Abstract
Staphylococcal enterotoxin B (SEB) is of concern to military and civilian populations as a bioterrorism threat agent. It is a highly potent toxin produced by Staphylococcus aureus and is stable in storage and under aerosolisation; it is able to produce prolonged highly incapacitating illness at very low-inhaled doses and death at elevated doses. Concerns regarding SEB are compounded by the lack of effective medical countermeasures for mass treatment of affected populations. This article considers the mechanism of action of SEB, the availability of appropriate experimental models for evaluating the efficacy of candidate medical countermeasures with particular reference to the need to realistically model SEB responses in man and the availability of candidate countermeasures (with an emphasis on commercial off-the-shelf options). The proposed in vitro approaches would be in keeping with Dstl’s commitment to reduction, refinement and replacement of animal models in biomedical research, particularly in relation to identifying valid alternatives to the use of nonhuman primates in experimental studies.
Keywords
Introduction
The staphylococcal enterotoxins (SEs) are protein exotoxins produced by Staphylococcus aureus. Of these, staphylococcal enterotoxin B (SEB) is one of the most frequently reported causes of toxin-associated food poisoning and is second only to SEA in this respect. 1 According to Le Loir et al., 2 the foods most often associated with S. aureus food poisoning are fermented milk products and cooked meals. There are some 15 enterotoxin serotypes, of which at least seven are produced by S. aureus (SEA, SEB, SEC1, SEC2, SEC3, SED and SEE 3 ). Although they have similar biological and structural properties, they are antigenically distinct with SEB, SEC1, SEC2 and SEC3 belonging to a different amino acid sequence homology set (serotype) to SEA, SED and SEE. 4 –6 They are relatively small proteins, with molecular weights of <30,000. 7 Following ingestion, the enterotoxins produce diarrhoea and vomiting responses. The staphylococcal enterotoxins can cause widespread damage to many organs in various host species via numerous biochemical activities. These can ultimately result in lethal shock if the dose is sufficiently high. 8
S. aureus can grow in a varied range of conditions and tolerates pH environments of 4.6–10 and temperature ranges of 7–47°C as well as sodium chloride concentrations of ≤20%. 9 S. aureus cultures can easily be grown using a variety of culture media, and various strains of the bacterium are available from commercial suppliers. 10 –12 The bacterium is also found on human skin and mucous membranes. 13 The bacterium is readily found in poultry processing plants and in such products as retailed raw chicken meat. 14 According to Le Loir et al., 2 S. aureus strains may be differentiated, based upon various biochemical characteristics (e.g. nitrate reduction capability, sugar fermentation characteristics, urease activity, haemolysis on blood agar 15 ), into biotypes depending on whether they originate from animal or human sources. Devriese 16 identified six biotypes: human, non-β-hemolytic, ovine, bovine, avian and nonspecific. S. aureus of human biotype origin are of greatest concern, as a high proportion of isolates (39%) from the human population may be enterotoxigenic. 17 SEs are generated by S. aureus during the log growth phase of bacterial cultures and also when growth transitions from log phase to the stationary part of the growth cycle. 7,18 –20 According to Onoue and Mori, 21 in five SE-producing strains of S. aureus, cystine and arginine are a requirement for bacterial growth and the synthesis of SEA, SEB and SEC.
SEB is relatively easy to produce in the form of crude bacterial culture supernatants, and one study reported a yield of 0.2 mg of SEB/mL of S. aureus (strain S-6) culture supernatant. 22 A culture batch of 20 L could therefore yield approximately 4 g of toxin. However, the generation of pure toxin required (in sequence) a combination of cation-exchange chromatography, isoelectric focussing and gel filtration chromatography, although this would not be required for the preparation to be potentially lethal (see below). Large volumes of S. aureus cultures could be generated by individuals with a relatively basic understanding of microbiology technique and with access to the required materials, although production of purified toxin would require higher level expertise and laboratory facilities.
SEB is very stable and easily aerosolized 23 and when inhaled at high doses may cause shock and death. 8 However, the toxin is generally regarded as an incapacitant, with inhalational exposure usually resulting in a highly incapacitating illness whose effects may last for up to 2 weeks. There is a paucity of data relating to the inhalation toxicity of SEB in models relevant to man, although an LD50 (estimated dose of a toxin that will be lethal for 50% of the exposed population) of 21.7–44.1 µg/kg by the inhalation route in Rhesus macaques is quoted in LeClaire et al. 24 The LCt50 (the estimated concentration of a toxin in air that will be lethal for 50% of the exposed population when inhaled for the specific period of time) for SEB 3 is 5 mg/min/m 3 , and this makes the toxin more potent than ricin by inhalation. Inhalation of aerosolised SEB results in severe respiratory distress, headache and fever, and in some subjects it will also cause nausea. 25,26
The sensitivity of humans to the incapacitating effects of SEB makes it a biological agent of significant concern to both military and civilian target populations, particularly from a terrorist and larger scale biological warfare perspective and consequently features on threat lists both in the United Kingdom and in the United States. In particular, its efficacy as an incapacitant at very low doses may lower the threshold for its use in warfare. According to Madsen, 27 the S. aureus enterotoxins have potential use as bioterrorism biowarfare agents as a consequence of their properties. This is compounded by the lack of established medical countermeasures that would be appropriate for mass treatment of exposed populations.
S. aureus is a widespread pathogen. It is known to cause toxic shock syndrome, wound sepsis and post surgery infection amongst other conditions. 28,29 Although it is a pathogen, S. aureus is a part of our bodily bacterial flora and is regarded as a commensal organism. 30 The integrity of the skin is essential in preventing infection of that tissue, otherwise both acute and chronic infections can occur. 31 In situations where the barrier function of the skin is impaired and the patients have reduced immunity, as in atopic dermatitis (AD), 32 one study by Nada et al. 33 reported that the skin of 87% of AD patients in their study was colonised by S. aureus, and of these patients, approximately 54% of the isolated strains were toxigenic with SEB being the most common superantigen gene found. Cole and Silverberg 34 have reported that AD subjects possess keratinocytes for which S. aureus has an increased affinity when compared with cells from normal individuals. In addition, Schlievert et al. 35 have suggested that S. aureus superantigens may facilitate the colonisation of the skin in steroid-resistant AD patients. In healthy individuals, S. aureus infection may occur as a result of activities such as contact sports and the use of shared toiletry equipment such as razors. 36
Estimates of the human population which harbour the bacterium (in the nares of healthy individuals) vary from 20 to 40%. 37 –39 In many cases, this may be an underestimate. In some studies, nares- and throat-derived S. aureus were found in 45% of the subjects studied. 40 Udo et al. 41 in a study of 250 healthy restaurant workers in Kuwait City found that 157 isolates of S. aureus could be obtained from the nares, but only 32 isolates could be cultured from their hands of which 12.5% synthesised either one or more SEs. Røder et al. 42 found that although 40% of nares-derived strains of S. aureus manufactured enterotoxins, of which SEB and SEC were the most commonly expressed, these staphylococcal toxins did not appear to have a significant role in the infection process.
This notwithstanding military operations in Iraq and Afghanistan and the large number of casualties with penetrating wounds that have arisen from these deployments have lent an additional urgency to the issue of medical countermeasures against infectious microorganisms, particularly with the development of antibiotic resistant strains 43 for such wounds provide a means of entry into the body for both staphylococcal and streptococcal infection. 44 S. aureus has a significant role to play in the infection of surgical sites and is responsible for infections in some 14% of such wounds. 45 Most infections in surgical sites are derived from the patients indigenous bacterial flora of which Staphylococcus spp. form only a part. 46 Indeed, one study indicated that this type of infection may involve numerous species, both aerobic and anaerobic, with on average up to six bacterial genera being involved, for example, Staphylococcus spp., Pseudomonas aeruginosa, Serratia marcescens, Corynebacterium spp., Peptoniphilus spp. and Prevotella spp. 47 In addition, the study identified two Bacteroidales spp. as being frequent colonisers of surgical site infections. In one study of war wounds in 415 patients treated in a Zagreb hospital between April and September 1991, infection occurred in some 15.7% of patients. 48 These infections were found to be mostly (in 90% of patients) due to aerobic bacteria, for example, S. aureus, P. aeruginosa and Acinetobacter spp., with only 9% being attributable to a combined aerobic and anaerobic bacterial population. It should be noted, however, that Wolcott et al. 47 have cautioned that the extent of involvement of anaerobic bacteria in surgical site infections may be grossly underestimated, for it was stated that <5% of bacterial species can be cultured easily using available media. Surgical site infection rates in war wounds can be >20%, if only primary closure of the wounds is employed, although this can be reduced to 3–4% with such approaches as debridement of nonviable tissue and delayed closure of the wound edges (typically at 4–6 days), together with optimal use of antibiotics. 49,50
The present situation regarding the status of SEB as a potential biowarfare agent clearly requires new experimental approaches that could be used to effectively screen potential candidate medical countermeasures. At the core of this issue is the debate associated with what constitutes an appropriate model which effectively and realistically reproduces essential aspects of the mechanism of action of SEB. This article considers the mechanism of action of SEB, the availability of appropriate experimental models and promising medical countermeasures which could be evaluated with appropriate in vitro systems for subsequent pull-through into appropriate animal models. This would have the added benefits of addressing ethical issues associated with the use of such in vivo models as nonhuman primates (NHPs) in terms of the need for reduction, refinement and replacement strategies.
Mechanism of action of SEB
SEB is a T-cell superantigen that has two domains in its structure (Figure 1), which allows it to bind to the variable region of the T-cell receptor β or γ chain on CD4+ (T helper) and CD8+ (cytotoxic T) cells. It simultaneously forms a bridge by cross-linking with major histocompatibility complex (MHC) class II molecules on antigen-presenting cells (APCs). 51,52 APCs most commonly include monocytes/macrophages, blood and lymph dendritic cells and vascular endothelial cells, 53 but also extend to include airway epithelial cells such as the type II pneumocytes of the alveoli. 54,55 The normal interaction between T-cells and APCs results in the stimulation of immune-cell expansion (e.g. proliferation of T-cells) and in the release of cytokines. In normal antigen-specific interactions, the CD4 molecule on T-cells has a stabilizing effect on interactions between antigen receptors on T-cells and the MHC molecules on APCs. However, staphylococcal enterotoxins and other superantigens may mimic the binding of CD4 and thus cause T-cell stimulation in a way that does not involve antigen recognition. 5

Comparison of binding interactions of the T-cell receptor, SEB and the MHC class II receptor: (a) is a ribbon representation of the interactions between the T-cell receptor (green/light green), a peptide antigen (yellow spacefill) and the MHC class II (red) complex. (b) is a ribbon representation of the interactions of T-cell receptor (green/light green), SEB (blue) and MHC class II (red) complex, showing the structural relationship of the proteins which results in an abnormal immune response. The image shows how the SEB molecule cross-links the MHC class II and T-cell receptor outside the normal binding cleft. (The location of the normal binding cleft is indicated by the presence of an antigen peptide (denoted by the yellow spacefill atom representation), which is retained in (b) for illustration purposes only.) The models were constructed by superposition from crystallography-derived data (Protein DataBank entries 3SEB, 2SEB, 1SBB and 2CBK), using SYBYL™ software (Tripos, 1699 South Hanley Road, St Louis, Missouri, USA). SEB: Staphylococcal enterotoxin B; MHC: major histocompatibility complex
The ability of superantigens such as SEB to mimic CD4 and related adhesion molecules effectively stabilises the complexes formed between MHC class II molecules on APCs and antigen receptors on T-cells. It is the ability of SEB to bind to monocytes at MHC class II molecules instead of their normal antigen-binding receptors that causes abnormal responses. 56 Normal processing of antigens involves uptake by APCs (such as monocytes) for proteolytic processing. Conventional antigens are subsequently presented for recognition by the T-cell receptor within the MHC II receptor binding cleft. However, SEB does not undergo processing by APCs and instead binds to the MHC class II site and the T-cell receptor complex. It is the association of SEB with these two components outside the binding cleft that causes the subsequent cytokine casade (Figure 2). 57 The SEB-induced proliferation of T-cells is particularly marked among cells that express specific variable domain (Vβ) elements on their cell-surface receptors, leading to activation and proliferation of T-cell subsets. 58 –61
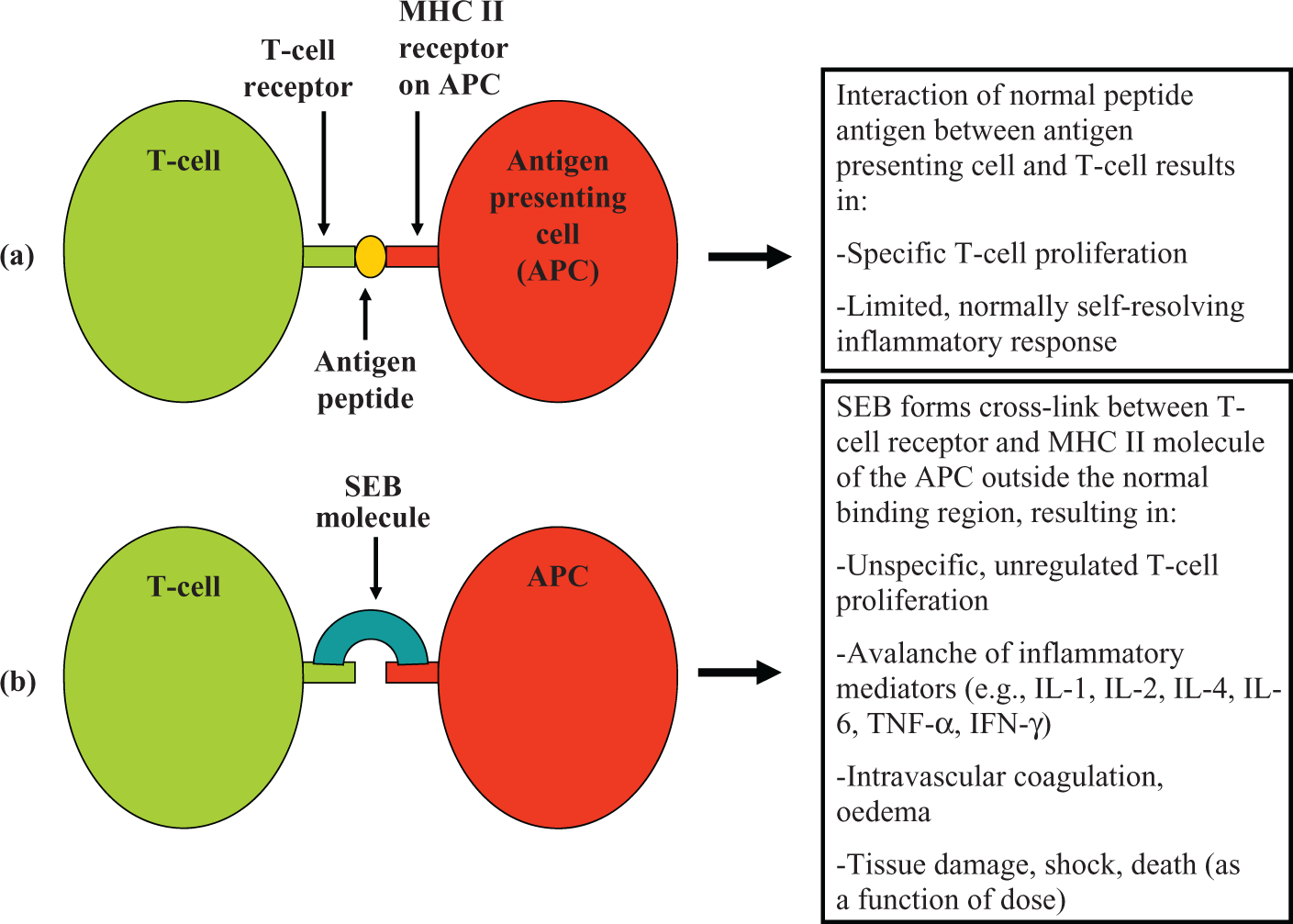
A diagrammatic representation contrasting (a) the normal process of antigen presentation by an APC and a T-cell via MHC II and (b) the nonspecific T-cell activation induced by SEB and its consequences. (The various components of the diagram are not to scale.). APC: antigen-presenting cell; SEB: Staphylococcal enterotoxin B; MHC: major histocompatibility complex
The uncontrolled expression of cytokines induced by SEB effectively bypasses conventional MHC-mediated antigen processing. The cell proliferation/cytokine release cycle causes an avalanche of products which results in intravascular coagulation, tissue damage and ultimately, organ dysfunction and in extreme cases, death. 8,62 SEB has been shown to increase expression of components of the arachidonic acid pathway such as cyclooxygenase and lipoxygenase metabolites, a process which results in inflammation and, subsequently, oedema and shock. 63,64 This is also accompanied by the uncontrolled expression of cytokines such as interleukin 1α (IL-1α) and IL-β, IL-2, IL-4, IL-6 and IL-10, interferon-γ (IFN-γ), tumour necrosis factor α (TNF-α), and TNF-β. 61,65 According to Calandra et al., 66 the levels of TNF-α and IL-1 in the circulation are correlated with clinical symptoms of septic shock. Studies by Nooh et al. 67 showed that SEB-exposure of spleen cells from human leukocyte antigen (HLA) class II transgenic mice resulted in progressive release of TNF-α, IFN-γ and IL-2 over time. DNA microarray transcript analysis of SEB-exposed HLA-DR3 transgenic mice indicated very large increases in IL-6 (700-fold), IFN-γ (360-fold), IL-2 (240-fold) and IL-4 (53-fold). 68 The consequence of this activity can range from self-limiting food poisoning episodes to potentially lethal toxic shock syndrome. 69 –71 The rapidity with which SEB can saturate MHC class II molecules would suggest that active immunity (i.e. the presence of circulating anti-SEB antibodies induced by a vaccine) may be the most appropriate defence against this agent. 3 However, this approach may not be feasible for all populations at risk. This necessitates consideration of postexposure therapeutic approaches.
One area that has not been adequately investigated in the pathology of SEB intoxication is the role of reactive oxygen species (ROS) following SEB exposure. APCs such as phagocytes may generate ROS such as the superoxide anion 72 and nitric oxide (NO). 73 The superoxide anion can also react with NO to generate the peroxynitrite radical; these highly active species can damage a variety of biologically important molecules. 72 ROS, NO and related species are normally utilised by white blood cells to kill microbial pathogens, but inappropriate activation could also compromise viable tissues. Interestingly, the activation of T-cells also results in the elevation of ROS levels in this cell type. 74 –76 The pulmonary oedema that follows inhalation exposure to SEB is probably a consequence of T-cell proliferation in the mucosal epithelium/alveolar epithelium of the respiratory tract, and the associated uncontrolled release of proinflammatory mediators, rather than a direct effect of the toxin on epithelial cells. 60 However, the potentially destructive effect of the release of particular proinflammatory mediators and related molecular species upon the architectural and functional integrity of structures associated with the blood/air interface (e.g. alveolar endothelium/epithelium) has not been formally tested. The use of appropriate in vivo and in vitro systems could address this issue.
In vivo and in vitro models
Issues associated with in vivo systems
A major problem associated with the use of such classical models as spleen cell cultures from the mouse is that although it is a well-established assay for analysing superantigens, 77 –79 mouse and rat-based models are regarded as being substantially less sensitive to SEB intoxication than NHP models. 8 One method to develop rodent models that are more sensitive to enterotoxins has been to utilise humanised mice that express HLA class II transgenes, resulting in T- and B-cell superantigen stimulated responses. 67 Alternatively, BALB/c mice have been used as a model for SEB and toxic shock syndrome toxin 1 (TSST-1, another superantigen toxin produced by S. aureus) pathology by injecting these toxins (intraperitoneal (i.p.)) at 1 µg/mouse, followed by a lipopolysaccharide (LPS) injection 4 h later (at a dose of 80 µg/mouse); these mice subsequently displayed symptoms classically associated with exposure to these toxins (e.g. elevated serum levels of TNF-α, IL-1α and IFN-γ). Huzella et al. 80 have also described a new intranasal (i.n.) and i.p. exposure model for SEB exposure based on C3H/HeJ mice. These mice responded to low microgram exposure levels of SEB with severe lung inflammation, hypothermia and, subsequently, death. The C3H/HeJ mouse was described as a more appropriate model for SEB studies as the signs and symptoms of SEB intoxication were associated with elevated blood serum and lung levels of IL-2 and monocyte chemoattractant protein-1 (MCP-1). More latter, Krakauer et al. 81 used transponders in C3H/HeJ mice to show that additional data such as body and chemokine (macrophage inflammatory protein-1 (MIP-1) α, MIP-1β and MCP-1) responses could be collected to assess the efficacy of potential therapeutic approaches. This strain did not require the use of potentiating agents such as LPS and may therefore be a more appropriate mouse model than BALB/c mice. SEs may cause diarrhoea and vomiting in man at ingested doses of <1 µg. 2,82 Dosing by the oral route at 5–20 µg/monkey resulted in emesis in an assay described by Bergdoll. 83 However, there are relatively few in vivo models of emesis that exist as alternatives for NHPs, although the house musk shrew has been employed in this capacity by Hu et al. 84 for studying the effects of administration of SEs by the oral and i.p. route. The ferret has also been used as a model of emesis, also following administration of SEB by the oral route. However, milligram quantities of the toxin were required, 85 so the relative insensitivity of the ferret to SEB may limit its value as a realistic exposure model.
Humans and NHPs are highly sensitive to SEB and other related toxins because of higher binding affinity of the MHC class II receptor for the toxin 3 ; so humanised mice may confer advantages to the study of SEB countermeasures. However, such models are costly to establish and maintain. The high level of sensitivity of humans to SEB via the inhalation route may also be attributable to the presence of MHC class II receptors on the surface of type II penumocytes and alveolar capillary endothelial cells. 86 More recently, Debbabi et al. 54 and Gereke et al., 55 using mouse models, demonstrated that type II alveolar epithelial cells could act as APCs via the MHC class II system. An ex vivo model comprised of whole blood from rhesus monkeys has been developed by Krakauer et al. 87 to study SEB responses. This study confirmed that whole blood, after exposure to SEB, releases several cytokines (e.g. IL-2, TNF-α and IFN-γ). However, data from all in vivo models needs to be carefully considered with respect to extrapolation to human responses.
A human cell-based SEB intoxication model
It would be potentially more relevant and ethically more acceptable to assess the efficacy of medical countermeasures using culture systems based on human T-lymphocytes, as this would model more relevant responses. The majority of in vitro studies utilise human peripheral blood mononuclear cells (PBMCs) from healthy human donors. This provides cell material to enable SEB-induced T-cell proliferation responses to be studied (in the presence of APCs such as monocytes and natural killer (NK) cells) and to permit analysis of the production and release of such cytokines as TNF-α, IFN-γ, IL-1β and IL-6. 4,81,88 –93 This approach could be developed further to permit direct analysis of the effects of T-cell activation upon such target epithelial types as human alveolar epithelium in mixed cell culture studies, so that medical countermeasures could be more effectively screened and down-selected for subsequent exploitation in relevant in vivo models. The ethical considerations associated with the use of NHPs to test anti-SEB countermeasures would be more appropriately addressed with the adoption of optimised in vitro systems to underpin the down-selection process of candidate medical countermeasures.
The use of human-based in vitro systems would allow the role of different immune cell subsets in the mediation of toxic effects of SEB to be assessed, for example, T-lymphocytes: CD4 and CD8, B-lymphocytes, NK cells, polymorphonuclear leukocytes, alveolar macrophages, and mononuclear APCs.
Clarification of the role of white blood cell species involved in SEB responses and of the inflammatory mediators they release, including ROS (as noted above), would allow a more focused approach to SEB countermeasures to be developed. This is because the release of cytokines varies as a function of cell-type. For example, following SEB exposure, T-cells and activated monocytes are the main sources of TNF-α, whereas IFN-γ is largely produced by T-cells and NK cells. IL-2 is generated by SEB-activated T-cells.
87
The extraction of PBMCs and isolation of subcellular fractions thereof (e.g. T-lymphocyte fractions), including the use of magnetic cell separation technologies, and the processes used to validate subtype purity and viability (by flow cytometry) have been described in Macildowie et al.
94
Mechanistic studies using coculture cell systems
A central issue associated with the pathology of inhaled SEB exposure relates to the damaging effects of the release of proinflammatory molecular species and how these are involved in pathological sequelae such as pulmonary oedema. This could be addressed using combined (coculture) systems based on human small airways epithelium, human alveolar epithelium or human vascular endothelium grown on semipermeable membranes. When confluent, these epithelial or endothelial cell systems could then be exposed to SEB-treated human-derived whole PBMC fractions or selected PBMC subfractions (see Figure 3 for an example of such an in vitro cell culture system). This would allow the role of alveolar APCs in the pulmonary pathology of SEB to be explored. See and Chow 95 have used human antigen-presenting monocytes cocultured with T-cells in a 1:1 ratio for their pyrogenic toxin studies. Current magnetic cell separation and purification systems would permit high purity fractions to be obtained from whole human blood and for the viability of these fractions to be assessed prior to use. Current studies indicate that T lymphocyte preparations with a purity of 99.4% (±0.4) can be routinely obtained from human PBMC preparations and that the T cells can be maintained at 100% viability for at least 24 h following isolation. 94 However, it should be noted that the majority of recent in vitro-based studies on SEB have used human whole PBMC preparations with no additional subfractionation to assess the contributions of T-cell subpopulations to SEB’s mechanism of action.

In a complex in vitro system for modelling SEB responses in human lung tissue (see picture (a)), cell culture inserts (IN) in combination with culture plates would be used (e.g. 6.5 mm diameter Costar® Transwell® inserts, Corning Inc.). Each insert would be placed in a well (W) of the plate. The alveolar epithelial cells would be grown on top of the semipermeable membrane at the bottom of the insert (see view looking into top of insert with cells stained purple in picture (b)). A photomicrograph of the membrane at the bottom of the insert is shown in (c), with human alveolar epithelial cells stained purple and the 8 µm pores in the membrane is shown as circular white holes (magnification, ×200). To complete the modelling of the alveolar structure as shown in (d), the alveolar cells (AC) would be grown on top of the insert membrane (M), with vascular capillary endodermal cells (EC) grown under the insert membrane. This would allow biochemical cross-talk between the cell types to occur before and after SEB challenge via pores (coloured grey) in the supporting membrane (M). Cross-talk would also encourage optimal cell differentiation to occur. When these cell types (or other cell combinations) had reached the required level of development, T-cells (TC) would then be added to the top of the membrane. To complete the exposure model, SEB would then be added to determine the effects of combining T-cells with various antigen-presenting cell types (e.g. AC and EC). The effects of candidate therapeutic compounds could then be more realistically and empirically modelled in vitro. The efficacy of these compounds would be judged on the basis of being able to inhibit such SEB-induced responses as: T-cell proliferation; cytokine/chemokine production and release, impairment of the structural integrity of the cell layers (representing the blood/air interface) and cell death. (The various components in (d) are not to scale.) The light orange line and small orange dots represent the level and location of nutrient medium in the system. SEB: Staphylococcal enterotoxin B.
Human microvascular endothelial cells could also be utilised to model effects of SEB on the pulmonary capillary bed as these are also an APC type. As noted above, the contributions of alveolar macrophages to SEB pathology should be assessed, and although obtaining these by pulmonary lavage presents major ethical and logistical problems, such cell types may now be obtained from human peripheral blood mononuclear fractions by stimulating them to differentiate along appropriate differentiation pathways using granulocyte–macrophage colony-stimulating factor. 96 Alternatively, human microvascular pulmonary endothelial cells and alveolar epithelial cells can be obtained from commercial sources (e.g. ScienCell Research Laboratories). Investigations with lung-derived target cells may be particularly relevant owing to the sensitivity of human subjects to inhalation exposure to SEB.
Such approaches could also resolve whether SEB-induced damage to the alveolar epithelium layer (and/or endothelial cells of the associated capillary bed) was a function of the SEB-induced release of active molecular species from white blood cell types found in inflammatory cell infiltrates and resident alveolar macrophage populations. At present, the precise role of ROS, cytokines and chemokines in the in vivo pathology associated with inhaled SEB remains unresolved. In vitro approaches would permit the relative importance of the role of different T-cell and APC combinations to be assessed, thus allowing a focussed approach to medical countermeasures to be adopted, for example, in the treatment of inhalation exposure to SEB. The use of candidate countermeasures such as selective inhibitors of different inflammatory mediators and ROS scavengers (see below) would also allow an insight into which molecular species caused the greatest tissue damage, and establish orders of priority for targeting their neutralisation. Interspecies comparisons of SEB-induced responses in improved in vitro models comprised of defined cell types (derived from relevant human, NHP and mouse tissues as noted above) would contribute to our understanding of the mechanism of action of this toxin and would assist in defining appropriate targets for intervention. This, together with an improved knowledge of the comparative efficacy of various candidate therapies for SEB intoxication using interspecies in vitro approaches, would inform extrapolation issues from commonly used in vivo models to likely efficacy in human subjects.
Candidate medical countermeasures approaches
The use of coculture systems based on relevant cell types of human origin would provide the opportunity to test a number of candidate countermeasures, including the efficacy of anti-SEB antibodies raised by vaccination regimes, and address whether windows of opportunity can be exploited for post-SEB exposure treatment. Using cells derived from human donors in vitro could, arguably, be of more scientific value than in vivo studies with nonhuman species. Reagents could, therefore, be screened to assess their efficacy in a system which was closer to humans.
Some studies in primates, being phylogenetically closer to the man, have offered potentially valuable information for treatment of humans. For example, in the study by Ulrich et al., vaccines developed against SEB have been found to protect immunised rhesus monkeys from inhalation challenge (30 × LD 50 ). 97 Interestingly, this group found that mixtures of genetically inactivated SEB and SEA, or other superantigens, were more protective in mice than single-toxin vaccines against individual toxins (administered i.p.) and also provided cross-protection against other superantigens. This may be a function of the amino acid sequence and structural homology of many superantigens. 97 It may, therefore, be possible to develop a broad coverage vaccine against these toxins.
The neutralising effect of antibodies generated in this way could be tested in human-derived cell systems. In vitro systems would allow such issues as windows of therapeutic efficacy and biocompatability of candidate countermeasures to be explored and pulled-through to in vivo efficacy assessment programmes. This is an important consideration, for small animal model data alone is of limited use for assessing the value of anti-SEB countermeasures owing to the large differences in sensitivity between man and rodent species. The development of an in vitro system based on relevant cell types from human tissue would permit analysis of the efficacy of some promising developments in the attenuation of the effects of SEB (and related superantigenic toxins).
In this context, the anti-inflammatory drug dexamethasone attenuates the SEB-stimulated release of proinflammatory mediators from human nasal epithelial cells 98 and also increased survival of SEB-exposed, LPS-sensitised BALB/c mice when administered no more than 4.25 h after SEB challenge, lowering proinflammatory cytokines. 99 IL-10 and IL-1 receptor antagonist and IFN-γ may also be a candidate anti-inflammatory tools. 100 Anti-SEB antibodies (administered intravenously) have also been reported to protect rhesus monkeys at 4 h after inhalation challenge with 5 × LD50’s of SEB. 3 Neutralising mouse anti-TSST-1 monoclonal antibody has been reported to cross-react with SEB so that it partially inhibits T-cell proliferation and TNF-α release. 59 Krakauer et al. 81 recently reported that rapamycin, an immunosuppressive drug, used in human organ transplantation procedures inhibited SEB-induced hypothermia and weight loss in SEB-exposed mice (i.n.) as well as reducing IL-6 and MCP-1 levels in the circulation. The drug was also found to inhibit SEB-induced T-cell proliferation and blocked SEB-stimulated manufacture of IL-1β, IL-2, IL-6, TNF-α, IFN-γ, MIP-1α, MIP-1β and MCP-1. Rapamycin was found to be more effective than dexamethasone when used to treat SEB-exposed mice as the former remained effective even when administered several hours after toxin exposure.
A range of potentially therapeutic molecules worthy of further investigation includes lactoferrin, 101 cell-permeant peptides 102 and several other drugs or compounds including caspase inhibitors. 68,88,92,93,103 Other inhibitors of SEB include a dodecapeptide (termed P72) that can protect mice against intoxication by SEA, SEB and SEC. 104 Other promising potential therapeutics include the broad spectrum antibiotic doxycycline, which has been reported to inhibit the SEB-stimulated production of various cytokines (IL-1β, IL-6, TNF-α and IFN-γ) and chemokines (MCP-1, MIP-1α and MIP-1β) from human PBMCs. 90 The antioxidants pyrrolidine dithiocarbamate and N-acetyl cysteine have been found to attenuate SEB-induced T cell proliferation and inflammatory mediator production in vitro, an effect attributed to the inhibition of gene activation pathways associated with immune system upregulation. 105 Pirfenidone treatment has also been reported to increase survival in SEB challenged mice from ≤10 to ≥80% even when administered up to 4.25 h after toxin exposure. 106 The vasodilatory drug pentoxifylline has been found to reduce SEB-stimulated proinflammatory cytokine release in human PBMCs and inhibits T-cell proliferation in vitro, but also improved survival from SEB challenge in vivo. It is a well-tolerated drug with few side effects. 107 It has also been found to attenuate the release of cytokines in whole blood cultures from rhesus macaques. 87 Additionally, plant extracts such as the flavone baicalin (extracted from the medicinal plant Scutellaria baicalensis Georgi) may also have value as anti-SEB therapeutics through inhibition of SEB-stimulated release of proinflammatory cytokines, in vitro. Similar effects were found for triptolide, a diterpene compound. This is found in Tripterygium wilfordii, a Chinese medicinal plant. 108 Finally, a therapeutic procedure at an advanced research stage is a blood filtration device that has been developed for the removal of proinflammatory cytokines by hemadsorption in order to reduce reperfusion injury following surgery. This has potential for the treatment of many serious inflammatory illnesses (Gourlay et al., personal communication).
In order to exploit developments in candidate therapeutic approaches to SEB exposure, and to provide a rational basis for screening these for pull-through to in vivo validation, it would be necessary to utilise model systems that permit realistic modelling of SEB/target cell interactions. Such approaches may also identify novel interactions that could develop into future potential countermeasures. The requirement to prioritise target cells associated with the inhalation route of exposure could be readily met as PBMC material is available from human donors. However, an issue that is not addressed in the literature is the modelling of the effects of these cascade components on the organs that are of greatest concern. In the biowarfare context that means human lung tissue and in particular, respiratory tract epithelium (particularly type II pneumocytes) and vascular endothelium within that tissue.
Summary
The inflammatory system clearly plays a major role in the pathology of SEB and other superantigenic toxins. Numerous in vitro and in vivo systems can be utilised to study not just the mechanism of action of these highly potent toxins, but also the potential efficacy of candidate therapeutic countermeasures.
However, although the involvement of the runaway immune cascade induced by superantigen exposure is largely established in terms of the production and release of cytokines and chemokines, it is still poorly characterised in terms of proof of the relative damage-inducing potential and mechanism of action of each component of that cascade.
The proposed in vitro systems have the potential to address these issues and open up new avenues for postexposure treatment options.
Footnotes
Acknowledgements
The authors thank Bry Lingard (Detection Department) for constructing the diagram of staphylococcal enterotoxin B interactions in Figure 1 and for supplying information for the legend and to Sarah Poole (Biomedical Sciences Department) for her contribution in the construction of the schematic representation of ![]() .
.
Funding
This study was supported by funding from the UK Ministry of Defence.
