Abstract
It has been hypothesized that oils containing high levels of omega-3 polyunsaturated fatty acids, such as canola and fish oil, could counteract some of the adverse effects induced by phthalates. In the present study, the influence of different oily vehicles on di-butyl phthalate (DBP)-induced testicular toxicity and lipid profile was investigated. Pregnant Wistar rats were treated by oral gavage from gestation days 13 to 20 with DBP (500 mg/kg/day) diluted in three different vehicles: corn, canola or fish oil. Male fetuses were analyzed on gestation day 20. DBP exposure lowered intratesticular testosterone levels and anogenital distance, regardless of the vehicle used. The percentage of seminiferous cords containing multinucleated gonocytes and cord diameter was increased in DBP-exposed groups, compared with vehicle controls, with no difference between the three DBP-exposed groups. Clustering of Leydig cells was seen in all DBP groups. Lipid profile indicated that administration of canola and fish oil can increase the content of omega-3 fatty acids in rat testis. However, content of omega-3 was diminished in DBP-treated groups. Overall, our results indicate that different oily vehicles did not alter fetal rat testicular toxicity induced by a high DBP dose.
Introduction
Certain phthalate esters, industrial chemicals mainly used to impart flexibility to polyvinyl chloride plastics, have been shown to disrupt the development of male reproductive tract when administered to rats during pregnancy. 1,2 The effects induced in rats following in utero exposure are remarkably similar to the disorders seen in the human testicular dysgenesis syndrome (TDS), a condition comprising hypospadias, cryptorchidism, low sperm counts and testicular cancer. 3,4 In this regard, the existence of a TDS-like condition, the so-called “rat phthalate syndrome” has been proposed, which could be used as a potential model for understanding the cellular and molecular mechanisms underlying human male reproductive disorders. 5,6
However, it is important to highlight that the spectrum and severity of toxic responses induced by phthalates during the prenatal period have been shown to vary significantly across different laboratories. 1,7,8 Although differences in rat strain sensitivity may be partially responsible for such variability, other factors such as the oily vehicle could also be involved.
A number of potential mechanisms have been proposed to explain the action of phthalate esters on the disruption of male reproductive tract development. In particular, phthalates are known to induce suppression of testosterone biosynthesis, 2 which appears to be the initial event underlying the induction of malformations. 9 In addition, dysfunction of Sertoli cells by active phthalates has been associated with testicular abnormalities, such as the presence of multinucleated gonocytes (MNGs) in fetal testis and low sperm counts at adulthood. 10,11 Nevertheless, the molecular targets leading to these changes remain unclear.
More recently, it has been proposed that the reproductive toxicity of phthalate esters could be related to inflammatory processes. 12,13 According to Latini et al., 12 induction of cyclooxygenases (COXs) by some phthalates could be implicated in intrauterine inflammatory responses and the consequent high incidence of preterm birth observed in some epidemiological studies. Xu et al. 14 also showed that phthalates can alter rat placental homeostasis of essential fatty acids (EFAs) via peroxisome proliferator–activated receptor overexpression, leading to a reduction in EFA transfer from the placenta to the fetus. In this regard, it has been hypothesized that dietary supplementation with oils containing high levels of omega-3 polyunsaturated fatty acids could counteract some of the adverse reproductive effects induced by phthalates. 12 Omega-3 fatty acids are found in fish and canola oil and in dark green vegetables, while omega-6 fatty acids are the components of vegetal oils and seeds, like corn, soy and others. 15 In contrast to omega-6 fatty acid derivatives (e.g. arachidonic acid) that generate major proinflammatory mediators when metabolized by clycloxygenases and lipoxygenases, omega-3 derivatives (e.g. eicosapentaenoic acid (EPA)) are believed to originate mainly anti-inflammatory compounds, such as series 3 prostaglandins.
In the present study, we investigated the influence of different oily vehicles on di-butyl phthalate (DBP)-induced testicular toxicity and lipid profile. For this purpose, phthalate effects were evaluated in animals receiving DBP diluted in corn oil, the usual vehicle for diluting phthalates in experimental studies, and two omega-3-rich vehicles, canola and fish oil.
Methods
Animals
The experimental protocol was approved by the Committee on Animal Research and Ethics of the Universidade Federal do Paraná (Curitiba, Brazil) under the number 421. Wistar rats were obtained from the stock of the Universidade Federal do Paraná and kept under a 12-h light/dark cycle and controlled temperature (22 ± 2°C). Standard pellet food (Nuvilab CR-1; Nuvital, Curitiba, Brazil) and tap water were provided ad libitum. Adult female rats were mated for 3 h during the dark cycle in the proportion of three females to one male. Vaginal smears were collected daily after mating and the day of sperm detection was considered as day 0 of gestation. 16 Clinical signs of general toxicity in dams, including body weight gain, piloerection, hyper- or hypoactivity and salivation, were evaluated.
Chemicals, dose selection and treatment
DBP, CAS number 84-74-2, purity 99%, was obtained from Sigma-Aldrich (Schnelldorf, Germany). Pregnant dams (n = 11–13/group) were treated daily by oral gavage from gestation days 13 to 20 with 500 mg DBP/kg body weight (bw)/day. Corn, canola or fish oil were used as vehicles and the administration volume was 5.0 mL/kg bw for all groups. 8 The selected treatment period, days 13–20 of gestation, corresponds to the period of prenatal androgen-dependent development in male rats, and it is believed to be the most critical window to phthalate exposure. 1,2 A total of six experimental groups were used: three control groups (vehicle only) and three DBP-treated groups that received DBP at 500 mg/kg/day diluted in corn, canola or fish oil. Fish oil was supplied by Herbarium Laboratório Botânico (Colombo, Brazil), while commercially available corn and canola oils were used.
The dose of 500 mg DBP/kg/day was based on the studies that demonstrate the induction of a wide spectrum of antiandrogenic effects and testicular disorders following in utero exposure. 1,7,17 In addition, this is the typical dose used to induce the rat phthalate syndrome, a proposed model for the human TDS.
Anogenital distance and collection of fetal testis
Dams were killed by decapitation on day 20 of gestation, 2 h after the last administration of DBP or vehicles. The fetuses were removed from the gravid uterus and weighed. The anogenital distance (AGD) was recorded for male and female fetuses using a digital caliper (TCM, Hamburg, Germany). To avoid errors caused by differences in body size, the AGD of each animal was divided by the cube root of body weight. 18
Fetuses were killed by decapitation. The right testes of three male fetuses per litter were collected and maintained at −80°C until analysis of testicular testosterone or lipid profile. The left testes were immersed fixed in Bouin’s solution for 2 h and routinely processed for histology or immunohistochemistry (immunostaining with anti-3β-hydroxysteroid dehydrogenase (anti-3β-HSD).
Lipid profile determined by HPLC
Lipid profile was determined by high-performance liquid chromatography (HPLC) as described by Naliwaiko et al. 19 Total lipids were extracted from oily vehicles, rat chow Nuvilab CR-1 (Nuvital, Curitiba, Brazil) and pools of fetal testis (n = 5 testis/group) using chloroform–methanol (2:1, vol/vol) according to Folch et al. 20 Extracts were saponified by 0.5 M sodium hydroxide in 90% methanol (v/v). Free fatty acids were collected by hexane and derivatized with 4-bromomethyl-7-coumarin. Separation was performed on a Varian pro-Star HPLC (Varian, Santa Clara, California, USA) using an octadecylsilica column (25 cm × 4.6 mm i.d.; particle size 5 μm). Fatty acids were resolved isocratically using an acetonitrile–water gradient (from 77:23 to 90:10, vol/vol) as mobile phase at a flow rate of 0.8 mL/min and detected by fluorescence (325 nm excitation; 395 nm emission).
Testicular testosterone
Each fetal testis was homogenized with 200 µL of phosphate buffered saline (PBS) and extracted three times with a total volume of 1.5 mL of diethyl ether. 1,21 The ether fraction was poured off into clean tubes and evaporated in a fume hood. Extracts were resuspended in 500 µL of PBS and analyzed by enzyme immunoassay using a polyclonal antitestosterone antibody (R 156/7, 1:7500 dilution) obtained from Coralie Munro at the University of California (Davis, California, USA), cross-reacting with testosterone 100.0%, 5-alpha-dihydrotestosterone 57.4%, androstenedione 0.27% and androsterone, dehydroepiandrosterone, cholesterol, oestradiol, progesterone and pregnenolone <0.05%. Serial dilutions of pooled testicular extracts produced displacement curves parallel to those of standards, and the assay sensitivity was 2.3 pg/well. All samples were analyzed in duplicate. The inter- and intra-assay coefficients of variation were 6.7 and 2.4%, respectively.
Percentage of cords containing MNGs and cord diameter
Testicular sections of 5 µm were stained with hematoxylin and eosin and analyzed microscopically. A total of 50 randomly selected seminiferous cords per testis were investigated at 400× magnification for the presence of Multinucleated gonocytes (MNGs). Results are expressed as percentage of cords containing MNGs. The diameter (micrometer) of 20 randomly selected round cords per testis was measured using the 3.0 version of UTHSCSA Image Tool software (UTHSCSA, San Antonio, Texas, USA). Images were acquired on a Leica microscope coupled with an imaging system (Leica Microsystems LAS Software, Version 3.5.0, Wetzlar, Germany) at 200× magnification.
Immunohistochemistry and evaluation of Leydig cell aggregation
For evaluation of Leydig cell aggregation, immunostaining with anti-3β-HSD was performed. Testicular sections of 5 µm were mounted onto a poly-
Quantification of Leydig cell clustering in testicular sections was undertaken using 3.0 version of UTHSCSA Image Tool software (UTHSCSA). Digital images were captured at 200× magnification, using a Leica microscope coupled with an imaging system (Leica Microsystems LAS Software, Version 3.5.0, Wetzlar, Germany). The Image Tool software was used to draw and calculate the area of 3β-HSD-immunopositive cell aggregates. A complete cross section from the center of the testis was analyzed for each animal (n = 5/group), and the area of individual clusters was expressed as a percentage of the total Leydig cell cluster area in that section. Clusters were then classified into three groups, according to Mahood et al. 22 : small clusters, accounting for 5% or less of the total Leydig cell cluster area; medium clusters, accounting for between 5.1 and 14.9%; and large clusters, which individually accounted for 15% or more of total cluster area.
Statistical analysis
Data were analyzed using GraphPad Prism 5.0 (GraphPad Software Inc., La Jolla, California, USA). Normality and homogeneity of variances were evaluated prior to statistical analysis. Data were analyzed by analysis of variance (ANOVA) or alternatively, by the Kruskal–Wallis test whenever data did not fit parametric conditions. Differences between groups were tested by Tukey’s multiple comparisons test (parametric data) or by Dunn’s test (nonparametric data). In all analyses, litters were used as the statistical units. Differences were considered to be statistically significant at a probability level of 5% (p < 0.05). All analyses were performed by investigators blind to experimental groups.
Results
Pregnancy data
No signs of general toxicity were observed in any experimental group. Maternal body weight gain was unaffected by DBP during treatment (gestation days 13–20), and no significant differences were observed in male and female fetal body weight at gestation day 20 for any treatment group (Table 1).
Litter size and body weight of dams and rat fetuses exposed to 500 mg DBP/kg/day from gestation days 13 to 20. a

DBP: di-butyl phthalate; GD: gestation day.
a Numbers are mean ± standard errors.
Anogenital distance
For female fetuses, AGD did not differ between experimental groups (data not shown). Nevertheless, DBP administered to pregnant rats caused a significant decrease in AGD of male fetuses when compared with animals whose mothers received the correspondent vehicles (Figure 1). However, there was no significant difference between DBP groups, regardless of the oily vehicle used.

Anogenital distance corrected for body weight (gestation day 20) in male offspring rats exposed in utero to DBP or oily vehicles. Rat dams were treated from 13 to 20 days of gestation. Values are litter means ± standard errors. There was no significant difference between control (vehicle only) groups. Likewise, DBP-treated groups did not differ significantly between them (ANOVA/Tukey). (a) p < 0.05 significantly different from corn oil control group. (b) p < 0.05 significantly different from canola oil control group. (c) p < 0.05 significantly different from fish oil control group. DBP: di-butyl phthalate; ANOVA: analysis of variance.
Lipid profile
Lipid profile is presented in Tables 2 and 3. For each oily vehicle and for the rat chow, three samples were analyzed, while for testicular samples, lipid content was determined in a pool of five testes per treatment group. For this reason, no statistical analysis was performed.
Fatty acid composition from oily vehicles, rat chow and fetal testis. a

DBP: di-butyl phthalate; EPA: eicosapentaenoic acid; DHA: docosahexaenoic acid; ND: nondetected.
a Data from oily vehicles and chow are presented as mean ± SD (n = 3). For testicular samples, fatty acids were obtained from a pool of five fetal testis per experimental group collected from rats exposed in utero to DBP or oily vehicles.
Content of EPA and DHA in fetal rat testis exposed to DBP diluted in different oily vehicles. a
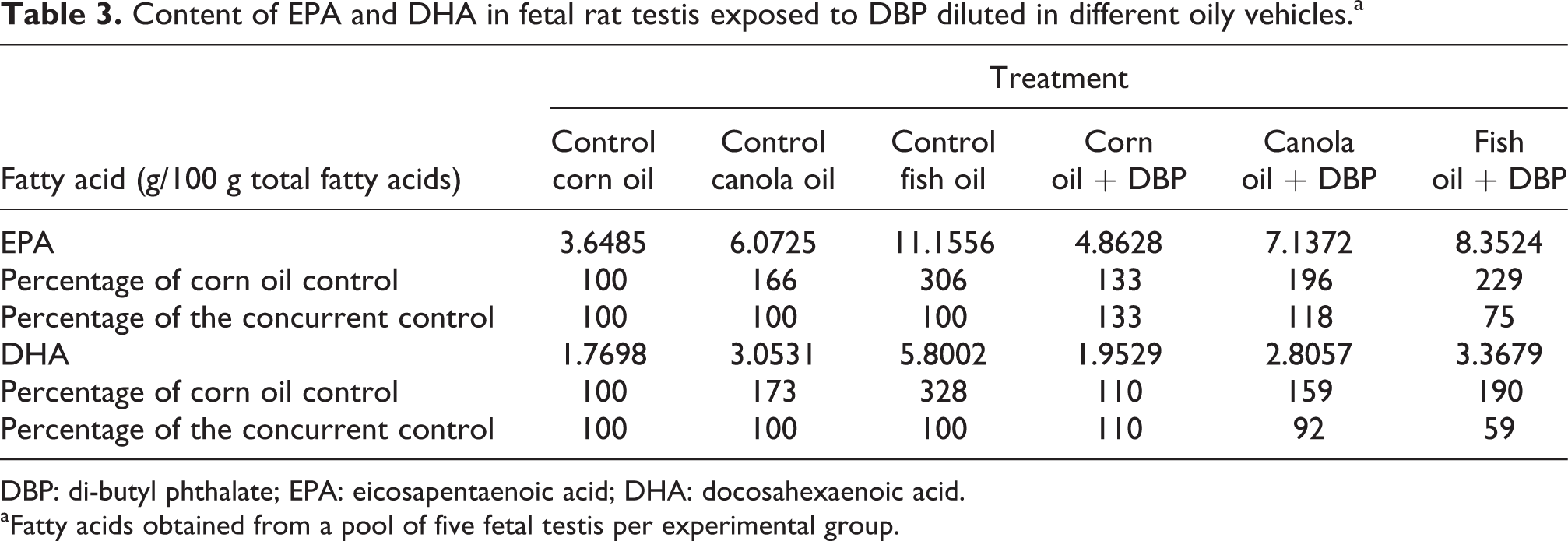
DBP: di-butyl phthalate; EPA: eicosapentaenoic acid; DHA: docosahexaenoic acid.
a Fatty acids obtained from a pool of five fetal testis per experimental group.
In contrast to corn and canola oil, fish oil presented high concentrations of EPA and docosahexaenoic acid (DHA; Table 2). Canola oil, on the other hand, presented the highest concentration of α-linolenic acid, an EFA that is converted in vivo into EPA and subsequently to DHA. As shown in Table 3, the concentrations of EPA and DHA in the fetal testes were higher in canola and fish oil groups, when compared with corn oil. Moreover, DBP exposure seemed to impair this increase. When compared to corn oil control group, DHA content was 73 and 228% higher in canola and fish oil control groups, respectively. In the groups treated with DBP diluted in canola and fish oil, the increase in DHA content was of 59 and 90%, respectively, when compared with corn oil control. When compared to their concurrent control groups, there was a decrease of 8 and 41% in DHA content in groups exposed to DBP diluted in canola and fish oil, respectively. On the other hand, for rats treated with corn oil and DBP, the concentration of DHA was 10% higher than in the group receiving corn oil alone. For EPA, another omega-3 derivative, the pattern was similar to that of DHA, particularly for groups treated with fish oil (Tables 2 and 3).
Fetal testicular testosterone
Exposure to DBP resulted in significant reductions in testicular testosterone content in relation to control groups. This reduction was similar in all DBP-treated groups (Figure 2).

Testosterone levels in fetal testes collected on gestation day 20 from control (oily vehicles) and DBP-exposed rats. DBP was diluted in three different oily vehicles and administered to pregnant rats from gestation days 13 to 20 at a dose level of 500 mg/kg/day. Values are litter means ± standard errors. There was no significant difference between control (vehicle only) groups. Likewise, DBP-treated groups did not differ significantly between them (ANOVA/Tukey). (a) p < 0.05 significantly different from corn oil control group. (b) p < 0.05 significantly different from canola oil control group. (c) p < 0.05 significantly different from fish oil control group. DBP: di-butyl phthalate; ANOVA: analysis of variance.
Percentage of cords containing multinucleated gonocytes and cord diameter
All groups, including controls, presented MNGs in seminiferous cords. In most cases, exposure to 500 mg DBP/kg/day caused a significant increase in the occurrence of MNGs, when compared with control groups (Figure 3). However, the group exposed to DBP diluted in canola oil did not differ statistically from its concurrent control group. Nevertheless, DBP-treated groups did not reveal any significant differences between them.

Percentage of cords containing multinucleated gonocytes (MNGs) in rat fetal testes collected on gestation day 20 following maternal treatment with DBP 500 mg/kg/day or oily vehicles from gestation days 13 to 20. Values are medians ± interquartiles (Q1, Q3). There was no significant difference between control (vehicle only) groups. Likewise, DBP-treated groups did not differ significantly between them (Kruskal–Wallis/Dunn). (a) p < 0.05 significantly different from corn oil control group. (b) p < 0.05 significantly different from fish oil control group. DBP: di-butyl phthalate.
Overall, histological analysis also showed an increase in the diameter of seminiferous cords in animals exposed to DBP, when compared with controls (Figure 4). Again, no significant differences were observed between DBP-treated groups. However, the group exposed to DBP diluted in fish oil did not differ from its concurrent control group (fish oil only; Figure 4).

Diameter of seminiferous cords in rat fetal testes collected on gestation day 20 after exposure to DBP 500 mg/kg/day or oily vehicles. Rat dams were treated from day 13 to 20 of gestation. Values are litter medians ± interquartiles (Q1, Q3). There was no significant difference between control (vehicle only) groups. Likewise, DBP-treated groups did not differ significantly between them (Kruskal–Wallis/Dunn). (a) p < 0.05 significantly different from corn oil control group. (b) p < 0.05 significantly different from canola oil control group. DBP: di-butyl phthalate.
Evaluation of Leydig cell aggregates
Abnormal distribution, or clustering, of Leydig cells was apparent in all DBP exposed groups. In DBP-treated animals, there were significant decreases in small clusters, regardless of the oily vehicle used. For medium and large clusters, the differences were not statistically significant in post hoc tests, even though overall ANOVA indicated significant differences (p = 0.012 for medium and p = 0.049 for large clusters; Figure 5).

Contribution (%) of small, medium and large clusters to total Leydig cell cluster area in rat fetal testes (n = 5/group) collected on gestation day 20 after in utero exposure to DBP or oily vehicles. Rat dams were treated from gestation days 13 to 20. Values are litter means ± standard errors. Small Leydig cell clusters were defined as those that accounted for 5% or less of the total Leydig cell cluster area per testis, medium clusters as those that accounted for 5.1–14.9%, and large clusters as those that individually accounted for 15% or more of total Leydig cell cluster area per testis. There was no significant difference between control (vehicle only) groups for any of the assigned categories. Likewise, DBP-treated groups did not differ significantly between them (ANOVA/Tukey). (a) p < 0.05 significantly different from corn oil control group. (b) p < 0.05 significantly different from canola oil control group. (c) p < 0.05 significantly different from fish oil control group. DBP: di-butyl phthalate; ANOVA: analysis of variance.
Discussion
The results of the present study indicate that different oily vehicles—corn, canola and fish oil—have no effect on DBP-induced fetal testicular toxicity in rats, following maternal exposure to a high dose (500 mg/kg/day). According to Mahood et al., 23 the fetal end points analyzed in the present study, including testicular testosterone levels, abnormal Leydig cell aggregation and gonocyte multinucleation, are the most sensitive markers of the rat phthalate syndrome, being able to predict the occurrence and severity of later effects.
The dose of 500 mg DBP/kg/day was selected on the basis of its ability to produce antiandrogenic effects and testicular disorders following in utero exposure and because, even so, there are still several discrepancies in the incidence and severity of effects reported in the literature. 1,7,17 In the present study, this DBP dose was not able to induce maternal toxic responses, regardless of the oily vehicle used. In addition, the vehicles alone were also unable to produce clinical signs of toxicity or changes in maternal and fetal body weight. In female fetuses, the AGD did not differ significantly between different experimental groups, confirming the absence of androgenic effects of DBP and the vehicles tested. However, male fetuses exposed in utero to DBP had decreased values of AGD, when compared to control animals. This effect was observed in all DBP groups, regardless of the oily vehicle used. Several studies have demonstrated that DBP and other phthalates can inhibit some enzymes in testosterone biosynthesis pathway, leading to alterations in male sexual development, including genital malformations and shortening of AGD. 8,7,17 The androgen insufficiency induced by DBP was confirmed in the present study by measuring intratesticular testosterone levels. Animals from experimental groups treated with 500 mg DBP/kg/day displayed reduced levels of testicular testosterone, when compared with groups that received only vehicles. However, there were no significant differences between DBP groups, indicating that the oily vehicles did not interfere in the androgen insufficiency induced by this high DBP dose. Taken together, these results indicate that the use of oily vehicles containing different ratios of omega-3 to omega-6 fatty acids do not alter the response of the fetal testis to a high DBP dose in regard to the androgenic status and, therefore, the vehicles are unlikely to be involved in the variability of responses seen in experimental studies.
However, beyond changes in androgenic status, possible effects of DBP in fetal testicular histology were also investigated. It is interesting to note that several histological alterations induced by phthalates in fetal testis, such as gonocyte multinucleation and enlargement of seminiferous cords, are believed to be unrelated to the decrease in testosterone production. 24,25 Gaido and coworkers 24 reported that DBP is able to significantly increase gonocyte multinucleation in mouse testis, without changing testosterone levels. In our study, DBP was able to induce a significant increase in the percentage of seminiferous cords containing MNGs, which is in agreement with earlier rat studies. 2,7,21 The group exposed to DBP diluted in canola oil, however, did not differ from its concurrent control (canola oil only). The biological significance of this finding is unknown but most likely reflects a normal variation in response in this variable. In addition, no significant differences were detected between DBP-treated groups. In relation to seminiferous cord diameter, a significant increase was seen in DBP-exposed animals, which is also a typical histological alteration induced by phthalates. For this histological parameter, there were no differences between the group exposed to DBP diluted in fish oil and its concurrent control group. The biological significance of this finding is also largely unknown. However, comparison of the three DBP-exposed groups did not show any statistically significant differences in cord diameter, regardless of whether corn, canola or fish oil were used, suggesting no relevant protective effect of any vehicle.
The analysis of the interstitial testicular compartment revealed the classical presence of large clusters of Leydig cells in DBP-exposed fetuses. A clear shift in the pattern of Leydig cell distribution was observed, with a significant decrease in the contribution of small clusters to the total cluster area in all groups exposed to DBP. These results are in agreement with a previous work reported by Mahood et al. 22
Several mechanisms of phthalate-mediated toxicity remain unclear, such as those leading to histological alterations. Some authors have proposed that phthalates could induce some of their toxic effects through an inflammatory process. 13,14,26 Given the anti-inflammatory properties of omega-3 fatty acid derivatives, Latini et al. 12 proposed the maternal dietary supplementation with omega-3 polyunsaturated fatty acids as a way to counteract some of the adverse effects of phthalates. Derivatives of omega-3 fatty acids, which are present in canola and fish oil, are able to act as substrates of COX enzymes, leading to the formation of anti-inflammatory compounds, while omega-6 derivatives, such as arachidonic acid, are major COX substrates for the generation of proinflammatory mediators. Determination of lipid profile in the present study revealed that administration of canola and fish oil in late gestation (days 13–20) was able to increase the content of omega-3 fatty acids, such as EPA and DHA, in rat fetal testis. As expected, administration of fish oil resulted in the highest contents of EPA and DHA. However, DBP diluted in fish oil has overall reduced the content of omega-3 derivatives incorporated into testicular tissue, when compared with its concurrent control (fish oil only). These results are in agreement with previous studies showing that phthalates like di-2-(ethylhexyl) phthalate are able to reduce the maternal-to-fetal placental transfer of EFAs, resulting in reduced content of omega-3 fatty acids like DHA in fetal plasma and brain. 27,28
The results of the present study indicate that administration of either canola or fish oil as vehicles is unable to alter DBP-induced testosterone insufficiency and its downstream consequences. In addition, no relevant effects were observed in the histological parameters investigated, indicating lack of any protective or aggravating effect of omega-3-rich oily vehicles on DBP-induced fetal testicular toxicity in rats. However, it seems that under certain circumstances, DBP is able to reduce the content of omega-3 derivatives in fetal testis, an effect that has also been observed in other tissues and deserves further investigation. Although our aim was to evaluate the effects of different oily vehicles at a high DBP dose (500 mg/kg/day), which is the common dosage used to induce the “rat phthalate syndrome,” it would be interesting to study the influence of these vehicles at lower doses. Besides, it would be valuable to investigate the possible protective role of oily vehicles containing omega-3 fatty acids following longer periods of supplementation (e.g. prior to mating and pregnancy).
Footnotes
Funding
This work was partially supported by Fundação Araucária – Brazil (Grant number 15190 – 219/2010); Ana Carolina S. Lourenço was a scholarship recipient from CAPES – Brazil (Master of Science).
Acknowledgments
The authors are grateful to Herbarium Laboratório Botânico for donation of fish oil and Dr. Ian Mason from University of Edinburgh for kindly providing anti-3β-HSD antibody.
Conflict of interest
The authors declared no conflicts of interest.
