Abstract
Oxidative stress has an important role in the pathophysiology of many occupational diseases. In this controlled exposure study, the intensity of oxidative stress biomarkers was assessed in the workers of natural sweet and sour gas refineries (SwGR and SoGR, respectively) and compared with controls. In addition, the role of gluthatione S-transferase M1 (GSTM1)-null and GSTT1-null polymorphisms on the intensity of oxidative stress and liver function tests (LFTs) was investigated. Blood samples were taken and measured for lipid peroxidation (LPO) level, total antioxidant capacity (TAC) and total thiol molecules (TTMs). GSTM1- and GSTT1-null genotypes were determined using polymerase chain reaction. LPO was significantly (p < 0.05) higher in the workers of SoGR. TAC was significantly lower in SwGR subjects (p < 0.001). TTMs were significantly lower in SoGR and SwGR subjects. Among LFTs, activities of aspartate aminotransferase and alanine aminotransferase but not alkaline phosphatase were elevated significantly (p < 0.001) in SoGR subjects. Multivariate linear regression revealed no association between studied polymorphisms, oxidative stress biomarkers, and LFTs. These results indicate that working in the SoGR and SwGR can lead to oxidative stress and abnormal LFTs. Continuous monitoring of natural gas workers for probable ongoing problems is therefore suggested.
Introduction
Natural gas is a source of potentially toxic substances like hydrogen sulfide (H2S) and benzene that endanger the workers’ health in workplace. However, several genetic pathways are involved in minimizing the adverse effects of these environmental insults. 1 It is well documented that these pathways are individually variable; so that understanding the role of these environmental responsive genes on human susceptibility to occupational diseases may help to protect susceptible persons in workplaces.1–3 Gluthatione S-transferases (GSTs) are a group of polymorphic enzymes of phase II metabolism, which catalyze conjugation of glutathione onto reactive electrophillic agents in exogenous and endogenous compounds. 4 Three more important polymorphisms of GSTs are M1 (GSTM1) and T1 (GSTT1) gene deletions and one transition point mutation of A1578>G that creates the GSTP1 Ile105Val polymorphism with reduced activity.5–7
Iran has the second largest reserves of proved natural gas in the world. The largest reserve of Iran’s natural gas is the offshore South Pars field. The produced natural gas in the South Pars field feeds some refineries and petrochemical complexes that are located in Asalouyeh, in southern Bushehr province. Removal of H2S, as the main impurity in natural sour gas, occurs in some refinery plants in Asalouyeh. This process of desulfurization or sweetening emits higher levels of H2S in the workplace.
Exposure to H2S can lead to a variety of adverse health effects ranging from minor symptoms, for example, headache and upper respiratory tract problems, to severe health defects, such as neurologic problems, pulmonary edema, immediate collapse (‘knockdown’) and even death.8–11
H2S also exhibits genotoxic effects that are mediated through free radicals.12–14 Previous studies in Iran have reported that living in the proximity of natural sour gas fields results in increase in the incidence of depression and changes in hematologic and cardiovascular parameters.15,16 Benzene is an ubiquitous carcinogen that can be emitted into the air and groundwater from natural gas. 17 One of the most important sources of benzene emission from gas industries is glycol dehydrators. Glycol dehydration facilities are built at or near natural gas fields, where the produced gas contains a small amount of water.
There is an experimental evidence that the induction of oxidative stress plays a key role in the toxic effects of benzene and H2S.12,18–22 Most of the effects of oxidative stress induction by H2S have been reported in in vitro studies, but to our knowledge, based on a search of literature, no studies have been conducted on the oxidative stress induction in exposed populations. This, coupled with the proposed role of GSTs in the detoxification of many reactive oxygen species (ROS), provides a strong rationale for the evaluation of oxidative stress-based biomarkers and liver function tests (LFTs) in people exposed to causative substances and studying the relationships of GSTM1 and GSTT1 polymorphisms with these markers.
Methods
We carried out a cross-sectional controlled exposure study. The exposed were all male employees who were recruited from sweet gas refinery (SwGR; n = 120) and sour gas refinery (SoGR; n = 110) in Asalouyeh, Iran. The two refineries are located in two discrete locations far apart. According to local authorities, the atmospheric concentrations of H2S in the two places were 10–15 ppm in the SoGR site and <1 ppm in the SwGR site. The atmospheric concentration of benzene in both the sites was undetectable. Controls were 76 healthy men living in Kerman province without any exposure to H2S. The inclusion criteria for cases were male gender and working for at least 5 years in the refineries, and the exclusion criteria was not intake of antioxidant supplements. Demographic data, general health conditions, lifestyle, years of employment and smoking habits were registered in a questionnaire. Written informed consents were obtained from the subjects after describing the aim of the study. The study was conducted according to the declaration of Helsinki and subsequent revisions and approved by the Ethical Committee of Kerman University of Medical Sciences (No: 206/88/k, KMU).
DNA extraction and GST genotyping
DNA was extracted from EDTA-anticoagulant venous whole blood according to the method of Miller et al. 23 After measuring the quality and quantity of extracted DNA by determination of A260/A280, aliquots of DNA were stored in Tris-EDTA buffer at −70°C until analysis. Polymorphisms of GSTM1 and GSTT1 were determined as described before.24,25 Briefly, deletions in GSTM1 (X68676.1, GenBank) and GSTT1 (AB057594.1, GenBank) genes were detected by a multiplex polymerase chain reaction (PCR). In this technique, in one reaction tube the following fragments were amplified synchronously: 219 bp, 450 bp and 764 bp fragments for GSTM1, GSTT1 and c-Abl genes, respectively. The c-Abl gene was used as internal control for the verification of PCR reaction and amplifiable DNA. The absence of the GSTM1 or T1-specific fragments indicated the corresponding null genotype.
Determination of oxidative stress biomarkers
Determination of total antioxidant capacity (TAC) was according to Benzi and Strain 26 method and on the basis of the ability of plasma to reduce Fe3+ to Fe2+. The complex between Fe2+ and 2,4,6-tripyridyl-s-triazine gives a blue colour with maximum absorbance at 593 nm.26,27 Total plasma sulfhydryl content was determined by the method of Hu 28 with minor modifications as described previously. 27 The basis for the determination of lipid peroxidation (LPO) in plasma was the reaction of 2-thiobarbituric acid (TBA) with 1,1,3,3′-tetraethoxypropane (malondialdehyde) and other lipid peroxides. 29 Briefly, plasma samples were mixed with 20% trichloroacetic acid and the precipitate was dispersed in H2SO4 (0.05 mM). TBA (0.2% in sodium sulfate) was added and heated for 30 min in a boiling water bath. LPO adducts were extracted by n-butanol and absorbance was measured at 532 nm.27,29
Statistical analysis
For the comparison of continuous variables, we first checked the assumption that they were normally distributed. If the distribution was normal, results were expressed as means and standard error (SE) and a classical t test or one-way analysis of variance was used accordingly. If the distributions do not meet normal distribution criteria, then a logarithmic transformation was attempted to normalize the data (e.g. AST and ALT). According to GSTM1 and GSTT1 polymorphisms, the study population was divided into two categories. First category consisted of individuals with deletion in the GSTM1 or GSTT1 genes (coded 0 in analysis, reference genotype). The second category included persons who had the genes (coded 1).
Multiple linear regression was used to best predict the value of the dependent variables such as LFTs and oxidative stress biomarkers using independent variables such as age, smoking, body mass index (BMI), exposure to H2S (location) and GSTM1 and GSTT1 polymorphisms. Those variables believed to potentially confound the association with oxidative stress biomarkers and the elevations of LFTs were included in the multivariate model. For all the tests, a p < 0.05 was considered significant. All analyses were conducted using SPSS (version 11.5).
Results
The demographics are reported in Table 1. Fifteen (12.5%) subjects from SwGR and four (3.6%) subjects from SoGR were active current smokers. A total of 76 residents of Kerman province, mostly college students and all males, were enrolled as control subjects. The active current smokers were 4% in control group. There was significant difference between age and BMI of cases and controls (Table 1).
Demographics of SwGR and SoGR workers and controls

SwGR: sweet gas refinery; SoGR: sour gas refinery; SE: standard error.
a p < 0.001 in comparison with control using one-way analysis of variance.
b p < 0.01 in comparison with control using one-way analysis of variance.
c p < 0.05 in comparison with SwGR using Chi-square test.
The oxidative stress biomarkers of studied groups are shown in Figure 1. Increase in LPO was observed in the workers of SwGR and SoGR. This increase was only significant for SoGR workers in comparison with controls (p < 0.05). TAC was significantly higher in the SoGR workers than controls (p < 0.001). However, TAC was significantly lower in the SwGRP workers than controls (p < 0.001). The concentration of total thiol molecules (TTMs) was significantly (p < 0.001) lower in the SwGR and SoGR workers in comparison with controls. Their concentrations were also significantly (p < 0.001) lower in the SoGR in comparison with SwGR workers. Simple and multivariate linear regressions were performed to analyze the association of GSTM1 and GSTT1 polymorphisms with oxidative stress biomarkers (Table 2). There was a negative weak association between GSTM1 polymorphism and LPO (B = −3.25, p = 0.06) indicating that LPO should be found lower in the subjects with GSTM1-present allele. This association was abolished (B = −0.70, p = 0.7) when multivariate regression was performed while controlling for location, age and BMI. Backward linear regression showed that only location was significantly associated with LPO (B = 3.3, p = 0.005). There was no association between GSTT1 polymorphism and LPO. Because of probable intervention of H2S in TAC measurement (see Discussion section), the statistical analysis was done only for controls and SwGR subjects. There was no association between GSTM1 and GSTT1 and TAC and it was not affected by controlling variables. The concentration of TTMs in plasma was significantly associated with GSTM1 (B = 0.04, p < 0.01), but this relationship was abolished when location entered into the analysis (B = 0.02, p > 0.05).

Lipid peroxidation (LPO), total antioxidant capacity (TAC) and total thiol molecules oxidative stress biomarkers in the sweet and sour gas refinery (SwGR and SoGR, respectively) workers and controls. *p < 0.05, ***p < 0.001 in comparison with controls, ¶¶¶p < 0.001 in comparison with SwGR using one-way analysis of variance. Data are expressed as mean ± standard error.
Association of GSTM1 and GSTT1 polymorphisms with oxidative stress biomarkers in the SwGR and SoGR workers and population controls

B: partial regression coefficient; CI: confidence interval; LPO: lipid peroxidation; TAC: total antioxidant capacity; TTM: total thiol molecules; GSTM1/GSTT1: gluthatione S-transferases M1/T1; SwGR: sweet gas refinery; SoGR: sour gas refinery.
aMultivariate regression. Parameters adjusted for age, smoking status, BMI and location.
bAnalysis was performed only for SwGR subjects.
Analysis of regression was also performed for determining the association of GSTM1/GSTT1 double deletion and TTMs and LPO levels. The LPO and TTMs were insignificantly associated with GSTM1/GSTT1 double deletion (B = −0.84, p = 0.3 and B = 0.06, p = 0.4, respectively for LPO and TTMs).
The correlation of serum AST (logarithmic) with the GSTM1 and GSTT1 polymorphisms, oxidative stress biomarkers, H2S exposure and potential confounders like age and BMI have been shown in Table 3. The only significant predictor for logAST in the subjects was the location of working (β = 0.51, p < 0.01). This indicates that logAST was significantly higher in the SoGR than SwGR. There were weak associations between GSTM1 (β = −0.01) and GSTT1 (β = 0.03) polymorphisms and logAST. Table 4 shows that H2S exposure (location) and GSTT1 polymorphism are significantly associated with serum logALT (β = 0.59, p < 0.01 and β = 0.12, p = 0.05, respectively). Table 5 shows the fitted models for ALP by using multivariate linear regression. The fitted model shows that there is no significant association between either of the variables and ALP.
Multiple linear regression models fitted for the levels of logAST using independent variables

B: partial regression coefficient; SE: standard error; CI: confidence interval; BMI: body mass index; TTM: total thiol molecules; LPO: lipid peroxidation; GSTM1/GSTT1: gluthatione S-transferases M1/T1.
aStandardized coefficient.
bFor model adjusted R 2 = 0.22; F = 8.21; df = 7, 183; p = 0.00.
Multiple linear regression models fitted for the levels of logALT using independent variables
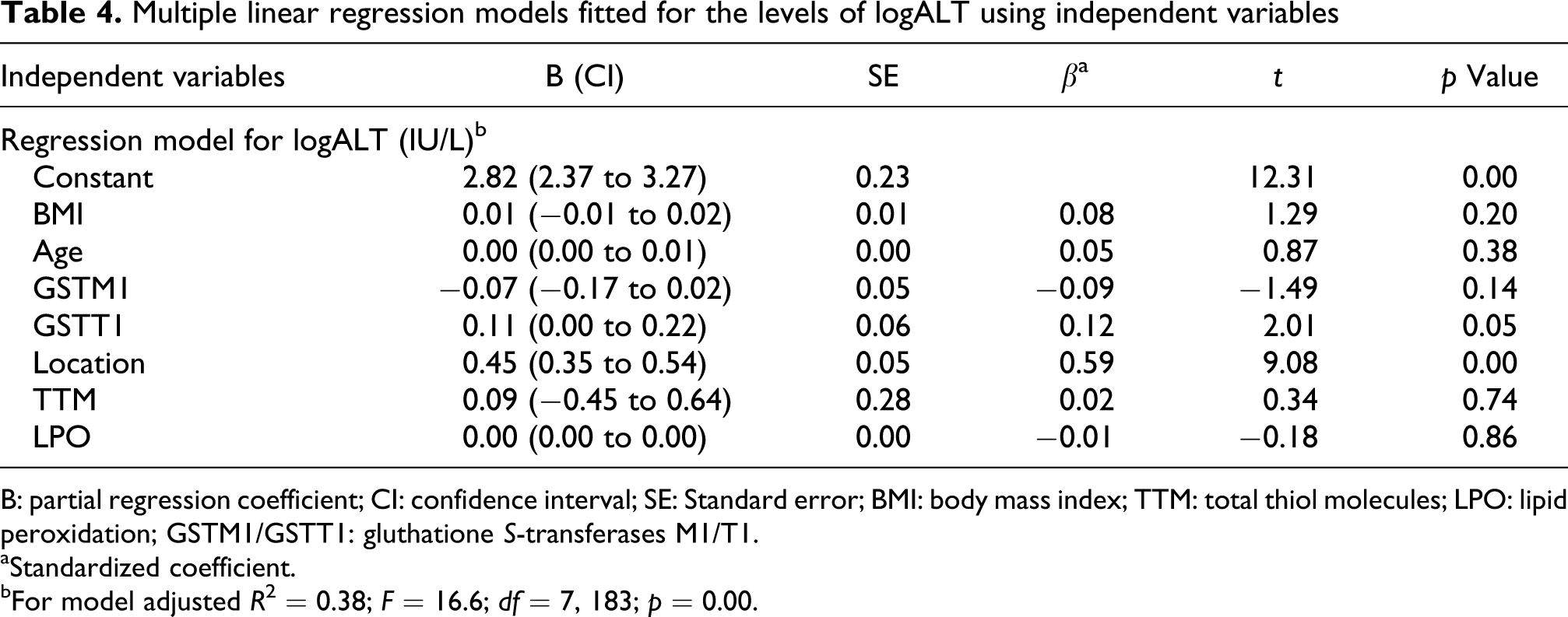
B: partial regression coefficient; CI: confidence interval; SE: Standard error; BMI: body mass index; TTM: total thiol molecules; LPO: lipid peroxidation; GSTM1/GSTT1: gluthatione S-transferases M1/T1.
aStandardized coefficient.
bFor model adjusted R 2 = 0.38; F = 16.6; df = 7, 183; p = 0.00.
Multiple linear regression models fitted for levels of ALP using independent variables
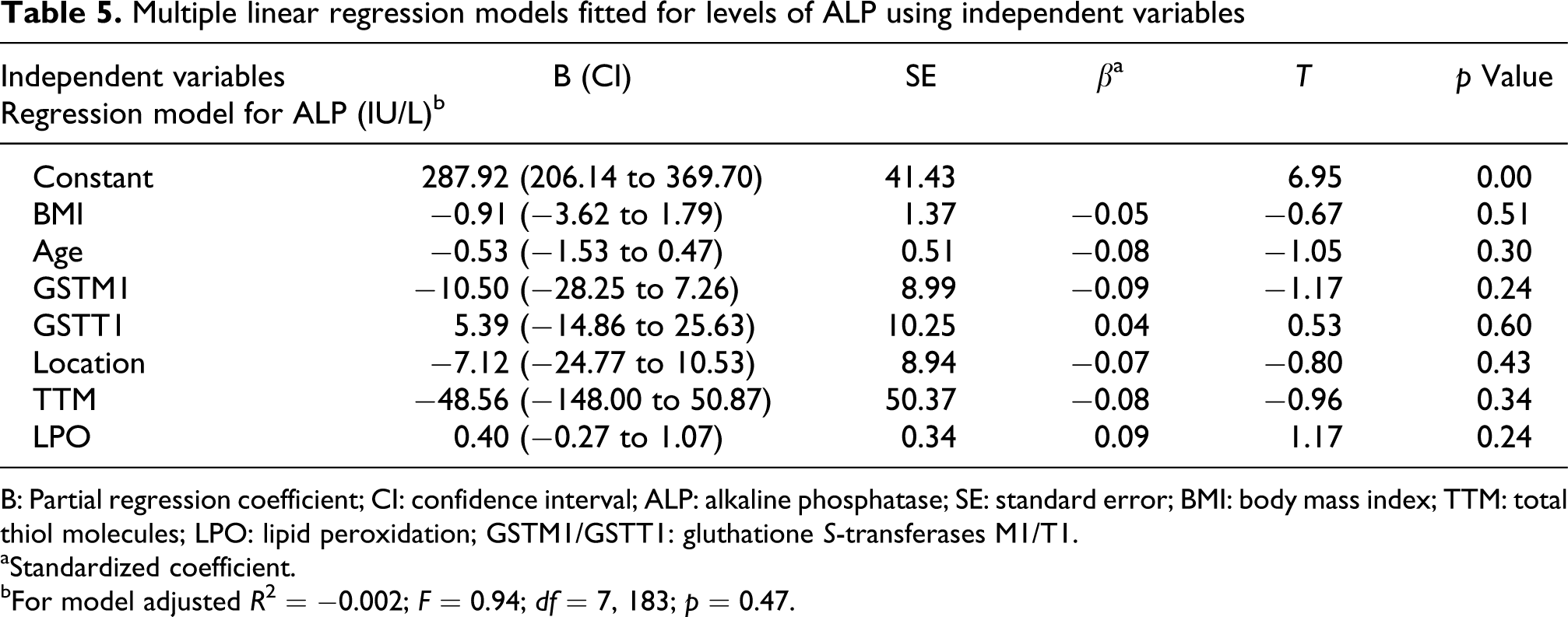
B: Partial regression coefficient; CI: confidence interval; ALP: alkaline phosphatase; SE: standard error; BMI: body mass index; TTM: total thiol molecules; LPO: lipid peroxidation; GSTM1/GSTT1: gluthatione S-transferases M1/T1.
aStandardized coefficient.
bFor model adjusted R 2 = −0.002; F = 0.94; df = 7, 183; p = 0.47.
Discussion
In this study, the intensity of oxidative stress was investigated in the subjects working in the SwGR and SoGR in comparison with controls. In addition, the effect of polymorphisms of GSTM1 and GSTT1 genes, which belong to a very important family of antioxidant enzymes, 30 on the oxidative stress intensity and the level of LFTs was evaluated. GSTs owe their importance to their role in defending against unwanted electrophillic metabolites resulted from free radicals and other reactive species avoiding their attack on cellular macromolecules. The removal is mediated through conjugating them with glutathione. 4
LPO as a marker of ROS concentration was markedly increased, while TAC and thiol groups as markers of serum antioxidant defence mechanisms were found significantly decreased in the SwGR and SoGR subjects. These changes were more prominent for SoGR subjects. Although multiple occupational and environmental factors related to gas refinery field such as CS2, benzene and H2S are known to be responsible for oxidative stress induction,8,12,13,17,20,31–39 only H2S was found to be of high concentration at the SoGR in our study. Taking all these into consideration, it can be expressed that the induced oxidative stress observed in this work is postulated to be due to H2S exposure. It is, however, noteworthy that the observed increase in TAC in the SoGR subjects can be attributed to the intervention of H2S in the determination of TAC. The basis for measurement of TAC is ‘the ferric reducing ability of plasma’ 40 that can be affected by strong reducing agents like H2S and its main metabolite in the body namely thiosulphate.41–44 This might justify the inconsistency of TAC results with LPO and TTMs in SoGR subjects. For this reason, we excluded SoGR from further statistical analysis on TAC data.
Moreover, we studied the role of GSTM1 and GSTT1 polymorphisms on the oxidative stress biomarkers. Multivariate linear regression controlling for age, BMI and location showed no significant associations between the polymorphisms and studied oxidative stress biomarkers (Table 2). The association of GSTM1/GSTT1 double deletion and total oxidative stress biomarkers showed an increase in the LPO of the workers with double deletion in M1 and T1(B = −0.84, p = 0.3). However, TTMs were positively associated with GSTM1/GSTT1 double deletion (B = 0.06, p = 0.4) suggesting a decrease in TTMs in the subjects with M1/T1 double deletion.
The role of the polymorphisms on the concentration of ALP, ALT and AST was also investigated in the SwGR and SoGR subjects. Multiple linear regression showed that ALP level was not associated with GSTM1 and GSTT1 polymorphisms. Neither did it show the association with location of working (Table 5). By contrast, association between AST and ALT levels and location of working was evident (Tables 3 and 4), so that AST and ALT significantly increased in the SoGR subjects. Elevation in the LFTs was too moderate to meet the criteria for ‘hepatocellular injury’.45,46 The two main mechanisms responsible for the cytotoxicity of H2S are inhibition of cytochrome oxidase39,47,48 and production of ROS. 12 ,20 Dorman et al. 39 showed that the main hepatotoxic mechanism of H2S is not the inhibition of cytochrome oxidase. On the other hand, Eghbal et al. 20 have suggested that CYP450-dependant metabolism of H2S is responsible for inducing ROS production by hepatocytes. Our results supported by this evidence demonstrate that the cellular damage caused by H2S-mediated ROS is the main mechanism of elevation in the LFTs in SoGR subjects. For this, association of oxidative stress and LFTs were evaluated by introducing LPO and thiol molecules as predictors of LFT levels. Although there was no correlation between these two oxidative stress biomarkers and the level of LFTs (Table 2), significant association was observed between GSTT1 polymorphism and ALT level (β = 0.12, t = 2.01, p = 0.05; Table 4). The latter, indicates that ALT level is lower in the GSTT1-null subjects. This is in line with the striking point that persons with a GSTT1-null genotype are at a lower risk of certain tumors.49–52
This work benefits from several strength points. Evaluating oxidative stress biomarkers in the vast population of natural gas refinery workforce as an at-risk community is absent from literature. Bringing together three populations different in their exposure to hazardous substances is another point, which is unique to our work so far as we are aware, adding to it the considerable size of recruited groups. Further strengthening factor comes from studying gene–environment interactions. It is however well worth to make recommendations which would be beneficial if similar studies to our work are to be carried out. Expanding on further oxidative stress markers is suggestible to widen the scope of investigations. A more well-matched control group than ours would have provided a much stronger conclusion through statistics. The results of this research indicate that working in natural gas refineries can increase serum biomarkers of oxidative stress. This may be valuable in deciding to add antioxidant supplements to their daily diet and also in assessing whether further reduction in worker exposures is needed.
Footnotes
Acknowledgments
The authors thank Ms V. Montazeri and Ms A. Haghdoost for their technical assistant.
Funding
This work was supported by a grant from Kerman University of Medical Sciences (to A.M.; grant number: 88/179).
