Abstract
Paracetamol was shown to induce hepatotoxicity or more severe fatal acute hepatic damage. Agomelatine, commonly known as melatonin receptor agonist, is a new antidepressant, which resynchronizes circadian rhythms with subjective and objective improvements in sleep quality and architecture, as melatonin does. In the present study, it was aimed to evaluate the hepatoprotective activity of agomelatine on paracetamol-induced hepatotoxicity and to understand the relationship between the hepatoprotective mechanism of agomelatine and antioxidant system and proinflammatory cytokines. A total of 42 rats were divided into 7 groups as each composed of 6 rats: (1) intact, (2) 40 mg/kg agomelatine, (3) 140 mg/kg N-acetylcysteine (NAC), (4) 2 g/kg paracetamol, (5) 2 g/kg paracetamol + 140 mg/kg NAC, (6) 2 g/kg paracetamol + 20 mg/kg agomelatine, and (7) 2 g/kg paracetamol + 40 mg/kg agomelatine groups. Paracetamol-induced hepatotoxicity was applied and liver and blood samples were analyzed histopathologically and biochemically. There were statistically significant increases in the activities of aspartate aminotransferase, alanine aminotransferase, levels of tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) and 8-iso-prostane, and decreases in the activity of superoxide dismutase and level of glutathione in the group treated with paracetamol. Administration of agomelatine and NAC separately reversed these changes significantly. In conclusion, agomelatine administration protects liver cells from paracetamol-induced hepatotoxicity via antioxidant activity and reduced proinflammatory cytokines, such as TNF-α and IL-6.
Introduction
Drugs are metabolized in the liver, which is the most important organ in which drug-related organ toxicity may develop. Kupffer cells are liver macrophages with a great capacity to eliminate endotoxins. 1,2 Once activated, Kupffer cells initiate the release of tumor necrosis factor-alpha (TNF-α), interleukin-1β (IL-1β), interleukin-6 (IL-6), and interleukin-8 (IL-8). 3 Recently, the role of Kupffer cells, TNF-α, and IL-6 in paracetamol-induced liver toxicity was evaluated. 4,5 Blazka et al. showed that paracetamol-induced production of TNF-α and IL-1β occurs in response to damage by paracetamol-induced hepatic intoxication, and selective neutralization of these cytokines changed during the pathophysiological response against paracetamol-induced hepatic intoxication. 4
Nonsteroidal anti-inflammatory drugs (NSAIDs) are the medications prescribed most often when patients need analgesic, anti-inflammatory and antipyretic effects. In Europe, NSAIDs make up 7% of all prescribed drugs. 6 Paracetamol has been in common use for the past 50 years, but it has only been widely known since 1983. The drug has analgesic and antipyretic but not anti-inflammatory effects. It differs from other NSAIDs in that paracetamol does not cause Reye syndrome in children and has no harmful effects on the gastrointestinal system (GIS); therefore, it is prescribed most commonly as an analgesic and antipyretic medication.
In many markets, the drug is often available as an over-the-counter (OTC) medication. In the United Kingdom, a study conducted among mothers with newborns indicated that 84% of those surveyed received a prescription for paracetamol during the first 6 months of their children’s lives. 7 In the United States, 100 million people take paracetamol at least once a year, and 50 million people use the drug at least once a week. According to the Food and Drug Administration, 70% of all paracetamol-containing products are sold as OTC drugs in the United States. 8
The risk of paracetamol toxicity is increased because it is so commonly used and easy to reach. Single doses in excess of 150 mg/kg in children and 7.5 g in adults 9 or a daily amount of 250 mg/kg in children and 12 g in adults 10 can induce paracetamol toxicity. This type of toxicity may be asymptomatic; in 1960, paracetamol was shown to induce hepatotoxicity or more severe fatal acute hepatic damage. 11 Currently, the most effective antidote for liver damage caused by paracetamol toxicity is N-acetylcysteine (NAC). 12 One study showed that 90% of patients with or without hepatotoxicity who overdosed on paracetamol healed completely after NAC treatment. 13 When NAC is administered in the first 12 h of hepatotoxicity, 80% of patients recover; this ratio decreases to 48% when NAC is not given. 14 At the same time, many pharmacologically active components (such as vitamins and medical plants), which possess antioxidant properties, have been studied for their utility to protect against paracetamol toxicity. 15,16
Agomelatine (S-20098, or N-(2-(7-methoxy-1-naphthyl) ethyl)acetamide) is a naphthalene derivative with a structure similar to that of melatonin. 17 Double-blind, randomized trials have shown agomelatine to be an effective antidepressant. Studies have also determined that agomelatine effectively resynchronizes circadian rhythms 18 with subjective 19 and objective 20 improvements in sleep quality and architecture, as does melatonin. Melatonin, synthesized from tryptophan in the pineal gland, plays a central role in fine-tuning circadian rhythms and seasonal changes in vertebrate physiology through its daily nocturnal increase in blood levels. 21 Matsura et al. suggested that orally administered melatonin significantly reduces hepatic lipid peroxidation induced by paracetamol administration. 22 However, no study to date has investigated the effects of agomelatine, a melatonin receptor agonist, on paracetamol-induced hepatotoxicity of reactive oxygen species and other free radicals.
In the present study, we investigated the hepatoprotective and antioxidative activity of agomelatine in comparison with NAC against paracetamol toxicity, which is a widely used model for investigating the acute hepatic drug injury response. To understand the relationship between the hepatoprotective mechanism of agomelatine and antioxidant system and proinflammatory cytokines, we measured superoxide dismutase (SOD) activity, levels of glutathione (GSH) and 8-iso-prostaglandin F2α (8-ISO) in hepatic tissue, and the plasma level of IL-6 and TNF-α.
Methods
Animals
In this study, we used 42 male Albino Wistar rats, which were obtained from the Medical Experimental Research Center, Ataturk University. The animals weighed between 240 and 260 g and were fed under normal temperature conditions (22°C) in separate groups before the experiments. The local committee of Ataturk University approved the design of the experiments, and our study was performed in accordance with the national guidelines for the use and care of laboratory animals.
Chemicals
All chemicals for laboratory experimentation (including paracetamol) were purchased from Sigma Chemical (Germany). We obtained thiopental sodium from IE Ulagay A.S. Istanbul, Turkey, and agomelatine (Valdoxan 25 mg Tb) was purchased from Servier, Turkey. NAC (Asist 200 mg capsules) was obtained from Husnu Arsan Drugs, Istanbul/Turkey.
Treatment
Animals fasted overnight and were divided into seven equal groups (n = 6). The effects of agomelatine on paracetamol-induced hepatotoxicity were studied in rats. Groups and details were performed as below. In paracetamol groups, drugs were administrated 1 h later paracetamol. The rats were allowed food postadministration of drugs for the next 24 h until they were sacrificed.
Group 1: intact;
Group 2: 40 mg/kg agomelatine, orally (po);
Group 3: 140 mg/kg NAC, po;
Group 4: 2 g/kg paracetamol (suspended in 1% carboxymethylcellulose (CMC) in 1× phosphate buffered saline (PBS) 2 ml per rat, po);
Group 5: 2 g/kg paracetamol (suspended in 1% CMC in 1× PBS 2 ml per rat, po) + 140 mg/kg NAC, po;
Group 6: 2 g/kg paracetamol (suspended in 1% CMC in 1× PBS 2 ml per rat, po) + 20 mg/kg agomelatine, po; and
Group 7: 2 g/kg paracetamol (suspended in 1% CMC in 1× PBS) 2 ml per rat, p.o. + 40 mg/kg agomelatine, po.
Twenty-four hours after the induction of paracetamol toxicity, the rats were anesthetized using a 50 mg/kg lethal dose of thiopental. Blood samples were collected into bottles by heart puncture. These specimens were used for the analyses of aspartate transaminase (AST), alanine transaminase (ALT), TNF-α, and IL-6.
Biochemical investigations
Sera were separated from blood by allowing the samples to clot followed by centrifugation at 4000 revolutions per minute (rpm) for 10 min at 4°C; samples were kept at −86°C until they were thawed for the assay.
Cytokine (IL-6 and TNF-α) serum measurements
In separated serum samples, IL-6 and TNF-α from each sample were measured in duplicate according to the manufacturer’s instructions with highly sensitive enzyme-linked immunosorbent assay (ELISA) kits containing RayBiotech-ELR-IL6.001 (United States) and Invitrogen-KRC3011 (United States), respectively, that were specifically designed for rats. All reagents, samples, and standards were prepared as instructed. Standard or sample, 100 μl, was added to each well coated with anti-rat IL-6 and TNF-α and was incubated 2.5 and 2 h at room temperature, respectively. After incubation, the liquid was discarded and the wells were washed and 100 μl biotin antibody was added to each well and incubated 1 h at room temperature with gentle shaking. After discarding the liquid and washing wells, 100 μl streptavidin solution was added and incubated 45 min at room temperature. After discarding the liquid and washing the wells, 100 μl 3,3′,5,5′-tetramethylbenzidine one-step substrate reagent was added to each well and incubated for 30 min at room temperature. Stop solutions, 50 and 100 μl, were added to each well containing IL-6 and TNF-α, respectively, and the wells were read at 450 nm immediately.
ALT and AST serum measurements
The separated serum samples were used for the determination of ALT and AST activity with an auto analyzer (Mindray-Perfect Plus Auto Analyzer).
Biochemical investigation of liver tissues
After macroscopic analyses, rat tissues were kept at −86°C; 100 mg of tissues from each rat were first perfused with PBS/heparin and then homogenized in a specific homogenate tampon (4.5 ml appropriate buffer) on ice by Ultra-Turrax after grinding in liquid nitrogen. The tissues were then centrifuged at 1200 rpm 10 min at 4°C. For our biochemical investigation, we measured the activity of SOD and the levels of 8-ISO and GSH from each supernatant in duplicate using Cayman-706002 (United States), Cell Biolabs-STA-337 (United States), and Cell Biolabs-STA-312 (United States) highly sensitive ELISA kits, respectively, which were specifically designed for rat tissue, according to the manufacturer’s instructions. All the data were presented as the mean ± standard deviation results based on per mg protein.
SOD assay
The diluted radical detector, 200 μl, and 10 μl of Standard and sample were added to each SOD standard and sample wells. Then 20 μl of diluted xanthine oxidase was added to all of the wells and the 96-well plate was incubated on a shaker for 20 min at room temperature. Wells were read by a plate reader. The absorbance was read at 440–460 nm immediately.
GSH assay procedure
GSH reductase solution, 25 μL, was added to each well in a 96-well plate and 25 μL of the NADPH were added onto it; 100 μL of the prepared GSH standards or samples were added to each well and mixed thoroughly. Then 50 μL of the chromogen was added and the absorbance was read at 405 nm.
8-ISO assay
Diluted anti-8-ISO antibody of 100 μL was added to the goat anti-rabbit antibody-coated plate and the plates were incubated for 1 h at 25°C on an orbital shaker. The solution was removed from the wells and the wells were washed five times with 300 μL 1× Wash Buffer per well. 8-ISO standard or sample of 55 μL and 55 μL of 8-ISO–horseradish peroxidase conjugate were combined in a microtube and mixed thoroughly. Combined solution of 100 μL was transferred per well, and the wells were incubated for 1 h at 25°C on an orbital shaker. After removing the combined solution, the wells were washed five times with 300 μL of wash buffer. Substrate solution of 100 μL was added to each well, and the wells were incubated at room temperature for 10–30 min on an orbital shaker. Enzyme reaction was stopped by adding 100 μL of stop solution to each well. Plate was read at 450 nm immediately.
Protein determination
The protein concentrations were determined by the Lowry method 8 using commercial protein standards (Sigma Aldrich, Total protein kit-TP0300-1KT, USA).
Histopathological study
At the end of the experiment, the rats were sacrificed. Their livers were removed, fixed in a 10% formalin solution for 24 h, dehydrated by an increasing-concentration alcohol series, and cleared by xylene series. After histological processing, the tissues were embedded in paraffin and sectioned to a thickness of 5 mm using a microtome (Leica RM2235, Leica Instruments, Nussloch, Germany) with disposable metal microtome blades (Leica 819, Leica Instruments, Nussloch, Germany). Sections were stained with hematoxylin and eosin (H&E) and observed under a light microscope. In addition, photomicrographs were obtained.
Statistical analyses
Data for the serum cytokine levels measured by ELISA, oxidant, and antioxidant enzymes were subjected to one-way analysis of variance using the Statistical Package for the Social Sciences (SPSS version 19.0) software. Differences among the groups were obtained using the Duncan’s multiple range test option and were considered significant at p < 0.05. All data were expressed as mean ± SD in each group.
Results
Biochemical results
Effects of agomelatine on serum TNF-α and IL-6 levels on paracetamol-induced hepatotoxicity in rats
In the present study, the serum levels of TNF-α and IL-6, which are inflammatory cytokines, were studied using a paracetamol-induced hepatotoxicity model based on the sera from rats (Table 1). The levels of both cytokines increased in the paracetamol group (TNF-α: 158.90 ± 28.20 pg/ml, IL-6: 341.00 ± 48.63 pg/ml) when compared with the healthy animals (control group) in which TNF-α was 33.95 ± 5.59 pg/ml and IL-6 was 76.50 ± 13.85 pg/ml, as shown in Table 1 (p < 0.05). In contrast with the paracetamol group, serum levels of TNF-α and IL-6 decreased as a result of the administration of all doses of agomelatine and NAC in the paracetamol condition (agomelatine-treated paracetamol groups and NAC-treated group) (p < 0.05). Administration of agomelatine at 20 and 40 mg/kg dosages and NAC decreased the serum levels of TNF-α to 71.61 ± 8.84 pg/ml, 62.72 ± 10.53 pg/ml and 74.38 ± 14.19 pg/ml, respectively. Administration of agomelatine (at 20 and 40 mg/kg dosages) and NAC decreased the serum levels of IL-6 to 150.63 ± 18.48 pg/ml, 132.75 ± 24.71 pg/ml and 170.25 ± 28.21 pg/ml, respectively. As shown in Table 1, the administration of agomelatine alone in the control rats did not affect the serum levels of either cytokine (TNF-α: 37.98 ± 6.50 pg/ml, IL-6: 82.25 ± 13.73 pg/ml) when compared to the control group.
Effects of Agomelatin treatments on changes in serum levels of ALT, AST, TNF-α and IL-6 in sera of rats.a
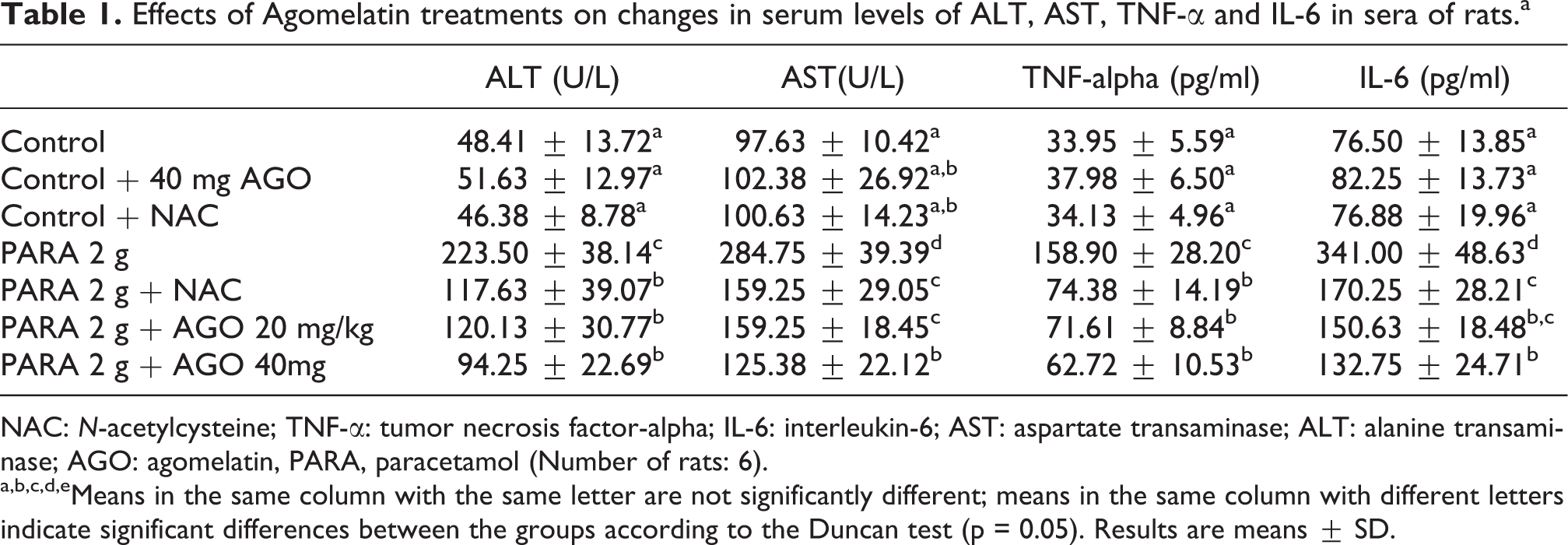
NAC: N-acetylcysteine; TNF-α: tumor necrosis factor-alpha; IL-6: interleukin-6; AST: aspartate transaminase; ALT: alanine transaminase; AGO: agomelatin, PARA, paracetamol (Number of rats: 6).
a,b,c,d,eMeans in the same column with the same letter are not significantly different; means in the same column with different letters indicate significant differences between the groups according to the Duncan test (p = 0.05). Results are means ± SD.
Effects of agomelatine on serum ALT and AST levels in paracetamol-induced hepatotoxicity in rats
The effects of agomelatine on the serum enzymes ALT and AST are shown in Table 1.
There were statistically significant (p < 0.05) increases in the plasma activities of AST and ALT in the group treated with paracetamol as compared with the control. Agomelatine administration significantly decreased the activities of the serum enzymes when compared with the control group (p < 0.05).
Effects of agomelatine on liver oxidant and antioxidant levels on paracetamol-induced hepatotoxicity in rats
The activity of SOD and the level of GSH, and the level of 8-ISO, was evaluated in all rat livers. Table 2 shows that SOD and GSH values for groups administered paracetamol were lower, and the 8-ISO level was higher than those of the control rat group. However, as compared with 8-ISO and SOD levels in agomelatine- and NAC-administrated rats, the opposite results were found for the levels of 8-ISO and SOD activities in liver tissue from the groups administered paracetamol. In contrast to the groups administered paracetamol, both doses of agomelatine and NAC increased SOD activity (p < 0.05), and both doses of agomelatine and NAC decreased the 8-ISO level (p < 0.05).
Effects of agomelatin treatments on changes in activities of SOD and with levels of 8-iso-prostaglandin F2α (8-ISO) and total GSH in liver tissues of rats.a

NAC: N-acetylcysteine; SOD: superoxide dismutase; GSH: glutathione; AGO: agomelatin, PARA: paracetamol (number of rats: 6).
a,b,c,d,eMeans in the same column with the same letter are not significantly different; means in the same column with different letters indicate significant differences between the groups according to the Duncan test (p = 0.05). Results are means ± SD.
Table 2 shows that the level of GSH decreased by paracetamol administration. The level of GSH increased by administration of both doses of agomelatine and NAC (p < 0.05), while the level of GSH elevated by paracetamol was more ameliorated by 40 mg/kg agomelatine (p < 0.05).
Histopathological results
Control group
When the control groups’ liver H&E-dyed sections were evaluated, the central vein and the nearby hepatocytes were seen as their natural structure. The sinusoids between hepatocytes had regular walls and natural endothelial cells. Additionally, the periportal area consisted of the bile duct, branches of hepatic arterioles and the portal vein, some connective tissue cells and the surrounding hepatocytes, all of which were determined to be normal (Figure 1).

Liver sections, control group. (a) Central region of the liver; central vein (cv) center of the hepatic lobule and hepatocytes, extending from the cv radially. (b) Portal region of the liver. Pa: portal area and its compounds, bile ducts and blood vessels. Dye: H&E, section thickness: 5 µm.
Agomelatine group
Hepatocytes present in both the central and the periportal area had similar structure as the control group histologically. Additionally, the sinusoids, endothelial cells, and compounds of portal areas did not differ from the control group (Figure 2).

Liver sections, agomelatin 40 mg group. (a) and (b) General view of liver. cv: central vein in the center of the hepatic lobule. Dye: H&E, section thickness: 5 µm.
Paracetamol group
Centrilobular necrosis was conspicuous at first glance. Inflammatory cell infiltration stimulated with necrotic hepatocytes was seen at the center of the necrotic foci. Sinusoids also had lost their regular walls. However, necrosis was not seen in periportal hepatocytes, but their eosinophilic dying pattern increased (Figure 3).

Liver sections, paracetamol group. (a) and (b) General view of the liver. Black arrow: necrotic focuses Dye: H&E, section thickness: 5 µm.
Paracetamol and 20 mg agomelatine group
In this group, liver section degeneration in hepatocytes was restricted with few cells in a line that surrounded the central vein (Figure 4(a)). Some of these hepatocytes had hyperchromatic nuclei and eosinophilic cytoplasm, while others had two nuclei that suggested evidence of regeneration (Figure 4(a)). Inflammatory cells in the sinusoidal areas around some central veins were also detected, although there were not as many as observed in the groups receiving only paracetamol (Figure 4(b)).

Liver sections, paracetamol and 20 mg agomelatin group. (a) Central regions of the liver, cv: central vein. Hepatocytes in this region had more acidophilic cytoplasm. White arrow: hepatocyte with double nuclei. (b) Portal and central regions of the liver, Pa: portal area. Inflammatory cells were noted around the central vein. Dye: hematoxylin and eosin (H&E), section thickness: 5 µm.
Paracetamol and 40 mg agomelatine group
The histopathological findings of this group’s liver sections were similar with the group that received paracetamol and 20 mg agomelatine, such as in the number of inflammatory cells remaining (Figure 5(a) and (b)). Nevertheless, degenerated hepatocyte lines were more prevalent in this group than in the former group.

Liver sections, paracetamol and 40 mg agomelatin group. (a) Central and portal regions of the liver, Pa: portal area, cv: central vein. Hepatocytes around the cv had more acidophilic cytoplasm. black arrow: necrotic hepatocytes. (b) Central region of the liver. Dye: hematoxylin and eosin (H&E), section thickness: 5 µm.
Paracetamol and NAC group
In this group, sections including a large number of hepatocytes with cytoplasmic vacuoles around the portal area were conspicuous (Figure 6(a) and (b)).

Liver sections, paracetamol and NAC group. (a), (b) Central and portal regions of the liver, cv: central vein, Pa: portal area. Many hepatocytes with cytoplasmic vacuoles were seen. Dye: hematoxylin and eosin (H&E), section thickness: 5 µm. NAC: N-acetylcysteine.
Discussion
In the present study, we evaluated the hepatoprotective activity of agomelatine on paracetamol-induced hepatotoxicity, which is a widely used model for assessing drug-induced hepatic injury. Paracetamol, which has been used for over 50 years, has only been widely known since 1983. It has analgesic and antipyretic but not anti-inflammatory effects. Because the drug is commonly used and easy to reach, the risk of paracetamol toxicity is generally higher than that of other NSAIDs.
At therapeutic doses, one of three processes occurs in the liver: the glucuronyl transferase enzyme conjugates paracetamol to glucuronic acid (60%), sulfonyl transferase enzyme conjugates it to sulfuric acid (35%), or it is conjugated to cysteine and excreted in the urine. Two percent of Paracetamol is excreted unchanged in urine. 13 However, a small amount of paracetamol is hydroxylated by N-hydroxylase and cytochrome p450 (CYP) enzymes to form a quite reactive end product, N-acetyl-p-benzoquinoneimine (NAPQI). This metabolite is a reactive electrophilic molecule that damages intracellular proteins by bonding with them covalently. In normal physiological conditions, NAPQI reacts with GSH and is excreted along with bile. 23,24 However, when administered in overdose quantities to humans, NAPQI consumes GSH stores in the liver and induces hepatotoxicity associated with oxidative stress. 25 This mechanism has been suggested to be a main pathway for paracetamol-induced hepatotoxicity. 24 Paracetamol toxicity in rats has been investigated in several studies. 8,26 Rats are the most known and used experimental animals and they have a very interesting metabolism. Heart rate of rats is five times higher than that of human beings and as a result liver metabolism changes. Therefore, the toxic doses of the drugs are different in humans and rats. Doses we used for rats are found after lots of trying and in accordance with literature mentioned above. 8,26
N-acetyl cysteine, cysteamine, and methionine are effective in neutralizing NAPQI. In clinical studies, each treatment was successful in preventing hepatotoxicity; however, methionine and cysteamine exerted greater side effects on the GIS and central nervous system. High doses of paracetamol administration in rats triggered the development of oxidative stress-induced damage in liver tissues. This effect was determined by increased lipid peroxidation, inhibited enzymatic antioxidants, and depleted nonenzymatic antioxidants (e.g. intracellular GSH) in the liver. The body has many antioxidant mechanisms for scavenging ROS to prevent oxidative stress. SOD is an antioxidant enzyme that eliminates ROS. A reduction in SOD activity in paracetamol toxicity may be due to the overproduction of superoxide radical anions. 27 Therefore, increased synthesis of superoxide, which is produced by a number of enzymatic reactions (including paracetamol oxidation to NAPQI) is a highly reactive, electrophilic molecule that binds with other intracellular proteins and may lead to lipid peroxidation. 28,29
Treatment with paracetamol caused a statistically significant decrease in hepatic SOD. Reduction in SOD activity seems to be a result of futile cycling of CYP caused by NAPQI, which used the reducing equivalent of NADPH with the concomitant reduction of molecular superoxide anion radical (O−). 24 The protective activities of the agomelatine against a paracetamol-induced decrease in hepatic SOD suggest that their hepatoprotective activity can be mediated at least in part via the preservation of normal levels of antioxidant activities. Additionally, 8-ISO is a lipid peroxidation biomarker produced from arachidonic acid that shows the oxidative stress status. Measurement of 8-ISO is one approach to quantitatively measure oxidative stress status in vivo. 30 Therefore, elevated 8-ISO has been widely used to determine the in vivo oxidative stress. 31 In the present study, the increase in serum 8-ISO levels after paracetamol administration was ameliorated by agomelatine and NAC administration, which exerted antioxidant effects.
Another defense mechanism of the liver against toxic metabolites is conjugation with GSH. 32 Agomelatine acts as an antagonist at the serotonin 5-hydroxytryptamine 2c receptor and with an even higher affinity as an agonist at the melatonin MT1 and MT2 receptors. 33,34 Melatonin shows a remarkable functional versatility, exhibiting immunomodulatory, 35 oncostatic, 36 antiaging, 37 and antioxidant 38 properties. Four mechanisms of action for melatonin have been described: (a) interaction with membrane receptors 39 ; (b) binding to nuclear receptors 40 ; (c) interaction with cytoplasmic proteins 41 ; and (d) antioxidant activity, including scavenging properties. 42 It has recently been reported that melatonin protects mice against paracetamol-induced hepatotoxicity probably via the inhibition of oxidative stress, including lipid peroxidation and protein oxidation. 43 Melatonin is known to induce c-glutamylcysteine synthetase, the rate-limiting enzyme of GSH synthesis, thereby increasing cellular GSH concentration in human vascular endothelial cells. 44 In vivo studies showed that melatonin exerts hepatoprotective effects against paracetamol toxicity by reducing both oxidative stress and inflammatory influx. 43,45 More recently, an in vitro study using primary cultured mouse hepatocytes reported that ROS generation followed by lipid peroxidation occurred in hepatocytes after paracetamol treatment and that melatonin pretreatment suppressed the increase in ROS and lipid peroxide in hepatocytes. 45 This hepatoprotective effect might be a result of stabilization in the redox state and maintenance of the antioxidant capacity offered by agomelatine. It could also be attributed to the melatonin receptor activity exerted by agomelatine.
In paracetamol overdose cases in clinical or experimental studies, the hepatic GSH store is depleted as a result of microsomal membrane damage and cell death. 46 NAC is therefore currently accepted as the most effective antidote for acute liver damage induced by paracetamol toxicity that leads to increased levels of GSH. 12 Toxic doses of paracetamol generate NAPQI, an oxidative product of CYP, in hepatocytes. GSH can neutralize the highly electrophilic NAPQI but in conditions of depleted GSH stores. In the current study, the level of GSH decreased following paracetamol administration. GSH levels increased after receiving both doses of agomelatine and NAC, while the level of GSH elevated by paracetamol was ameliorated by 40 mg/kg agomelatine.
Depletion of GSH strongly sensitizes hepatocytes to the exacerbating effects of TNF-α. 47,48 High doses of paracetamol in the rat are also associated with an increase in IL-1. 4,49 Blazka et al. demonstrated that the administration of anti-TNF-α antibodies to animals receiving paracetamol ameliorated the high enzyme level of the intoxication. 4 From this study, we can conclude that TNF-α may participate in the processes that cause liver failure. Our study investigated the role of agomelatine on TNF-α and IL-6 in the pathogenesis of paracetamol-induced hepatic injury in rats. The results of our study indicate that the levels of both cytokines increased in the paracetamol group, and the serum levels of TNF-α and IL-6 decreased as a result of the administration of all doses of agomelatine and NAC in the paracetamol group. This conclusion has been derived from a number of experimental findings. They principally contribute to the protection of cells and tissues against deleterious effects. A dosage of 2 g/kg paracetamol resulted in profound hepatotoxicity and correlated well with the rise in serum AST (284.75 ± 39.39 IU/l) and ALT (223.50 ± 38.14 IU/l) levels.
Test compounds were administered orally, and the serum enzyme levels were monitored as markers for hepatic conditions. ALT is a marker for liver injury in humans and is also used in preclinical toxicity studies. Serum levels of ALT are elevated in pathological conditions of the liver, such as viral infections, cirrhosis, nonalcoholic steatohepatitis, and drug toxicity. Liver injury caused by drugs is a term that describes a condition of abnormalities in liver tests, often shown by an increase in serum ALT levels. Nevertheless, in the current study, agomelatine administration significantly decreased serum enzyme activity when compared with the paracetamol group. The ability of agomelatine to prevent an increase in the activities of these enzymes is the primary evidence of their hepatoprotective activity. Increases in the serum activities of AST and ALT in the groups treated with 2 g/kg paracetamol indicated that the tested dose was capable of causing P450-dependent centrilobular hepatotoxicity in rats. 50 Toxic doses of paracetamol can cause ROS production that modifies proteins and macromolecules of liver cells and results in severe centrilobular hepatotoxicity and acute liver failure. 24,51 The histopathogenesis of paracetamol-induced hepatotoxicity is well defined and has been associated with liver congestion and necrosis. Furthermore, our histological study of tissue damage induced by paracetamol in the liver confirmed previous findings that overdose causes centrilobular necrosis that appears conspicuous at first glance. Inflammatory cell infiltration stimulated with necrotic hepatocytes was seen at the center of the necrotic foci. Sinusoids also lost their regular walls. Treatment with agomelatine or NAC improved these histological changes in the liver.
In conclusion, the current study suggested that rats given a high dose of paracetamol experienced altered biomarkers related to liver oxidative stress, decreased antioxidant activity, elevated serum TNF-α and IL-6, as well as induced cell injury. Agomelatine administration protects liver cells from paracetamol-induced hepatotoxicity via antioxidant activity and reduced proinflammatory cytokines, such as TNF-α and IL-6. Data from our study support the notion that agomelatine has biological properties without any antidepressant effect and may be useful in the prophylaxis and/or treatment of drug-induced hepatotoxicity. As a future consideration, further studies should try to determine the specific mechanisms involved in the hepatoprotective and antioxidant mechanisms of agomelatine without central nervous system effects.
Footnotes
Funding
This study was supported by the Scientific Research Council of Atatürk University with the number of 2011/266.
Acknowledgment
This research was conducted in the laboratories of Ataturk University, School of Medicine, Department of Pharmacology; and School of Veterinary Medicine, Department of Pharmacology and Toxicology, 25240 Erzurum/Turkey.
Conflict of interests
None of the authors has a commercial interest, financial interest, and/or other relationship with manufacturers of pharmaceuticals, laboratory supplies, and/or medical devices or with commercial providers of medically related services.
