Abstract
A solid phase extraction method for enrichment-separation and the determination of cobalt (Co2+), copper (Cu2+), nickel (Ni2+), zinc (Zn2+) and lead (Pb2+) ions in real samples has been proposed. The influences of some analytical parameters like pH, flow rate, eluent type and interference of matrix ions on recoveries of analytes were optimized. The limits of detection were found in the range of 1.6–3.9 µg L−1, while preconcentration factor for all understudy metal ions were found to be 166 with loading half time (t 1/2) less than 10 min. The procedure was applied for the enrichment-separation of analyte ions in environmental samples with recoveries higher than 94.8% and relative SD <4.9% (N = 5).
Keywords
Introduction
Because of possible negative roles of heavy metal ions in animal and human life, their environmental pollution is one of the important human problems.1–6 Their determination at trace levels, especially in environmental complicated matrices samples, is restricted by two main problems, including over concentration than the detection limit by flame atomic absorption spectrometer (FAAS, as most prominent technique) and the interferences sourced from the matrix. In such cases, the direct evaluation of heavy metal ions is impossible. For accurate, reliable and sensitive quantification of the metal ions performing a prior preconcentration and separation procedure to enrich analytes from original sample solution is required.7–13
Solid-phase extraction (SPE) is a popular technique for enrichment and separation of metal ions because of its advantages such as simple operation, high preconcentration factor, rapid phase separation and the ability of combination with different detection techniques such as FAAS.
Generally, the design of stable and efficient sorbent for separation and enrichment purpose depends on the surface area, nature and activity of solid support and properties of the organic complexing agent loaded on the solid substrate. In this regard, various support in our laboratory applied for the preconcentration–separation of heavy metal ions.11–18
This research focuses on the modification of Diaion HP-2MG with 2-(2,6-dichlorobenzylideneamino)benzenethiol (DCBABT) and its efficient application to enrich and preconcentrate understudy metal ions. To obtain maximum recoveries for understudy metal ions, the influence of variables like pH, amount of DCBABT, condition of eluent and flow rates on method performance was examined.
Experimental
Instruments
The measurements of analyte elements were performed with Shimadzu 680 AA atomic absorption spectrometer equipped with a hollow cathode lamp and a deuterium background using an air–acetylene flame at instrumental parameters ordered by the manufacturer. A Metrohm 691 pH/ion meter with a combined glass–calomel electrode was used for the adjustment of test solution pH.
Reagent and solutions
Acids, bases and nitrate salts of all investigated cations with the highest purity available are purchased from Merck (Darmstadt, Germany), while the solution was prepared in double-distilled deionized water. The pH adjustment was carried out by the addition of dilute nitric acid (HNO3) and/or potassium hydroxide (KOH) to obtain the desired pH solution.
Synthesis of DCBABT
The ligand was synthesized according to a similar method in our previous report. 19 The physical and spectral data of present ligand is as follow: elemental analysis, % C13H9Cl2NS: calculated: C, 55.33; H, 3.21; N, 4.96 found: C, 54.8; H, 3.4; N, 4.7. IR (KBr, cm− 1): 3444 (bs, νOH), 3054 (w, CH-Aromatic), 2915 (w, CH-iminic), 1623 (–C=N), 1565 (C=C), 1461 (m, C=C), 1430 (m), 1373 (w), 1268 (m, νC–N), 1199 (w), 1095 (m, C–O), 1041 (m), 964 (m), 887 (s), 759 (s, C–S), 555 (m). Schematic expectable diagram of complexation of ligand with divalent metal ions is presented in Figure 1.

Shematic diagram of complexes formation(ML and/or ML2).
Preconcentration procedure
Understudy metal ions solution (0.2 µg mL−1) of 250–1500 mL at pH 4.5 is passed through the DCBABT loaded on Diaion HP-2MG at a flow rate of 2 mL min− 1. All understudy metal ions were enriched due to chelation with proposed ligand. The enriched analytes were subsequently eluted with 6 mL of 4 mol L− 1 HNO3. Analytes levels in the eluent were determined by FAAS.
Pretreatment of real samples
Cheese and/or animal butter (20 g) purchased from Yasouj, Iran, is completely grinded to small pieces. The grinded small size samples were heated in silica crucible on a hot plate for 3 h and the overcooked material was transferred to furnace and heated overnight at 650°C. Then, the residue was cooled and treated efficiently with 10 mL concentrated HNO3 and 3 mL 30% (w/w) hydrogen peroxide and again kept in furnace for 2 h at same temperature until finishing evolution of organic compound. The final residue was treated with 3 mL concentrated hydrochloric acid (HCl; 36%, w/w) and 2–4 mL 70% (w/w) perchloric acid and evaporated to fumes that lead to changing all metals content to respective ions. The supernant solution and residues dissolved in water and filtered and their pH was kept at 4.5 by the addition of KOH and HCl and/or diluted to mark in 250 mL volumetric flask. These samples were subjected to the described procedure mentioned in above section for triplicate and the analyte contents and recoveries were evaluated by standard addition method.
Results and discussion
Influences of pH
The most important factors in SPE procedure is the pH of the working media.20–23 Also, the reaction between analytes and the complexing agent significantly depends on the pH value. The pH influences on the chelation of understudy metal ions with DCBABT. Therefore, the effect of pH in the range of 3–8 at constant values of other parameters on the recoveries of understudy metal ions was investigated.
The obtained results (Figure 2) indicate that the understudy metal ions could be retained quantitatively by the modified solid phase at pH 4.5. The decrease in signal at pH >4.5 is probably due to the precipitation of ions as their related hydroxide or mixed charged unextractable hydroxy complexes of understudy metal ions and at pH <4.5 may be due to hydronium ion competition with metal ions for complexation with DCBABT. Therefore, pH of 4.5 was selected.

Influences of pH on the recoveries of analyte metal ions.
Influences of amounts of DCBABT and adsorbent
In preliminary experiments, some similar experiments in the absence and presence of proposed ligand were undertaken. It was found that low recoveries of understudy metal ions in the absence of chelating agent can be significantly improved via impregnation of DCBABT on Diaion HP-2MG. Therefore, the studies were performed with various amounts of DCBABT. The results are depicted in Figure 3. The recoveries of analytes increased with increasing amount of DCBABT and reached constant and maximum value (>95%) using at least 40 mg. A 40-mg ligand was used for all further works.
The effects of the amounts of proposed (DCBABT) solid phase Diaion HP-2MG at fixed optimum values of ligand were investigated and respective results display that up to 0.3 g of solid phase efficiency of recoveries of understudy metal ions probably due to increase in significantly enhanced reactive sites. In the proposed procedure, 0.3 g of solid phase is recommended (Figure 4).

Influences of amount of ligand on the recoveries of analyte ions.
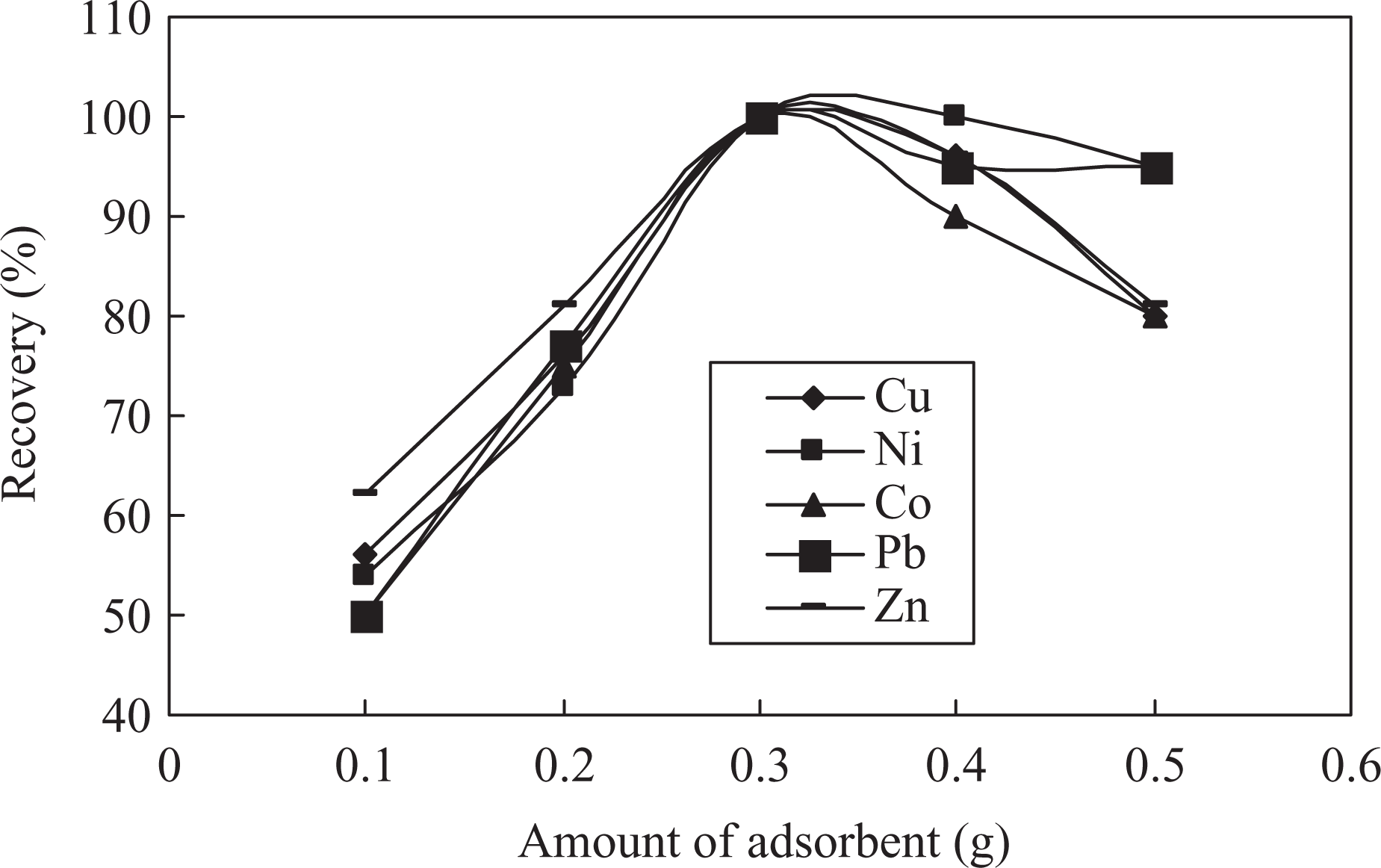
Influences of amount of solid phase.
Eluent conditions
For SPE study, the choice of a suitable eluent is very important for the elution of retained metal ions that is compatible with measurement system. 24 ,25 Different acids at various concentrations and volumes were applied to desorb the understudy metal ions (Table 1). It was observed that quantitative recoveries for analyte ions were obtained using 4 mol L− 1 HNO3, while lower recoveries were obtained using other eluents.
Influences of various eluents on the recoveries of analytes.

Cu: copper; Ni: nickel; Co: cobalt; Pb: lead; Zn: zinc; HNO3: nitric acid; CH3COOH: acetic acid; HCl: hydrochloric acid; H2SO4: sulfuric acid.
The volume of eluent was examined by changing the volume of 4 mol L−1 HNO3 in the range of 2–10 mL. The smallest efficient volume of 4 mol L−1 HNO3 for quantitative elution of the metal ions was 6 mL. Therefore, 6.0 mL of 4 mol L−1 HNO3 was selected for subsequent experiments to obtain high enrichment factor.
Effects of flow rates
The influences of the sample and eluent flow rates on the investigated metal ion recoveries were separately examined in the flow rates in the range of 1–5 mL min−1. The flow rates of the solutions were controlled using the stopcock of the column. When the influences of flow rates of sample was investigated, the eluent flow rate was kept constant as 1 mL min−1. The retentions for the analytes were quantitative up to a sample flow rate of 2 mL min−1.
Effect of sample volume
The measurement of the breakthrough volume (highest sample volume that their analytes content can be enriched without the loss of recoveries) was examined by dissolving 50 μg of metal ions in various sample volumes in the range of 200–1100 mL. It was found that analyte contents quantitatively enrich on the solid phase until 1000 mL. The efficient elution of retained analytes via 6 mL of 4.0 mol L−1 allow a preconcentration factor of 1000/6(167).
Matrix effects
Before the application of the preconcentration method for the determination of analyte ions in samples with complicated matrix, the influences of some alkaline and alkaline earth and some transition metal ion on the recoveries of the analyte ions were also investigated and respective results are given in Table 2. The tolerance limit is defined as (ion concentration lead to relative error smaller than 5%) the combination of preconcentration and determination of analytes. Metal ions were quantitatively recovered in the presence of large amounts of alkaline and alkaline earth ions and some transition and heavy metal ions.
Effects of the matrix ions on the recoveries of the examined metal ions (N = 3).

Application to environmental samples
The test procedure was applied to different solutions containing various amounts of analytes and blank solutions. The response of proposed method was plotted according to the initial concentration and characteristic performance of the method, which is presented in Table 3. The experimental preconcentration factor (ratio of slope of calibration curve after to before preconcentration) as the ratio of calibration curve slope before to respective one after preconcentration and the preconcentration factor as the phase volume ratio (V initial/V eluent) for all ions was presented in Table 3. The limit of detection of proposed method according to three times the SDs of the blank (n = 10) and the empirical limit of quantification as lowest accurate and precise measurable level of analytes were presented in Table 3.
Specification of presented method at optimum conditions for each element.

Cu: copper; Ni: nickel; Co: cobalt; Pb: lead; Zn: zinc; RSD: relative standard deviation.
We have explored the feasibility of the methodology given in Experimental section using enrichment of analyte ions in different environmental matrices by standard addition method, while the accuracy was investigated by spiking a known quantity of analytes to the sample. The recoveries and relative SD of application of proposed method for analytes evaluation in cheese and animal butter is presented in Table 4. The high values of recoveries in spiked samples satisfactorily show the capability and applicability of the system for the determination of analyte ions. The obtained recoveries (higher than 95%) confirm that the accuracy of proposed method is independent of the matrix effects.
Recoveries of analyte ions from spiked samples (N = 3).
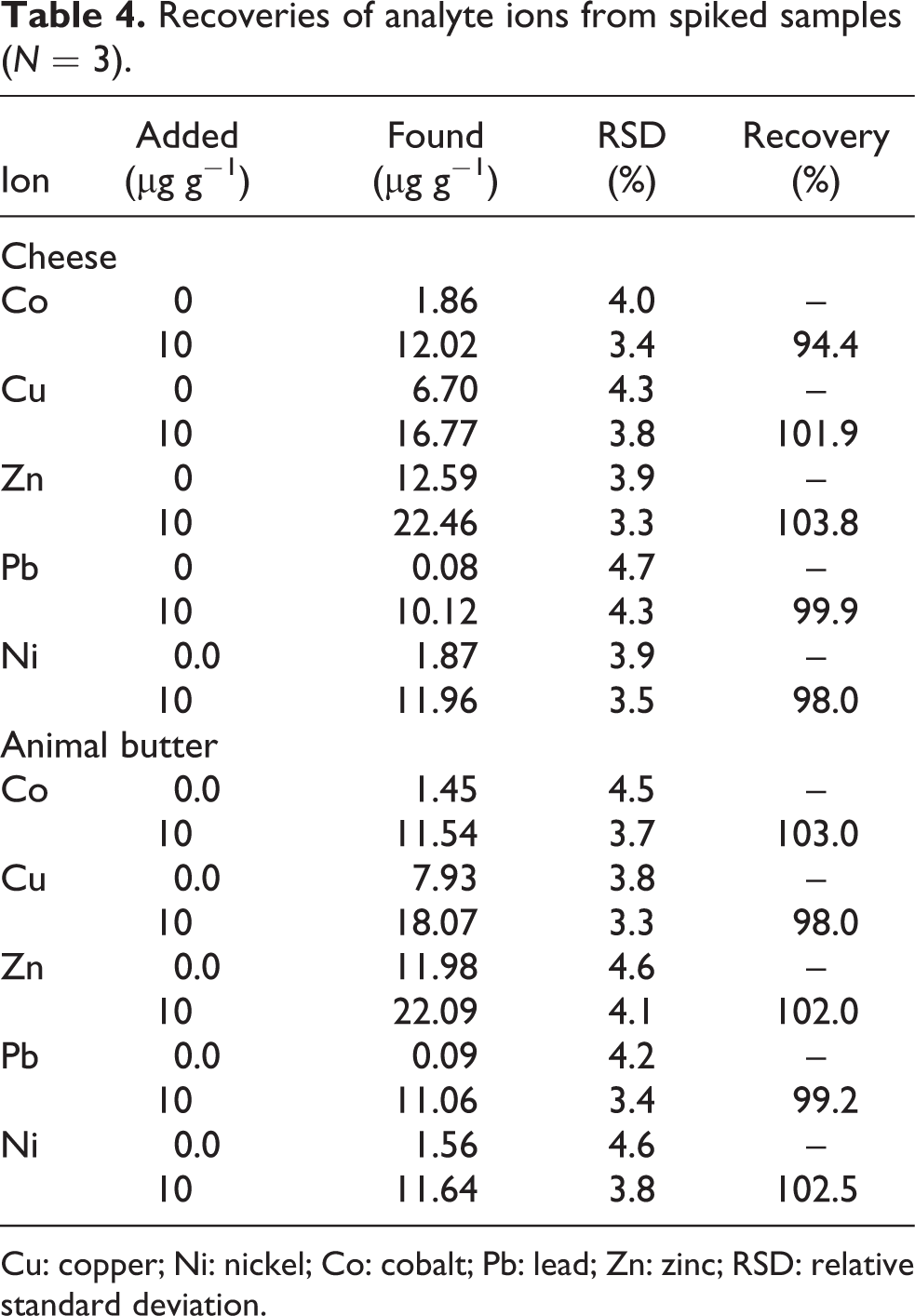
Cu: copper; Ni: nickel; Co: cobalt; Pb: lead; Zn: zinc; RSD: relative standard deviation.
Conclusions
A solid phase extraction technique was established based on the enrichment of Co2+, Cu2+, Ni2+, Pb2+ and Zn 2 . The conditions for quantitative and reproducible preconcentration, elution and FAAS determinations were studied (Table 5). The effects of matrix ions were found tolerable. The method is simple, economic and accurate and can be applied for the determination of these understudy metal ions in environmental samples. The system showed reproducibility and reliability in analytical data, with a relative SD value of <5% on triplicate experiment. The system was successful in preconcentration of analytes in large sample volume (1000 mL).
Comparative data for the preconcentration of simultaneous preconcentration of some metal ions using chemically modified sorbents and detection by FAAS.

FAAS: flame atomic absorption spectrometer; Cu: copper; Ni: nickel; Co: cobalt; Pb: lead; Zn: zinc; Mn, manganese; Fe, iron; LOD: limit of detection; DHMP: 4, 6-dihydroxy-2-mercaptopyrimidine; PT: preconcentration time; CI: cumulative index.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
