Abstract
Ketamine is increasingly used in clinical practice, and ketamine addiction is common in young individuals. There are limited reviews on the chronic effects of ketamine on the testes. Three groups of rats received saline or ketamine 50 mg/kg/day intraperitoneally for 6 weeks with or without a subsequent 4-week drug-free period. Serum follicle-stimulating hormone, luteinizing hormone, prolactin, and testosterone levels, as well as testicular malondialdehyde concentrations, were measured. Epididymal sperm parameters were assessed. Testicular tissues were examined by hematoxylin and eosin staining and immunohistochemical staining using caspase-3 and vimentin antibodies. Chronic ketamine injection significantly decreased the levels of the examined hormones and adversely affected sperm parameters. Testicular tissue showed a significant increase in caspase-3 expression. In addition, Sertoli cell shape and position were disrupted. These effects disappeared 4 weeks after drug withdrawal. Chronic ketamine treatment has revisable hazardous effects on the rat reproductive function. There is a need to increase the knowledge of physicians and the public regarding these harmful effects of ketamine.
Introduction
Ketamine is a noncompetitive glutamate N-methyl-D-aspartate receptor blocker related to phencyclidine. It was first synthesized in 1962 and was approved for medical use by the Food and Drug Administration in 1970. 1 As a nonrespiratory and noncardiac depressant, ketamine at anesthetic doses is preferred over other anesthetics. It is commonly used as an anesthetic in lower- and middle-income countries because it requires less postoperative monitoring equipment. In addition, it is widely administered in veterinary practice to large and small domestic animals and wildlife. 2
In addition to its use in anesthesia, ketamine has various other indications in clinical practice. It is used for both acute and chronic pain management, 3 and it has been studied as an analgesic for pain both related and unrelated to cancer. 4 Extensive studies have described ketamine use as an antidepressant. 5 Ketamine has a rapid onset of action in contrast to other conventional antidepressants. 6 Another therapeutic use of ketamine is as an anticonvulsant, principally to manage refractory status epilepticus. 7 Moreover, it has been suggested as a treatment for alcohol and heroin addiction and for anorexia. 8
Since its synthesis, ketamine has been used for recreational purposes. It produces addiction in both animals and humans. 9,10 Ketamine is known among abusers by common street names such as K hole, Special K, Super K, Kit-Kat, Cat Valium, Cat Tranquilizer, and others. 11 In several countries, ketamine abuse is not common, but there are high rates of ketamine addiction in Hong Kong. 12 The growth of both ketamine uses clinically (beyond its traditional anesthetic use) and nonmedical recreational ketamine exposure should prompt additional studies focusing on its toxicity in various organs.
Chronic ketamine intake has several detrimental effects, including neuropsychiatric manifestations, apoptotic neurodegeneration, intracranial hypertension, and a decrease in the gray matter. Ketamine affects cardiac function in the form of tachycardia, hypertension, and cardiac muscle damage. 8 Moreover, ketamine can evoke hepatorenal injury. 13 It is well known to produce uropathy. 14
Accurate assessment of the testicular function necessitates histopathological investigation. 15 In addition, hormones have a vital role in spermatogenesis. This includes follicle-stimulating hormone (FSH), which is essential for Sertoli cell function. LH stimulates Leydig cells to synthesize steroids particularly testosterone. In addition, LH has a partial stimulating effect on sperm maturation. Testosterone deficiency interrupts the major processes in the testicular tissue that are mandatory for spermatogenesis and normal male fertility. 16
Tissue malondialdehyde (MDA) is a good indicator of the oxidative condition of the testes. 17 Apoptosis has a crucial role in spermatogenesis in pathological conditions. 18 So, the assessment of testicular hormones, MDA, cellular apoptosis, and histopathological examination are used to assess testicular function in this study. This study aimed to investigate the toxic action of ketamine on the testicular function and whether these effects are reversible.
Materials and methods
The local ethical committee of the Faculty of Medicine, Minia University, approved all procedures of this study. All steps were carried out in agreement with the Ethical Committee of Minia University for animal care. Thirty adult male Wistar albino rats were obtained from the Faculty of Agriculture, Minia University. Animals were housed in polypropylene cages (five per cage to avoid overcrowding) at 25 ± 5°C. Animals were fed laboratory chow and water ad libitum. Rats were acclimated to the experimental conditions for 1 week before starting the experiment. Ketamine was purchased as Ketalar® (ketamine hydrochloride 50 mg/ml, Parke-Davis, NJ, USA).
Rats were divided into three groups (10 rats each). Group 1 (control group): Rats received normal saline intraperitoneally (IP). Five rats were euthanized after 6 weeks (control group A), and the other five rats were maintained under the same experimental conditions for an additional 4 weeks (control group B). Group II (ketamine group): Animals received ketamine IP at a subanesthetic dose (50 mg/kg/day) for 6 weeks. Group III (withdrawal group): Each animal received ketamine 50 mg/kg/day IP for 6 weeks, followed by a 4-week drug-free period to test the reversibility of ketamine toxicity. Ketamine is used as an anesthetic agent in rats at a dose of 75–100 mg/kg. 19 The selected subanesthetic dose was chosen according to Wang et al. 20
Throughout the study, the rats were weighed weekly (to adjust the calculated dose) and before euthanizing. Testes, epididymis, seminal vesicles, and prostate were dissected, cleared from adjacent tissues and weighed, and the relative organ weights were calculated using the following formula: organ weight (g)/body weight × 100. 21
At the end of the experiments, the rats were euthanized by decapitation. Blood samples were collected via cardiac puncture and centrifuged (4000 r/min for 10 min) to obtain serum for hormone assays. Samples were stored at −20°C till analysis.
Serum FSH, LH, prolactin, and testosterone were measured. All hormones were assayed using enzyme-linked immunosorbent assay kits (Microwell kits, Syntron Bioresearch, Inc., CA, USA). 22
Testes were divided into small 1 × 1 cm2 segments for tissue homogenization, followed by testicular tissue MDA assessment, or hematoxylin and eosin (H&E) staining and immunohistochemistry.
Sperm parameters
The epididymis of each rat was opened using a surgical blade and rinsed with physiological saline. A sperm suspension was collected via vigorous pipetting in phosphate-buffered saline (PBS) solution at 37°C and filtered through an 80-µm stainless steel mesh. Sperm motility, count, and head abnormality were assessed in the obtained suspension.
The mean sperm count was determined as described previously. 23 Ten microliters of the suspension were loaded into the Neubauer hemocytometer (deep 1/10, Labart, Munich, Germany). The settled spermatozoa were counted by a light microscope (400×) after 5 min and expressed as a million/ml suspension.
Sperm motility was evaluated immediately after animal dissection according to El-Wakf et al. 24 In brief, two drops of the epididymal suspension were placed on a slide and coverslipped. The number of motile sperm was counted at 40× magnification and divided by the total number of spermatozoa, and the results are presented as a percentage. Five slides from each sample were examined.
One drop of the epididymal suspension was mixed with 1% eosin Y solution on a glass slide and allowed to air dry for 30 min. Five smears were prepared from each sample and examined under 40× magnification. The number of abnormal sperm heads among 200 spermatozoa was counted, and the results are presented as a percentage. 25
Assessment of testicular tissue MDA concentration
Tissue MDA was assessed by the thiobarbituric acid method according to the steps described by Noiri et al. 26 A homogenized sample of testicular tissue was mixed with 2.5 volumes of 10% trichloroacetic acid. After centrifugation, thiobarbituric acid (0.67%) was added to the supernatant. The mixture was placed in a boiling water bath for 30 min. The absorbance was read at 532 nm, and the MDA concentration (nmol/mg protein) was calculated from the standard curve.
Histological procedures
Testis segments were fixed with Bouin’s solution to be followed by ordinary H&E staining. Five-micrometer-thick sections were cut and stained with H&E. 27
Immunohistochemistry
Other 5-μm-thick sections were used for immunohistochemical staining for caspase-3 and vimentin using polyclonal antibodies. Briefly, sections were hydrated and washed in 0.1 M phosphate buffer. Sections were treated with 0.01% trypsin for 10 min at 37°C, washed with phosphate buffer for 5 min, and soaked in absolute methanol containing 0.3% hydrogen peroxide for 30 min at room temperature. Nonspecific binding was blocked by normal goat serum. Tissues were incubated overnight at 4°C with primary antibody (caspase-3, 1:1000; vimentin, 1:500) and then for 60 min with biotinylated goat antirabbit immunoglobin serum. Sections were washed with PBS and incubated with avidin/biotin peroxidase complex (Vector, Burlingame, California, USA). Peroxidase binding was detected using the chromogenic 3,3′-diaminobenzidine tetrahydrochloride substrate. Tissue sections were counterstained with hematoxylin. 28 Caspase-3-positive cells showed brown staining in the cytoplasm and/or nucleus, while vimentin-positive cells showed brown cytoplasmic staining.
Image capture
H&E and immunostained sections were examined using an Olympus light microscope (Olympus, Tokyo, Japan). Images were captured using a digital camera mounted on the microscope and connected to a computer. Image analysis and measurement were performed using ImageJ software (National Institutes of Health, Bethesda, MD, USA).
Morphometric study
In H&E-stained sections, 10 adjacent nonoverlapping fields were randomly selected at 400× magnification. The mean seminiferous tubule diameter, lumen diameter, and epithelium height were measured.
Caspase-3 and vimentin immunopositive areas were measured in 10 adjacent nonoverlapping fields of each slide. Immunostained areas were calculated as a percentage of the total field area using the following formula 29 :
Data analysis and statistics
Quantitative data analysis was carried out using SPSS version 20 (IBM Corp., Armonk, NY, USA). Data are presented as mean ± standard deviation. Statistical significance was tested by one-way analysis of variance followed by Tukey honestly signficant difference (HSD) post hoc test for comparing between groups. A p value of less than 0.05 was considered significant.
Results
Table 1 provides changes in the rat body weight throughout the study. Chronic ketamine administration induced a significant reduction in body weight. Four weeks after stopping ketamine administration, body weight increased to be insignificant in comparison with the weight of the appropriate control rats (control group B). Table 2 illustrates the absolute and relative weights of sex organs of rats treated with ketamine and that of the recovery group.
Body weights of male rats exposed to a subanesthetic dose of ketamine for 6 weeks with or without a 4-week drug withdrawal period (mean ± SD).a
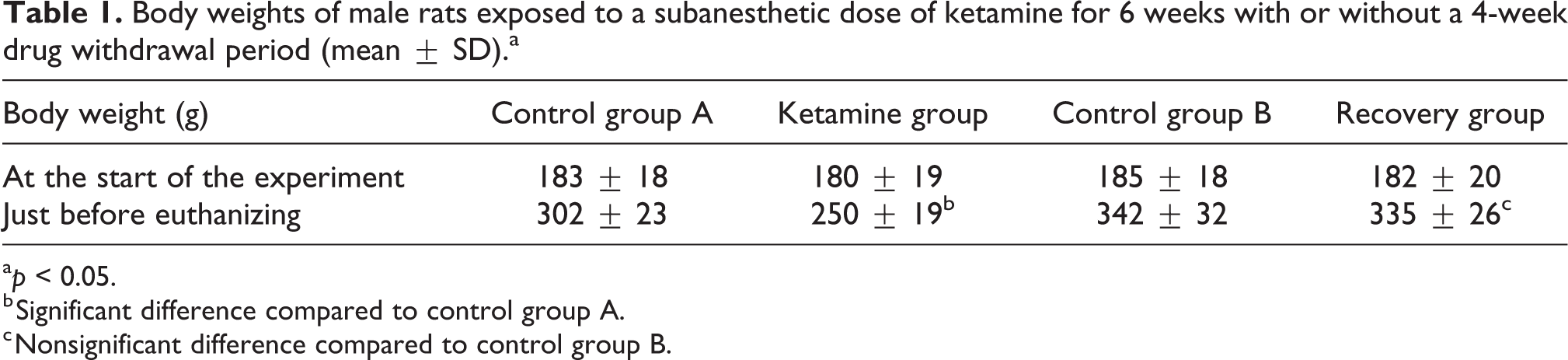
a p < 0.05.
b Significant difference compared to control group A.
c Nonsignificant difference compared to control group B.
Absolute and relative weights of the reproductive organs of male rats exposed to a subanesthetic dose of ketamine for 6 weeks with or without a 4-week drug withdrawal period (mean ± SD).a

SD: standard deviation.
a Absolute weight in grams while relative weight is calculated as organ weight/animal weight × 100 g.
b Significant difference compared to control group A. p < 0.05.
Table 3 shows that the IP injection of ketamine at a subanesthetic dose adversely affected sex hormones. Serum LH, FSH, prolactin, and testosterone decreased significantly (p < 0.05) in the ketamine group compared with the control group A. After ketamine withdrawal, the examined hormones returned to the normal values.
Serum reproductive hormones and testicular MDA concentrations in male rats exposed to a subanesthetic dose of ketamine for 6 weeks with or without a 4-week drug withdrawal period (mean ± SD).

LH: luteinizing hormone, FSH: follicle-stimulating hormone; MDA: malondialdehyde; SD: standard deviation.
a Significant difference compared to control group A. p < 0.05.
Chronic ketamine administration produced a significant increase in testicular MDA levels compared with its levels in the control group A (p < 0.01). Four weeks after stopping the ketamine injections, MDA concentration decreased to that in the control group B (Table 3).
Sperm parameters (count, motility, and abnormal heads) were significantly altered in the epididymis of rats that received ketamine compared with those that received saline (p < 0.05) (Table 4). Four weeks after ketamine withdrawal, all sperm parameters improved.
Semen parameters of rats treated with ketamine IP for 6 weeks with or without a 4-week drug withdrawal period.

a Significant difference compared to control group A. p < 0.05.
H&E results
Microscopic examination of testicular tissue from the control groups (A and B) revealed a normal histological structure of the seminiferous tubules and interstitial tissues. The seminiferous tubules were lined by several rows of Sertoli cells and spermatogenic cells at different stages of maturation. The spermatogenic cells are the oval-shaped spermatogonia resting on the basal lamina, primary spermatocytes with large nuclei containing dark clumps of chromatin, spermatids with rounded nuclei, late spermatids with dense and elongated nuclei, and mature sperm in the lumen of the tubules. Sertoli cells have irregular pale nuclei located basally. The interstitial tissue contained Leydig cells (Figure 1).

Photomicrographs of rat testis (a, b) control group, (c, d) ketamine group, and (e, f) recovery group. (a) Seminiferous tubules (T) and interstitial tissue containing Leydig cells (L). (b) Oval-shaped spermatogonia (1) resting on the basal lamina of the seminiferous tubules, primary spermatocytes (2) with large nuclei containing dark clumps of chromatin, spermatids (3) with rounded nuclei, late spermatids (4) with dense and elongated nuclei, and sperm in the tubule lumen (5). Note the Sertoli cells (S) with basally located nuclei. (c) Congested subcapsular blood vessel (C) and distortion of some seminiferous tubules (T). (d) Interstitial deposition of acidophilic vacuolated hyaline material (arrows and inset). (e, f) Most seminiferous tubules (T) appear normal, with restoration of the germinal lining (l) thickness at different stages of maturation, and sperm (s) in the lumen of most tubules. Note the few tubules with desquamated cells in the lumen (circle) and the minimal vascular congestion (c, in inset). H&E: a, c, d, e: ×100, scale bar 200 µm; b, f insets: ×400, scale bar 50 µm.
Ketamine administration for 6 weeks produced variable sporadic changes. Vascular congestion and seminiferous tubule distortion were noted. In some disorganized tubules, the germ cells were separated from the basal lamina. Other tubules showed germ cell exfoliation, with thinning of the epithelium lining. Few or no sperm were detected in the lumina of these tubules. Disruption of the basement membrane, without the loss of contact between germ cells and the basal lamina, was also observed. Some areas showed widening and edema of the interstitium, while other zones showed interstitial deposition of acidophilic vacuolated hyaline material (Figures 1 and 2).

Photomicrographs of testes sections from rats treated with ketamine (50 mg/kg IP) for 6 weeks showing the following additional changes: (a) Congested interstitial blood vessel (C) and widening of the interstitium (*) between some tubules. (b) Other tubules with germ cells separated from the basal lamina (arrows). (c) Other disorganized tubules (T) with germ cell exfoliation (*) and a thinner epithelium lining (arrows); disruption of the basement membrane with germ cells in the interstitium (thick arrow). Note the limited number or absence of sperm in these tubules. (d) Higher magnification images show disorganized tubules (T) with a thinner epithelium lining (arrows) and limited sperm in the lumen (*). H&E (a–c): ×100, scale bar 200 µm; (d) ×400, scale bar 50 µm.
Four weeks after ketamine withdrawal, most seminiferous tubules appeared normal with the restoration of germinal lining thickness. Spermatogenic cells at various stages of maturation and sperm could be identified. Only a few tubules contained some degenerated germinal epithelium, and minimal congestion was observed (Figure 1).
A morphometric study of H&E testis sections showed that chronic ketamine administration had adverse effects on seminiferous tubule diameter, lumen diameter, and epithelium height. There was a significant decrease in these three parameters in the testes of rats that received ketamine compared with that of the control A. The three parameters approached normal values 4 weeks after stopping ketamine administration (Table 5).
Morphometric analysis of testes from rats exposed to a subanesthetic dose of ketamine for 6 weeks with or without a 4-week drug withdrawal period (mean ± SD).

SD: standard deviation.
a Significant difference compared to control group A. p < 0.05.
Immunohistochemical results
A caspase-3 antibody was used to identify apoptotic cells. The control group showed scarce caspase-3-positive interstitial Leydig cells. The ketamine group showed extensive immunostaining of germinal cells in the nucleus and/or cytoplasm. In addition, caspase-3-positive interstitial cells were observed. The ketamine withdrawal group showed few caspase-3-positive spermatogonia (Figure 3).

Photomicrographs of rat testis immunostained for caspase-3. (a) Control group with scarce caspase-3-positive interstitial Leydig cells (arrow). (b) Ketamine-treated group with extensive immunostaining of germinal cells in the nucleus (thick arrow), cytoplasm (circle), or both (thin arrows). Note the immunopositive interstitial cells (curved arrow). The inset shows immunostaining of the oval-shaped spermatogonia (arrow). (c) Recovery group (4 weeks after ketamine cessation) with few caspase-3-positive spermatogonia (arrow) in the rat testes. Immunohistochemistry, counterstained with H: ×400, scale bar 50 µm.
A vimentin antibody was used to identify Sertoli cells (the only vimentin-immunoreactive cells within seminiferous tubules). In the control groups (A and B), there were three sections of Sertoli cells with different configurations in the basal compartment, the nuclear zone, lateral, and apical protrusions. The border between Sertoli cells and neighboring spermatogenic cells appeared highly variable, and lightly immunostained interstitial cells were noted. Ketamine injection for 6 weeks produced atrophy in the seminiferous tubules, which contained many vimentin-positive cells. The Sertoli cells were irregularly shaped, and their nuclei were surrounded by a thick layer of vimentin-immunoreactive material. Most tubular lumina were filled with numerous Sertoli cells. Extensive immunostaining of interstitial cells was observed. Similar to the control group, group III showed reduced immunoreactivity of Sertoli cells (Figure 4).

Photomicrographs of rat testis immunostained for vimentin. (a) Staining of samples from the control group revealed the presence of Sertoli cells, the only vimentin-immunoreactive cells within the seminiferous tubules. Three groups of immunoreactive Sertoli cells were observed in different configurations: (1) the basal compartment, (2) the nuclear zone, and (3) the lateral and apical protrusions. The border of Sertoli cells along neighboring spermatogenic cells appeared highly variable. Note the lightly immunostained interstitium (I). (b) Rats injected with 50 mg/kg ketamine IP for 6 weeks showed atrophic seminiferous tubules (T) with numerous irregular vimentin-immunostained cells. The nuclei of these positive cells were surrounded by a thick layer of vimentin-immunoreactive material (arrows). Most tubules were filled with Sertoli cells. (c) Extensive immunostaining of interstitial cells (arrow) in ketamine-injected rats (group 2) was apparent. (d) Rats examined 4 weeks after ketamine withdrawal showed reduced immunoreactivity of Sertoli cells, which appeared normal (arrows). Immunohistochemistry, counterstained with H: ×400, scale bar 50 µm.
A morphometric study of testicular tissue using caspase-3 and vimentin showed that chronic ketamine administration significantly increased caspase-3- and vimentin-immunopositive areas compared to control group A (p < 0.01). However, 4 weeks after ketamine withdrawal, there was a significant reduction in the immunostained areas compared with the time point immediately after the cessation of ketamine injections (Table 5).
Discussion
Ketamine is a commonly misused anesthetic in parties and clubs that is predominantly abused by young people. 30,31 This dissociative anesthetic drug has become one of the most common substances of addiction worldwide. In China, ketamine use ranks third among illicit drugs. In Hong Kong, ketamine is the number one abused drug by individuals younger than 21 years. 32
The testis is considered the most important organ in the male reproductive system. It has two main functions: steroid hormone synthesis and sperm production. Various factors affect spermatogenesis, including medicines and toxic elements in environmental pollution. 33 Histopathological examination of the testes provides valuable information regarding the assessment of the severity of toxicity, the cellular site of damage, and the possibility of recovery. 34
There have been few studies concerning the toxic effects of chronic ketamine on reproductive function. Apart from two studies 22,35 that examined the toxic effects of ketamine for 2 or 3 weeks, the literature has scant data on the long-term effect of ketamine on the testes. In addition, there is no information about the reversibility of these hazardous effects.
Chronic administration of ketamine decreased the weight of male reproductive organs, which agrees with the finding of Qi et al. 35 This effect was produced by ketamine administration for 6 weeks in this study and for 3 weeks in the study by Qi et al. 35 The weights of the testes and accessory sexual organs reflect the androgenic status of the animal. Products of normal spermatogenesis and fluids excreted by Sertoli cells are the main contributors to testicular weight. 36,37 The reduced testosterone levels and disrupted spermatogenesis and Sertoli cells contributed to the decreased testicular weight in this study. In addition, Venâncio et al. 38 reported that chronic ketamine administration induced a reduction in body weight, which contributed to a decrease in testicular weight.
Ketamine influences the gonadotropic hormones FSH and LH as well as prolactin and testosterone. The hormonal effect of ketamine was also reported previously. 22,35 Qi et al. 35 noticed a dose-dependent decrease in LH, FSH, and testosterone in response to ketamine. The decreased serum testosterone levels are partially due to decreased gonadotrophic hormones and the direct detrimental effect of ketamine on Leydig cells. Ketamine-induced lipid peroxidation and apoptosis are possible mechanisms for the effects of this drug on Leydig cells. 39 The effect of ketamine on FSH, LH, and prolactin may be attributed to the direct depressant action of ketamine on the pituitary gland. Qi et al. 35 noticed that ketamine administration depressed messenger RNA expression of gonadotrophic hormones in the pituitary gland.
It is well known that increased reactive oxygen species (ROS) adversely affect sperm parameters, and this detrimental effect can be explained by several mechanisms. ROS attack the sperm plasma membrane and cause lipid peroxidation. In addition, free radicals induce DNA fragmentation and decrease sperm motility. 40 –42 The study presented herein showed that ketamine caused lipid peroxidation, which mirrored increased testes MDA levels in rats treated with ketamine for 6 weeks. This effect disappeared 4 weeks after the cessation of ketamine injections. Araghi et al. 22 reported similar findings.
In the present study, variable and patchy histological changes in testicular tissues were observed. Vascular congestion and distortion of the seminiferous tubules appeared with basement membrane disruption, a decrease in germ cells in some tubules (hypoplasia), desquamation of germ cells lining the seminiferous tubules, and widening of intercellular spaces (edema of the interstitium). The most notable histopathological finding was the reduction in normal spermatogenic cells and, accordingly, in spermatozoa in many tubules. This finding agreed with the results reported by Araghi et al. 22 and, to a lesser degree, Qi et al. 35 Four weeks after the cessation of ketamine administration, intact spermatogenic cells at different stages of maturation were restored in the testes, which then appeared similar to those of the control rats.
The immunohistochemical results of this study using the caspase-3 antibody confirmed the H&E results regarding the degeneration of spermatogenic cells. The testicular tissue of rats that received ketamine for 6 weeks showed a significant increase in caspase-3-positive cells compared to that of control rats. However, 4 weeks after ketamine withdrawal, there was a significant reduction in caspase-3-positive cells compared to control. This result indicates that ketamine increased apoptosis in testicular tissues, which is in line with the results of other studies, which showed that ketamine evoked apoptosis in other tissues, such as hepatic cells, 43 the brain, 44 and the urinary bladder. 45
Apoptosis, necrosis, and autophagy are the main three pathways of cells to die. Apoptosis is a genetically programmed pathway that results from external and/or internal factors that end with cell death. Caspases and the B-cell lymphoma 2 (Bcl-2) proteins play a cornerstone role in apoptosis. 46 It is well established that ketamine induces apoptosis via multiple mechanisms. Ketamine induces DNA fragmentation. In addition, ketamine inversely affects mitochondria and initiates Bcl-caspase apoptosis pathway. 47 Furthermore, increased lipid peroxidation that was evident in this study enhances apoptotic cell death.
Since Sertoli cells were discovered by Enrico Sertoli in 1865, they have been shown to play a central role in spermatogenesis. Without both the physical and metabolic maintenance of these cells, spermatozoa production will not occur. Sertoli cells have a well-built cytoskeleton with intermediate filaments (vimentin type) that surround the nucleus and radiate upward to form thin columnar rows. The integrity of this cytoskeleton plays a crucial role in Sertoli cell function. 48 In this study, although chronic exposure to ketamine caused massive germ cell loss, the number of Sertoli cells did not decrease significantly; rather, there was an unexpected significant increase in vimentin expression compared to the control group. Sertoli cell shape and positions were both obviously irregular. A similar finding was reported by Dalgaard et al., 49 who showed prominent vimentin expression in seminiferous tubules in rats exposed to diphthalate. However, the increased vimentin expression may be attributed to the progressive collapse of Sertoli cells and not to an actual increase in the amount of vimentin filaments. This is an important observation in this study, as it provides more explanation of the mechanism by which ketamine adversely affects reproduction.
Ketamine treatment of rats at a subanesthetic dose for 6 weeks markedly decreased sperm count and mobility compared with control. Ketamine administration increased the number of abnormal sperms. These effects were reversed and approached normal 4 weeks after stopping ketamine administration. The only report discussing the effect of ketamine on sperm was written by Tan et al., 50 who found that ketamine induced a decrease in sperm motility. It is well known that sperm count is the cumulative result of all phases of spermatogenesis and is one of the most sensitive tests for evaluating the infertility status of animals. 51 In addition, normal sperm motility plays a vital role in sperm fertilizing capability. 52 Ketamine-induced apoptosis, hormonal changes, and increased lipid peroxidation collectively adversely affected the process of spermatogenesis.
These adverse effects of ketamine on the testicular tissue should be kept in mind in clinical uses of this drug. This is worth noting as ketamine is increasingly used in clinical practice for various chronic diseases. 5,53 In addition, ketamine addiction is common, chiefly in young individuals. 30
In conclusion, chronic ketamine treatment has hazardous effects on reproductive function. Ketamine adversely affects sex hormones and sperm parameters. These effects occur via multiple mechanisms involving lipid peroxidation, apoptosis, and Sertoli cell disruption. There is a need to increase the knowledge of physicians and the public regarding these harmful effects of ketamine.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

