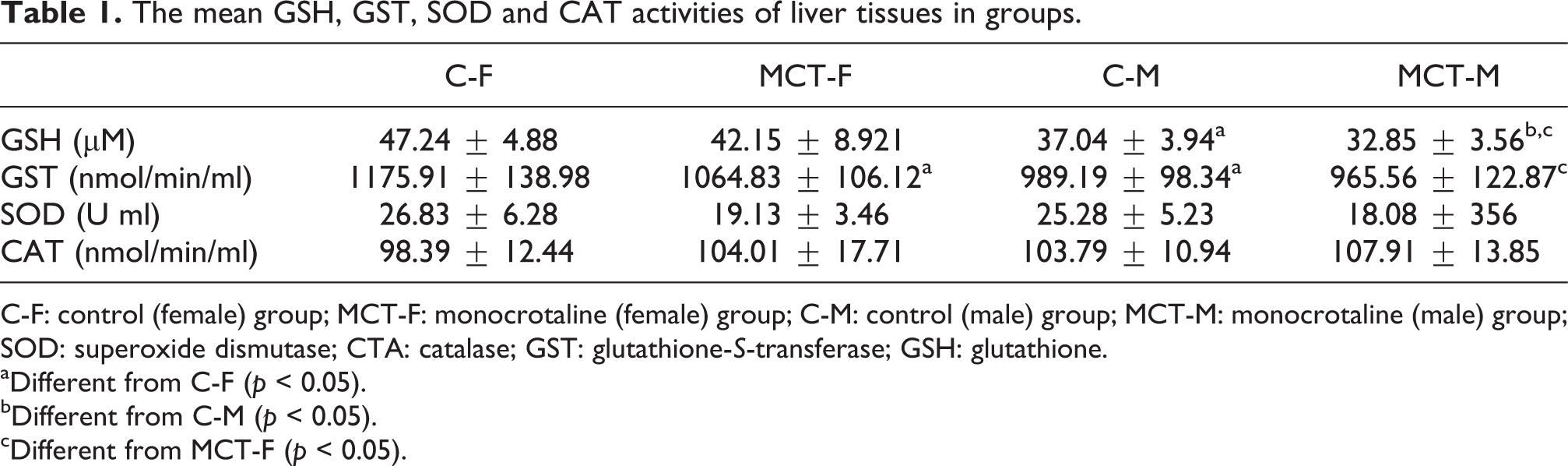
C-F: control (female) group; MCT-F: monocrotaline (female) group; C-M: control (male) group; MCT-M: monocrotaline (male) group; SOD: superoxide dismutase; CTA: catalase; GST: glutathione-S-transferase; GSH: glutathione.
aDifferent from C-F (p < 0.05).
bDifferent from C-M (p < 0.05).
cDifferent from MCT-F (p < 0.05).
Similar to the liver tissue, CAT activity of lung tissue increased in male and female rats in MCT groups compared to control groups (p = 0.07). There were no significant differences between male and female rats in both MCT and control groups. The GSH level and the GST, SOD and CAT enzyme activities in the lung tissue of animals from both groups are shown in Table 2.
The mean GSH, GST, SOD and CAT activities of lung tissues in groups.

C-F: control (female) group, MCT-F: monocrotaline (female) group, C-M: control (male) group, MCT-M: monocrotaline (male) group; SOD: superoxide dismutase; CAT: catalase; GST: glutathione-S-transferase; GSH: glutathione.
aDifferent from C-F (p < 0.05).
bDifferent from C-M (p < 0.05).
cDifferent from MCT-F (p < 0.05).
Discussion
The differences between the sexes in severity and prevalence of cardiovascular and cardiopulmonary diseases have been established in previous studies. 1 –4 The differences identified between the male and female gender cannot be explained only with classical cardiovascular risk factors such as age, diet and lifestyle. 33,34 On the other hand, the mechanism of these differences between the sexes is not yet fully clarified. However, oxidative stress that plays a role in the development and progression of vascular diseases may account for this difference and sex hormones are thought to play an important role in regulating vascular oxidative stress. 17
The significantly increased RVPs found in male and female rats compared to the control animals in the present study were also shown in other studies. 35 As is known, pulmonary endothelium releases mitogenic and antimitogenic mediators, and vasoconstrictors and vasodilators, all of which play a role in regulating vascular tone. The continuous changes occurring in the production of these vascular cell mediators impair the homeostatic balance, affect the vascular tone and induce the development of vascular remodeling. 36,37 Therefore, in the present study, RVP might be increased in the MCT groups as a result of impaired homeostasis in pulmonary vasculature due to MCT-induced endothelial damage.
Right ventricular hypertrophy develops in later stages of the disease as an adaptive response against increased pulmonary vascular resistance in pulmonary hypertension. In the present study, the right ventricular hypertrophy/HW ratio, which is the measure of right ventricular hypertrophy, significantly increased in male and female MCT rats compared to control rats. Previous studies have also reported the development of right ventricular hypertrophy in rats treated with MCT. 23,35
The increased RVPs and right ventricular/HW ratio found in male rats compared to females in the MCT group can be considered as that MCT-induced pulmonary hypertension is more severe in male rats. The differences found between the sexes in the clinical course of disease are possibly related to sex hormone levels. Previous studies have demonstrated that estrogen mediates the decreased vascular oxidative stress through regulating the expression and function of antioxidant enzymes. 6,17,35 It was also reported that estrogen decreases pulmonary artery pressure, right ventricular hypertrophy and vascular remodeling, and it has preventive and therapeutic effects on MCT-induced pulmonary hypertension in rats. 38,39 In a study, estrogen levels may help to avoid the appearance of pulmonary edema, the characteristic of MCT model and right ventricular failure. 40 Based on these results, it can be considered that the gender difference found in MCT-induced pulmonary hypertension resulted from higher estrogen levels in female rats than in males.
The antioxidant enzymes in the cell such as GST, CAT and SOD and complex antioxidant mechanisms protect the cells against reactive oxygen species. 17,19 Under physiological conditions, O2 − is rapidly metabolized to hydrogen peroxide (H2O2) by an important cellular antioxidant defense mechanism, SOD. Then, the CAT enzyme converts H2O2 to water and molecular oxygen. 19,41,42 The GST is a multifunctional enzyme that plays an important role in cellular detoxification. It protects the cells against toxic substances through conjugating them to GSH. Then, it neutralizes the nucleophilic parts of these toxins to render them more soluble in water. 19,41 GSH is a nonenzymatic antioxidant, which plays an important role particularly in the protection of hepatocytes against exogenous toxins. Depletion of cellular GSH stores is known to cause oxidative stress. 19 Activities of these enzymatic and nonenzymatic antioxidant systems are thought to be regulated by hormones and nutrients. 19,41,42
In the present study, the GSH level and GST activity significantly decreased in both male and female rats treated with MCT. On the other hand, MCT decreased SOD enzyme activity in both sexes (except for lung tissue of male rats) and increased CAT enzyme activity. These differences observed between MCT and control groups were statistically significant for SOD enzyme activity but not for CAT enzyme activity. The studies of various tissue homogenates from rats with MCT-induced pulmonary hypertension have reported inconsistent results. In a rat model of MCT-induced pulmonary hypertension, SOD, CAT and GSH activities in liver and lung tissue were found to be significantly increased. 43 On the other hand, another study found decreased SOD and increased CAT activities at week 3 after the MCT injection. 44 In another study by Amin et al., the decreased GSH level and GST activity and increased SOD and CAT activities were found in rats treated with MCT. 28 Farahmand et al. injected MCT to the rats and found that CAT and SOD activities in the RV slightly increased after 1 week, significantly increased after 2 weeks and significantly decreased after 6 weeks. 45 In a study of MCT-induced right ventricular failure, myocardial CAT and SOD activities were not found to be significantly different from the control group. 26 Cellular enzymatic/nonenzymatic defense mechanisms against oxidative stress induced in tissues by the electrophilic metabolite of MCT are thought to be depleted in male and female rats treated with MCT. Reduced GSH levels and GST and SOD enzyme activities found in our animal groups treated with MCT may be interpreted as oxidative stress plays a role in the pathogenesis of MCT-induced pulmonary hypertension. In studies with rat MCT model, gender differences and underlying cause regarding antioxidant enzyme status in the target organs were not investigated. Therefore, our study might present a definite report in this subject.
In terms of gender, control female rats had higher GSH level and higher GST and SOD activities in the lung and liver tissues compared to male controls. For the CAT enzyme activity in lung and liver tissue, no statistically significant difference was found between the two control groups. In a study of Hackenhaar et al., SOD and CAT activities were found to be significantly higher in lung tissue of female rats when they were compared with male rats. 42 Also Yamamoto et al. reported decreased GSH and increased CAT activity in liver tissue of male rats when compared with female rats. 46 In another study, liver GST levels were lower in male control rats than females. 47 Wang et al. (2011) found higher CAT, GST and SOD activities in the liver tissue of female rats than in that of males. This was considered to be the effective mechanism in the protection of female mice from the liver injury induced by Dioscorea Bulbifera L. rhizome. 19 Antioxidant status between genders in rats was rarely investigated and showed conflicting results in each study. Our study showed similarity in terms of enzyme profile with the majority of previous studies we indicated above. In a previous study, SOD and CAT activities were found to be higher in macrophages of female rats than in that of males and this was explained by the regulatory effect of estrogen on activities of these enzymes. 41 In another study by Malorni et al., 18 smooth muscle cells derived from aorta of male rats and GSH, SOD and CAT enzyme activities were found to be higher in cell lines derived from the female rats. The authors suggested that this difference observed in antioxidant defense systems between the sexes at baseline status might be a cause of well-known gender differences in cardiovascular disease. 18 In a sucrose-induced rat model of metabolic syndrome, Perez-Torres et al. 48 determined an antioxidant activity for estrogen and a peroxidant activity for androgens. The authors concluded that sex hormones have a regulatory effect on SOD and CAT enzyme activities, with estrogen affecting these enzymes positively. 48
In conclusion, compared to male rats, the higher antioxidant enzyme/enzymatic activity in females can be considered as a defense mechanism against many diseases including oxidative stress-induced cardiovascular diseases. The low activity defense system observed in lung and liver tissues of control rats may be considered as a cause of aggressive course of MCT-induced pulmonary hypertension in male rats. The positive effect of estrogen on GSH level and GST and SOD enzyme activities as well as its claimed antioxidant property may have a protective effect against MCT-induced pulmonary toxicity in female rats. Further studies are warranted to evaluate the effects of sex hormones on oxidative stress and antioxidant defense system in oxidative stress-induced cardiovascular damage models and should also include the measurement of estrogen levels. The role of gender and related antioxidant system in the MCT model in rats was investigated in our study and the results seem to fortune that the female gender is less likely to develop cardiovascular disease, it should also be kept in mind that not only the gender factor but other risk factors such as age, nutrition, genetic factors and lifestyle are also effective in the initiation and progression of these diseases. Future studies should shed light on the differences between the sexes by enzymatic/nonenzymatic activity measurements in male and female rats with standardized risk factors.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
