Abstract
Oxidative stress caused by sodium valproate (SV) is known to play a key role in the pathogenesis of brain tissue. The present study was designed to evaluate the protective effect of
Introduction
Epilepsy is one of the chronic neurological diseases that could be the most frequent to happen after stroke. 1 The person, who suffers from epilepsy, is liable to many difficulties and psychological problems. 2 Seizures, epilepsy, and status epilepticus are more common in the elderly than in younger adults and result in greater mortality. 3
Valproic acid (VPA) is primarily used to treat epilepsy, migraine, and cancer. 4 Unfortunately, the drug high doses are required to allocate clinical effects because of ineffectual delivery of VPA to the brain. Moreover, there are side effects of VPA due to its permanent need. 5 Scism et al. 6 found that the discharge of VPA from the blood–brain barrier was 2.7-fold. VPA distribution in the brain is less than that of other anticonvulsants because it has the ability to combine with plasma protein more than the other antiepileptic drugs and the transport of valproate from the brain to blood is unequal to that from the blood to brain. 7,8
The brain injury resulting from epilepsy could be due to many reasons: genetic factors, disturbances in the intracellular electrolyte, oxidative stress, and increased concentration of cytokines which in turn cause neuronal cell death. 9 Oxidative stress arises as a result of an imbalance between the production of reactive oxygen species (ROS) and the defense system’s ability to remove the reactive intermediates. Oxidative stress has been implicated in the progression of different neurodegenerative diseases as it leads to free radical attack on neural cells. 10
Ahmed et al.
15
evaluated the antioxidant capacity of many antioxidants including vitamin C, N,N′-diphenyl-p-phenylenediamine (DPPD), and LC against testicular oxidative damage induced by cisplatin in rats. They found that LC reduced the testicular damage effect of cisplatin on rats, similar to that of DPPD on lipid peroxidation (LPO), nitric oxide (NO), superoxide anion, and superoxide dismutase (SOD). Moreover, N-acetyl-
Cathepsin A can act as a protective protein, interacting with other enzymes to prevent them from breaking down prematurely. 18 Based on this protective function, this enzyme is also called protective protein/cathepsin A. 19 Most of the studies assessing oxidation-induced changes in lysosomal proteolytic behavior have focused on the analysis of the enzymatic activity of cathepsins, the lysosomal proteases. Oxidizing conditions result in increased or decreased cathepsin activity depending on the cellular conditions. 18 Neurodegeneration is accompanied by pronounced reactive astrocytosis and is preceded by an accumulation of ultrastructurally and biochemically unique lysosomal bodies in large cortical neurons and by axonal enlargements. 20
However, no studies have been found in the literature that has examined the effect of LC supplementation on the improvement of the antioxidant and histological structure of the brain affected by sodium valproate (SV) in male rats. Because there is limited information on the levels of neurotransmitters and signaling molecules, particularly involving cathepsin activity and neuronal NO level, the present study was undertaken to determine the effects of two doses of SV with/without LC on these parameters and redox state as well as malondialdehyde (MDA) levels, SOD, catalase (CAT), and glutathione peroxidase (GPx) activities and the histological changes in the brain tissue of rats.
Materials and methods
Chemicals
SV was purchased from Sigma Chemical Co., St. Louis, Missouri, USA. The test dose of SV (100 and 500 mg/kg body weight) was chosen based on the previous studies of Khan et al. 21 LC was purchased from Sigma Chemical Co., St. Louis, Missouri, USA. It was given in a dose of 100 mg/kg. 15 All other chemicals used in the experiment were of analytical grade.
Experimental animals
The animals were obtained from the animal house of Faculty Veterinary Medicine, Zagazig University. Sixty adult male albino rats weighing approximately 200 g were housed in metal cages, bedded with wood shavings, and kept under standard laboratory conditions of aeration and room temperature of about 25°C with 12/12 light and dark cycle. The animals were allowed free access to standard diet and water ad labium throughout the experimental period. We have followed the European community Directive (86/609/EEC) and national rules on animal care that was carried out in accordance with the National Institutes of Health (NIH) Guidelines for the Care and Use of Laboratory Animals, Eighth edition. The animals were accommodated to the laboratory conditions for 2 weeks before being experimented, one group served as control and five as treatment groups.
Experimental design
The animals were divided into six groups (n = 10) as follows: control group 1 was treated with saline as vehicle, groups 2 and 3 were treated with low and high doses of SV (100 and 500 mg/kg, respectively) according to Khan et al., 21 group 4 was treated with LC (100 mg/kg), 15 and groups 5 and 6 were treated concomitantly with low-dose SV + LC and high-dose SV + LC, respectively. All the groups were treated orally by gastric tube for 30 successive days.
Organosomatic index (OSI) of brain
The body weight of each animal was noted before treatment and also on day 30. After the rats were euthanized, the brain weight of each animal was also recorded. From these values, OSI of the brain was calculated according to the following formula: 22
Preparation of brain homogenate
The brain tissues were cut coronally to get the cerebral cortex. The brain samples were divided into two sets; the first set was used in the preparation of brain homogenates. The samples of brain cerebral cortex were used in the analysis of oxidative stress and antioxidant parameters. The brain cerebral cortex was homogenized in 5 mL of cold buffer per gram tissue and centrifuged at 5000 r/min for 30 min. The resulting supernatant was transferred into Eppendorf tubes and preserved in a deep freezer until used for various antioxidant assays. The second set was used for histological examination.
Determination of oxidative biomarkers
LPO level
The extent of LPO was estimated in the cerebral cortex of brain tissues as the concentration of thiobarbituric acid reactive product MDA by using the method of Ohkawa et al. 23 MDA concentrations were determined using 1,1,3,3-tetraethoxypropane as standard and expressed as µmol/g tissue.
NO level
NO level in the cerebral cortex of brain tissue of rat was measured. 24 The 5% tissue homogenate was made in 0.25 M sucrose solution and centrifuged at 6000 r/min at 4°C for 10 min to obtain a clear free supernatant. An equal volume of 0.25 M sucrose solution, 1% sulfanilamide, and 0.1% naphthyl ethylenediamine hydrochloride was then added to the supernatant and mixed well. After 20 min of incubation, the optical density was measured spectrophotometrically at 550 nm.
Tissue-free hydroxyl radical (–OH) production
For free hydroxyl radical estimation, the animals of each group were treated with 30% dimethyl sulfoxide at a dose of 0.4 mL per 100 g body weight for 2 h before sacrifice. After sacrifice, a 5% homogenate (in distilled water) of brain tissue was prepared and used to determine the free hydroxyl radical formation. 25 The homogenate was treated with 10 N H2SO4 to allow precipitation of protein. The protein-free filtrate was then taken for extraction of methane sulfonic acid, which was produced from dimethyl sulfoxide by the action of free hydroxyl radicals. Fast blue 4′-Amino-2′,5′-diethoxybenzanilide (BB) salt was used for the production of a yellow-colored product by reaction with methane sulfonic acid, which was measured at 425 nm spectrophotometrically.
Determination of enzymatic antioxidant biomarkers
SOD activity
SOD activity was measured 26 in the cerebral cortex of the brain tissues by assaying the autoxidation of pyrogallol at 440 nm for 3 min. One unit of SOD activity was calculated as the amount of protein that caused the inhibition of 50% pyrogallol autoxidation. A blank without homogenate was used as a control for nonenzymatic oxidation of pyrogallol in Tris–ethylenediaminetetraacetic acid (EDTA) buffer (50 Mm Tris, 10 mM EDTA, pH 8.2). The SOD activity is expressed as U/mg protein.
CAT activity
CAT activity was measured 27 in the brain tissues by assaying the hydrolysis of H2O2 and the resulting decrease in absorbance at 240 nm over a 3 min period at 25°C. Before determining the CAT activity, samples were diluted 1:9 with 1% (v/v) Triton X-100. CAT activity is expressed as mmol/mg protein.
GPx activity
GPx activity was determined 28 in the brain tissues. The peroxide substrate (ROOH), glutathione reductase (GRx), and nicotinamide adenine dinucleotide phosphate (NADPH) are included in the reaction mixture. The formation of the oxidized form of glutathione (GSSG) that is catalyzed by GPx is coupled to the recycling of GSSG back to the reduced form of GSH using GRx. NADPH is oxidized to NADP+. The change in A 340 due to NADPH oxidation is monitored and is indicative of GPx activity. The activity of GPx was expressed in terms of mmol GSH consumed/min/g wet weight tissue.
GRx activity
GRX activity was determined 29 in the brain tissues. The enzymatic reaction proceeded at a rate directly proportional to GSH concentration. The unit of GRx activity was defined as an increase in the log GSH of 0.001 per min.
Total antioxidant level
Total antioxidant capacity (TAC) was determined in the cerebral cortex of the brain tissues using ferric reducing antioxidant power assay according to the method of Prieto et al. 30 ferric reducing antioxidant power (FRAP) reagent (300 mM acetate buffer, pH 3.6, 10 mM 2,4,6-tri(Z-pyridyl)-S-(triazine, 99%) in 40 mM HCl and 20 mM FeCl3.6H2O in the ratio of 10:1:1) was prepared. To 50 µL of brain homogenate, 1.5 mL of FRAP reagent was added and incubated at 37°C for exactly 5 min. The change in absorbance was measured at 593 nm due to the formation of a blue-colored FeII-tripyridyl triazine complex from the colorless oxidized FeIII form by the action of electron-donating antioxidants. The absorbance of the sample was read against a reagent blank (1.5 mL FRAP reagent and 50 µL distilled water) at 593 nm.
Cathepsin activity
Cathepsin activity of rat brain was measured 31 in the brain tissues. The 5% tissue homogenate was incubated with 4% hemoglobin substrate at 37°C for 60 min. The reaction was stopped by addition of 8.0% trichloroacetic acid (TCA). In a similar way, a buffer blank was also prepared in which TCA was added before addition of tissue homogenate and the substrate. All the tubes were centrifuged to get the supernatant, and readings were taken in a UV–vis spectrophotometer at 280 nm wavelength. The enzyme activity was expressed in terms of tyrosine per minute per mg protein.
Histological evaluation
For histological examination, the second portion of cerebral cortex was fixed in 10% neutral buffered formalin, sectioned, and stained with hematoxylin and eosin. 32 The sections were examined with a light microscope and photographed. Histological activity index (HAI) was assessed based on the degree of microscopic lesions (oval nuclei and glial cells, glial fibers, atrophic neurons, pyknotic nuclei, and congestion with a mild area of hemorrhage).
Statistical analysis
Data are expressed as mean values ± standard error (n = 6). Statistical analysis was performed using one-way analysis of variance to assess significant differences among treatment groups. For each significant effect of treatment, the post hoc Tukey’s test was used for comparisons. The criterion for statistical significance was set at p < 0.05. All statistical analyses were performed using SPSS statistical version 20 software package (SPSS Inc., USA). Data of histological analysis were based on the sum of the scores histological criteria (overall score) using Kruskal-Wallis test in SPSS (n = 10).
Results
The results of OSI of brains of rats administered SV in different doses are presented in Figure 1. The mean brain weight of rats administered the two doses of SV was significantly (p < 0.01) reduced than in rats administered saline. There was no significant difference in OSI of the brain of rats administered LC. The coadministration of VS and LC did not change the OSI of the brain as compared to its related treated group with SV only.
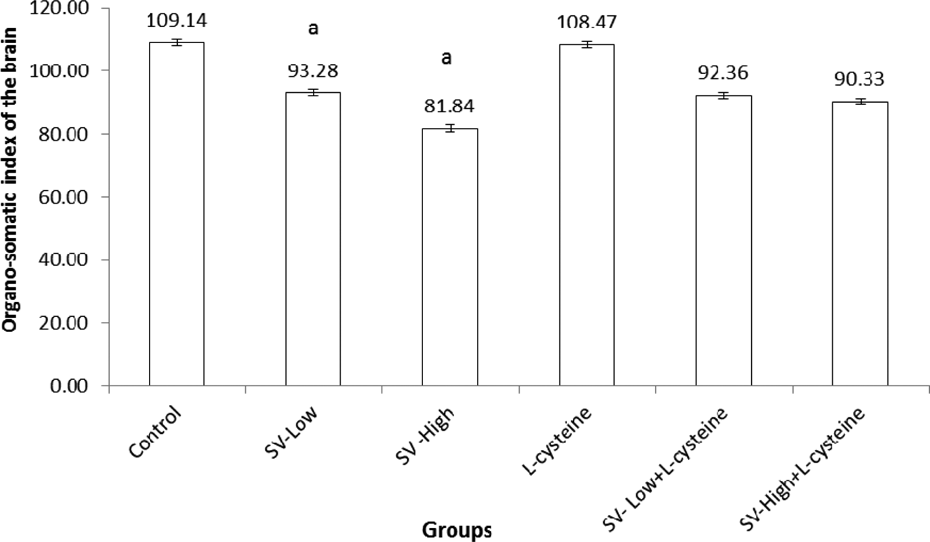
Effect of
Our finding showed that the levels of NO of brain tissue in SV-treated groups had a tendency to increase significantly as compared to the control animals (Figure 2); however, it significantly decreased by using LC with SV administration. Also, our data showed that levels of NO of brain tissue in the group that received LC only had decreased compared to the control group (Figure 2).

Effect of
Hydroxyl radical levels of brain tissue in SV groups were significantly higher by increasing the dose of SV than control rats, but it significantly decreased by using LC with different doses of SV (Figure 2).
In chronic SV treatment, significant alterations in the activity of cathepsin A were observed by increasing the dose of SV. However, on combining the SV with LC, the activity of cathepsin was improved when compared with SV-High (Figure 2).
In the SV-Low and SV-High groups, LPO levels were increased significantly by 2.0- and 2.5-fold, respectively, as compared to the control (Table 1). Treatment of SV groups with LC decreased the LPO by 18.8% and 16.7% for SV-Low + LC and SV-High + LC as compared to its related group of SV only, respectively.
Effect of

LPO: lipid peroxidation; SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase; GRx: glutathione reductase; TAC: total antioxidant capacity. Values are expressed as means ± SE; n = 10 for each treatment group.
aSignificant difference as compared to control.
bSignificant difference as compared to SV-Low.
cSignificant difference as compared to SV-High.
dSignificant difference as compared to SV-Low + LC.
In the SV groups, all the enzymatic antioxidants and TAC were decreased in a dose-dependent manner of SV; these antioxidant responses were reversed significantly in coadministration of SV and LC (Table 1).
Cross section of rat brain of control group showing neuron bodies with rounded to oval nuclei and glial cells in the fibrillary background showed the following normal layers: outer molecular (I), external granular (II), external pyramidal (III), internal granular (IV), internal pyramidal (V), and polymorphic (VI; Figure 3A). Pyramidal neurons with rounded to oval nuclei are shown in Figure 3A. No morphological abnormalities were observed in control rats. Normal cerebral cortex of brain tissues formed of round and pyramidal-shaped neurons surrounded by eosinophilic glial fibers were also observed in rats treated with LC (100 mg/kg; Figure 3B).

(a) Cross section of cerebral cortex of rat brain in control group showing the following normal layers: outer molecular (I), external granular (II), external pyramidal (III), internal granular (IV), internal pyramidal (V), and polymorphic (VI). Pyramidal neuron with rounded to oval nuclei (NCB, blue arrow) and different types of glial cells with different shaped nuclei (GC, red arrow) are observed in the fibrillary background. Normal blood vessel with perivascular area (blue arrow; H&E, ×200). Pyramidal layer with appearance of normal perivascular neurons and appearance of normal blood vessel with normal width (yellow arrow). (b) Cross section of rat brain (cerebral cortex) of the group treated with
Atrophic brain tissues of the cerebral cortex with few atrophic neurons with pyknotic nuclei and surrounded by excessive glial tissue with cerebral cortex layers are shown in Figure 3C. All cerebral cortex layers of the brain are showing dilated and congested vascular vessel. In the molecular layer and granular layer, there are dilated congested vessels. However, the third layer (III) shows deeply stained atrophic neurons with pyknotic nuclei. All the previous observation resulted when the animals treated with SV-Low dose (Figure 3C). SV-High dose (500 mg/kg) caused congestion with a mild area of hemorrhage between the compartments of brain tissues with more atrophic neurons in the congested area (Figure 3D). Treatment of the rats with SV-Low dose and LC showed the normal appearance of sulci of cerebral cortex of brain tissues with very mild hemorrhage in between the compartments of brain tissues with severe congested blood vessel (Figure 3E). The combination of SV-High dose with LC improved the structure of brain tissues and fragment separated by very mild congested area (hematoxylin and eosin, ×200).
HAI in cerebral cortex hemisphere was assessed based on the degree of microscopic lesions (oval nuclei and glial cells, glial fibers, atrophic neurons, pyknotic nuclei, and congestion with a mild area of hemorrhage), as shown in Table 2.
Histological activity index (HAI) was assessed based on the degree of microscopic lesions in cerebral cortex brain tissue as the effect of sodium valproate and

−−−−−− Indicates absence of the change in the animals of the studied group. ++++ A change which was often found in all the studied animals of a group. +++− A change which was observed in almost all the studied animals of a group. ++−− A change not so often observed in all animals of a group. −−−+ A change which was rare within a group (n = 10).
Discussion
Antioxidant therapies have received considerable attention in the treatment of epilepsy. 33 The potential role of oxidative stress in injury associated with SV suggests that antioxidants LC may enhance the efficacy of the treatment protocol designed to mitigate SV-induced toxicity. The present study, as far as we are aware, is the first to report altered levels of NO, OH−, and activity of cathepsin in rat brain following SV treatment. Moreover, no studies have been found in the literature that has examined the effect of LC on antioxidant and histological structure of the rat brain treated with SV.
The significant decrease in OSI of the brain was observed in SV-treated rats in this study. OSI can be described as the ratios of organs to body weight; measured organ in relation to body mass can be directly linked to toxic effects of the chemical on the target organ.
The present study also revealed alterations in the levels of antioxidants in brain tissue as a response to the two doses of SV. The decrease in antioxidant enzymes, SOD, CAT, GPx, and GRx, accompanied by an increase in the prooxidative markers LPO, NO, and OH− as well as decreasing the cathepsin activity in the SV-treated rats is suggestive of oxyradical release. The previous studies carried out on cytotoxic activity of valproate are the result of the generation of hydrogen peroxide and the production of highly reactive hydroxyl radicals. 34 This could lead to the increased levels of thiobarbituric acid reactive substances (TBARS) and hydroperoxides and decreased levels of enzymatic antioxidants (SOD, CAT, and GPx) and TAC. 35 Further, SV treatment through a process of free radical damage causes functional damage to the liver. 36,37
The results obtained in the present study, regarding the LPO level, are in agreement with the previous studies which reported that LPO increased and TAC decreased significantly in the hippocampus homogenate of epileptic rats 38 and in patients. 39 This may be explained by the fact that epilepsy increased the content of ROS and superoxide generation in the brain. Free radical generation can induce seizure activity by direct inactivation of glutamine synthase, thereby permitting an abnormal buildup of excitatory neurotransmitter glutamic acid. 40 The convulsions in animals are correlated with a decrease in the cerebral content of neurotransmitter gamma-aminobutyric acid due to the inhibition of the enzyme glutamate decarboxylase by ROS. 41
Increasing LPO levels in the liver as the effect of SV in rats 35,37 in coincidence with the present results suggest that SV enhances oxidative stress in the brain, thereby disturbing the antioxidant defense. Therefore, increased oxidative stress could be one of the mediating factors in the pathogenesis of SV toxicity in the brain as demonstrated here with a dose-dependent effect of SV on HAI of brain lesions.
LC in the present study offers protection against oxidative damage of SV by participating in the nonenzymatic oxidative decarboxylation in the hydrogen peroxide decomposition process. Reports have also shown that LC is a precursor for GSH formation. 11,13 Its maintenance of free sulfhydryl groups is considered crucial to the biological functions of many proteins. 14
It is known that the ammonia-induced inhibition of antioxidant enzymes is mediated by the activation of NO synthase and the formation of NO, which inhibits the activities of antioxidant enzymes. 42 SV increased the level of NO and this could be the reason for the decrease in the activities of antioxidant enzymes in the current study.
There is no study on the effect of SV on cathepsin in experimental rats. The process of neurodegeneration in epilepsy can result from the accumulation of the cell debris, 44 which could be removed by cathepsin D. The deficiency of cathepsin D in mice caused neurodegeneration and accumulation of lipofuscin pigments in brains. 43,44 Furthermore, the elevation of cathepsin A and carboxypeptidase in neuropathological conditions has been reported. 45 One of the previous studies indicated that the induction of oxidative stress stimulated protein degradation in skeletal muscle by changing cathepsins, calpains, and the ubiquitin–proteasome system. 46 In the present study, LC improved the activity of cathepsin A in SV-treated animals that are parallel with the histological finding and enhancement of antioxidant parameters.
Brain tissues of rats treated with SV showed marked astrocyte swelling, damaged glial cells, pyknotic and shrunken neurons, inflammation, and severe extracellular edema. These changes may be linked to oxidative stress occurring following SV, leading to the abnormal morphology of astrocytes and neurons. Similar histopathological findings have been reported previously. 47 NAC supplementation to hepatic encephalopathy rats was able to reverse the structural deficits as evident from reduced astrocyte swelling and improved tissue morphology. 48 Cerebellum sections also showed signs of abnormal morphology following bile duct ligation, whereas on NAC supplementation animals showed reduced inflammation, edema, and improvement in structural deficits.
In conclusion, these results suggest that the SV drugs enhanced hydroxyl radical (OH.) production and effect on the cerebral cortex of brain tissues and offer a possible mechanism of cellular toxicity by this compound. Moreover, the data clearly demonstrate that the protective effect of LC in brain tissue of rat is mediated through attenuation of oxidative stress, suggesting a therapeutic role for LC in the side effect of epilepsy, when treated with SV.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

