Abstract
Neuroendocrine regulation is disrupted by di-n-butyl phthalate (DBP) when exposure occurs during the critical periods of fetal development, which can lead to neurological disorders. To evaluate the toxic potential of DBP, it is necessary to conduct teratological studies, which could determine impacts on the development of the fetus. The present study was designed to understand the sequelae of neuroendocrine regulation in one-month-old pups when rats were exposed to DBP (F1–F3) in utero and during lactation. The rats received DBP (500 mg/kg BW/day) dissolved in olive oil through oral gavage from gestation day 6 to postnatal day 30, while the control group received the olive oil (vehicle) during the same timeline. Following the exposure, thyroid profile and estradiol, which were measured at GD-19, exhibited a significant decrease (P < 0.05) in dams (F0–F2). The exposure resulted in developmental outcomes, including underdeveloped fetuses, and a notable number of resorptions in experimental rats. The one-month-old pups were assessed for serum thyroid profile and testosterone and neurotransmitters in discrete brain regions, cerebral cortex, cerebellum, and hippocampus for up to three generations. The levels of dopamine and cortisol showed a significant increase (P < 0.05), but serotonin levels decreased when examined in distinct brain regions of the experimental group as compared to the control. DBP, which is considered an endocrine disruptor, had the most impact on the third generation in this study, leading to a significant decrease in testosterone levels. In summary, in utero exposure to DBP impaired the neuroendocrine system and had an antiandrogenic effect in the three successive generations.
Introduction
The growth of new neurons, their migration, and the formation of synaptic connections all rely on adequate levels of thyroid hormones. 1 The lack of thyroid hormones during pregnancy can be attributed to multiple factors; the underlying cause could be either insufficient iodine intake or being exposed to chemicals that interfere with the endocrine system. 2
Di-n-butyl phthalate (DBP) is widely recognized as an endocrine disruptor that can lead to negative reproductive health effects.3,4 It is commonly used in personal care items 5 like cosmetics, nail polish, lipstick, and various medical products. 6 These items, when discarded, release phthalates into the environment, exposing individuals to phthalates in unavoidable and continuous ways through eating, breathing, or skin contact. 7 The emerging evidence from the literature suggests that there is an association between prenatal exposures to phthalates and adverse reproductive health outcomes 8 such as altered male reproductive tract development 9 and decreased testosterone synthesis. According to Moog et al, 10 fetal developmental programming relies on the maternal hormone milieu (thyroid and cortisol) which should be in harmony and are found to be a crucial factor required for its development. Further, the fetus was found to be at higher risk when exposed to environmental pollutants, 11 particularly phthalates, than adults. In addition, DBP was considered a developmental toxicant with the documented outcomes such as undescended testes, retained nipples, shortened ano-genital distance, 9 cryptorchidism, 12 and poor learning ability. 13 DBP is metabolized into MBP (monobutyl phthalate). Both DBP and MBP differ in their potencies in inhibiting androgen production, with MBP being more potent than DBP in suppressing steroidogenesis and androgen production. 14 The free form of phthalate metabolites is more biologically active than their glucuronidated form when compared to the parent compound. 15 DBP is lipophilic and capable of crossing the blood–brain barrier 16 allowing it to affect neuronal tissue. The intrauterine developmental programming of the fetus was found to be very susceptible upon exposure to DBP leading to impaired neuronal development, learning disability, and altered behavioral attributes. For instance, in utero exposure to DBP was shown to induce developmental abnormalities in the brain of the fetus subsequently leading to cognitive impairment 17 and lower IQ scores and attention deficit hyperactive disorder in children. 18 In addition, the development of fetus depends on the level(s) of glucocorticoid, viz., cortisol. 19 The level(s) of cortisol would be extensive when exposed to phthalate leading to the high stress in rats demonstrating a link exists between the phthalate exposure and cortisol level(s). 20
The sex hormones govern the brain functions by regulating behavior and cognition. 21 Krolick et al 22 demonstrated that estrogen levels modulate neurotransmitter levels in discrete brain regions that regulate mood, reward, and motivation, which differ between the sexes. While existing research has extensively examined the impact of phthalates on thyroid function, there is a lack of knowledge regarding the long-term effects of DBP on thyroid hormone levels, testosterone, and cortisol over multiple generations, as well as their effects on neurotransmitters like dopamine and serotonin. This study was initiated to address this gap.
Materials and Methods
Animals
The albino rats of Wistar strain were procured from Sri Raghavendra Enterprises, Bangalore. They were acclimatized for a week in the animal house and maintained at the temperature of 25 ± 2°C with relative humidity of 50% under a controlled 12-hour light–dark cycle. The animals were fed with standard rodent diet purchased from Amrit Feeds, India, and water ad libitum throughout the experiment. The design and protocol of the study were approved by the Institutional Animal Ethics Committee, Bangalore University, Bangalore (Vide no. CPCSEA No. 402, File No. 25/525/2009 dated 23.03.2015).
Chemicals
Di-n-butyl phthalate (DBP, 99% purity), with CAS number: 84–74-2 and molecular weight 278.34 g/mol in the form of liquid, was procured from Sigma-Aldrich Ltd. Acetic acid, Alumina, EDTA, HCl, n-Butanol, n-Heptane, sodium acetate, O-phthaldehyde, dopamine, serotonin, homovanillic acid, 5-hydroxyindole acetic acid, and other standards of analytical grade (AR) were purchased from Sigma Pvt. Ltd.
Experimental Design
The study design was initiated with 26 adult albino rats (200–250g) of Wistar strain (20 females and 6 males). Rats were housed in cages with a ratio of 3 females:1 male for a week and were examined for the vaginal plugs to confirm mating on a daily basis. Out of 18 females, 12 females were confirmed to have mated and were divided into two groups. Group-I consisted of control rats (n = 6) and were administered 0.1 mL olive oil, and the experimental group-II (n = 6) received DBP [500 mg/kg body weight (BW)/day] dissolved in 0.1 mL olive oil through oral gavage from gestation day (GD) 6–19 and during lactation. The pregnant rats were considered parental generation (F0) and dams were housed individually until parturition and their pups were labelled as the F1 generation. The F0 dams of control group and experimental group received olive oil and DBP throughout lactation, respectively. The pups born to them, the F1 generation of both control and experimental groups, were sexed on PND 21, received olive oil and DBP (500 mg/kg BW/day
23
which is equivalent to 1/16 of the LD50) dissolved in olive oil, respectively, until termination (PND 30). Breeding of siblings was avoided. Similarly, the experimental design was carried out in three consecutive generations in both the groups, the ratio of mating 3 females:1 male was maintained. The control group rats were reared along with the experimental groups to undertake the comparative study for three generations (F1, F2, and F3). One-month-old male rats (F1, F2, and F3) were euthanized by cervical dislocation under 1% pentobarbital sodium (0.4 mL/100g BW) anaesthesia, and discrete regions of the brain, namely, cerebral cortex (cc), cerebellum (cb), and hippocampus (h), were excised to assess the levels of neurotransmitters, dopamine, and serotonin studied for three generations (control and experimental groups). The experimental design is shown in Figure 1. Schematic diagram showing the experimental design and the number of animals (control and experimental) used to raise F1, F2, and F3 progeny.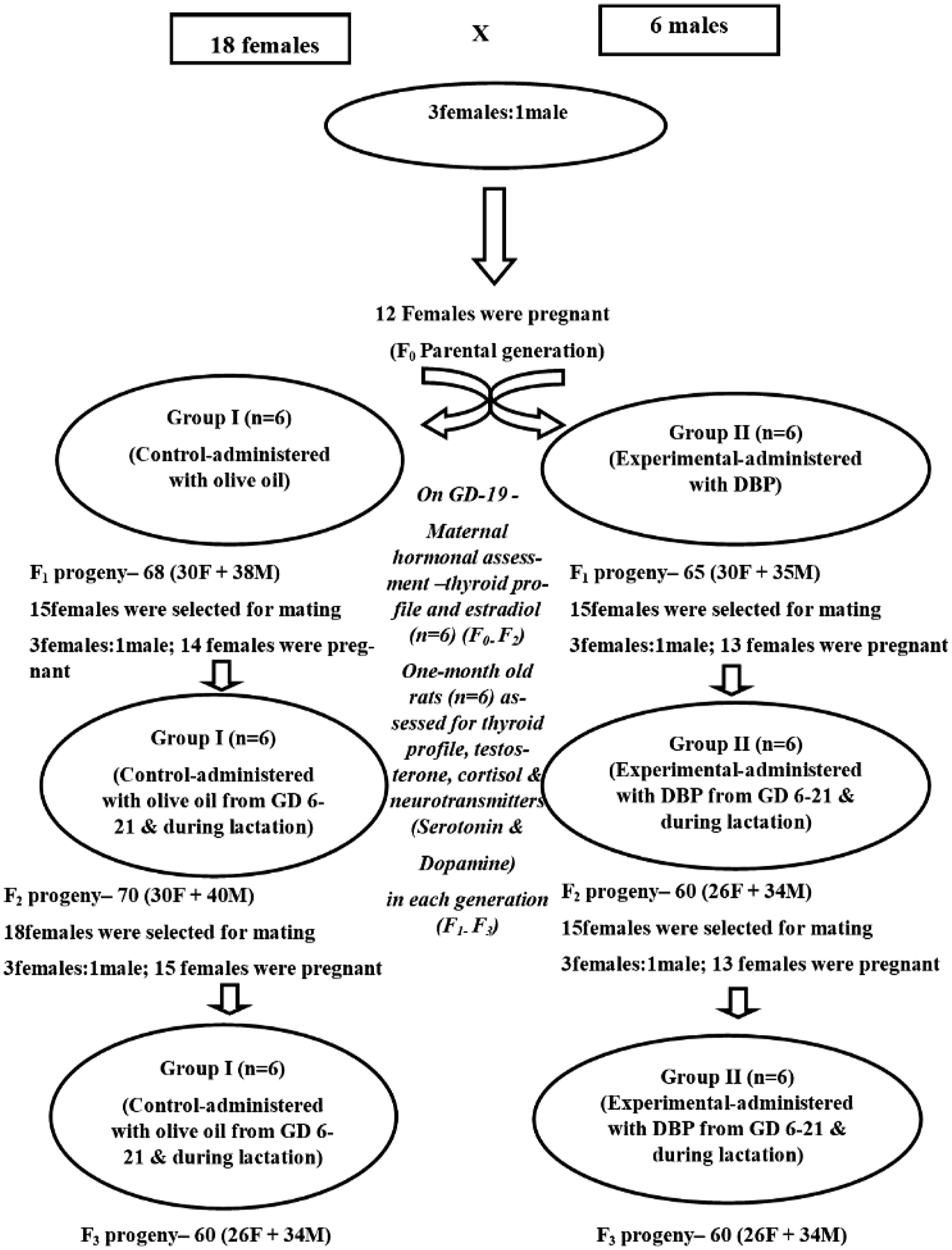
Hormonal assay
On GD-19, the gestational rats were euthanized by cervical dislocation under 1% pentobarbital sodium (0.4 mL/100 g BW) anaesthesia to draw blood from a cardiac puncture using a disposable heparinized syringe. The serum samples were prepared by routine methods and stored at −20°C until assayed. Serum samples were prepared for assessing thyroid profile, viz., serum free triiodothyronine (FT3), free tetraiodothyronine (FT4), thyroid-stimulating hormone (TSH), and estradiol by fully automated bidirectional interfaced chemiluminescent immunoassay technique 24 (CLIA). In addition, the fetuses were examined for developmental indices. Similarly, on PND 30, the sera of rat pups were assessed for thyroid profile (FT3, FT4, TSH), testosterone, and cortisol using CLIA technique.
Memory Retention Test
The rats were subjected to spatial learning and memory using the T-maze as in our previous study. 25 One-month-old male pups [control and DBP-treated groups (F1–F3)] were assessed for acquisition and performance of rewarded alternation task in T-maze as shown in Table S1. In addition, the rats were subjected to memory retention test following the acquisition and allowed to perform 30 trials per session, and the number of errors committed by each rat subject was recorded and correlated with neurotransmitters (dopamine and serotonin) in discrete brain regions.
Preparation of Tissue Homogenates for Neurotransmitters Assay
The three major regions of brain, namely, cerebral cortex (cc), cerebellum (cb), and hippocampus (h) tissue homogenates were prepared in ice-cold acid (10 mM HCl)—n-butanol (0.85 mL of 37% HCl in 1 L of n-butanol) and centrifuged at 3000 rpm for 10 min at 4oC. To 4 mL of supernatant, 10 mL heptane and 2 mL distilled water were added, and tubes were shaken mechanically for 10 min and centrifuged for 10 min at 3500 rpm. Upper and lower organic phases were distinct; the lower aqueous phase was used for measurement of dopamine and serotonin.
Measurement of Dopamine and Serotonin
The catecholamine, dopamine, was measured by adopting the method given by Kari et al. 26 To 2 mL of the lower aqueous phase, 200 mg of activated alumina and 1.5 mL of 2 M sodium acetate were added and mixed well. Upon adjusting the pH to 8.0 with 0.1 N sodium hydroxide, the solution was vortexed for 10 min and centrifuged at 3500 rpm for 5 min. The assay of serotonin was carried out by taking 3 mL of the supernatant in the tubes containing 3 g sodium chloride, 1 mL of 0.35 M borate buffer (pH 10.0), and 6 mL of n-butanol, and the tubes were vortexed for 10 min and further centrifuged at 3500 rpm for 5 min. The volume of 5 mL organic phase was transferred to a centrifuge tube containing 6 mL n-heptane and 0.5 mL hydrochloric acid (0.1 N). Tubes were vortexed for 10 min and centrifuged for 5 min. Upper organic phase was aspirated and discarded. A 0.4 mL aliquot of the aqueous phase was mixed with 0.6 mL O-phthalaldehyde reagent and heated in water bath for 10 min. Tubes were cooled and read excitation at λ360 nm and emission at λ470 nm. The activated alumina that was added in the above reaction mixture was washed and vortexed twice with 2 mL distilled water, and centrifuged at 3500 rpm for 5 min. The water as supernatant was discarded. The walls of the tubes were blotted with strips of filter paper, and the alumina was shaken for 5 min with addition of 2 mL acetic acid (0.2 N) to elute the catecholamine, followed by centrifugation at 3500 rpm for 10 min. To 1 mL supernatant, 0.2 mL EDTA (0.1 M) was added to adjust pH 6.5. After addition of 0.1 mL iodine (0.1 N) solution, the mixture was mixed and incubated at room temperature for 2 min followed by the addition of 0.2 mL alkaline sulphate solution. After mixing, the solution was allowed to stand for 2 min at room temperature and the pH adjusted 5.4 with 5N acetic acid. For dopamine assessment, the same solution was heated again for 5 min in a boiling water bath; tubes were cooled and assayed by measuring the fluorescence excitation at λ320 nm and emission at λ370 nm.
Statistical Analysis
The data were analyzed by Student’s t-test to compare experimental and control groups, and the inter-generational analysis was done using one-way analysis (ANOVA) followed by post-hoc Tukey’s test by using SPSS software (version 20.0). All the values were expressed as mean ± SE of six animals. The P value <.05 was considered statistically significant.
Results
The dose of 500 mg DBP/kg body weight (BW), 23 1/16 LD50, was appropriate, as there was no maternal toxicity in the three generations.
Changes in Maternal Hormonal Milieu (Gestational Rats)
The gestational rats exhibited a decrement in thyroid profile and decrease in the FT3 and FT4 levels in experimental rats, while a significant (P < .05) increase was observed in the TSH levels as shown in Figure 2. In addition, the levels of estradiol indicated a significant decrease (P < .05) compared to their respective control groups (Figure 2(d)). Variations in the thyroid profile and estradiol hormone levels in gestational rats (n = 6) upon DBP exposure: A three-generational comparative study. Values are mean ± SE of six dams (n = 6) indicate the status of hormonal milieu measured on GD-19. Symbol ‘*’- significantly different from their respective controls as determined by Student’s t-test, P < 0.05; different alphabets ‘a’, ‘b’ and ‘c’ are significantly different when comparisons made among DBP treated groups as analyzed by one-way ANOVA followed by post-hoc Tukey’s HSD test, P < 0.05. F0, F1 and F2 represent parental, first and second generation during gestation period. C- Control, E- Experimental. a) FT3 - Free triiodothyronine; b) FT4 - Free thyroxine; c) TSH- Thyroid stimulating hormone; d) Estradiol.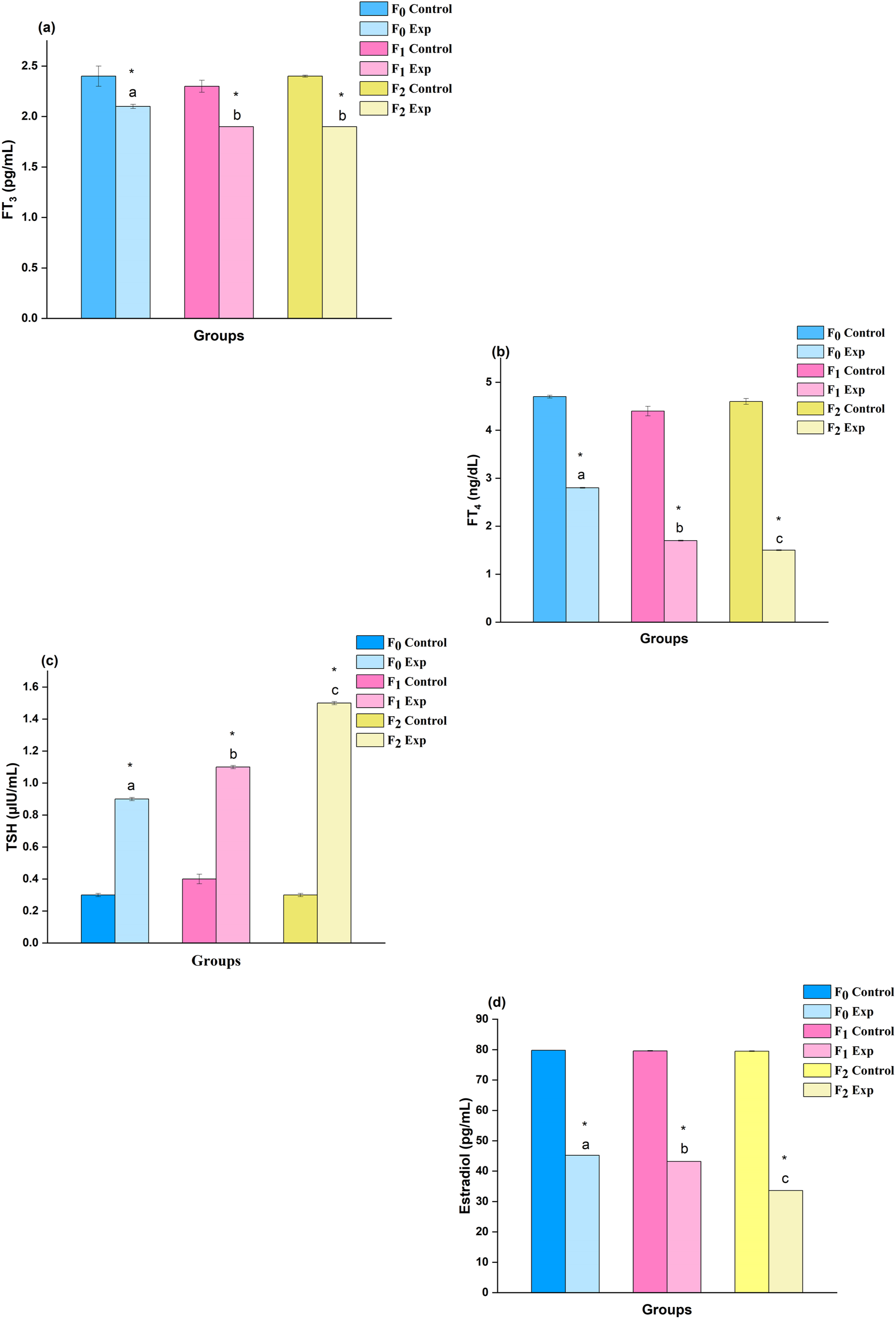
Developmental Indices
Figure 3(A) shows a rat with healthy uterine horns (control), and Figure 3(B) shows the resorptions of F2 dam upon DBP exposure. 2% of the F2 fetuses were underdeveloped (Figure 3(C)) and 16% of the fetuses displayed unilateral anophthalmic condition (Figure 3(D)) in one-month-old rats when compared to the control groups. The findings were at laprohysterectomies, but the study was not significantly powered to infer a causative effect. Photographs depict the changes in the developmental indices of rat fetuses (n = 6) upon DBP exposure. CL- Corpora lutea, LF- Live fetus, R-Resorptions; A- Control and B-DBP treated during the gestation period; C– Photograph depicts the status of rat fetuses on GD-19 wherein incidence of underdevelopment was evident (2 %) in F2 fetus upon DBP exposure made for three generations during the fetus development period; D– One-month old rat showing unilateral anophthalmia condition (lack of one eye) during the post-weaning period.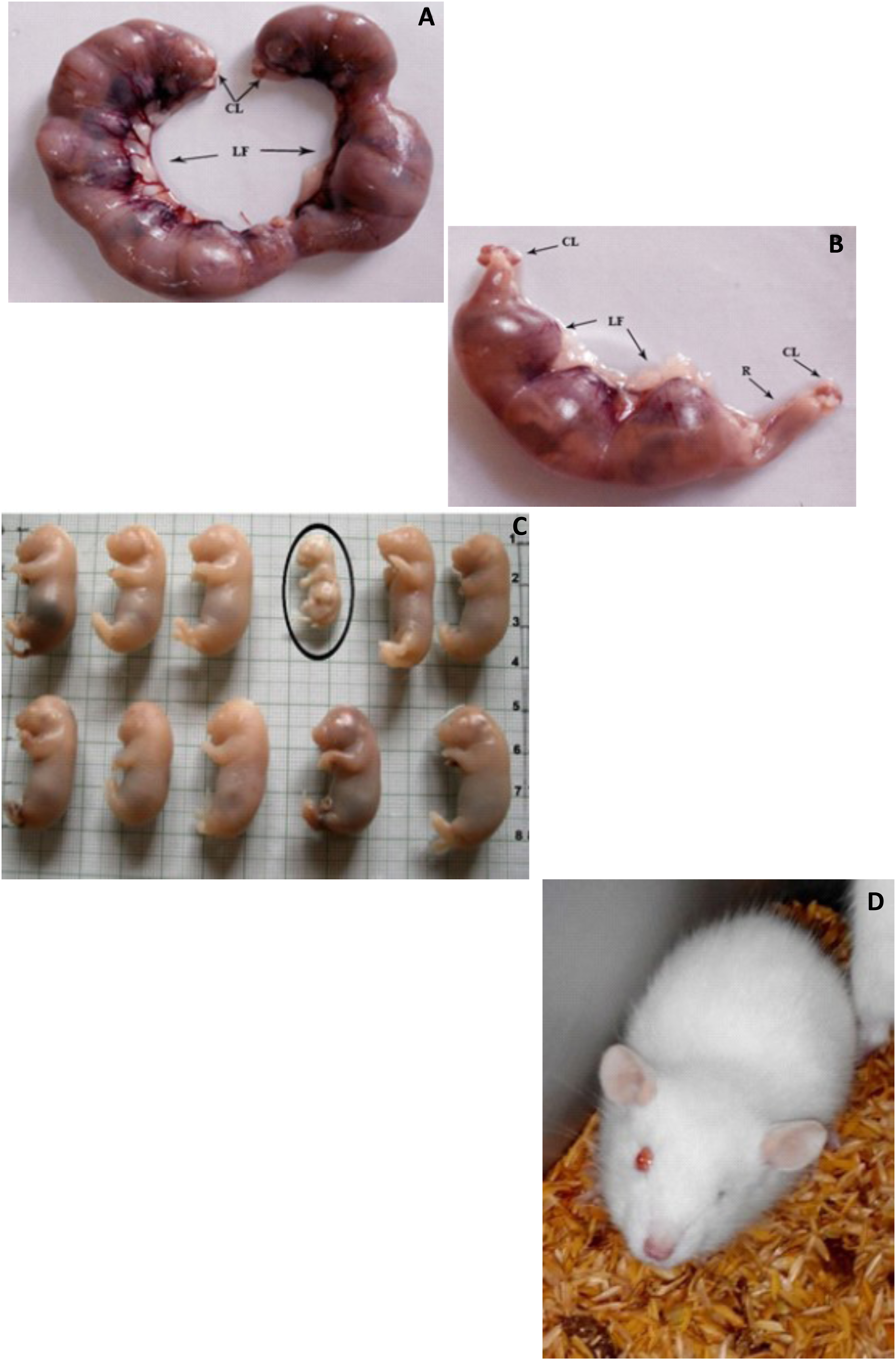
Changes in Hormonal Milieu (one-month-old Rats)
The serum thyroid profile (TSH, FT3, and FT4), testosterone, and cortisol assessments that were measured in 1-month-old male rat pups are shown in Figure 4. The results show that DBP exposure caused considerable decrements (P < .05) in the levels of thyroid (TSH, FT3, and FT4) and testosterone hormones. The serum FT3 levels were found to be affected by −29.7, −31.1, and −40.8% change in DBP-exposed rats of F1, F2, and F3 generations, respectively, while the concentrations of FT4 were affected by −18.8, −35.4, and −37.6%. Further, TSH levels showed decrements in experimental groups by −33.0, −36.8, and −36.9% change over their control groups. The levels of cortisol increased in serum by + 26.0, +105.7, and +233.8% while the serum testosterone levels were affected by −41.5, −46.6, and −52.7% in F1, F2, and F3 rats, respectively, when compared to their respective control groups. Changes in the level of hormonal milieu assessed in one-month old rats (n = 6) upon the exposure of DBP: A three-generational comparative study. Values are mean ± SE of six rats (n = 6) indicate the status of hormonal milieu during the post-weaning period. Symbol ‘*’- significantly different from their respective controls as determined by Student’s t-test, P < 0.05; different alphabets ‘a’, ‘b’ and ‘c’ are significantly different when comparisons made among DBP treated groups as analyzed by one-way ANOVA followed by post-hoc Tukey’s HSD test, P < 0.05. F1, F2 and F3 represent first, second and third generation. C- Control, E- Experimental. a) FT3 - Free triiodothyronine; b) FT4 - Free thyroxine; c) TSH- Thyroid stimulating hormone d) Cortisol e) Testosterone.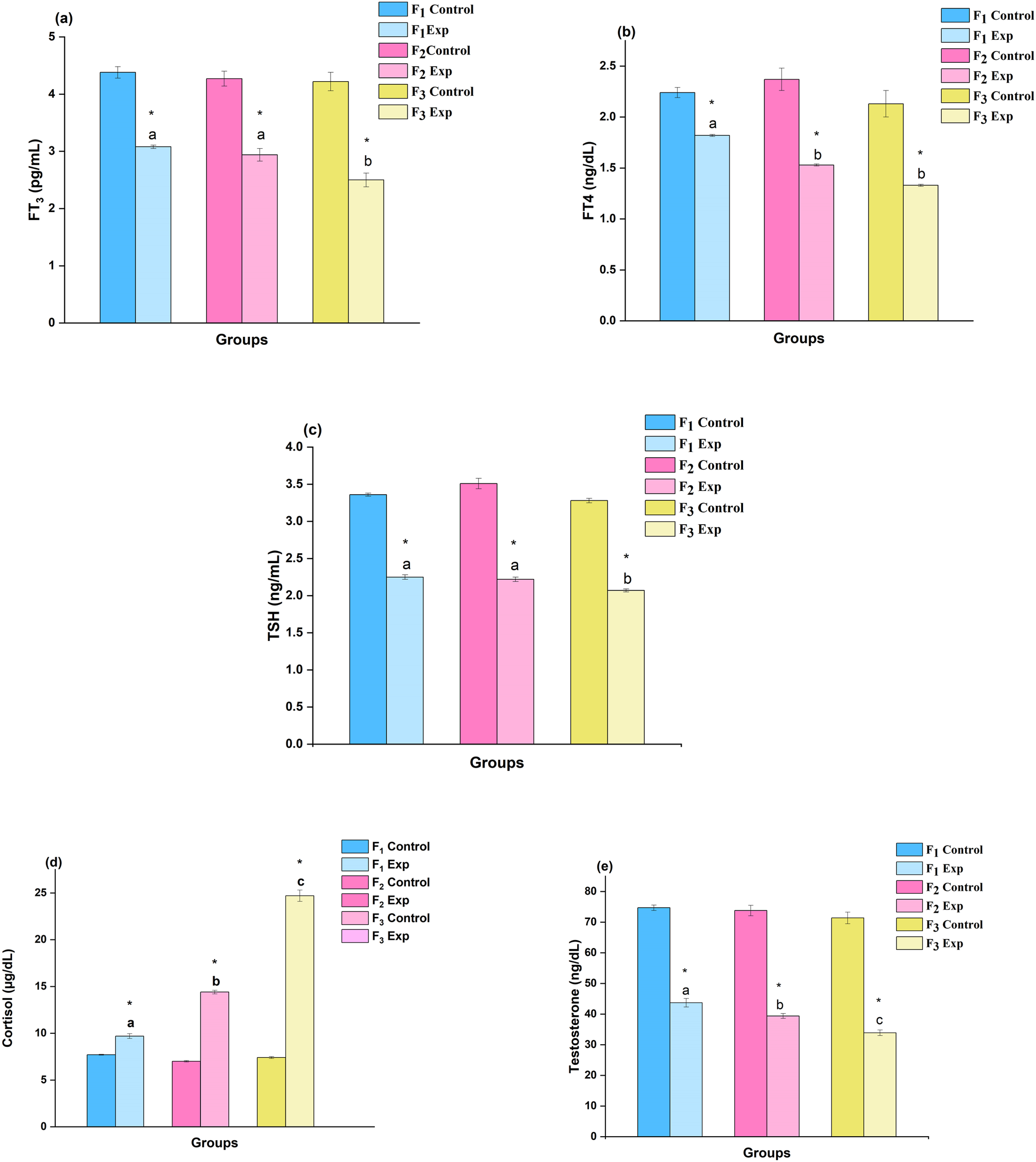
Memory Retention Test
The DBP-treated rats that were subjected to acquisition required more sessions to learn the left–right arm discrimination with a forced alternation, and more errors were committed in the memory retention test as shown in Table S1.
Neurotransmitter Assessment
Assessment of Dopamine
The disparities in the dopamine level upon DBP exposure made for three generations are represented in Figure 5(a). The brain regions, viz., cc, cb, and h, of DBP-exposed rats exhibited significant (P < .05) changes in the level of dopamine content, and the percent (%) increase was found to be +114.0, +40.5, and +49.5 in F1; +111.9, +44.2, and +58.9 in F2; and +132.7, +52.4, and +58.7 in F3 rats, respectively. The cc region was observed to be more sensitive and exhibited higher dopamine content compared to the control group rats. Changes in the level of (a) dopamine and (b) serotonin in discrete brain regions of rats upon DBP exposure: A comparative study of three generations. Values are mean ± SE of six rats (n = 6) of one-month-old during the post-weaning period. Symbol ‘*’ - significantly different from their respective controls as determined by Student’s t-test, P < 0.05; alphabets ‘a’ and ‘b’ are significantly different among experimental groups in the inter-generational analysis as determined by one-way ANOVA followed by post-hoc Tukey’s HSD test, P < 0.05. F1, F2 and F3 represent first, second and third generation respectively. cc– cerebral cortex; cb– cerebellum; h-hippocampus.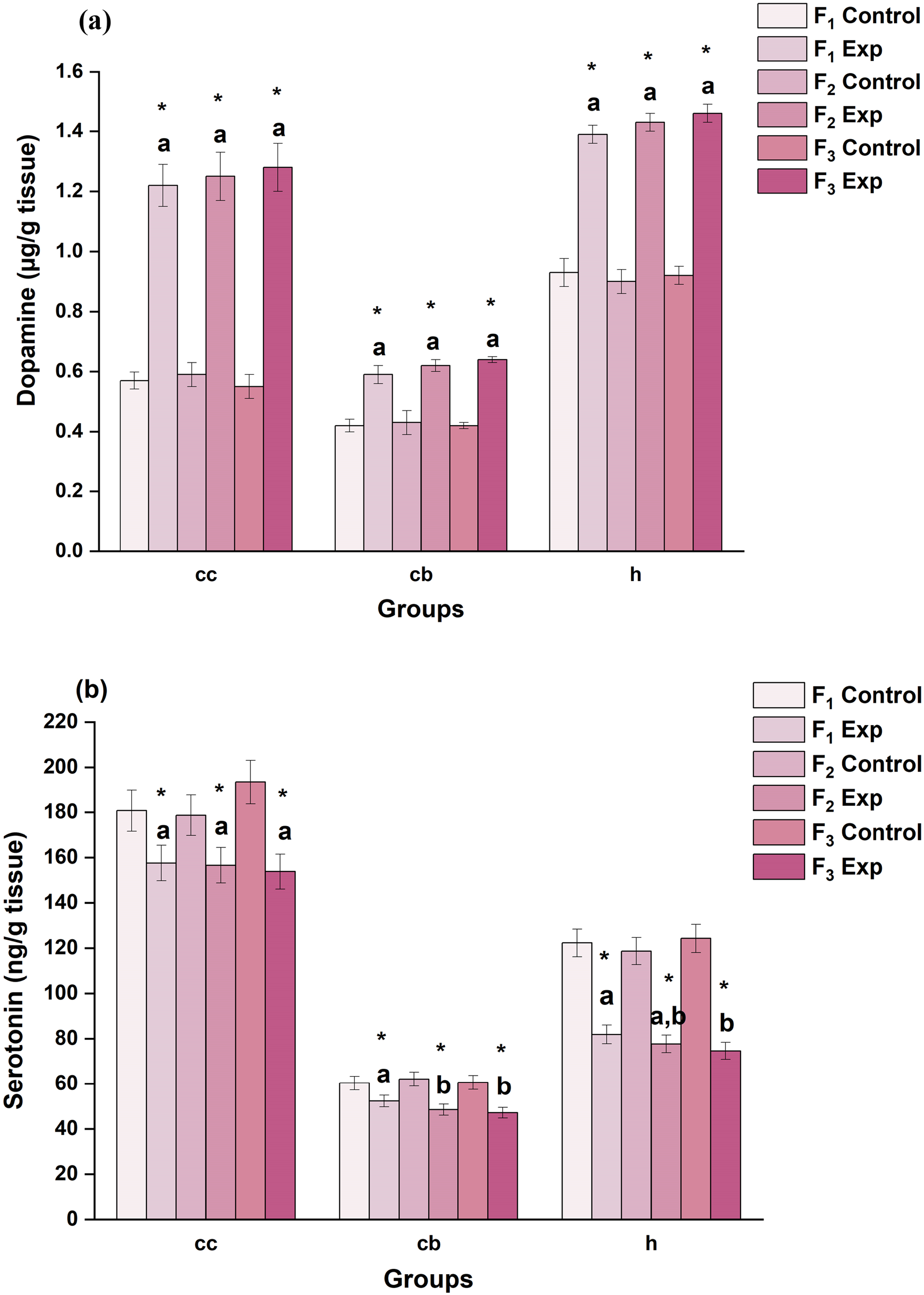
Assessment of Serotonin
The variations that occurred in serotonin levels are depicted in Figure 5(b). The brain regions, viz., cc, cb, and h, of experimental group rats exhibited decrements in serotonin content, and the percent (%) decrease was found to be −12.8, −13.3, and −33.0 in F1; −12.4, −21.9, and −34.5 in F2; and −20.5, −22.3, and −40.0 in F3 rats, respectively. The h region exhibited significantly the greatest decrease in serotonin content when compared amongst all the three brain regions of the experimental group.
Correlates Between Errors Committed and the Neurotransmitters (Dopamine and Serotonin)
Correlates Between the Errors Committed by DBP-Intoxicated Rat Progeny During Memory Retention Test and Neurotransmitters (Dopamine/ Serotonin) in Discrete Brain Regions: A Comparative Study of Three Generations.
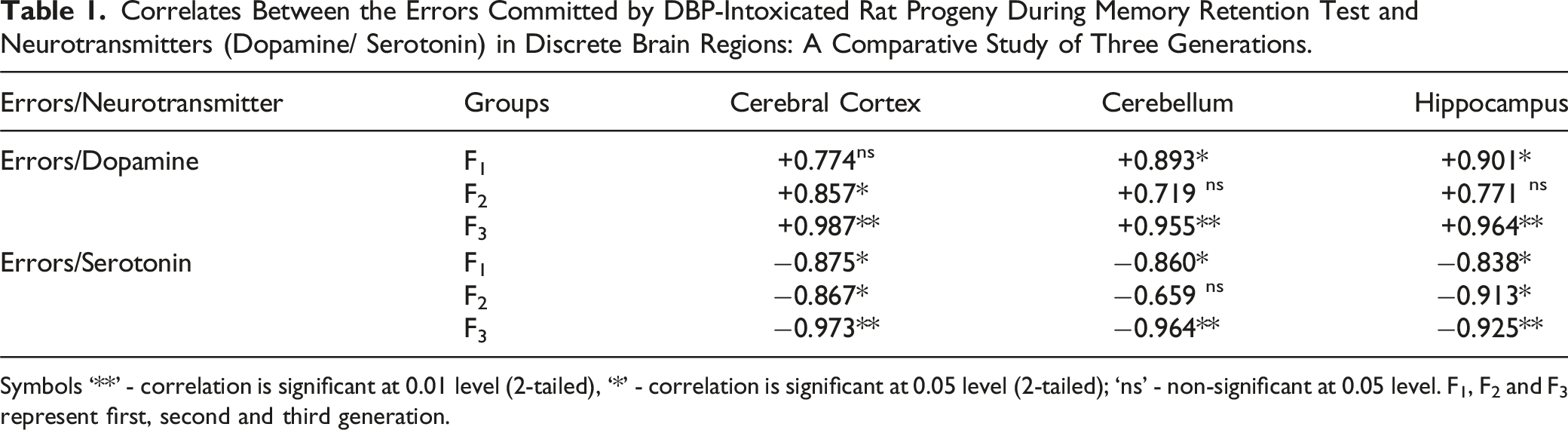
Symbols ‘**’ - correlation is significant at 0.01 level (2-tailed), ‘*’ - correlation is significant at 0.05 level (2-tailed); ‘ns’ - non-significant at 0.05 level. F1, F2 and F3 represent first, second and third generation.
Discussion
Phthalates are the common contaminants in soil and water due to their widespread usage in various products and their persistence in the environment, potentially leading to adverse health outcomes. Since phthalate and their metabolites have the ability to cross the placenta27-30 owing to their lipophilic nature, there is a concern that in utero exposures may be linked to altered gestational duration, 31 reduced ano-genital distance in males, 32 impaired behavior, altered executive functioning skills, 33 and reproductive, 29 respiratory, 34 metabolic, and thyroid adverse effects, 35 in different animal models.28,36 Hormones and neurotransmitters have a great impact on the behavioral attributes. 37 Further, the dual-hormone hypothesis proposed by Zilioli et al 38 postulates that cortisol and testosterone share a compounded effect on the behavioral traits. In addition, exposures to environmental chemicals (phthalates) have been demonstrated to alter thyroid hormone homeostasis and testosterone, leading to neurobehavioral deficits. 39 Further, phthalates affect the genital system during prenatal and postnatal periods of development. 10 Research to date has indicated that in utero exposure to phthalates caused adverse effects on the development of the fetus and cognitive deficits in the later phase of life. 18 The following sections represent the present study results with a comparative analysis of three generations.
Maternal Hormonal Assessment
In this study, the fetal outcomes were assessed in rats for three consecutive generations with exposure to DBP during the critical periods of fetal development (in utero and during lactation) posing a great health impact in F1–F3 generations. The results indicated decrements in maternal serum thyroid profile (FT3 and FT4) and estradiol levels in rats upon DBP exposure during gestation period (GD-19) while there was an increment in TSH level of DBP-exposed dams (F0-F2) as feedback mechanism. Before the fetal thyroid gland becomes functional, it is necessary to have maternal thyroid hormones for fetal brain development and protection. Studies of Morreale et al 40 demonstrated that any discrepancy in maternal thyroid hormone supply, a critical factor for prenatal brain development, disrupts processes such as proliferation, and differentiation followed by neurogenesis. Another study by Shanmugam et al. 41 indicated the decrement in the levels of progesterone and estradiol and their cognate receptors (PR and ERα) in the uterus of rats upon di-(2-ethylhexyl) phthalate (DEHP) exposure. In the present study, Figure 2 shows the increased TSH with decreased FT3 and FT4 levels in dams relative to control, which could be due to antagonistic effect of DBP on thyroid function. The earlier studies showed similar variations in gestational thyroid profile reflecting the underdeveloped fetus; likewise, the low levels of estradiol during the gestation upon DBP exposure led to poor fetal growth development. 42
Thyroid Profile, Testosterone, and Cortisol Assessment in 1-Month-Old Rat Pups
According to findings, DBP, an environmental endocrine disruptor, limits the activity of hormones because it has antiandrogenic characteristics. Moog et al 10 findings corroborate the present study results by demonstrating the role of thyroid in neuronal functioning as well as brain development and maturation. Likewise, Bernal 43 research suggested that thyroid hormones are necessary for normal brain function by influencing cellular interactions. Furthermore, changes in the maternal thyroid hormones caused disturbances in fetal neurogenesis, leading to cognitive impairment in the offspring, as reported in Mohan et al 44 studies. The present study findings are an extension of our previous studies to correlate the status of neurotransmitters with the behavioral attributes recorded in one-month-old rats using T-maze, 25 and it was evident from the assessments that the insufficiency in maternal thyroid hormones contributed a low learning in progeny and demonstrated poor memory as the errors committed during memory retention test profoundly by the experimental groups (F1–F3). Bernal 45 reported neurological discrepancies in the progeny upon maternal thyroid hormone deprivation, and his findings advocated that the development of the cerebrum and cerebellum (motor performance) was affected due to the impaired T3 binding to the mutant receptor due to hypothyroidism. Corroborating the findings of Bernal, 45 the present study findings draw the inference that the behavioral attributes were suppressed due to hypothyroidism leading to poor learning ability and memory retention. Besides, the elevated cortisol levels and decreased serotonin in cc, cb, and h regions signify low learning and memory in DBP-exposed rats, establishing a potential link between phthalate and cortisol, which appears to be a stress-related outcome. The rise in the level of cortisol was found to reduce the electric signals in the discrete brain regions which suppresses the functioning of hippocampus and affects spatial navigation. 25 The results further ascertained that the DBP induced increments in cortisol levels with suppression in testosterone appears to inhibit the growth of new neurons, synaptogenesis, dendritic branching, and axon development. The phthalate-induced stress was found to bring changes in the neuronal architecture and activity in terms of neuronal proliferation and neurogenesis and reduced the hippocampal volume. 25 The current research also showed that rats (F1–F3) exposed to DBP had elevated cortisol levels that were correlated to a significant decrease in testosterone levels. Both cortisol and testosterone are produced from the same precursor, resulting in competition for binding sites as described in the research by Brownlee et al. 46 The current study findings show that the decrease in testosterone levels in experimental rats (F1–F3) may be caused by higher cortisol levels resulting from the combined impact of DBP’s antiandrogenic properties and elevated cortisol levels.
Neurotransmitter Assessment
The steroid hormones that regulate neurotransmitters are active in the nervous system and are considered neurosteroids. 47 Testosterone being associated with male sexual development that interacts with neurotransmitters such as dopamine and serotonin in the brain influences behavioral patterns. The decrease in testosterone levels in the present study was associated with the secretion of monoamine neurotransmitters and increased dopamine synthesis in the midbrain’s limbic system, which was consistent with the results of Yuan et al 48 and Alderson and Baum. 49 In addition, there was inhibition in the re-uptake of serotonin which altered the behaviour 50 thereby making more errors observed in our previous study investigations. 24 The DBP-exposed rats (F1–F3) had a decrement in the level of estrogen which was similar to the studies of Wang et al 51 which showed the decrease in estrogen receptor as well as the dopamine receptors. Results of Tafet et al 52 indicated that high cortisol levels due to stress increased the serotonin uptake besides depression. The early life exposure to DBP alters the gonadal hormones that regulate neurotransmitters, resulting in significant changes in the behavioral traits of rats (F1–F3) exposed to DBP.
Conclusion
The results suggest a complex feedback cycle in which hormonal and neurochemical factors appear to have interacted and caused changes in the behavioral characteristics of the experimental groups (F1–F3). Additionally, studying DBP’s ability to modulate the neuroendocrine system through changes in neurotransmitter levels and hormone status reveals its potential long-term health effects on both dams and fetuses across three consecutive generations. In summary, maternal exposure to DBP mimicked hormone action and regulated neurotransmitters as an endocrine disruptor. This had the most significant impact on the third generation of rats exposed to DBP, affecting the fetal brain through an interaction between thyroid and stress hormones. Simply put, the research showed that DBP can lead to neurotoxicity in the developing rat brain.
Supplemental Material
Supplemental Material - In Utero Exposure to di-n-butyl Phthalate Causes Modulation in Neurotransmitter System of Wistar Rats: A Multigenerational Assessment
Supplemental Material for In Utero Exposure to di-n-butyl Phthalate Causes Modulation in Neurotransmitter System of Wistar Rats: A Multigenerational Assessment by Radha and Mahaboob P. Basha in International Journal of Toxicology
Footnotes
Author Contributions
Radha, M.J. and Basha, P Mahaboob contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript, and gave final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support from University Grants Commission, New Delhi, India (F.No. 41-31 / 2012 (SR) dated 10-07-2012), and also funds from Bangalore University Research Grants (Ref-DEV: D2a:BU-RP:2020-21 dtd 24.02.2023).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
