Abstract
The aim of this study was to examine the antitumour activity of resveratrol in human colorectal cancer cell lines (HCT116 and Caco2) and to explore its mechanism of action assuming that it is by calorie-restriction effect. Resveratrol inhibited the proliferation of colon cancer cells with half maximal inhibitory concentration (IC50) equal to 50 and 130 μM for HCT116 and Caco2, respectively. Caco2 cells appeared with significant time-dependent increase in the glycolytic pathway, a behaviour that was absent in HCT116 cells. Resveratrol (100 μM) significantly decreased the glycolytic enzymes (pyruvate kinase and lactate dehydrogenase) in Caco2 cells, while an increase in citrate synthase activity and a decrease in glucose consumption were observed in both cell lines. Moreover, resveratrol downregulated the expressions of leptin and c-Myc, and decreased the content of vascular endothelial growth factor. The apoptotic markers, caspases 3 and 8, were activated and the Bax/BCl2 ratio was increased. The study suggested a promising anticancer activity of resveratrol, calorie-restriction pathway may be one of the driving forces for this activity.
Introduction
Colorectal cancer is the third most frequent cancer in the Western countries according to the American cancer society in 2008. It is also responsible for about 10% of all cancer-related deaths in the United States. 1 The incidence is now rapidly increasing in areas that were previously at low risk such as Latin America, Asia and Africa. 2 Worldwide variations in the incidence provide evidence that environmental factors influence the risk of the disease. 3
Obesity, physical inactivity and diets high in total calories seem to be the pre-disposing factors of the disease. 4 There is evidence that the action of many of these risk factors is mediated through hyperinsulinaemia and that insulin may stimulate the growth of colon tumours. 5
Chemotherapy contributes to overall patient’s survival with compromised quality of life. Naturally occurring substances, derived from plant-based diet, appear to be promising strategy for cancer treatment. These compounds act by the mechanism that targets multiple pathways rather than a single pathway, as do many synthetic compounds. 6
Resveratrol (3,5,4′-trihydroxystilbene) is a naturally occurring polyphenolic compound present in grapes, berries, peanuts and red wine. 7 It has broad-spectrum health beneficial effects such as antioxidant, anti-inflammatory, cardioprotective and anticarcinogenic effects both in vivo and in vitro. 8,9 It was found to have chemoprevention\chemotherapeutic activity against all the three major stages of carcinogenesis, that is, initiation, promotion and progression. 10,11 Resveratrol can block tumour initiation process by targeting cytochrome p450 enzymes that activate procarcinogens. 12 It also induces several phase II detoxification or antioxidant enzymes. 13 In vitro, it has been found to have proapoptotic effects against tumour cells and induces apoptosis. 14 Although, several studies suggested multiple molecular targets of resveratrol to prevent cancer, Baur and Sinclair 8 have proposed that resveratrol might follow the same pathway as does calorie restriction. However, how exactly resveratrol can follow this mechanism is not yet investigated. The objective of this study was to test the growth inhibitory effect of resveratrol on colon cancer cell lines, Caco2 and HCT116, and to confirm the hypothesis of dietary restriction.
Methods
Drug
Resveratrol was obtained from Sigma Aldrich Chemical Co. (St Louis, Missouri, USA). It was freshly dissolved in dimethyl sulphoxide (DMSO) and then diluted with incubation medium, Roswell Park Memorial Institute 1640 (RPMI-1640), before each experiment, so that the final concentration of DMSO was not more than 0.1% (v/v).
Chemicals and antibodies
DMSO, RPMI-1640 medium, sulphorhodamine-B (SRB) and lactate dehydrogenase (LDH) were all purchased from Sigma Aldrich. Phosphoenolpyruvate (PEP) was obtained from Fluka Biochemika (St. Gallen, Switzerland) and also nicotinamide adenine dinucleotide (NADH) from Roth (Karlsruhe, Germany). Protease inhibitor cocktail was obtained from Biobasic (Markham, ON, Canada). Monoclonal anti-human caspases 3 and 8 were obtained from R&D Systems (Minneapolis, USA), monoclonal anti-BCl2 antibody and β-actin were obtained from Sigma Aldrich. Polyclonal anti-human leptin and c-Myc were obtained from Koma Biotech (Gayang dong, Seoul, Korea). All other chemicals were of reagent grade and used without further purification.
Cell lines and culture conditions
Human colorectal carcinoma cell lines, Caco2 and HCT116, used in this study were obtained from the American Type Culture Collection (Manassas, USA). They were maintained and grown at the Egyptian National Cancer Institute (Cairo, Egypt) in RPMI-1640 supplemented with 10% fetal bovine serum, 2 mM
Cytotoxicity assay
Cytotoxicity was evaluated using the SRB assay. 15 Briefly, exponentially growing cells were seeded in 96-well microtitre plates at an initial density of 5 × 103/well. After 24 h, resveratrol was added to each well at various concentrations (12.5–200 μM) and incubated in a humidified, 5% CO2 atmosphere at 37°C for 48 and 72 h. Cells were fixed with 10% trichloroacetic acid for 1 h at 4°C and stained with 0.4% SRB for 30 min. The wells were then washed four times with 1% acetic acid, air-dried and the dye was solubilized with 10 mM Tris base (pH 10.5). The optical density (O.D.) was measured spectrophotometrically at 570 nm with the microplate reader (Tecan Sunrise™, Männedorf, Switzerland). The experiment was repeated three times. The percentage of cell survival was calculated as follows: survival fraction = O.D.(treated cells)/O.D.(control cells).
Determination of IC50 values
The half maximal inhibitory concentration (IC50) values (the concentration of resveratrol required to produce 50% inhibition of cell growth) of the two human cancer cell lines after 48 and 72 h treatment were calculated using sigmoidal dose response curve-fitting models (Graphpad Prizm Software, version 3, GraphPad Software, Inc. Avenida de la Playa La Jolla, USA).
Glucose consumption
Cells were plated in media without phenol red (Biowest, Nuaille, France) in six-well plates in which the control and the treated wells were incubated with 0 and 100 µM resveratrol for 48 and 72 h. The glucose concentration was determined colorimetrically at 500 nm using the Diamond Diagnositics kit (Fiske St, Holliston, USA). 16
Immunocytochemistry
Insulin expression in control and 48 h-treated cells was determined by an immunocytochemistry technique. Insulin antibody (Koma Biotech) is bound to intracellular insulin protein. Insulin antibody is located by the streptavidin–biotin kit (Thermo Fisher Scientific, Geel, Belgium) and cells were photographed with a light microscope (200×).
Citrate synthase activity measurement
Control and treated cells were harvested, washed twice with ice-cold phosphate buffered saline and pelleted at 1200 r/min for 5 min. The cells were then lysed in lysis buffer containing 150 mM sodium chloride, 10 mM Tris, 0.2% Triton X-100, 0.3% NP-40, 0.2 mM sodium vanadium oxide and protease inhibitor cocktail, pH 7.7. The supernatants were collected after centrifugation at 14,000 r/min for 15 min at 4°C, and the protein content was determined by the Bradford method. 17 Citrate synthase activity was measured according to kit instructions (Sigma Chemical Co., St Louis, Missouri, USA). 18 The yellow product (5-thio-2-nitrobenzoic acid) is observed spectrophotometrically by measuring the increase in the absorbance at 412 nm. Enzyme activity was expressed in units per milligram of protein.
Pyruvate kinase activity measurement
Cell lysates were prepared as mentioned previously and pyruvate kinase (PK) activity was measured using PEP as a substrate. 19 The assay mixture consisted of 200 µl of 50 mM 4-(2-hydroxyethyl)-1-piperazineethanesulphonic acid, 328 µl of distilled H2O, 100 µl of 100 mM magnesium chloride, 100 µl of 500 mM potassium chloride, 50 µl of 40 mM adenosine diphosphate, 100 µl of 100 mM PEP, 50 µl of 10 mM NADH, 22 units LDH and 50 µl of sonicated protein in a final volume of 1 ml. The decrease in A 340 for 4–5 min was recorded and ΔA 340 per minute from the initial linear portion of the curve was calculated. Enzyme activity was expressed in units per milligram of protein.
LDH activity measurement
Intracellular LDH activity was determined after 48 and 72 h of resveratrol treatment. LDH activity was measured using readymade kit (Spectrum Diagnostics, Rosewood Ln N, Plymouth, USA). 20 The oxidation of NADH was measured at 340 nm for 5 min and enzyme activity was expressed in units per milligram of protein.
SDS-polyacrylamide gel electrophoresis and immunoblot analysis
Aliquots of protein supernatants containing equal amounts of protein and sodium dodecyl sulphate (SDS)-reducing buffer were boiled for 5 min, electrophoresed on SDS-polyacrylamide gels and transferred to polyvinylidene difluoride membranes. The membranes were blocked with 5% non-fat dry milk and probed with specific primary antibodies of monoclonal anti-human caspases 3 and 8, monoclonal anti-BCl2 and Bax and polyclonal anti-leptin and c-Myc antibodies followed by incubation with peroxidase-conjugated secondary antibodies. The blots were developed with Amersham ECL Western Blotting Detection Reagents (GE Healthcare, Amersham Place, Little Chalfont, UK) according to the manufacturer’s protocol. The blots were quantified by Scion image software (Scion Corporation, version 0.4.0.3, Maryland, USA) and protein loading was corrected for β-actin as loading control.
DNA degradation
DNA was extracted according to the instruction of genomic DNA extraction and purification kit (Bioflux, Hangzhou, China). The required amount of DNA was separated on 1% agarose gel with 0.5× Tris/borate/ethylenediaminetetraacetic acid (EDTA) buffer (44.5 mM Tris base, 44.5 mM boric acid and 1 mM EDTA, pH 8.0). Ethidium bromide-stained DNA in the gel was visualized under ultraviolet light and photographed.
Measurement of VEGF secretion
Cells were seeded in 12-well plates and incubated for 24 h before treatment with resveratrol for the indicated time intervals. Phenol red-free culture supernatants were collected and assessed for vascular endothelial growth factor (VEGF) levels by ELISA using the Quantikine Human VEGF Immunoassay kit (R&D Systems, McKinley Place NE, Minneapolis, USA) according to the manufacturer’s protocol. 21 The O.D. of each well was measured at 450 nm within 30 min.
Statistical analysis
All data are expressed as mean ± SD. Differences between the tested groups were tested for statistical significance using two-way analysis of variance (ANOVA) in the SRB cytotoxicity study and one-way ANOVA followed by the Tukey–Kramer test for post hoc analysis in rest of the experiment. Non-parametric ANOVA was carried out for comparison between three blots of Western blotting followed by the Kruskal–Willis test for post hoc analysis. The 0.05 level of probability was used as the criterion of significance using GraphPad InStat, version 4.0 (GraphPad, San Diego, California, USA).
Results
Inhibition of the proliferation of HCT116 and Caco2 cells by resveratrol
The cytotoxic effect of resveratrol on HCT116 cells was more pronounced than in Caco2 cells. The IC50s of resveratrol of HCT116 cell line were 50 and 40 µM, while of Caco2 cells were 131 and 115 µM after 48 and 72 h, respectively, as shown in Figure 1.
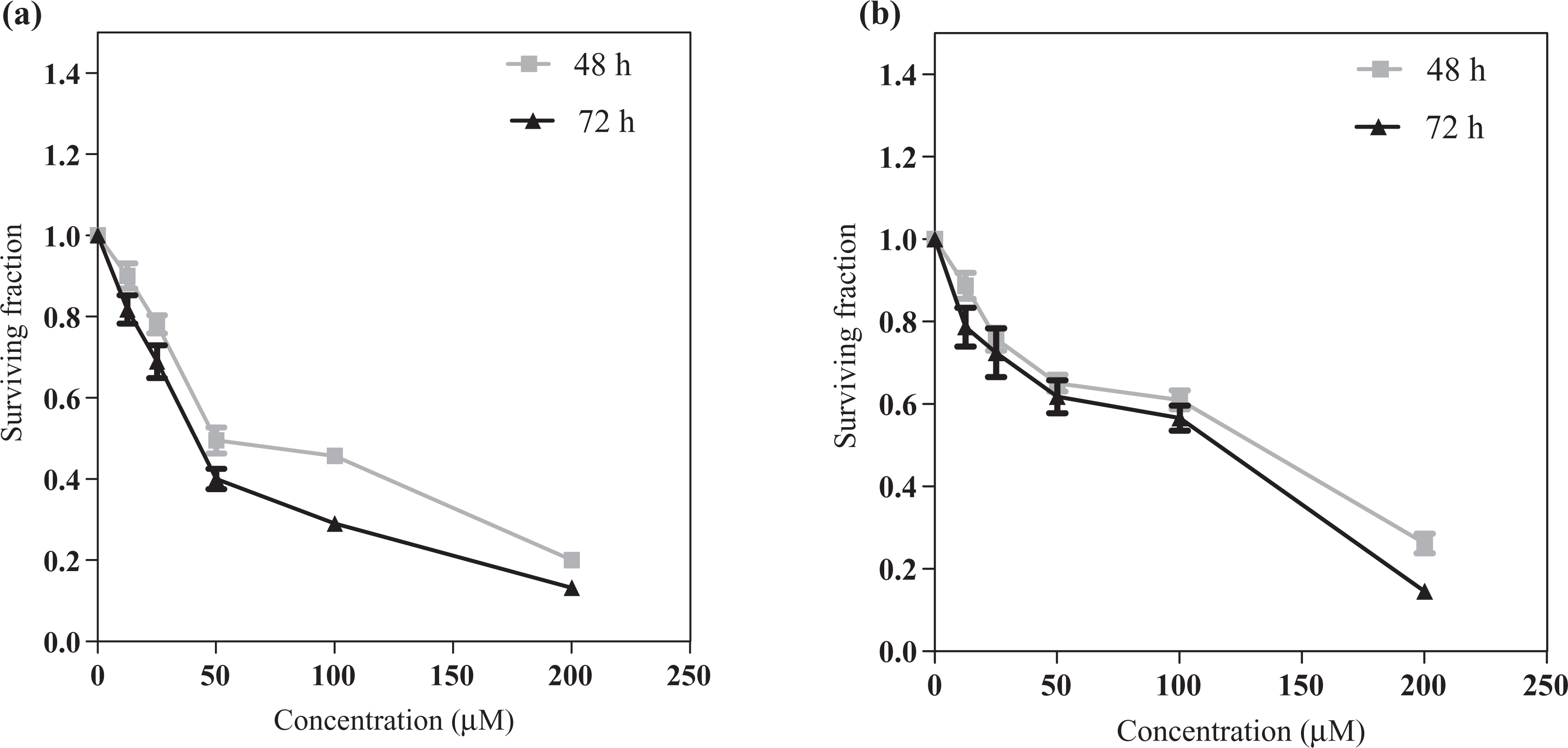
Effect of resveratrol on proliferation of HCT116 (a) and Caco2 (b) cells treated with increasing concentrations of resveratrol (12.5–200 µM) for 48 and 72 h. All data are expressed as mean ± SD from three independent experiments, each performed in triplicate.
Resveratrol decreases glucose consumption
A significant time-dependent increase in glucose consumption in untreated control cells of both cell lines was found (Figure 2). On the other hand, after incubation with resveratrol, glucose consumption was shown to be significantly reduced after 72 h compared to 48 h in both cell lines. In addition, there was a significant reduction in glucose consumption after resveratrol treatment of HCT116 cells at 72 h and of Caco2 cells after 48 and 72 h compared with their respective controls.

Effect of 100 µM resveratrol on the percentage of glucose consumption of colon cancer cell lines HCT116 (a) and Caco2 (b) after 48 and 72 h. (1) Data were expressed as means ± SD of three independent experiments, each performed in triplicate. (2) Differences between different time intervals were tested for statistical significance using one-way analysis of variance followed by the Tukey–Kramer test for post hoc analysis. a,bSignificantly different when control and treated groups at 72 h are compared with their respective one at 48 h at p value <0.05. cSignificantly different when treated groups compared with their respective control at p value <0.05.
Resveratrol decreases glycolytic enzymes (PK and LDH)
PK and LDH activity showed significant increase with time in control Caco2 cells. After resveratrol treatment, PK activity significantly decreased after 72 h of treatment in HCT116 cells and Caco2 cells compared with 48 h. When compared with their respective control, Caco2 cells showed significant decrease in PK activity at 48 and 72 h and HCT116 at 72 h only. However, LDH activity in Caco2 cells significantly decreased after 48 and 72 h (Figure 3).
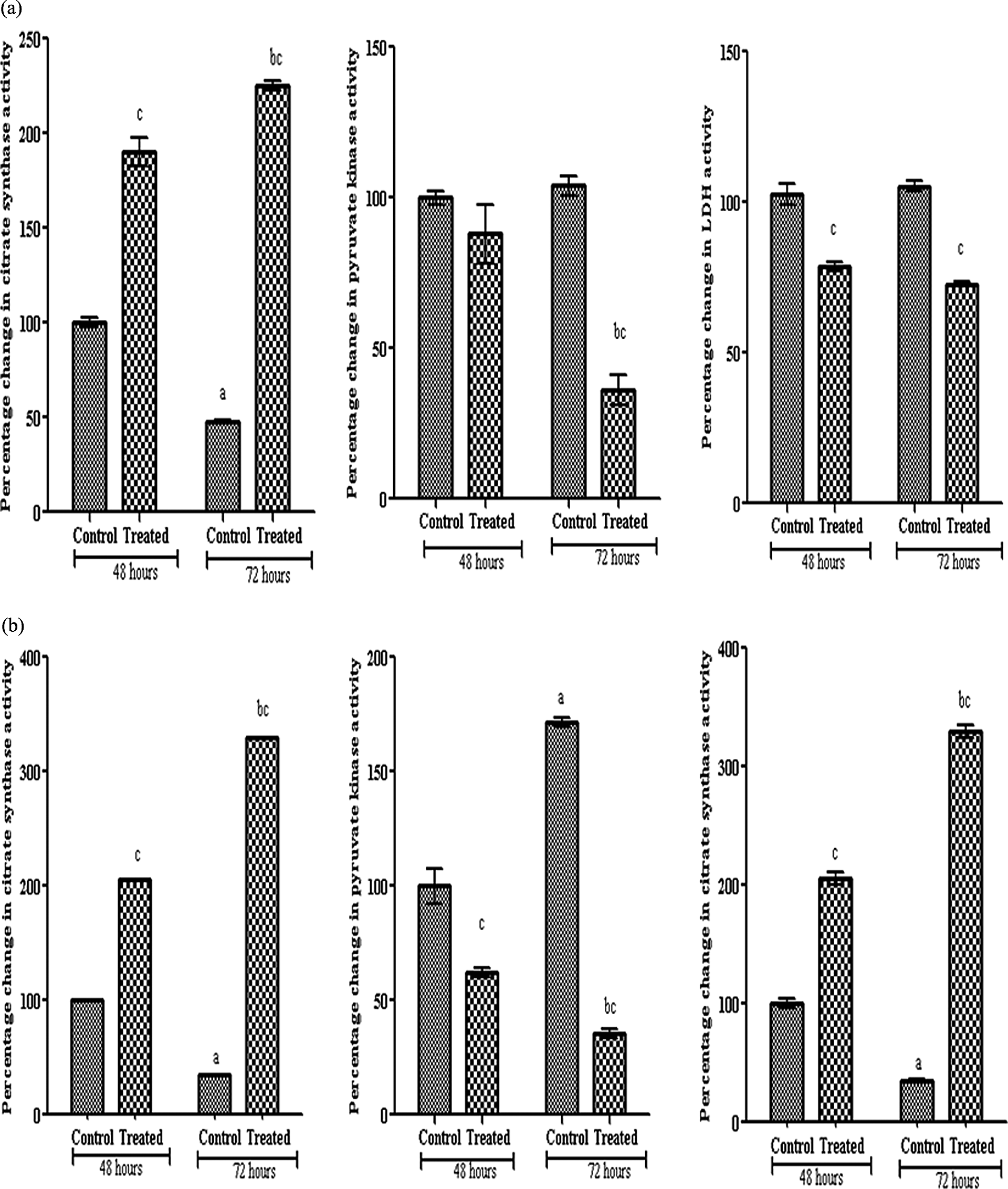
Percentage change in citrate synthase, pyruvate kinase and LDH activities in HCT116 (a) and Caco2 (b) cells after 48 and 72 hours of 100µM resveratrol treatment. (1) Data were expressed as means ± SD of three independent experiments, each performed in triplicate. (2) Differences between different time intervals were tested for statistical significance using one way analysis of variance (ANOVA) followed by Tuky Krammer test for post-hoc analysis. a,bSignificantly different when control and treated groups at 72 hours compared with their respective one at 48 hours at p value < 0.05. cSignificantly different when treated groups compared with their respective control at p value < 0.05. LDH: lactate dehydrogenase.
Resveratrol increases citrate synthase activity
Resveratrol of 100 µM significantly decreased the citrate synthase activity in control untreated cells of both cell lines. The enzyme activity was decreased significantly after treatment with resveratrol for 72 h compared to 48 h and compared with their respective controls in both cell lines (Figure 3).
Resveratrol induces insulin expression
Increase in insulin expression (brown dots in cytoplasm) in HCT116 and Caco2 cells after 48 h of resveratrol treatment was determined by the immunocytochemistry technique (Figure 4).

Effect of 100 µM resveratrol on insulin expression (brown dots in yellow cytoplasm and nuclei stained purple) as determined by an immunocytochemistry technique, streptavidin–biotin–peroxidase method in HCT116 (a) and Caco2 (b) cells after 0 and 48 h and photographed with a light microscope (×200).
Decreased leptin and c-Myc expressions by resveratrol
As shown in Figure 5, leptin expression was not detected in HCT116 cells, while it was expressed in Caco2, which decreased significantly after treatment with resveratrol. Moreover, c-Myc protein expression significantly decreased after 48 h of resveratrol treatment in both HCT116 and Caco2 cell lines.

Effect of 100 µM resveratrol on leptin and c-Myc protein levels (a′), percentage of c-Myc (b′) and leptin (c′) expressions in HCT116 (a) and Caco2 (b) cells after 24 and 48 h using the Western blot technique. Data were expressed as mean ± SD of three blots. (1) Band intensities were measured using Scion image program. (2) Differences between different time intervals were tested for statistical significance using one-way analysis of variance followed by Dunette’s test for post hoc analysis. a p value <0.05 was considered a significant difference when 24 and 48 h-treated groups are compared with 0 h control group.
Resveratrol induces activation of intrinsic and extrinsic apoptotic pathways
The effect of resveratrol treatment on apoptotic markers, BCl2, Bax, along with caspases 8 and 3 is shown in Figure 6. The Bax/BCl2 ratio increased in both cell lines. Furthermore, resveratrol treatment resulted in the activation of caspase 8 in Caco2 cell line only, while activation of executioner caspase 3 was in both cell lines as shown in Figure 7.
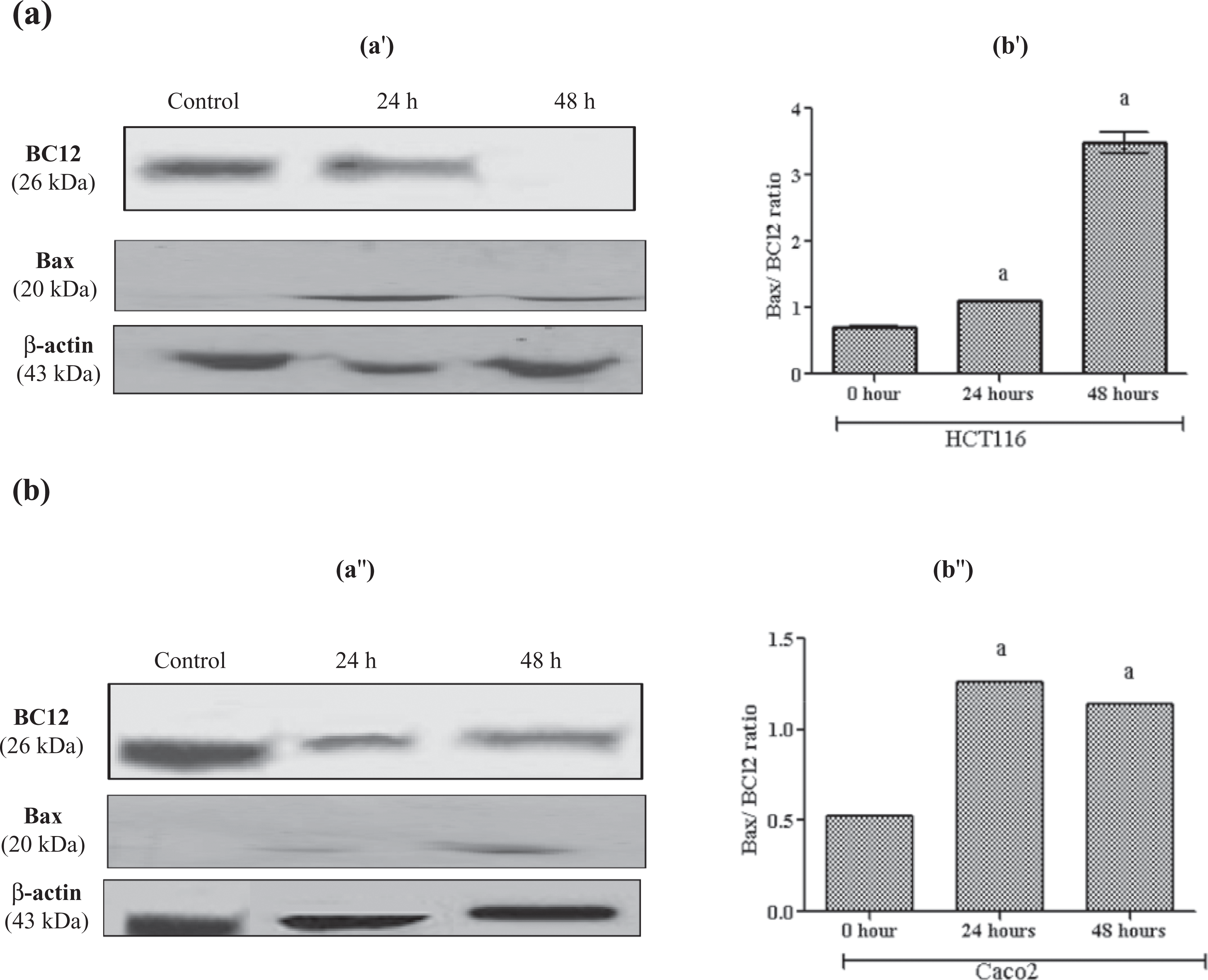
Effect of 100 µM resveratrol on BCl2, Bax protein levels (a′), Bax/Bcl2 ratio (b′) in HCT116 (a′) and Caco2 (b′) cells after 24 and 48 h using the Western blot technique. The legend as in Figure5.
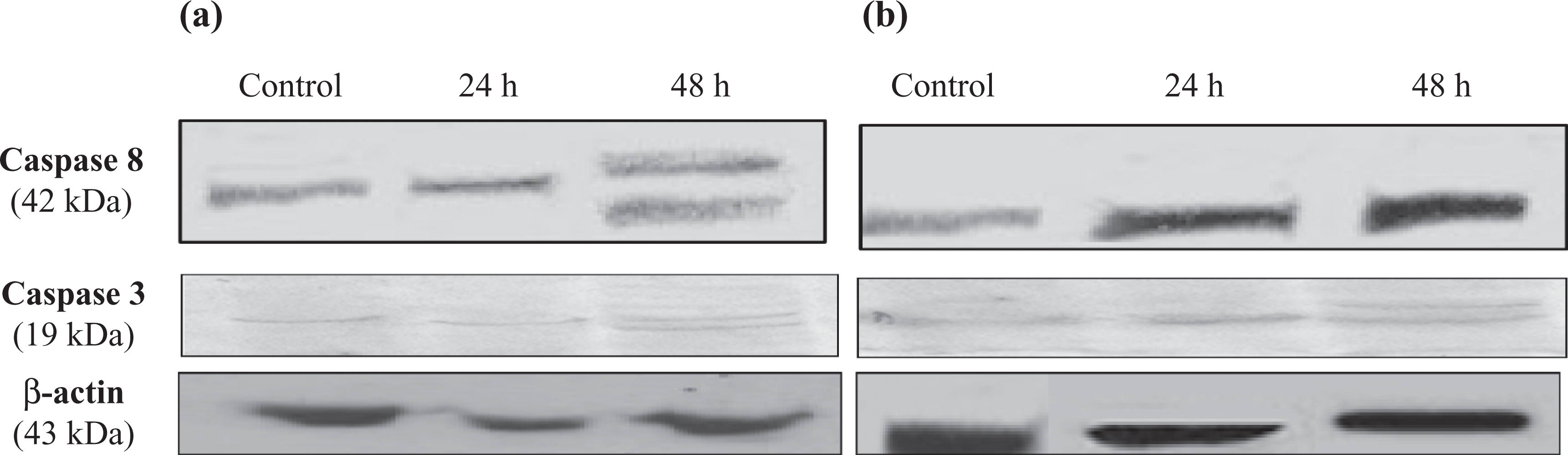
Effect of 100 µM resveratrol on caspase 8 and 3 activations after 24 and 48 h using the Western blot technique in HCT116 (a) and Caco2 (b) cells.
Resveratrol did not induce DNA fragmentation
Degradation of DNA into oligonucleosomal fragments is a hallmark of apoptosis. Gel electrophoresis revealed that 100 µM resveratrol induced no DNA degradation after 48 and 72 h in both HCT116 and Caco2 cell lines as shown in Figure 8.
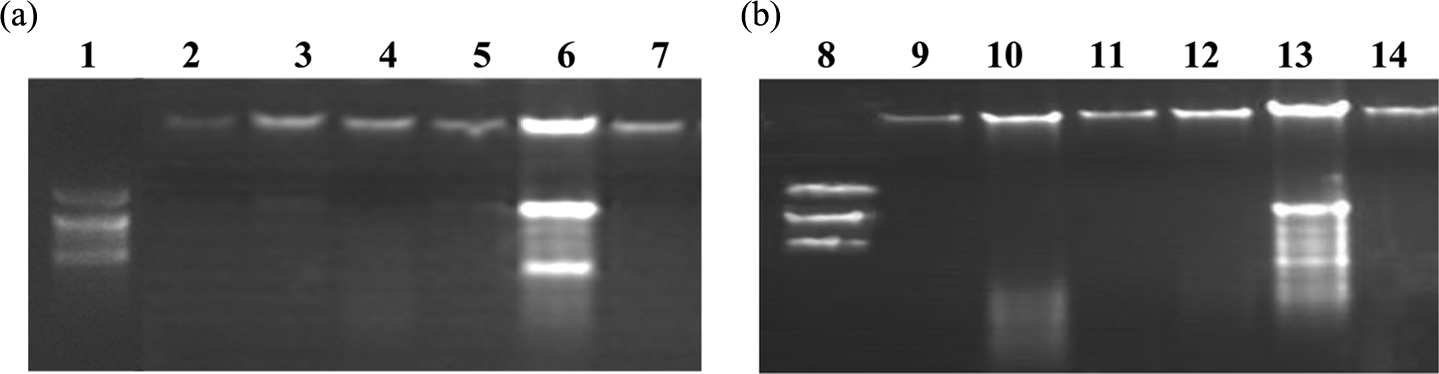
Effect of 100 µM resveratrol on DNA of HCT116 (a) and Caco2 (b) cells at 48 and 72 h. Lanes 1 and 8: DNA ladder. Lanes 2, 5, 9 and 12: control cells treated with vehicle for 48 and 72 h, respectively. Lanes 3, 6, 10 and 13: positive control cells treated with 10 µM cisplatin for 48 and 72 h, respectively. Lanes 4, 7, 11 and 14: cells treated with 100 µM resveratrol for 48 and 72 h, respectively.
Resveratrol decreases the VEGF level
Caco2 cells produced significant increase in VEGF secretion with time. After resveratrol treatment, significant reduction in the VEGF level in HCT116 and Caco2 at 72 h compared with 48 h is shown in Figure 9.
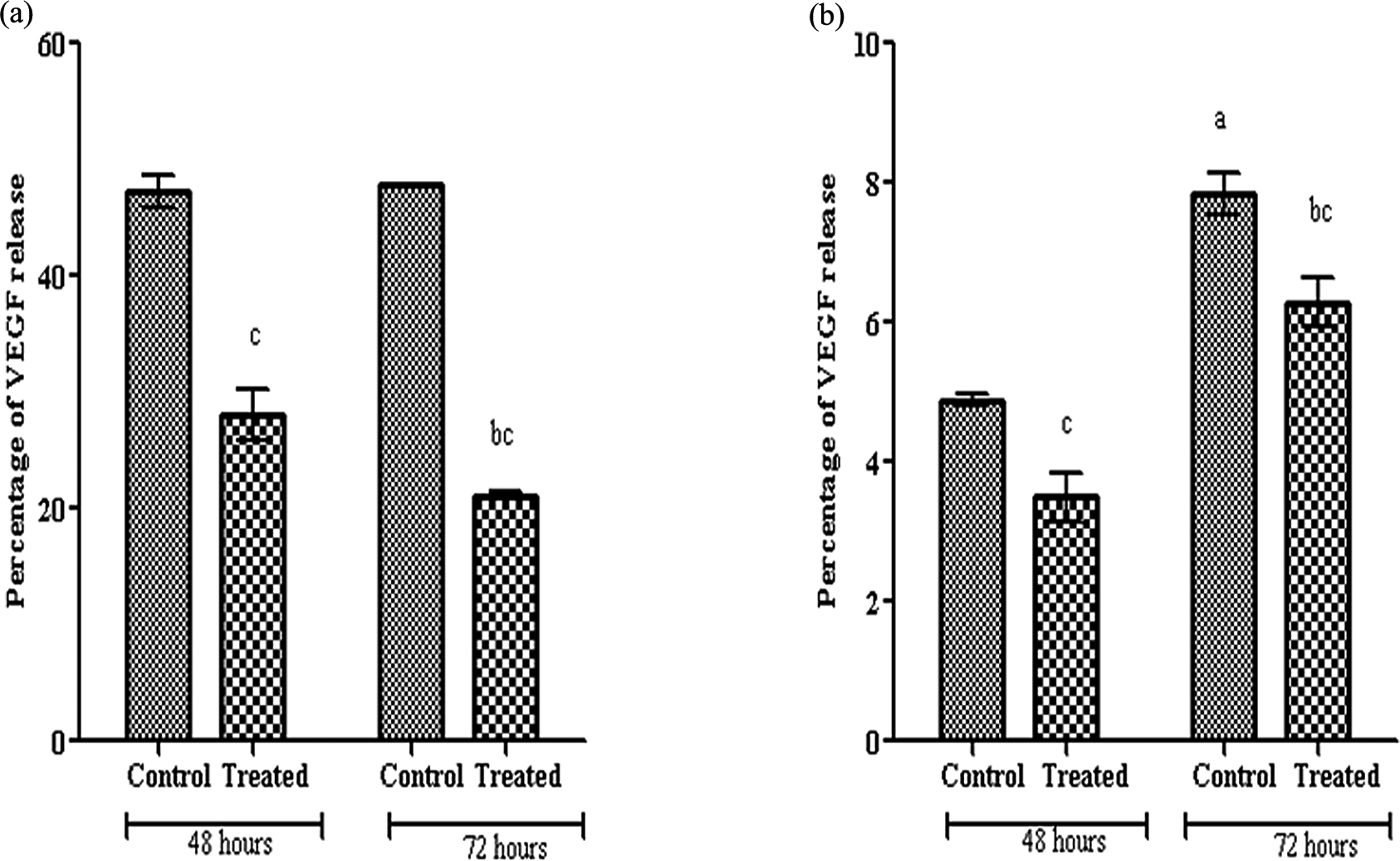
Percentage of VEGF release in HCT116 (a) and Caco2 (b) cells treated with 100 µM resveratrol for 48 and 72 h. (1) All data are expressed as mean ± SD from two independent experiments, each performed in duplicate. (2) The legend as in Figure 2. VEGF: vascular endothelial growth factor.
Discussion
The growth inhibition by resveratrol of various cancer cell types including liver, 22 breast, promyelocytic leukaemia 23 and colon 24 was detected. The current study showed growth inhibition of human colon carcinoma cell lines, HCT116 and Caco2, by resveratrol. The anticancer activity of resveratrol was suggested to be due to the decrease in the activity and expression of ornithine decarboxylase (ODC) and polyamines synthesis 25,26 as well as the inhibition in DNA synthesis mainly ribonucleotide reductase and polymerase activities. 27,28
Tumours have permanent impairment of oxidative metabolism resulting in a compensatory increase in glycolytic flux 29 due to reduced number of mitochondria and increased prevalence of mutations of mitochondrial DNA. 30 HCT116 cells treated with the inhibitor of H+-ATP synthase caused rapid increase in glucose consumption and rates of aerobic glycolysis. 31 However, inhibition of glycolysis effectively increases sensitivity and killing of HCT116 and lymphoma cells to anticancer agents like taxol, doxorubicin, arsenic trioxide, vincristine and ara-C. 32 Our study showed time-dependent increase in glucose consumption and glycolytic enzymes, while decrease in citrate synthase with no insulin expression in the non-treated colon cancer cell lines.
Resveratrol is considered as a promising mitochondrial-targeted drug, which mimics caloric restriction in metazoans, resulting in extended lifespan. 33 This calorie-restricted state was reported as one of the most effective ways known to prevent breast cancer in animal models 34 and in humans as well. 35 These data are consistent with our results that showed decrease in glycolytic enzymes activities and glucose consumption, while enhancement in citrate synthase activity and insulin expression by resveratrol in human colon cell lines. The inhibition of glycolytic enzymes forces cancer cells to revert to oxidative phosphorylation in order to reoxidize NADH and produce ATP. 36 The effect of resveratrol in decreasing glucose consumption could be explained by inhibiting glucose metabolism 37,38 or/and decreasing glucose uptake through reduction in the levels of phosphorylated Akt and mammalian target of rapamycin (mTOR), creating a state that mimics glucose deprivation. 38 In addition, the combination of glucose uptake inhibitor (2-deoxyglucose) and the IGF1-receptor inhibitor II promoted significant apoptosis in HCT116. 39 On the other hand, Faber et al. 40 suggested that the mechanism did not involve diminished facilitated glucose uptake in lymphoma cells by resveratrol. On the contrary, Barger et al. 41 suggested that resveratrol had calorie-restriction effect by enhancement of insulin-mediated glucose uptake in mouse muscles and plasma. In vivo studies showed that dietary resveratrol exerts hypoglycemic effects in diabetic rats, 42 prevents the establishment of insulin-resistance and postpones – or even prevents – the onset of type 2 diabetes initiated in humans. 43 An effect that was attributed to the potentiation of insulin secretion 44 and sensitivity 45 leads to an increase in the tissue responses. 46,47 However, other investigators like Zhang 37 found that resveratrol inhibits insulin responses in H4IIE cells, HepG2 cells, HEK-293 cells and also in Caenorhabditis elegans leading to significantly prolonged lifespan.
c-Myc protein was expressed in a variety of human tumour cells, including transformed colorectal cancer cell lines, 48 to regulate the increases in LDH expression, 49 cell cycle, apoptosis, differentiation, cellular metabolism and genomic instability. 50 c-Myc represents a therapeutic target of chemoprevention and its downregulation might contribute to inhibitory effect exerted by resveratrol. 26 The expression of c-Myc protein was examined in this study and it was found that c-Myc was expressed in both cell lines HCT116 and Caco2 and the expression was decreased after resveratrol treatment with time. Similar effect was also found by Wolter et al. in Caco2 cells, in which the transcription factor c-Myc, a positive regulator of ODC gene, was attenuated by resveratrol treatment. 26
Cancer cells have the ability to produce high amount of lipids rather than utilization of it. 51 In this work, leptin hormone, the product of obesity gene, was found to be expressed in Caco2 cells, while no expression was found in HCT116 cells. Although Amemori et al. 52 and Aloulou et al. 53 explored leptin receptors to be expressed in both cell lines HCT116 and Caco2, leptin was demonstrated to be secreted in the first stage of colon cancer development activating the nuclear factor-kappaB and inducing epithelial wall damage and neutrophil infiltration leading to the inflammatory bowel diseases 54 and it is also involved in breast cancer development. 55 After incubation of Caco2 cells with resveratrol, leptin expression was decreased in this study. Similar effect of resveratrol was found by Szkudelska et al. 56 and Rayalam et al., 57 who documented reduction in leptin secretion in isolated rat adipocytes. They reported that induction of apoptosis was accompanied with inhibition of adipogenesis in 3T3-L1 adipocytic cells. Moreover, it was found that resveratrol reduced the hyperleptinaemia in obese Zucker rats. 58
The induction of apoptosis selectively in cancer cells is regarded as an important strategy for cancer prevention as well as therapy. The calorie-restriction effect of resveratrol was the driving force for its apoptotic and antiangiogenic actions. 59 Herein, our results revealed that resveratrol-cytotoxicity involved apoptotic machinery via both intrinsic pathway (Bax/BCl2 ratio increased) and extrinsic pathway (caspase 8 activation) leading to the activation of caspase 3 in both colon carcinoma cell lines. In agreement with our data, Mahyar-Roemer et al. 60 reported upregulation of Bax in HCT116 cells suggesting that the apoptosis was mediated primarily by mitochondria. In addition, an increase in caspase 3 activity and cell cycle arrest in S to G2 phases were found by Wolter et al. 61 in HCT116 and Caco2. Moreover, resveratrol activated both caspases 3 and 9 and increased the expression of proapoptotic Bax in human glioma U251 cells 62 and activated caspases 8 and 3 and induced the redistribution of Fas receptor on the cell membrane in SW480 human colon cancer cells. 63 The resveratrol apoptotic effect was attributed to the lysosomal cathepsin D that acts upstream for the cytosolic caspases activation in colon cancer cells, HT29 and DLD1. 64
Furthermore, in our study, resveratrol did not induce DNA degradation in both colon cancer cell lines, HCT116 and Caco2, suggesting that it acts as cytostatic and not cytotoxic or it may be very late event. Similarly, no DNA laddering was detected by the study of Schneider et al. in Caco2 cells. 25
Degradation of nuclear DNA is one of the hallmarks of apoptotic cell death. It is a secondary consequence, rather than an integral cause, of apoptosis. It occurs late in response to various apoptotic stimuli. Caspase 3 in particular allows caspase-activated DNase to cleave DNA. So, we suggest that DNA fragmentation is a late process after activation of caspases.
VEGF is one of the important angiogenic factors secreted by the tumour cells, which stimulates tumour neo-angiogenesis and vascular permeability. 65 Our results represent significant increase in VEGF protein secretion from Caco2 cell with time contrary to HCT116, which was insignificant. This aggressive behaviour of Caco2 cells may be related to the increased glycolytic enzymes activity observed in our data by Caco2 cells compared to HCT116 leading to an increase in tumour growth, vascularization and metastatic potential. Resveratrol treatment significantly reduced the level of VEGF in both cell lines with time. Similar effect of resveratrol on VEGF was found in human leukaemia (U937) cells, 66 ovarian carcinoma cells 3 67 and breast cancer cells (MDA-MB-231). 68 In addition, a decrease in the expression of protein and mRNA of VEGF was documented in human tongue squamous cell carcinomas 9 and hepatoma cells (HepG2). 69 Cao et al. 67 and Zhang et al. 69 reported that the mechanism by which resveratrol decreased the VEGF level was related to the inhibition of Akt and MAP kinases and enhancement of proteasomal degradation of HIF-1α protein resulted in the inhibition of VEGF expression. Resveratrol inhibits various types of matrix metalloproteases like MMP2 and 9 67 and suppresses both cyclooxygenase and ODC enzymes that have a role in vascularization and tumour growth 8 in vitro. Moreover, it increased p53 and p21 expression and perturbed cell cycle progression of endothelial cells or smooth muscle cells. 70 Also, it inhibited capillary-like tube formation of human umbilical vein cells and inhibited its binding to VEGF. 71
In conclusion, resveratrol shows great promise in the treatment of obesity and cancer, the most leading causes of morbidity and mortality . Resveratrol exerts cytotoxic, apoptotic and antiangiogenic effects, which may be attributed partially to its calorie-restriction effects on colon carcinoma cell lines.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
