Abstract
The widest distribution and the highest uptake of methamphetamine (MA) in the human body occurred in the lungs, so that more and more attention should be paid to MA-induced pulmonary toxicity. MA induces the release of serotonin, which is an important mediator in pulmonary disease. The purpose of this study is to investigate the chronic response of the lung to MA and its potential mechanism in rats. Models of the chronic toxicity of MA were established with MA of 5 mg/kg and 10 mg/kg (intraperitoneally, twice per day) for 5 weeks. It was found that the high dose of MA induced rat pulmonary toxicity: crowded lung parenchyma, thickened septum, reduced number of alveolar sacs, inflammatory cell infiltration, and pulmonary arteriolar remodeling. In addition, MA resulted in a significant increase in the lung serotonin concentration and the marked upregulation of tryptophan hydroxylase 1, vesicular monoamine transporter 2, serotonin transporter, and downregulation of monoamine oxidase-A. These findings suggest that MA induced chronic pulmonary toxicity, which is concerned with the elevated serotonin concentration in rat lungs by increased synthesis, reduced metabolism, augmented accumulation, and promoted release of serotonin.
Keywords
Introduction
Methamphetamine (MA) is a highly addictive and toxic drug of abuse. 1 There is a growing body of evidence to show that MA abusers suffer from a variety of deficits in nervous and cardiovascular system: in brain, these include psychoses and memory deficits; in heart, these include coronary heart disease and cardiomyopathy. 2 These changes are associated with MA facilitating release and stopping the metabolism and reuptake of dopamine (DA), noradrenaline (NA), and serotonin. 3 Especially, increasing evidence of MA-induced neurotoxicity was focused on serotonin neurotoxin. 4
Serotonin is catalyzed by tryptophan hydroxylase (TPH) for biosynthesis and subsequently transported by vesicular monoamine transporters (VMATs) into storage vesicles for subsequent exocytotic release by serotonin transporter (SERT) into synaptic cleft. 5,6 Accumulated intracellular serotonin is metabolically degraded into 5-hydroxyindole acetic acid by monoamine oxidase (MAO). 6,7 Serotonergic dysfunction ultimately resulted in the imbalance of serotonin, which was involved in MA-induced neurotoxicity. 8
Except the central nervous system diseases, serotonin is also associated with many peripheral vascular diseases, including atherosclerosis and hypertension. 9 Pulmonary arterial hypertension (PAH) is mainly caused by the pulmonary functional and structural changes: sustained pulmonary vasoconstriction, excessive pulmonary vascular remodeling, and perivascular inflammation. 10 Remodeling and inflammation are the hallmark pathological changes in PAH. 11 Serotonin exerts mitogenic and comitogenic effects on pulmonary artery endothelial cells (PAECs) and pulmonary artery smooth muscle cells (PASMCs). 12 Pulmonary vascular remodeling occurs through serotonin uptake by the SERT, internalization of serotonin and subsequent stimulation of superoxide anion, and increased tyrosine phosphorylation of guanosine triphosphatase-activating protein. 13 Serotonin as an inflammatory mediator induced pulmonary inflammation associated with SERT-induced rapid activation of extracellular signal-regulated kinase 1/2. 14 Our previous studies demonstrated that serotonin played an important role in the development of PAH. 15 –19
Epidemiological study showed that MA abuse significantly increased the risk of developing PAH. 20 An acute inhalation exposure to the vaporized MA causes lung injury in mice. 21 Clinically, chest pain and dyspnea can be caused within seconds by smoking MA, which is associated with pulmonary edema. 21,22 Pharmacokinetics data showed that the widest distribution and the highest uptake of MA in the human body occurred in the lungs. 2 Taken these together, we proposed the notion that more and more attention should be paid to the pathogenesis of MA-induced pulmonary toxicity. Pathologically, pulmonary toxicity is characterized by parenchymal damage, recruitment of inflammatory cells, and progression of the inflammatory process, 23 which are the partial pathological changes in PAH. But there remains the question about the pathogenesis of chronic pulmonary toxicity induced by MA. Serotonin has been used as a tool to induce pulmonary diseases for many years. 24 Therefore, the purpose of this study is to investigate the chronic response of the lung to MA and whether serotonin mechanism is involved in this process after long-term exposure of rats to MA.
Materials and methods
Animal study
A total of 45 male Wistar rats (180 ± 10 g) from the Animal Resource Center, China Medical University (certificate number: Liaoning 034) were divided into three groups: control group, 5 mg/kg MA group (M5), and 10 mg/kg MA group (M10). The rats in MA-treated groups were intraperitoneally (i.p.) injected with 5 mg/kg and 10 mg/kg of MA (China Criminal Police University, China), respectively (twice per day for 5 weeks). The rats in the control group were administrated with an equivalent volume of 0.9% saline (i.p.) everyday. All the rats were housed in a controlled humidity (50–70%) and temperature (18–22°C) room and were given free access to food and water ad libitum in an alternating 12-h light/dark cycle over a period of 5 weeks. All experimental protocols for the study were approved by the Institutional Animal Care and Use Committee of China Medical University (Heping, Shenyang, China).
Hemodynamic measurement and tissue collection
Rats in all the groups were anesthetized by 3% pentobarbital sodium (40 mg/kg; i.p.). The polyvinyl (PV-1) tube and the polyethylene (PE-50) tube were filled with saline containing 1% heparin before catheterization The method of hemodynamic measurement using PV-1 tubes to measure pulmonary arterial pressure (PAP) and PE-50 tubes to measure systemic arterial pressure (SAP) was carried out as described previously. 15 SAP and PAP were recorded by Polygraph System (RM6000, Nihon Kohden, Tokyo, Japan).
Of the 45 rats, 15 rats (five per group) were perfused with paraformaldehyde. The right lower lung tissues were dissected and then they were paraformaldehyde-fixed and paraffin-embedded. The lung sections (4 μm thick) were prepared for hematoxylin–eosin (H&E) staining and immunohistochemical (IHC) staining.
The other rats were then killed by overdose pentobarbital sodium. The rat lungs were quickly dissected and removed on ice and stored at −80°C until further use for enzyme-linked immunosorbent assay (ELISA) and Western blotting. The hearts were dissected and taken out. Right ventricle (RV) and left ventricle plus interventricular septum (LV + S) were dissected and weighed separately to evaluate the magnitude of the right ventricular hypertrophy expressed as right ventricular index (RVI) by the following equation
Morphological analysis
Lung sections (4 μm thick) were stained with H&E to watch and analyze under light microscopy (Metamorphy/BX41, UIC/Olympus, New York/Tokyo, USA/Japan). The changes in rat lung parenchyma were evaluated by the number of alveolar sacs (three randomly selected sites were analyzed in each section, 200×). The remodeling of pulmonary arterioles (external diameter < 100μm, 400×) was calculated as the percentage of medial wall thickness (MT%) and the percentage of wall area (WA%) by the following equation
Enzyme-linked immunosorbent assay
Samples of lung tissue from rats in each group were homogenized using a Polytron homogenizer (Kinematical, Lucerne, Switzerland) to extract protein. The homogenate was centrifuged at 15,000g for 30 min at 4°C, and the supernatant was collected and stored at −80°C for ELISA. Serotonin concentration in the lung tissues was measured using rat serotonin ELISA Kits (R&D Systems Inc., Minneapolis, Minnesota, USA), according to the manufacturer’s instructions. The limit of detection of the assay for serotonin protein was 0.02 ng/L.
Immunohistochemistry
After the tissues had been processed and embedded in paraffin, lung sections were cut and then stained immunohistochemically using Ultrasensitive S-P and diaminobenzidine staining kits (Maxin-Bio Co., Fuzhou, China). IHC staining followed a basic indirect protocol using a citrate antigen-retrieval method. The primary rabbit polyclonal antibodies against VMAT-2 (Santa Cruz Biotechnology, Inc., California, USA) and SERT (Beijing Biosynthesis Biotechnology Co., Ltd., Beijing, China) were diluted 1:200, respectively. As a negative control, samples were incubated with 0.01 mol/L of phosphate-buffered saline (PBS) instead of the primary antibody. Digital images were analyzed under a BX51/MetaMorphy microscope (Olympus). The VMAT-2 and SERT protein content was calculated as average optical density.
Western blotting
Protein extraction buffer was added to the frozen lung tissues and the sample was completely homogenized on ice, and the protein concentration was determined using ultra spectrophotometer. Electrophoresis was carried out in a 10% sodium dodecyl sulfate–polyacrylamide gel using 50 µg of total protein in each lane. After electrophoresis, proteins were transferred to a polyvinylidene difluoride membrane (Bio-Rad, Hercules, USA). After the transfer, the membranes were incubated in blocking buffer for 1 h (5% nonfat dry milk, PBS, and 0.1% Tween). The membranes were probed with primary rabbit polyclone anti-TPH1 (1:200), anti-SERT (1:200), anti-MAO-A (1:200) (Beijing Biosynthesis Biotechnology Co., Ltd.), anti-VMAT-2 (1:400), and mouse monoclonal anti-β-actin (1:3000) (Santa Cruz Biotechnology, Inc.) overnight at 4°C. After washing with PBS–0.1% Tween, the membranes were incubated in the presence of goat anti-rabbit secondary antibody for TPH1, VMAT-2, SERT, and MAO-A at a dilution of 1:2000 for 2 h at room temperature, and rabbit anti-mouse secondary antibody for β-actin at a dilution of 1:5000 for 2 h at room temperature, followed by enhanced chemiluminescence. The relative protein expression was quantified by densitometry using image quant software (Molecular Dynamics, Boston, USA). The result of the protein expression of TPH1, MAO-A, VMAT-2, and SERT was represented by the relative yield to the β-actin.
Data analysis
All the data are presented as means ± SD. Statistical analysis was performed by one-way analysis of variance, followed by Fisher’s least significant difference post hoc multiple comparison with SPSS 13.0 software. Values of p < 0.05 were considered statistically significant.
Results
Effects of MA on weight and hemodynamic indexes in rats
The weight of rats was increased in all the groups, but the percentage of weight changes was different between the groups after 5 weeks. The percentage of weight change was significantly lower in M10 group than in control group (p < 0.05, M10 vs. control; Table 1).
Effects of MA on the rat weight in different groupsa

M5: 5 mg/kg methamphetamine; M10: 10 mg/kg methamphetamine; MA: methamphetamine.
aData are means ± SD.
b p < 0.05, M10 vs. control group.
MA has no significant effect on heart rate and SAP. In M10 group, the PAP and RVI were subtly increased, but there was no statistical significance between the groups (Table 2).
Effects of MA on HR, mSAP, mPAP, and RVI in different groupsa
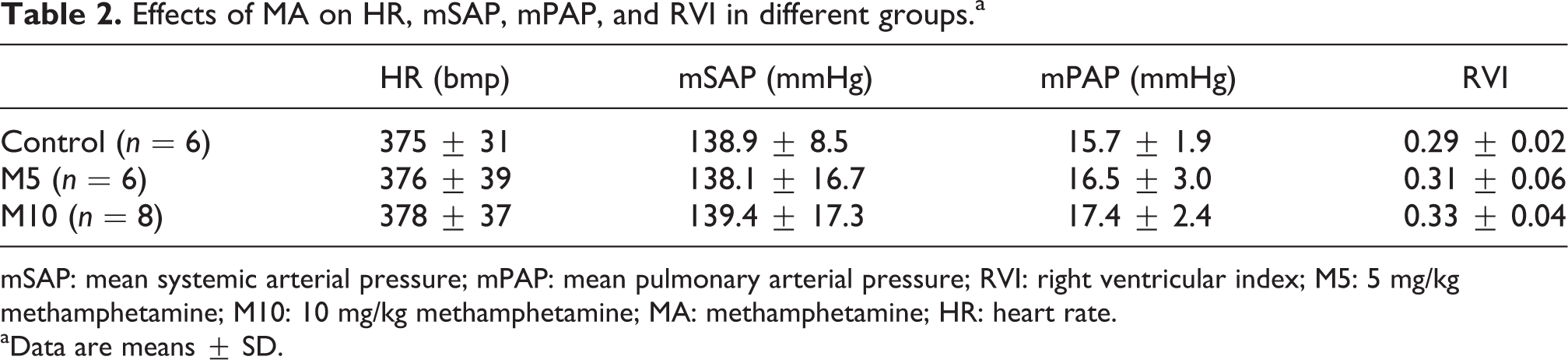
mSAP: mean systemic arterial pressure; mPAP: mean pulmonary arterial pressure; RVI: right ventricular index; M5: 5 mg/kg methamphetamine; M10: 10 mg/kg methamphetamine; MA: methamphetamine; HR: heart rate.
aData are means ± SD.
MA-induced changes in lung parenchyma
Representative H&E sections from control, M5, and M10 groups indicate that 5 mg/kg MA caused subtle lung injury (Figure 1(b)), but 10 mg/kg MA induced detectable lung injury (Figure 1(c)) when compared with rat lung tissue in the control group (Figure 1(a)). Microscopically the lung tissue of rat was infiltrated by the inflammatory cells, and the lung parenchyma was more compact with the thickened septum in M10 group than that in the control group. In addition, the number of alveolar sacs was substantially lower in M10 group than that in the control group (p < 0.05, M10 vs. control; Figure 1(d)).

MA-induced changes in lung parenchyma in different groups. H&E staining (magnification, ×200) illustrated that more compact lung parenchyma with thickened septum and remarkably reduced number of alveolar sacs were seen in the M10 group (c), compared with the control group (a) and the M5 group (b). (d) The number of alveolar sacs in different groups. Data are presented as means ± SD. n = 5. *p < 0.05, M10 vs. control. M5: 5 mg/kg methamphetamine; M10: 10 mg/kg methamphetamine; MA: methamphetamine; H&E: hematoxylin–eosin.
Assessment of pulmonary arteriolar remodeling
Medial hypertrophic and hyperplastic changes in the pulmonary arterioles (external diameter < 100 μm) were prominent in the M10 group by H&E staining (Figure 2). But at 5 mg/kg, MA had no significant effect on the morphological changes in the pulmonary arterioles compared with the control group. The MT% increased from 14.2 ± 4.6% in the control group to 37.5 ± 8.2% in the M10 group (p < 0.05, M10 vs. control). The WA% increased markedly from 29.4 ± 5.6% in the control group to 52.7 ± 9.7% in the M10 group (p < 0.05, M10 vs. control).

Pulmonary arteriolar remodeling in different groups. The pulmonary arterioles (diameter < 100 μm) are illustrated by H&E staining (magnification, ×400) from (a) control group, (b) M5 group, and (c) M10 group. (d) Comparison of the medial wall thickness percentage and the wall area percentage in different groups. Data are presented as means ± SD. n = 5. **p < 0.01, M10 vs. control; # p < 0.05, ## p < 0.01, control vs. M5. M5: 5 mg/kg methamphetamine; M10: 10 mg/kg methamphetamine; H&E: hematoxylin–eosin.
Serotonin concentration in rat lungs
To evaluate the role of serotonin in MA-induced lung injury, we measured lung serotonin concentration in different groups using ELISA (Figure 3). In the control group, lung serotonin concentration was 173.7 ± 26.8 ng/L. In M5 group, lung serotonin concentration was slightly increased to 202.3 ± 13.3 ng/L. But compared with the control group, lung serotonin concentration in M10 group was significantly elevated to 260.2 ± 67.6 ng/L (p < 0.05, M10 vs. control).

Lung serotonin concentration in different groups. Data are the mean ± SD (n = 6). *p < 0.05, M10 vs. control. M5: 5 mg/kg methamphetamine; M10: 10 mg/kg methamphetamine.
Western blot analysis of TPH1 expression in rat lungs
Western blotting analysis revealed that the TPH1 protein expression was significantly upregulated in the lungs of rats in the M5 and M10 groups when compared with the expression in the control group (0.78 ± 0.34 vs. 0.34 ± 0.10 and 0.82 ± 0.28 vs. 0.34 ± 0.10, respectively; p < 0.05; Figure 4).

Western blot analysis of TPH1 expression in the rat lungs in different groups. Data are the mean ± SD (n = 5). *p < 0.05, M10 vs. control; # p < 0.05, control vs. M5. M5: 5 mg/kg methamphetamine; M10: 10 mg/kg methamphetamine; TPH1: tryptophan hydroxylase 1.
Western blot analysis of MAO-A expression in rat lungs
Western blotting analysis revealed that the MAO-A expression was not markedly changed in the M5 group in the rat lungs when compared with the control group. Yet the MAO-A protein expression in the lungs of rats was significantly downregulated in the M10 group when compared with the expression in the control group (0.49 ± 0.19 vs. 0.86 ± 0.26, p < 0.05, and M10 vs. control; Figure 5).

Western blot analysis of MAO-A expression in the rat lungs in different groups. Data are the mean ± SD (n = 5). *p < 0.05, M10 vs. control; # p < 0.05, control vs. M5. M5: 5 mg/kg methamphetamine; M10: 10 mg/kg methamphetamine; MAO-A: monoamine oxidase-A.
Effect of MA on VMAT-2 protein expression
IHC analysis showed that VMAT-2 protein was predominantly expressed in the endothelium and in the media of the pulmonary arteries in rats. Compared with that in the control group, VMAT-2 protein expression in the M5 group was not significantly increased, but that was markedly increased in the M10 group (p < 0.05, M10 vs. control; Figure 6).

Immunohistochemical determination of VMAT-2 immunoreactivity in different groups. Cross-sections were stained with rabbit anti-VMAT-2 (brown) and counterstained with hematoxylin (blue) in different groups (magnification, ×400). VMAT-2 protein expression was illustrated by optical density average. Data are means ± SD. n = 5. *p < 0.05, M10 vs. control; M5: 5 mg/kg methamphetamine; M10: 10 mg/kg methamphetamine; VMAT-2: vesicular monoamine transporter 2.
Results from Western blot analysis showed that VMAT-2 protein expression was significantly increased in the M10 group (p < 0.05, M10 vs. control), yet VMAT-2 expression was not markedly changed in the M5 group in the rat lungs (Figure 7). The findings in the rat lungs were coincident with the results from IHC analysis of VMAT-2 in the pulmonary arteries of the rats.

Western blot analysis of VMAT-2 expression in rat lungs in different groups. Data are means ± SD. n = 5. *p < 0.05, M10 vs. control; M5: 5 mg/kg methamphetamine; M10: 10 mg/kg methamphetamine; VMAT-2: vesicular monoamine transporter 2.
Effect of MA on SERT protein expression
IHC analysis showed that SERT protein was predominantly expressed in the endothelium and in the media of the pulmonary arteries in rats. Compared with that in the control group, SERT protein expression in the M5 group was increased, but there was no significance between them. Yet SERT protein expression was markedly increased in the M10 group (p < 0.05, M10 vs. control; Figure 8).
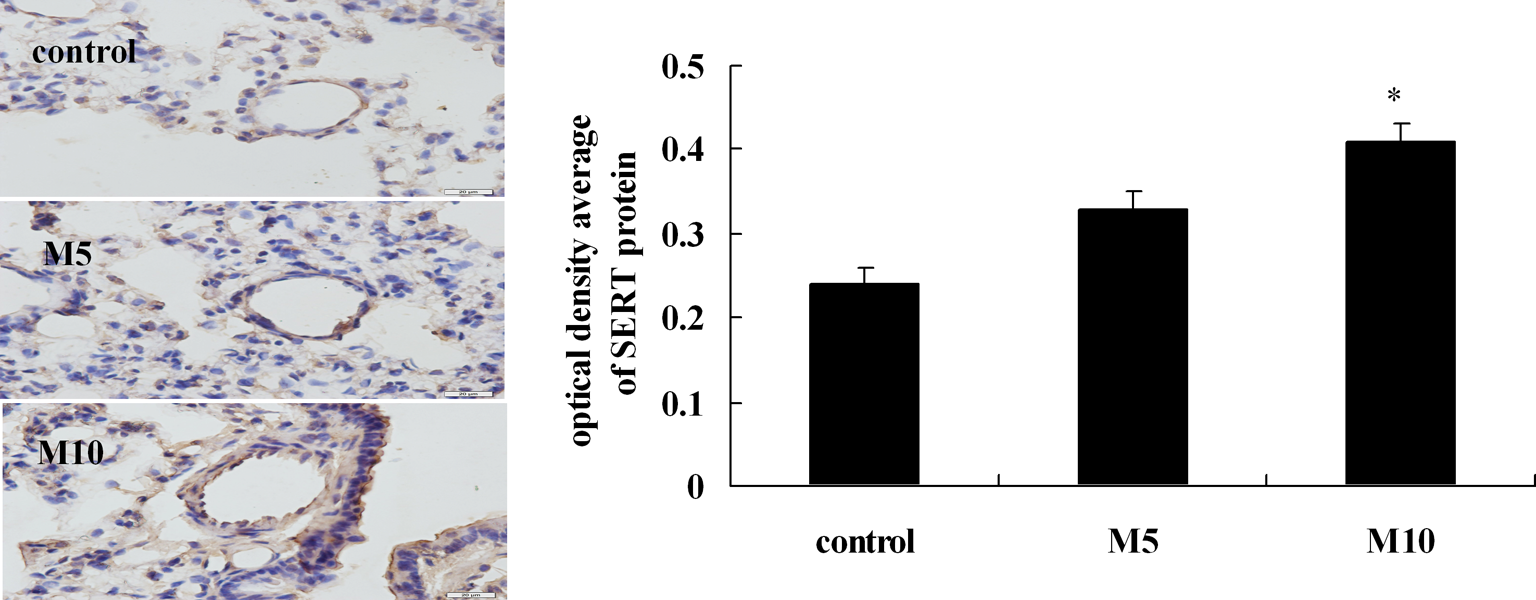
Immunohistochemical determination of SERT immunoreactivity in different groups. Cross-sections were stained with rabbit anti-SERT (brown) and counterstained with hematoxylin (blue) in different groups (magnification, ×400). SERT protein expression was illustrated by optical density average. Data are means ± SD. n = 5. *p < 0.05 M10 vs. control. M5: 5 mg/kg methamphetamine; M10: 10 mg/kg methamphetamine; SERT: serotonin transporter.
Results from Western blot analysis showed that SERT protein expression was significantly increased in the M10 group (p < 0.05, M10 vs. control); yet SERT expression was not markedly changed in the M5 group in the rat lungs (Figure 9). The findings in the rat lungs were similar with the results from IHC analysis of SERT in the pulmonary arteries of the rats.

Western blot analysis of SERT expression in rat lungs in different groups. Data are means ± SD. n = 5. *p < 0.05 M10 vs. control. M5: 5 mg/kg methamphetamine; M10: 10 mg/kg methamphetamine; SERT: serotonin transporter.
Discussion
This study focused on the chronic response of the lung to long-term exposure of rats to MA. Results from our study indicated that the high dose of MA induced obvious pulmonary toxicity, which showed that the rat lung tissue was infiltrated by the inflammatory cells, that the lung parenchyma was more crowded with thickened septum, that the number of alveolar sacs was substantially reduced, and that pulmonary arterioles were induced markedly hypertrophic or hyperplastic changes in the medial walls. High dose of MA resulted in the significant increase in serotonin concentration in the lung tissue and the marked upregulation of TPH1, VMAT-2, and SERT and downregulation of MAO-A. These findings suggest that MA-induced chronic pulmonary toxicity is associated with the high serotonin level in the rat lungs by the increased synthesis, reduced metabolism, augmented accumulation, and promoted release of serotonin.
Circulating serotonin is produced mainly in the enterochromaffin cells of the intestine and is stored within the platelet. 25 Serotonin storage in platelet was released from pulmonary neuroendocrine cells and neuroepithelial bodies distributed throughout the airways. 26 Increased platelet consumption by the lung enhanced plasma concentration of free serotonin. 27 Evidence that the elevation in plasma serotonin has been implicated in the pathogenesis of pulmonary disease and has been accumulating for decades. 28 –30 Serotonin is a mitogen for a wide variety of cell types, including rat and human pulmonary endothelial cells, smooth muscle cells, and myofibroblasts. 29 Serotonin plays a role in both the proliferative and functional components of the pathogenesis of pulmonary hypertension. 27 Elevated serotonin levels in plasma enhance the risk of pulmonary toxicity. 31 But the present findings showed that the serotonin level in the rat lungs was also increased significantly after long-term and high dose of MA, and that the pulmonary toxicity was obviously appeared: crowded lung parenchyma, thickened septum, reduced number of alveoli, and pulmonary arteriolar remodeling. Alteration of the local 5-hydroxytryptamine concentration could influence the functions of 5-HT. 9 Thus, the elevated 5-HT concentration in the lung plays an important role in MA-induced chronic pulmonary toxicity.
Enzyme TPH catalyzes the rate-limiting step in serotonin biosynthesis. 32 Two isoforms of TPH have been characterized, TPH1 and TPH2, with TPH1 expressed in the periphery and TPH2 in the brain. 33 Although peripheral serotonin is synthesized chiefly by the enterochromaffin cells in the gut, PAECs produce serotonin and express the TPH1 isoform. 14,34 In this study, both serotonin concentration and TPH1 expression are increased in the lung tissue obtained from rats given with the high dose of MA compared with controls, which demonstrated that the increased TPH1 increasingly prompted the synthesis of serotonin and resulted in the elevated serotonin level in the lungs. In addition, serotonin concentration is also accommodated by serotonin metabolism. On the basis of substrate specificity, MAO exists in two forms, MAO-A and MAO-B. 35 MAO-B has a high affinity for phenylethylamine, whereas MAO-A preferentially metabolizes norepinephrine and serotonin. 35 Reduced MAO-A levels in the lung suggest a more generalized role for the inhibition of MAO-A activity in physiological effects in the lung. 24 The present study showed that high dose of MA led to MAO-A inhibition in the lung and resulted in the reduced serotonin metabolism, and then increased the serotonin potentially available for release. The results suggested that the increased TPH1 and the reduced MAO-A take an important part in MA-mediated changes in the serotonin concentration of the lungs.
VMATs mediate the accumulation of monoamines, such as serotonin, DA, adrenaline, NA, and histamine, from the cytoplasm into storage organelles with an absolute dependence on a vacuolar ATPase-generated proton gradient to transport the cationic amine substrates into the storage organelle in exchange for protons. 36 It was reported that amphetamine analogs, including MA, affect the function of VMAT-2 associated with serotonergic neuron. 37 The VMAT-2 has a crucial role in the storage and synaptic release of serotonin. 38 By interacting with VMAT-2, MA increases cytosolic serotonin concentrations available for translocation by SERT to the extracellular compartment. 39 The present study showed that VMAT-2 protein was predominantly expressed in the endothelium and in the media of the pulmonary arteries, and that the lung VMAT-2 protein level was significantly increased by 10 mg/kg MA in rats. VMAT-2 is a principal regulator of cytoplasmic serotonin concentrations. 37,40,41 Thus, these findings demonstrated that the increased immunoactivity and protein expression of VMAT-2 by MA augmented the serotonin storage in the lungs.
SERT is the major protein responsible for uptake and release of serotonin, transporting serotonin in either direction. 9,42 It was reported that drugs interacting with SERT can be divided into two basic classes: reuptake inhibitors and substrate-type releasers. 43 But MA, as a substrate for SERT, differs from selective serotonin reuptake inhibitor, in that MA not only inhibits serotonin reuptake but also stimulates serotonin release. 37,44 Accordingly, SERT contributes in regulating the level of activation of serotonin receptors via modulation of extracellular and intracellular serotonin concentrations. 9 It has been demonstrated that SERT plays a crucial role in serotonergic neurotransmission as it controls the concentration of free active neurotransmitter outside the neurons and consequently it is the molecular target for clinically effective drugs as well as for drugs of abuse. 45 Furthermore, it was reported that SERT is expressed in high levels in lungs. 24,46 The present study showed that the lung SERT expression was significantly increased after long-term administration of MA, and the histological finding revealed that SERT was predominantly expressed in PAECs and PASMCs. Taken together, the results demonstrated that MA facilitated SERT-mediated efflux of cytoplasmic serotonin and then results in the elevated serotonin concentration in the lung tissue.
In summary, long-term and high-dose administration of MA induces the chronic pulmonary toxicity in rats, and the effects of MA are due to the elevated serotonin concentration in the rat lung by increased synthesis, reduced metabolism, augmented accumulation, and promoted release of serotonin, which supports that the serotonin mechanism could be involved in MA-induced chronic pulmonary toxicity.
Footnotes
Conflict of Interest
The authors declared no conflicts of interest.
Funding
This work was supported by National Natural Science Foundation of China (grant numbers 30973533 and 30572194).



