Abstract
The present study focused on investigating whether the inhibitory effect of di (n-butyl) phthalate (DBP) on testosterone (T) biosynthesis was mediated by the glucocorticoid (GC) pathway in prepubertal male rats and T production after the exposure to DBP ceased. Prepubertal male rats were administered DBP in corn oil orally at 0, 250, 500, 1000, and 2000 mg/kg daily for 30 days. Serum T and GC were measured by radioimmunoassay and enzyme-linked immunosorbent assay, respectively. The responses, including glucocorticoid receptor (GR), type I 11β-hydroxysteroid dehydrogenase (11β-HSD1), and steroidogenesis acute regulatory protein (StAR) in the testes tissues, were determined by Western blotting and reverse transcriptase PCR. DBP exposure resulted in testicular toxicity, such as seminiferous tubule degeneration and a decrease in the number of spermatogenic cells. T was decreased and GC was increased in a DBP concentration-dependent manner in the exposure group. The expression of GR and 11β-HSD1 was significantly increased, with an associated decrease in expression of StAR. Neither the expression of the GR nor 11β-HSD1 and StAR were statistically significantly different in the postexposure group compared with the control. However, the weight and morphology of the testes did not recover in the postexposure group. These data suggest that DBP inhibits testosterone production through a GC-mediated pathway in prepubertal male rats, and after exposure to DBP ceases, testosterone biosynthesis returns.
Keywords
Di (n-butyl) phthalate (DBP) is used as a plasticizer and a stabilizer in numerous consumer products, including hair sprays, cosmetics, latex adhesives, inks, and food packing material. 1 About 9 million tons of DBP were produced globally in 2007. Therefore, because of their widespread use, there is a high potential of risk to humans, particularly among those using polyvinyl chloride-based medical devices. 2,3 The estimate of human exposure to DBP ranges from 0.84 to 113 μg/kg/d. 4,5 DBP does not bioaccumulate in the body and metabolizes in the gut to mono-n-butyl phthalate (MBP) and phthalic acid. The toxicity of DBP is ascribed to MBP. 1,6 The half-lethal doses of DBP in rats and mice are 8 g/kg body weight (BW) and 5 g/kg BW, respectively. DBP can result in many bioeffects, including hepatic toxicity, embryotoxicity, and especially damage to the male reproductive system, such as testicular atrophy and infertility. 7,8 In previous studies, administration of DBP to female rats at a dose of 500 mg/kg/d during pregnancy for 14 days caused a variety of male reproductive malformations, including underdeveloped or absent reproductive organs (such as reductions in anogenital distance), malformation of genitalia (such as hypospadias), cryptorchidism, decreased anogenital distance, diminished sperm counts, and Leydig cell adenomas. 9,10
DBP is regarded as an endocrine-disrupting chemical. 11 Typically, endocrine-disrupting chemicals are thought to disrupt at the level of the estrogen or androgen receptor. However, DBP and its metabolite do not interact with the androgen receptor. 12 Other studies have suggested that DBP can disrupt testosterone (T) synthesis in the testes, through diminished expression of several genes in the cholesterol transport and T biosynthesis pathways. 13,14 In addition, evidence has also shown that DBP can interfere with energy supply, metabolism, and transfer in spermatogenic cells and in the induction of apoptosis. 15 Many studies of reproductive toxicity of DBP focused on exposure of fetal male rats in utero and exposure during lactation, but there were few studies focusing on exposure of prepubertal male rats.
Glucocorticoids (GCs) directly regulate T production in testes via a glucocorticoid receptor (GR). However, GC access to GR is controlled by the local expression of 11β-hydroxysteroid dehydrogenase 1 (11β-HSD1). We hypothesized that DBP would inhibit T production by regulating the GC-GR access by controlling the expression of 11β-HSD1. Thus, in this study, we focused on (1) the inhibitory effect of DBP on T biosynthesis in prepubertal rats mediated by the GC-GR pathway and (2) T production after the termination of DBP exposure by monitoring the gene expression levels of 11β-HSD1, steroidogenesis acute regulatory protein (StAR), and GR in the testes.
Materials and Methods
Chemicals
Di (n-butyl) phthalate (CAS registry Number, 84-74-2; purity, 99.5%) was purchased from North Fine Chemicals Co, Ltd (Beijing, China). Anti-glucocorticoid receptor polyclonal antibodies and anti-β-actin antibodies were bought from Santa Cruz Biotechnology (Santa Cruz, California) and the alkaline phosphatase-conjugated secondary antibodies from Promega Biotechnology (Madison, Wisconsin). The RNA extraction kit and reverse transcriptase PCR kits were purchased from TaKaRa Biological Co, Ltd (Dalian, China). Testosterone and corticosterone hormone detection kits were purchased from North Institute of Biological Technology (Beijing, China).
Animals and Housing Conditions
The protocol for this study was approved by the Animal Care and Utilization Committee of Harbin Medical University in China. Sprague-Dawley rats were purchased from the Laboratory Animal Center of Jilin University (Changchun, China) and housed in stainless steel cages (4 rats per cage) in the animal facility under the conditions of controlled temperature (23 ± 2°C), humidity (55% ± 5%), and lighting (12-hour light/dark cycle), and they were given free access to the basal diet and tap water.
Animal Treatments
Eighty 5-week-old male rats were randomly divided into 5 groups according to body weight. Rats were given, by gavage, 250, 500, 1000, and 2000 mg/kg/d DBP or corn oil as control. Body weights were recorded twice each week. Eight rats from each group were killed after 30 days by an overdose of sodium pentobarbital; blood was collected from the abdominal artery during euthanasia for measuring serum T and GC. At necropsy, testes, epididymides, and adrenals were immediately removed and weighed. One of the testes was fixed in 4% buffered formalin for histopathological examination, whereas the other testis was frozen with liquid nitrogen and stored at –80°C for measuring the levels of GR protein, 11β-HSD1 mRNA, and StAR mRNA levels. The remaining 8 rats in each group were fed for an additional 15 days without DBP exposure, and termination procedures were similar to those animals described above.
Measurement of Serum Hormone
The levels of T and GC were measured by radioimmunoassay and an enzyme-linked immunosorbent assay (ELISA), respectively (North Institute of Biological Technology, Beijing, China).
Histopathologic Examination
Fixed tissues were dehydrated, processed, and embedded in paraffin. Then, 5-mm serial sections were cut and mounted onto slides. Tissue sections were stained with Harris's hematoxylin and eosin (HE) and examined for histopathologic changes.
RT-PCR
Total RNA was extracted from testes homogenates using an RNA extraction kit. RNA concentration was quantified spectrophotometrically (HITACHI, Tokyo, Japan). Then, 10 to 20 μg of total RNA was separated on 1% agarose gel and visualized to confirm integrity according to the production size of electrophoresis. First-strand cDNA synthesis was performed using an RT-PCR kit. After an initial incubation at 94°C for 15 minutes, PCR was done for 30 cycles with denaturation at 94°C for 45 seconds, annealing at 53.4°C (11β-HSD1), 59.2°C (StAR), and 53.7°C (β-actin), respectively, for 45 seconds and extension at 72°C for 45 seconds on a Gradient Cycler (Eppendorf, Germany). A final extension at 72°C for 10 minutes was conducted after 30 cycles. After amplification, samples were subjected to electrophoresis on 2% (w/w) agarose gel containing ethidium bromide, visualized under ultraviolet light, and photographed using an Alpha-Imager 3400 (Bio-rad, Hercules, California). The β-actin gene was used as a housekeeping reference gene to normalize expression levels between the samples. All the data were collected in triplicate and expressed as relative to β-actin. The following primers were used: β-actin: F-5′ GTA AAG ACC TCT ATG CCA ACA 3′, R-5′ GGA CTC ATC GTA CTC CTG CT 3′, 227-bp product (accession no. NM_007393); 11β-HSD1: F-5′ GCA GAG CGA TTT GTT GTT 3′, R-5′ TGT CTA TGA AGC CGA GGA 3′, 364-bp product (accession no. NM_017080); StAR: F-5′ CCC AAA TGT CAA GGA AAT CA 3′, R-5′ TGG CTG GCG AAC TCT ATC T 3′, 366-bp product (accession no. NM_031558).
Western Blotting
Testes were homogenized with a homogenizer in ice-cold 50 mM Tris-HCl buffer (pH 7.4) containing 0.25 M sucrose and a 1% protease inhibitor cocktail. The homogenate was centrifuged for 10 minutes at 10 000 g, 4°C, and the supernatant was collected. Protein concentrations were determined with a bicinchoninic acid (BCA; 2, 2′-bicinchonic acid) assay. Equal amounts of protein (60 μg) were resolved by SDS-PAGE (sodium dodecyl sulfate polyacrylamide gel electrophoresis), followed by transferring to Immobilon-p transfer membranes. The blots were blocked in 3% nonfat dry milk in phosphate-buffered saline (PBS) for 1 hour at room temperature. After the blockage, the membranes were incubated overnight at 4°C with rabbit polyclonal anti-GR antibody (diluted 1:1000) and then were incubated with secondary antirabbit IgG conjugated with alkaline phosphatase (diluted 1:2000) in PBS with 3% nonfat milk powder for 1 hour under agitation at room temperature. Visualization of the protein was performed by an enhanced chemiluminescence (ECL) reaction. The densities of the specific protein bands were quantified using SIM Gel Imaging Analysis System (Bio-pro, Ashbourne, UK).
Statistics
Data were subjected to analysis by 1-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparisons testing to identify significant differences between groups. All data are expressed as mean ± SD. Differences were regarded as significant at P < .05.
Results
General State of Rats
During the exposure and postexposure periods, there were no deaths observed in any group, but there were 4 rats whose normal activity, judged by visual observation, decreased compared with the control group after 17 days of exposure at 2000 mg/kg. Body weights increased weekly, and there were no significant differences between DBP treatment groups and the control group.
In either the exposure or the postexposure group, the organ/body weight ratio of testes significantly decreased (P < .05) at doses of 500, 1000, and 2000 mg/kg DBP, respectively, and the organ/body weight ratio of epididymis significantly decreased (P < 0.05) at doses of 1000 and 2000 mg/kg DBP. However, there was no effect on the organ/body weight ratio of the adrenals, as shown in Table 1 .
Organ/Body Weight Ratio in the Rats Treated by DBP (n = 8, Mean ± SD)
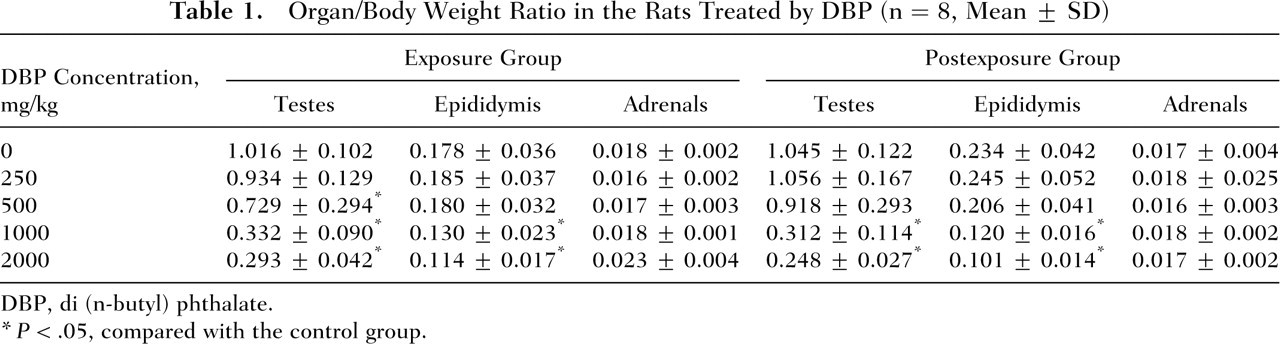
DBP, di (n-butyl) phthalate.
P < .05, compared with the control group.
Histopathologic Changes
Histopathological examination of the HE-stained tissue sections of the testes from rats in the exposure group showed degeneration of seminiferous tubules and a decrease in the amount of different spermatogenic cells, concomitant with the reduction of Leydig cell number, in a dose-dependent manner at doses of 50, 1000, and 2000 mg/kg DBP (Figure 1). However, at 250 mg/kg DBP, no histopathological effects were seen. Moreover, these changes observed did not significantly improve in the postexposure groups; conversely, Leydig cell production increased. There were no histopathological alterations of the adrenals observed.
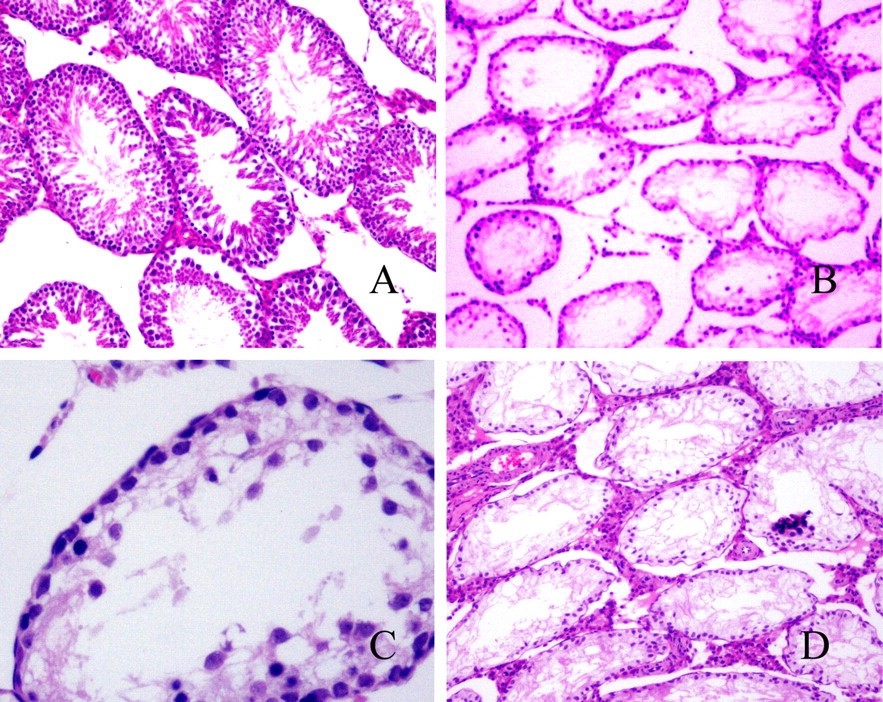
Exposure group: histopathologic changes of testes treated by di (n-butyl) phthalate (DBP). (A) Control group with normal spermatogenesis. Spermatogenic cells in different developmental stages in the seminiferous tubules (original magnification ×100, hematoxylin and eosin [HE] stain). (B) The 2000-mg/kg DBP group. Seminiferous tubules of testes degenerated, spermatogenic epithelium was thinner, and spermatogenic and Leydig cells decreased (original magnification ×100, HE stain). (C) The 2000-mg/kg DBP group. Spermatogenesis was incomplete and few spermatogenic cells observed (original magnification ×400, HE stain). (D) Postexposure group: 2000-mg/kg DBP group. Spermatogenesis was incomplete, spermatogenic epithelium was thin, and Leydig cells increased (original magnification ×00, HE stain).
Changes of Serum T and GC Concentration
As shown in Figure 2, in the exposure group, DBP significantly decreased (P < .05) the serum T concentrations at doses ≥500 mg/kg DBP and significantly increased (P < .05) the serum GC levels at 1000 and 2000 mg/kg DBP. However, compared with the control group, the levels of T and GC had no significant differences in the postexposure group.
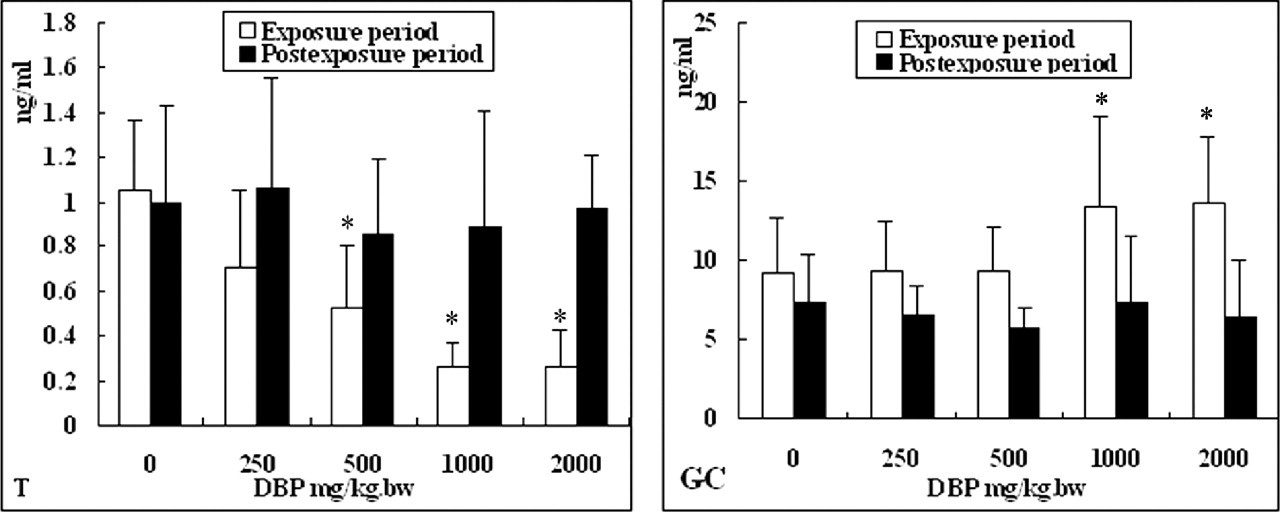
The changes in hormone levels in the serum of rats treated with di (n-butyl) phthalate (DBP). The decline of testosterone (T) concentration and the increase of glucocorticoid (GC) concentration caused by DBP were dose dependent in the exposure group. The concentration of T and GC had no significant changes in the postexposure group compared with controls. *P < .05.
The Expression of 11 β-HSD1 Gene and GR Protein Promoted by DBP
DBP exposure resulted in the enhanced expression of 11β-HSD1 mRNA in the exposure group (Figure 3A). The results from correlation and regression analysis showed that there was a linear correlation between the genetic expression levels of 11β-HSD1 (Y) and DBP dose (X) (regression equation was Y = 0.562 + 0.274X [R = 0.766]). In the postexposure group, the expression levels of 11β-HSD1 had no differences between different DBP-treated groups and the control group (Figure 3B).
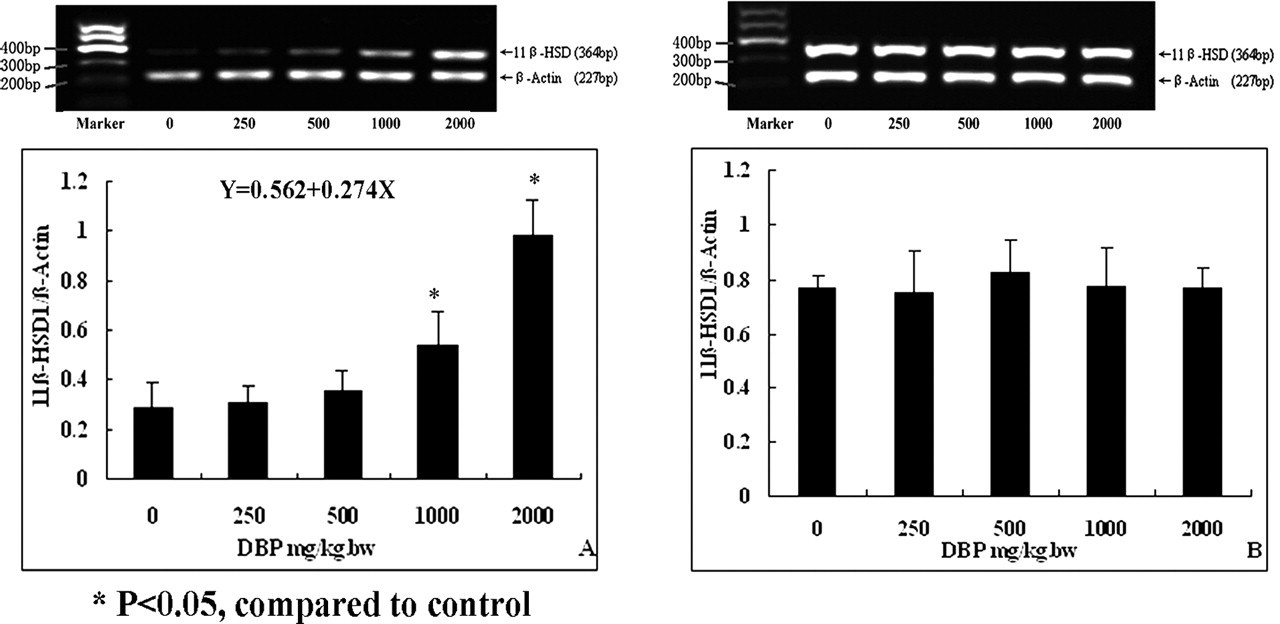
Expression levels of 11β-HSD1 gene in the testes of rats after exposure to di (n-butyl) phthalate (DBP). (A) Exposure group: a dose-dependent increase in gene expression of 11β-HSD1 after DBP exposure. (B) Postexposure group: no effects of DBP on gene expression of 11β-HSD1.
DBP exposure also resulted in a dose-dependent increase in the expression of GR in the exposure group (Figure 4A). The regression equation between the protein expression levels of GR (Y) and DBP dose (X) was Y = 0.485 + 0.464X (R = 0.790). The expression levels of GR in the postexposure groups had no statistically differences between the DBP-treated groups and the control group (Figure 4B).
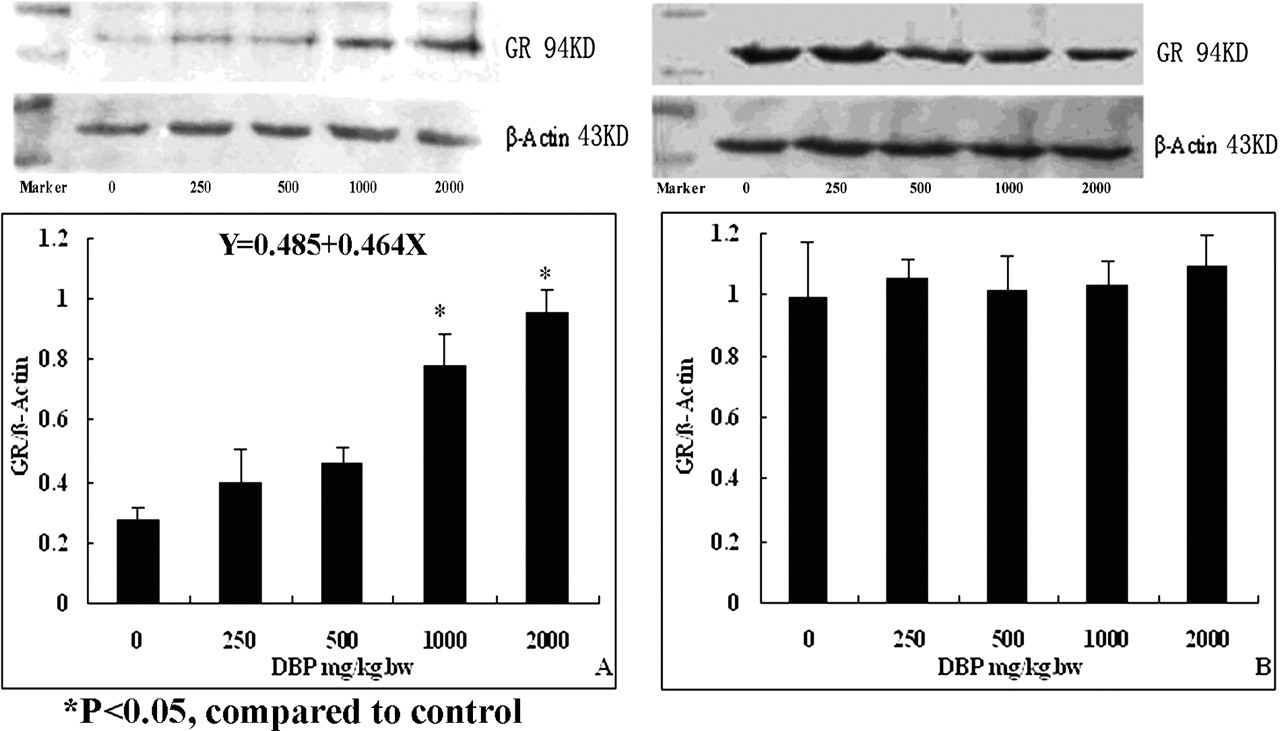
Expression levels of glucocorticoid receptor (GR) protein in testes of rats after exposure to di (n-butyl) phthalate (DBP). (A) Exposure group: the expression of GR protein increased dose dependently after exposure to DBP. (B) Postexposure group: no effects of DBP on expression of GR protein determined.
DBP Inhibited the Expression of StAR Gene
The expression levels of StAR in the testes significantly declined (P < .05) in response with 1000 and 2000 mg/kg DBP in the exposure group compared with the control group, as shown in Figure 5A. In the postexposure group, there were no obvious changes in the DBP-treated groups (Figure 5B).
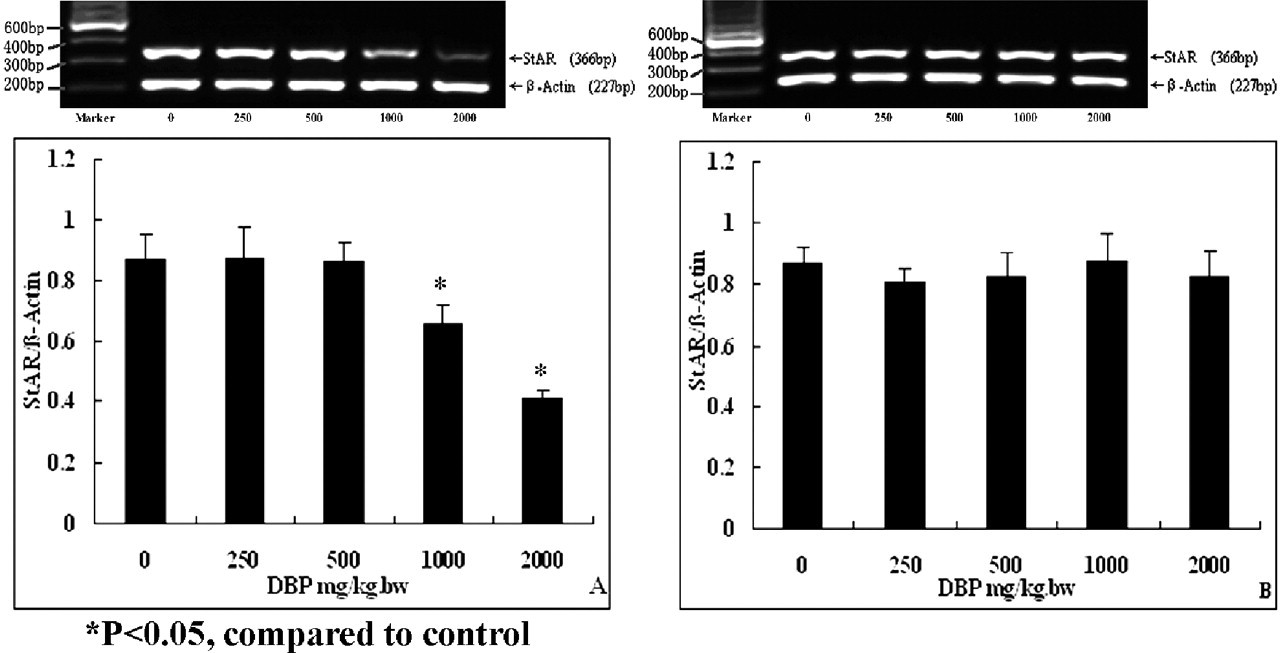
Expression of the steroidogenesis acute regulatory protein (StAR) gene in the testes of rats after exposure to di (n-butyl) phthalate (DBP). (A) Exposure group: DBP inhibited the expression of StAR gene. (B) Postexposure group: no effects of DBP on expression of StAR gene determined.
Discussion
Many studies have focused on reproductive toxicity in fetal male rodents exposed to DBP in utero and during lactation. For example, exposure to 250 mg/kg and higher DBP during sexual differentiation causes male reproductive tract malformations, including malformations of the epididymis and vas deferens, hypospadias, and reductions in anogenital distance. 16 At the same time, exposure to 250 mg/kg and higher DBP also decreased T production in fetal male rats. 17 Nevertheless, the reproductive toxicity of DBP in prepubertal or pubertal male rats is less reported. Because male reproductive toxicity of DBP is based on age-dependent sensitivity, in this study, we selected 2000 mg/kg DBP as the maximum exposure in prepubertal rats. The results showed that 1000 mg/kg and higher DBP caused a decrease in testes and epididymides weight in prepubertal rats, as well as morphological changes of testes, such as seminiferous tubule degeneration and a decrease in the number of different spermatogenic cells, but we did not examine for histopathological changes of the epididymis and epididymal sperm. Moreover, 500 mg/kg and higher DBP decreased T production by downregulation of StAR mRNA expression, which was the key enzyme involving steroidogenesis. However, there were no significant changes in T production and expression of StAR in the postexposure groups when compared with the control group (P > .05), except that histopathological changes of the testes remained similar to those in the exposure groups. Therefore, based on T and expression of StAR in the postexposure group, it is thought that testicular toxicity in prepubertal male rats could be reduced to some extent after terminating DBP exposure.
Studies with a targeted GR deletion or GR overexpression in mice have revealed the role of GC in many tissues and organs, including brain, fat, and the immune and reproductive systems. 18,19 The effects of GC on T inhibition act through the classic GC-GR model. GC binds, as a ligand, to the GR protein, which binds specific DNA sequences (glucocorticoid response elements). Leydig cells contain GR and are responsive to GC in the testes. GC can inhibit T production through suppressing cAMP formation, which is mediated by GR. The present study showed that DBP increased GC levels in the serum of male rats and promoted GR protein expression in the testes in a dose-dependent manner. The main consequence of GR-mediated action was a transcriptional repression of genes that are involved in steroidogenesis, including StAR. These results suggested that DBP might exert an inhibitory effect on T biosynthesis through a GC signaling pathway, which may be a novel finding in this study. However, why DBP increased the GC concentration in rats needs to be further elucidated.
The evidence we accumulated suggests that the actions of GC in target tissues are determined by the expression of not only GR but also GC-metabolizing enzymes, known as 11β-HSD. 20 Moreover, GC access to GR is controlled prior to the GR by a metabolizing pathway mediated by 11β-HSD. 11β-HSD has 2 known isoforms: type I 11β-HSD (11β-HSD1) and type II 11β-HSD (11β-HSD2). Type I 11β-HSD (11β-HSD1) is a bidirectional nicotinamide adenine dinucleotide phosphate (NADP+)/nicotinamide adenine dinucleotide phosphate reduced (NADPH)–dependent oxidoreductase. In contrast, type II 11β-HSD (11β-HSD2) is a unidirectional oxidase. 20 11β-HSD1 is localized in Leydig cells, where it is expressed and may catalyze the oxidative inactivation of GC in the testes of rats. In previous studies, it has been reported that 11β-HSD1 can couple with 17β-hydroxysteroid dehydrogenase 3 (17β-HSD3), one of the steroidogenic enzymes. 21 Thus, the physiological concentrations of androstenedione serve as a substrate for 17β-HSD3 using NADPH to generate NADP+, which drives 11β-HSD1 in Leydig cells primarily as an oxidase to eliminate the adverse effects of GC on T production. At the same time, 11β-HSD1 generates NADPH, which promotes T biosynthesis by stimulating 17β-HSD3 in a cooperative cycle. 22 In this study, our results showed that 11β-HSD1 expression was gradually increased with increasing concentration of DBP. Thus, we thought that 11β-HSD1 mediated the GC inhibition of T production, which might have a threshold, because 11β-HSD1 cannot completely inactivate the GC in the process of GC inhibition of T production. Consequently, 11β-HSD1 failed to display this regulation, and the increase of 11β-HSD1 expression may be the result of a compensational response, whereby increasing 11β-HSD1 induces the inactivation of GC. Furthermore, the decrease in T after DBP exposure may be a result of (1) impairment of cholesterol transport, therefore inhibiting steroidogenesis, or (2) that DBP increased GC concentration in rats, and the elevated GC concentration perhaps exceeded 11β-HSD1 capacity for inactivating GC, thus resulting in the suppression of T production mediated by GR.
In conclusion, our present data suggest that DBP could inhibit T production in prepubertal male rats through a GC-GR-mediated mechanism and that T production could recover after terminating exposure to DBP.
Footnotes
Notes
Acknowledgment
We thank Dr Zhang Lei from the Field of Pathology, Harbin Medical University of China for reviewing and assessing the histopathology of tissue.
