Abstract
Introduction
Cancer is a growing health problem due to the changes in lifestyle such as inappropriate diet, obesity, infections, radiation, stress, use of tobacco, lack of physical activity and exposure to the environmental pollutants. 1 The risk of cancer can be reduced either by eliminating the identified carcinogens or by minimizing exposure to them, for instance, by dietary habits. Epidemiological studies indicated that many dietary agents are well-known chemopreventive agents. 2 These agents have the ability to modulate at more than one critical pathway of carcinogenesis. So, the chemopreventive intervention using the naturally occurring dietary substance is extremely desirable to delay the process of carcinogenesis. 3
The development of skin cancer is a multistage process that involves initiation, promotion and progression. 4,5 In the mouse model of skin cancer, tumor initiation can be caused by a single topical application of 7,12-dimethylbenz[a]anthracene (DMBA; a carcinogen that mutates H-Ras proto-oncogene). 6 Subsequently, the repeated applications of 12-O-tetradecanoylphorbol 13-acetate (TPA) trigger tumor promotion, involving clonal expansion of initiated cells. 6 The mutated Ras protein gets farnesylated by the enzyme farnesyl transferase (FTase). The farnesylated Ras protein then gets attached to the cell membrane which leads to the activation of Ras/Raf/extracellular signal-regulated protein kinase 1/2 (ERK1/2) cascade. 7,8
Applications of TPA also induced the generation of reactive oxygen species (ROS) which induces DNA strand breaks and oxidation of DNA bases, leading to mutagenesis. 5,9 Increased level of ROS damages almost all biomolecules such as DNA, RNA and proteins, thus playing a key role in the pathophysiology of a number of degenerative and chronic diseases, including cancer. 9 During oxidative stress, a reduction in the activities of antioxidative enzymes viz. catalase (CAT), glutathione reductase (GR), glutathione peroxidase (GPX), glutathione S-transferase (GST) and reduced glutathione (GSH) content has been found with an increase in the level of ROS in murine skin. 9 The status of intracellular redox is regulated by antioxidant enzymes (CAT, GR, GPX and GST) and nonenzymatic antioxidants (GSH and vitamin C). 10 They also activate proinflammatory genes such as in the expression of cyclooxygenase-2 (COX-2) and various transcription factors. 11
In the present study, we investigated the effect of
Materials and methods
Chemicals
The chemicals used in this study such as
Animals
Female Swiss albino mice (6–8 weeks old; 20–25 g) were obtained from Central Animal House Facility of Hamdard University, New Delhi. The mice were housed in a well-ventilated room at 25 ± 2°C under 12-h light:12-h dark cycle. The mice were fed pellet diet (Hindustan Lever Ltd, Bombay, India) and drinking water ad libitum. The study protocols (CAHF file no. 283) were approved by the Committee for the Purpose of Control and Supervision on Experiments on Animals (CPCSEA), India. The mice were treated and killed according to approved ethical guidelines. The dorsal skin of the mice was shaven with an electric clipper (Oster A2, USA) followed by the application of hair-removing cream (Anne French, Geoffery Manners, Bombay, India) at least 2 days before treatment.
Treatment schedule for investigating the effect of d -limonene on TPA-induced tumor promotion biomarkers in the skin
Mice were randomly divided into five groups of six each. Mice of group 1 received topical treatment of 200 μl acetone and served as vehicle control group. Mice of group 3 were topically applied with
For estimation of ODC activity and [3H] thymidine incorporation, the treatment schedule and allotment of mice were exactly same as above. To measure the ODC activity, the animals of all groups were killed after 6 h of last TPA treatment. For the [3H] thymidine incorporation into epidermal DNA, mice of all the groups were injected intraperitoneally with [3H] thymidine (20 μCi/mouse/200 μl saline) 18 h after the last TPA treatment and were killed after 2 h of [3H] thymidine treatment.
Mouse skin edema and histopathological studies
The estimation of skin edema was done per the method described by Afaq et al.
4
A skin punch of 1 cm diameter was made fat-free and weighed quickly. The skin punch was dried for 24 h at 50°C and reweighed. The loss of water content was determined. The extent of edema was calculated as the difference in water gain between control and TPA-treated groups. The inhibitory effect of
For histopathological study, the dorsal skins were removed and fixed in 10% neutral buffered formalin. The skins were then embedded in paraffin wax as standard procedure and 5 µm sections were cut. The sections were stained with hematoxylin & eosin. The infiltrations of leukocytes in dermis were observed and epidermal thickness (as an indicator of hyperplasia) was measured at least in six different regions using microscope (Motic Digital Microscope).
Immunohistochemical analysis
The dorsal skins of mice were excised and fixed in 10% buffered formalin. The skins were embedded in paraffin wax. Five-micrometer thick sections were cut and mounted on silanized glass slides. The sections were deparaffinized in xylene and dehydrated in graded ethanol. For antigen retrieval, the sections were kept in 10 mM citrate buffer (pH 6.0) at 95°C for 6 min. Each section was treated with 3% H2O2 for 20 min and then with blocking solution (1% non-fat skimmed milk) for 30 min. Sections were incubated with monoclonal anti-COX-2 antibody at room temperature for 2 h. Thereafter, they were incubated with secondary antibody at room temperature for 1 h. In order to develop the color, DAB was applied on each section for 3–5 min. Sections were then rinsed with distilled water and counterstained with hematoxylin. Finally, they were microscopically (Motic Digital Microscope) analyzed at least in six different regions.
Tissue preparation and assays of nonenzymatic and enzymatic antioxidants
The dorsal skins of mice were minced into small pieces and homogenized (Kinematica Polytron PT 3100 Homogenizer, Switzerland) in chilled phosphate buffer (0.1 M, pH 7.4). The epidermal homogenates were centrifuged at 10,000g for 30 min to get the post-mitochondrial supernatant, which were used for the estimation of GSH, GPX, GR, GST and CAT. GSH was determined by the method of Jollow et al. 19 GR and GPX activities were measured by the method of Mohandas et al. 20 GST activity was estimated by the method of Habig et al. 21 CAT activity was assayed per the method of Claiborne, 22 and the level of malondialdehyde (MDA) as an end product of lipid peroxidation was assessed according to the method of Wright et al. 23
Estimation of ODC activity
The dorsal skins were minced into small pieces and were homogenized in ice-cold Tris-HCl buffer (50 mM, pH 7.5) containing 0.4 mM EDTA, 0.32 mM pyridoxal phosphate, 0.1 mM PMSF, 1 mM 2-merceptoethanol, 4 mM dithiothreitol and 0.1% Tween-80. The homogenates were then centrifuged at 10,000g for 30 min and a portion of supernatant was centrifuged at 105,000g for 60 min at 4°C to obtain the microsomal pellet and cytosol. The cytosol was used for the estimation of ODC activity per the method of Verma et al. 24 In brief, the reaction mixture (495 μl) contained 400 μl of cytosolic enzyme solution and 95 μl of co-factor mixture containing 0.32 mM pyridoxal phosphate, 0.4 mM EDTA, 4 mM dithiothreitol, 0.4 mM ornithine, 0.02% Brij-35 and 0.05 μCi DL [14C] ornithine. After adding the enzyme solution and cofactor mixture, the test tubes were immediately closed with rubber stopper and the central well assemblies were containing 0.2 ml ethanolamine and methoxyethanol mixture in 2:1, v/v ratio. The reaction mixture was kept in water bath at 37°C for 1 h. The reaction was arrested by injecting 1.0 ml of 2.0 M citric acid solution along side of glass tubes and the solution was kept for 1 h to ensure complete absorption of CO2. Finally, solution in central well was transferred to vial containing 2 ml ethanol and 10 ml scintillation fluid. Radioactivity was counted in scintillation counter (Beckman LS 6500 multipurpose). ODC activity was expressed as pmol 14 CO2 released per hour per mg protein.
Assessment of [3H] thymidine incorporation
Assessment of incorporation of [3H] thymidine was done per the method of Smart et al. 25 Skins were quickly removed and the extraneous materials including dermis were cleaned, and homogenates (10%, w/v) were prepared in ice-cold water. To the homogenate, an equal volume of ice-cold 10% TCA (trichloroacetic acid) was added and centrifuged at 2500g for 10 min. The supernatant was then discarded and the precipitate was washed with cold 5% TCA and centrifuged. The precipitate thus obtained was incubated with cold 10% perchloric acid (PCA) at 4°C overnight. The suspension was centrifuged, washed with 5% cold PCA and dissolved in warm 10% PCA, followed by incubation in a boiling water bath for 30 min. It was then filtered through Whatman 50 filter paper to get clear nucleotide solution, which was taken for DNA estimation by DPA (diphenylamine) method. A fixed volume of filtrate was used for [3H] thymidine counting in liquid scintillation counter (Beckman LS 6500 multipurpose). The amount of [3H] thymidine incorporated was expressed as dpm/μg DNA.
Two-stage mouse skin tumorigenesis
The mice were divided in four groups of 20 mice each. Mice of group 1 received 200 μl acetone only and served as vehicle control. The treatment of the dorsal skin of mice of groups 2, 3 and 4 was initiated by single topical application of 50 μg DMBA in 200 μl acetone. One week later, tumor growth was promoted by repeated topical applications of TPA (2 mg in 200 μl acetone). Mice of group 2 were treated with TPA alone and served as positive control. However, mice of groups 3 and 4 topically received
Preparation of skin/tumor lysates for western blot analysis
The excised dorsal skin/tumors were cleaned to remove the extraneous materials with a sharp scalpel blade in ice-cold phosphate-buffered saline and pooled in vial. The skin/tumor lysates were prepared as described by Kundu et al., 26 with minor modifications. Briefly, the pooled skins/tumors were minced and homogenized in ice-cold lysis buffer (20 mM Tris-Cl, pH 7.5; 10 mM KCl, 0.1 mM EDTA, 2 mM MgCl2, 1 mM PMSF, 1 mM DTT, 0.1% nonylphenol ethoxylate (NP)-40 and freshly prepared 1 mM each of pepstatin, leupeptin and aprotinin). Then, the homogenate was centrifuged at 10,000g for 30 min at 4°C, and the supernatant was collected as cytosol fraction and aliquoted in 1 ml tubes. The pellet was processed for membrane fraction, resuspended in buffer (20 mM Tris-Cl, pH 7.5; 0.1 mM PMSF, 0.1% sodium dodecyl sulfate (SDS) and 1% NP-40) and centrifuged at 14,000g for 10 min at 4°C. The supernatant was collected as membrane fraction and aliquoted in 1 ml tubes. All aliquots were stored at −80°C. The protein content in each sample was assayed according to the method of Bradford 27 using bovine serum albumin as standard.
Western blot analysis
For Western blot analysis, 50–75 µg of the proteins were resolved by 10–12% SDS-polyacrylamide gel electrophoresis and blotted onto nitrocellulose membrane. The membranes containing transferred protein were blocked with 5% (w/v) non-fat dry milk in TTBS buffer (0.1% Tween-20, 20 mM Tris base and 137 mM NaCl, pH 7.6) for 1 h at room temperature followed by incubation with appropriate primary antibody (Ras, Raf, p-ERK1/2, Bax, Bcl-2 and β-actin) for 2 h at 4°C. After washing the membrane three times in TTBS for 10 min each, the membrane was incubated with horseradish peroxidase conjugated with anti-mouse IgG or anti-rabbit IgG secondary antibodies for 2 h. The membrane was washed 3 times with TTBS buffer for 10 min each and detected by DAB in 0.1 M Tris-Cl pH 7.5 in the presence of H2O2. To quantify equal loading, identical gels were run simultaneously and probed with β-actin antibody in the same manner as described above.
Statistical analysis
The difference between various groups was analyzed by Student-Newman-Keuls t test after the application of one-way analysis of variance by InStat software. Values of p < 0.05 were considered statistically significant.
Results
Effect of d -limonene on TPA-induced mouse skin edema and skin morphological changes
Earlier studies suggested that TPA treatment to mouse skin causes cutaneous edema, hyperplasia and infiltration of leukocytes, mainly neutrophils and monocytes, in the dermis
4,8
which play an important role in tumor promotion. After 12 h of topical treatment of TPA, the skin punch weight was significantly increased upto 48%; p < 0.001 as compared to acetone treatment (Table 1). Whereas, the pretreatment with
Effect of limonene on TPA-induced histological changes (epidermal thickness, number of epidermal layers and leukocyte infiltration) in mouse skin a

TPA: 12-O-tetradecanoylphorbol-13-acetate.
aEach value represents the mean ± SE of six regions in the slides of each groups. Treatment protocols and dose regimen are described in Material and methods section. (++++) severe infiltrations; (++) moderate infiltrations; (+) mild infiltrations; (−−−) no infiltration. Groups 5 (limonene, 100 mg/kg body weight) and 2 (TPA-treated; 2 µg/200 µl acetone/mouse) are compared to the control group 1 (200 µl acetone/mouse). Groups 3 (limonene, 50 mg/kg body weight + TPA) and 4 (limonene, 100 mg/kg body weight + TPA) are compared to group 2 (TPA-treated).
b p < 0.05.
c p < 0.01.
d p < 0.001.
eNot significant.
The effect of

Effect of pretreatment of
Effect of d -limonene on TPA-induced expression of COX-2 enzyme in mouse skin
TPA application induces COX-2 expression in epidermal cells which plays an important role in inflammation, cell proliferation and skin tumor promotion.
28
By immunohistochemical analysis, we demonstrated the effect of pretreatment of

Effect of pretreatment of
Effect of d -limonene on TPA-mediated decrease in CAT, GSH and its redox enzymes in mouse skin
Topical application of TPA to mouse skin has been reported to reduce the activity of CAT, GSH and its redox enzyme.
9,29
As compared to acetone-treated mice, the activity of CAT in TPA-treated skin exhibited a decrease of 58% (p < 0.001). Whereas
Effect of pretreatment of limonene on TPA-induced alterations in cutaneous levels of reduced glutathione (GSH) and activities of antioxidant enzymes a

CAT: catalase; CDNB: 1-chloro 2,4-dinitrobenzene; GPx: glutathione peroxidase; GR: glutathione reductase; GSH: reduced glutathione; GST: glutathione S-transferase; MDA: malondialdehyde; NADPH: reduced nicotinamide adenine dinucleotide phosphate; TPA: 12-O-tetradecanoylphorbol-13-acetate.
aEach value represents the mean ± SE of six animals. Treatment protocols and dose regimen are described in Material and methods section. Groups 5 (limonene, 100 mg/kg body weight) and 2 (TPA treated; 2 µg/200 µl acetone/mouse) are compared to the control group 1 (200 µl acetone/mouse). Groups 3 (limonene, 50 mg/kg body weight + TPA) and 4 (limonene, 100 mg/kg body weight + TPA) are compared to group 2 (TPA-treated).
b p < 0.05.
c p < 0.01.
d p < 0.001.
eNot significant.
Effect of d -limonene on TPA-induced epidermal ODC activity and [3H] thymidine incorporation
Studies suggested that TPA application to mouse skin results in overexpression of ODC enzyme.
4,30
As shown in Figure 3(a), application of TPA to mouse skin significantly increased the ODC activity by 733% (p < 0.001) as compared to acetone-treated mice. However, the application of
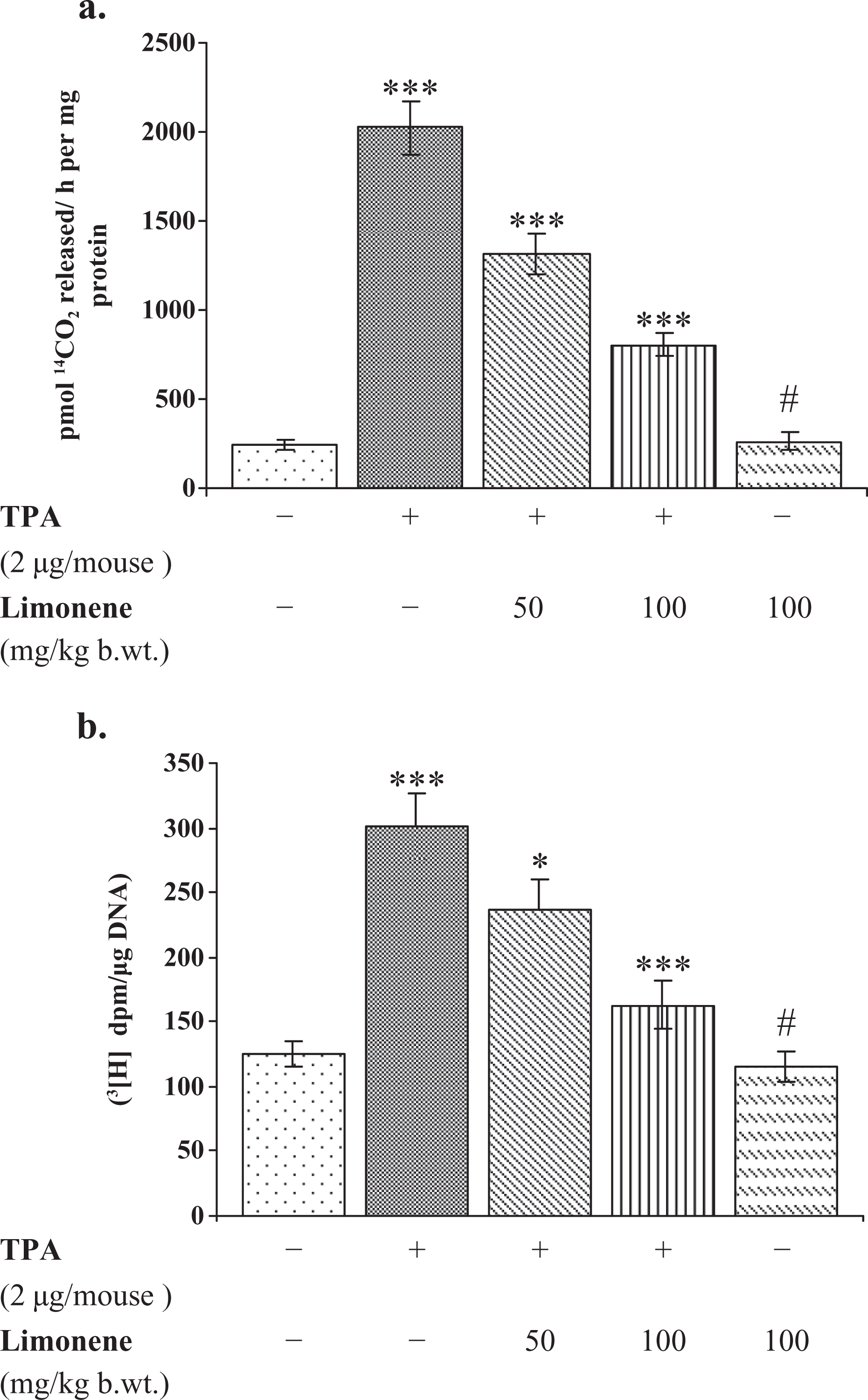
Effect of pretreatment of
Effect of d -limonene on DMBA/TPA-mediated skin tumor promotion
The effect of

Effect of pretreatment of
Effect of d -limonene on DMBA/TPA-induced expression of Ras, Raf and p-ERK1/2 in skin/tumors
To determine whether DMBA/TPA could induce the activation of Ras/Raf/ERK1/2 signaling pathway under in vivo condition, Western blot analysis was performed using Ras, Raf and phospho-specific ERK1/2 antibodies. The level of membrane-bound Ras protein in

Effect of pretreatment of
Raf protein expression was analyzed in connection with Ras protein analysis. An elevated level of Raf protein was observed in DMBA/TPA-mediated skin tumor in comparison with acetone-treated skin (Figure 5). Whereas, pretreatment of
Effect of d -limonene on DMBA/TPA-induced expression of Bax and Bcl-2 in skin/tumors
To find out the role of Bax and Bcl-2 protein expression for apoptosis, we performed Western blot. It was observed that DMBA/TPA treatment downregulated Bax expression in comparison to acetone treatment. However, pretreatment with
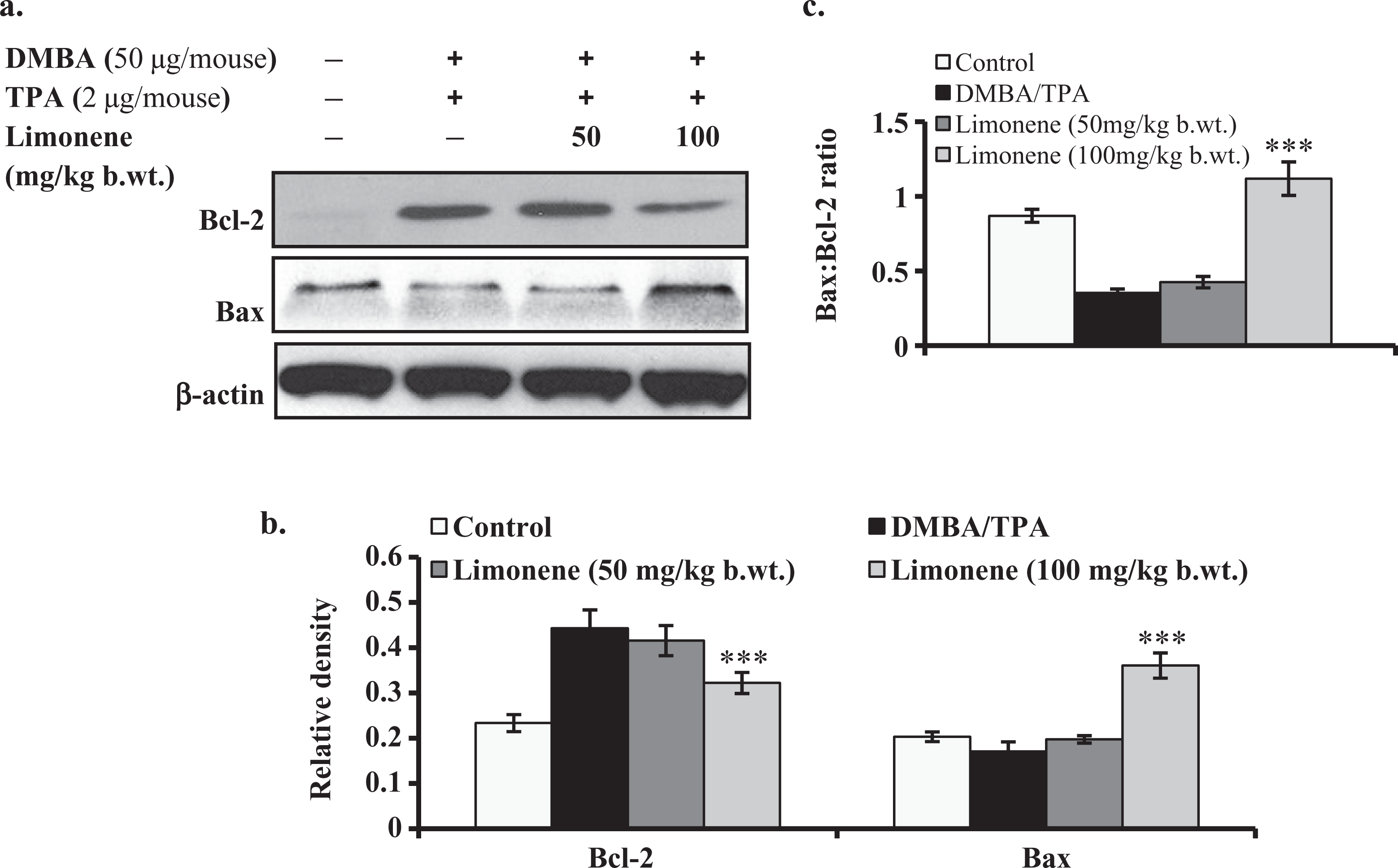
Effect of pretreatment of
Discussion
The promotion stage in multistep tumorigenesis offers an opportunity to delay the process of cancer development. It provides a realistic path to understand the mechanism of action of chemopreventive dietary agents. Multiple lines of studies showed that phorbol-type tumor promoters induce inflammatory responses like induction of edema, hyperplasia and COX-2 expression in epidermal cells leading to malignant transformation.
4,8
Histological changes (edema and hyperplasia) of skin and infiltration of polymorphonuclear (PMN) leukocytes in dermis are responsible for increasing the risk of carcinogenesis. TPA-induced upregulation of COX-2 and subsequently production of prostaglandins (PGs) indicate its role in tumor promotion and in tumorigenesis.
31
Maier et al.
32
reported that interleukin-1a induces PG synthesis and primes leukocytes to enhance the production of reactive oxygen intermediates in epidermal keratinocytes; the latter have been implicated in the induction of tumor formation. However, our result showed that pretreatment with
Accumulating evidences showed that the tumor promoter induces infiltration and activation of leukocytes which induce the production of ROS in the cells.
9,33
According to earlier observations,
ODC is a rate-limiting enzyme of polyamines (putrescine, spermidine and spermine) biosynthesis that is essential for normal cell growth and differentiation.
35
Elevation in the activity of ODC has been detected in transformed cell line and experimental animal tumors.
30
Application of tumor promoter induced the activity of ODC and incorporation of thymidine into DNA, thereby enhancing the growth and differentiation of cell leading to development of cancer. Our data showed that
We demonstrated that
Inhibition of Ras pathway alters the expression level of many intermediate apoptotic proteins like Bim, Bax, Bcl-2 and so on, thereby leading to apoptosis.
37
The Bcl-2 gene product is a negative regulator of apoptosis, which forms a heterodimer complex with Bax and neutralizes the effect of pro-apoptosis. Thus, the ratio of Bax-to-Bcl-2 protein plays a crucial role in apoptosis in DMBA/TPA-treated mouse skin.
38
In the present study, we found that
In conclusion, the topical treatment of
Footnotes
Acknowledgements
The authors are thankful to the Indian Council of Medical Research (ICMR), New Delhi, for providing the fellowship to the first author during the course of study. The authors are also thankful to Dr Usha Agrawal, Institute of Pathology (IOP), New Delhi, for providing necessary suggestions in histopathology analysis.
Declaration of Conflicting Interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
